43f84d44-18eb-7f88-e063-6394a90ab3d5
34390-5
HUMAN OTC DRUG LABEL
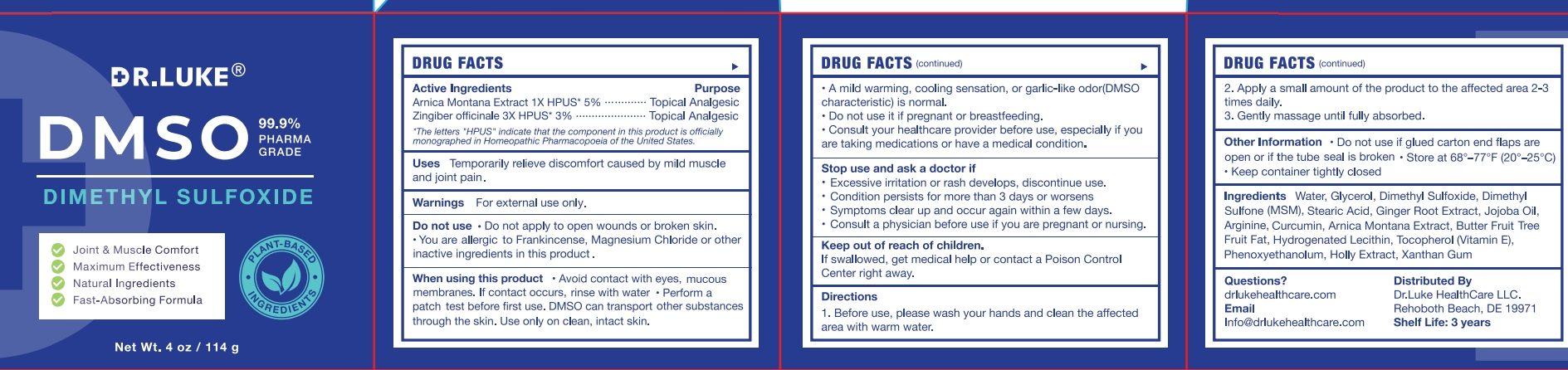
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Arnica Montana Extract 1X HPUS 5% Zingiber officinale 3XHPUS 3%
Medication Information
Warnings and Precautions
- For external use only.
Indications and Usage
Temporarily relieve discomfort caused by mild muscle and joint pain
Dosage and Administration
Apply a small amount of the product to the affected area 2-3 times daily.
Description
Arnica Montana Extract 1X HPUS 5% Zingiber officinale 3XHPUS 3%
Section 50565-1
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
- excessive irritation or rash develops, discontinue use.
- condition persists for more than 3 days or worsens
- symptoms clear up and occur again within a few days.
Section 50567-7
- Avoid contact with eyes, mucous membranes. If contact occurs, rinse with water
- Perform a patch test before first use.DMSO can transport other substances through the skin. Use only on clean, intact skin.
- A mild warming, cooling sensation, or garlic-like odor(DMSO characteristic) is normal.
- Do not use it if pregnant or breastfeeding.
- Consult your healthcare provider before use, especially if you are taking medications or have a medical condition.
Section 50568-5
consult a physician before use if you are pregnant or nursing.
Section 50570-1
- Do not apply to open wounds or broken skin.
- You are allergic to Frankincense, Magnesium Chloride or other inactive ingredients in this product
Section 51727-6
Water
Glycerol
Dimethyl Sulfoxide
Dimethyl Sulfone
Magnesium Chloride
Stearic Acid
Jojoba Oil
Arginine
Curcumin
Frankincense Extract
Hydrogenated Lecithin
Vitamin E
Xanthan Gum
Section 51945-4
Section 55105-1
Topical Analgesic
Section 55106-9
Arnica Montana Extract 1X HPUS 5%
Zingiber officinale 3XHPUS 3%
Section 59845-8
Before use, Please wash your hands and clean the affected area with warm water.
Apply a small amount of the product to the affected area 2-3 times daily.
Gently massage until fully absorbed.
Section 60561-8
- Do not use if glued carton end flaps are open or if the tube seal is broken.
- Store at 68°-77°F (20°-25C).
- Keep container tightly closed
Structured Label Content
Indications and Usage (34067-9)
Temporarily relieve discomfort caused by mild muscle and joint pain
Dosage and Administration (34068-7)
Apply a small amount of the product to the affected area 2-3 times daily.
Warnings and Precautions (34071-1)
- For external use only.
Section 50565-1 (50565-1)
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
- excessive irritation or rash develops, discontinue use.
- condition persists for more than 3 days or worsens
- symptoms clear up and occur again within a few days.
Section 50567-7 (50567-7)
- Avoid contact with eyes, mucous membranes. If contact occurs, rinse with water
- Perform a patch test before first use.DMSO can transport other substances through the skin. Use only on clean, intact skin.
- A mild warming, cooling sensation, or garlic-like odor(DMSO characteristic) is normal.
- Do not use it if pregnant or breastfeeding.
- Consult your healthcare provider before use, especially if you are taking medications or have a medical condition.
Section 50568-5 (50568-5)
consult a physician before use if you are pregnant or nursing.
Section 50570-1 (50570-1)
- Do not apply to open wounds or broken skin.
- You are allergic to Frankincense, Magnesium Chloride or other inactive ingredients in this product
Section 51727-6 (51727-6)
Water
Glycerol
Dimethyl Sulfoxide
Dimethyl Sulfone
Magnesium Chloride
Stearic Acid
Jojoba Oil
Arginine
Curcumin
Frankincense Extract
Hydrogenated Lecithin
Vitamin E
Xanthan Gum
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Topical Analgesic
Section 55106-9 (55106-9)
Arnica Montana Extract 1X HPUS 5%
Zingiber officinale 3XHPUS 3%
Section 59845-8 (59845-8)
Before use, Please wash your hands and clean the affected area with warm water.
Apply a small amount of the product to the affected area 2-3 times daily.
Gently massage until fully absorbed.
Section 60561-8 (60561-8)
- Do not use if glued carton end flaps are open or if the tube seal is broken.
- Store at 68°-77°F (20°-25C).
- Keep container tightly closed
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:17.085337 · Updated: 2026-03-14T23:10:33.744870