Sodium Bicarbonate 10 Gr Tablets, Usp
4493d7c5-f2c3-416e-a5c1-35398fc7f61d
34390-5
HUMAN OTC DRUG LABEL
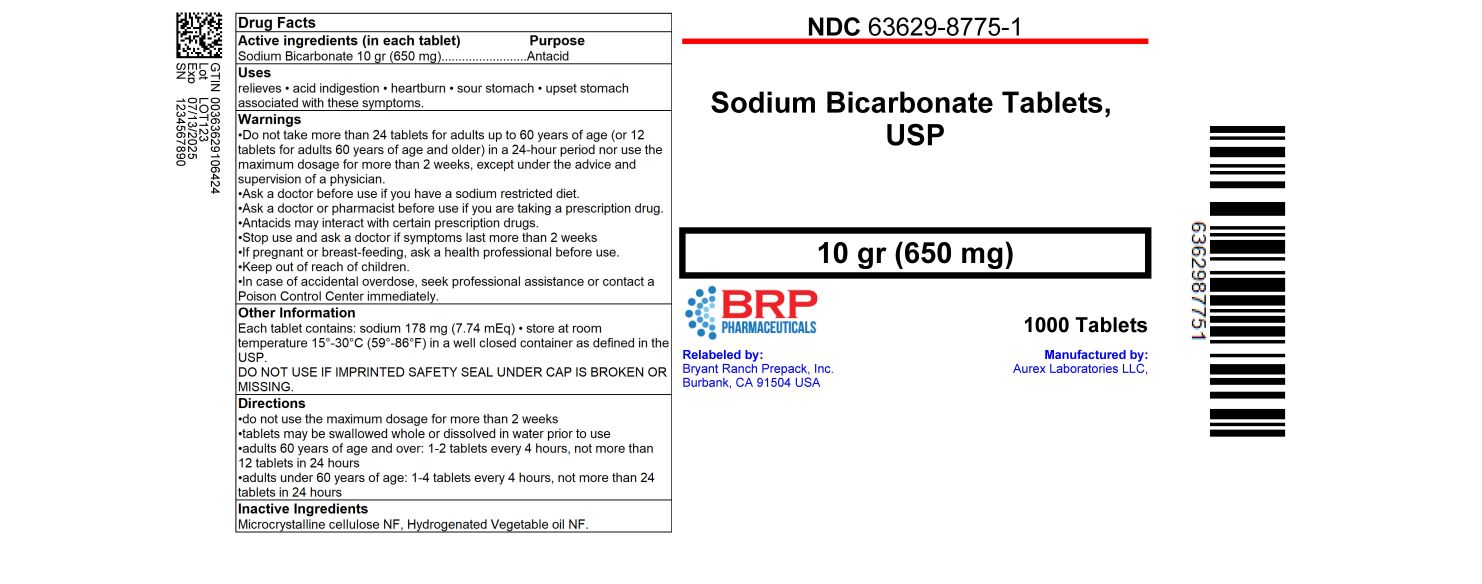
Drug Facts
Composition & Product
Identifiers & Packaging
Description
DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
How Supplied
NDC 63629-8775-1: 1000 Tablets in a BOTTLE Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
Medication Information
Recent Major Changes
DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
Warnings and Precautions
Warnings
Do not take more than 24 tablets for adults up to 60 years of age (or 12 tablets for adults 60 years of age and older) in a 24-hour period nor use the maximum dosage for more than 2 weeks, except under the advice and supervision of a physician.
Ask a doctor before use if you have a sodium restricted diet.
Ask a doctor or pharmacist before use if you are taking a prescription drug.
Antacids may interact with certain prescription drugs.
Stop use and ask a doctor if symptoms last more than 2 weeks
If pregnant or breast-feeding, ask a health professional before use.
Indications and Usage
Uses: relieves • acid indigestion • heartburn • sour stomach • upset stomach associated with these symptoms
Dosage and Administration
Directions
- do not use the maximum dosage for more than 2 weeks
- tablets may be swallowed whole or dissolved in water prior to use
- adults 60 years of age and over: 1-2 tablets every 4 hours, not more than 12 tablets in 24 hours
- adults under 60 years of age: 1-4 tablets every 4 hours, not more than 24 tablets in 24 hours
How Supplied
NDC 63629-8775-1: 1000 Tablets in a BOTTLE
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504
Description
DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
Section 42229-5
Rev. 11/19
Manufactured for:
Rising Pharma Holdings, Inc.
2 Tower Center Blvd,
East Brunswick, NJ 08816
Manufactured by:
Aurex Laboratories LLC,
East Windsor, NJ 08520
Section 44425-7
Other Information: Each tablet contains: sodium 178 mg (7.74 mEq) • store at room temperature 15°-30°C (59°-86°F) in a well closed container as defined in the USP.
Section 50565-1
Keep out of reach of children.
In case of accidental overdose, seek professional assistance or contact a Poison Control Center immediately.
Section 51727-6
Inactive Ingredients: Microcrystalline cellulose NF, Hydrogenated Vegetable oil NF
Section 51945-4
Sodium Bicarbonate 650 mg Tablet
Section 53413-1
Questions or Comments? 1-866-562-4597
Section 55105-1
Purpose
Antacid
Section 55106-9
Drug Facts
Active ingredients (in each tablet)
Sodium Bicarbonate 10 gr (650mg)
Structured Label Content
Recent Major Changes (34066-1)
DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
Indications and Usage (34067-9)
Uses: relieves • acid indigestion • heartburn • sour stomach • upset stomach associated with these symptoms
Dosage and Administration (34068-7)
Directions
- do not use the maximum dosage for more than 2 weeks
- tablets may be swallowed whole or dissolved in water prior to use
- adults 60 years of age and over: 1-2 tablets every 4 hours, not more than 12 tablets in 24 hours
- adults under 60 years of age: 1-4 tablets every 4 hours, not more than 24 tablets in 24 hours
Warnings and Precautions (34071-1)
Warnings
Do not take more than 24 tablets for adults up to 60 years of age (or 12 tablets for adults 60 years of age and older) in a 24-hour period nor use the maximum dosage for more than 2 weeks, except under the advice and supervision of a physician.
Ask a doctor before use if you have a sodium restricted diet.
Ask a doctor or pharmacist before use if you are taking a prescription drug.
Antacids may interact with certain prescription drugs.
Stop use and ask a doctor if symptoms last more than 2 weeks
If pregnant or breast-feeding, ask a health professional before use.
Section 42229-5 (42229-5)
Rev. 11/19
Manufactured for:
Rising Pharma Holdings, Inc.
2 Tower Center Blvd,
East Brunswick, NJ 08816
Manufactured by:
Aurex Laboratories LLC,
East Windsor, NJ 08520
Section 44425-7 (44425-7)
Other Information: Each tablet contains: sodium 178 mg (7.74 mEq) • store at room temperature 15°-30°C (59°-86°F) in a well closed container as defined in the USP.
Section 50565-1 (50565-1)
Keep out of reach of children.
In case of accidental overdose, seek professional assistance or contact a Poison Control Center immediately.
Section 51727-6 (51727-6)
Inactive Ingredients: Microcrystalline cellulose NF, Hydrogenated Vegetable oil NF
Section 51945-4 (51945-4)
Sodium Bicarbonate 650 mg Tablet
Section 53413-1 (53413-1)
Questions or Comments? 1-866-562-4597
Section 55105-1 (55105-1)
Purpose
Antacid
Section 55106-9 (55106-9)
Drug Facts
Active ingredients (in each tablet)
Sodium Bicarbonate 10 gr (650mg)
How Supplied (HOW SUPPLIED)
NDC 63629-8775-1: 1000 Tablets in a BOTTLE
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:49.772252 · Updated: 2026-03-14T23:02:34.788055