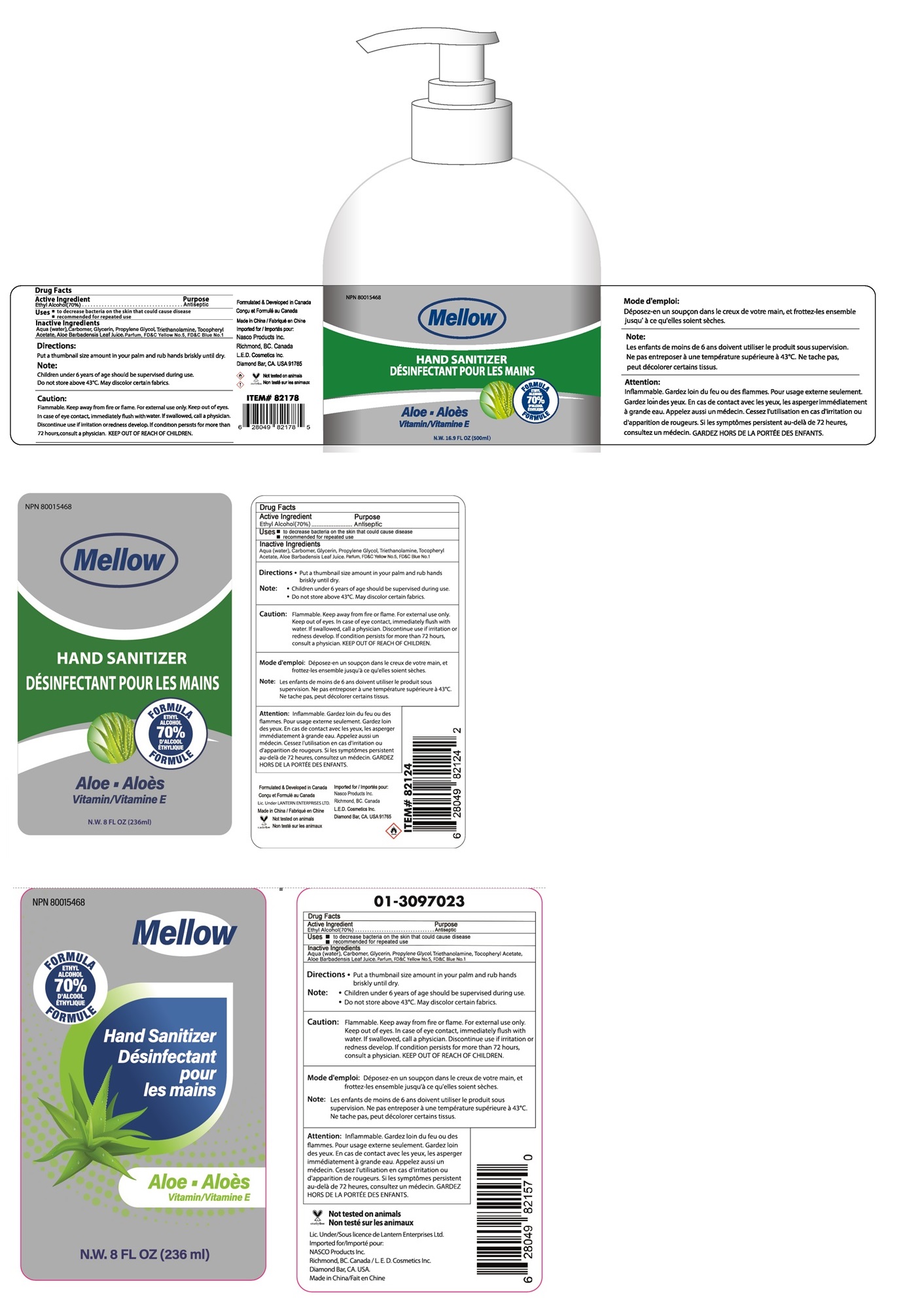
mellow Hand Sanitizer
4377ba5e-21f6-407e-9b8c-25e3c6321a5b
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ethyl Alcohol(70%)
Purpose
Antiseptic
Medication Information
Warnings and Precautions
Caution: Flammable. Keep away from fire or flame. For external use only. Keep out of eyes. In case of eye contact, immediately flush with water.
If swallowed, call a physician. Discontinue use if irritation or redness develop. If condition persists for more than 72 hours, consult a physician.
Indications and Usage
Uses • to decrease bacteria on the skin that could disease
• recommended for repeated use
Purpose
Antiseptic
Dosage and Administration
Directions • Put a thumbnail size amount in your palm and rub hands briskly until dry.
Note : • Children under 6 years of age should be supervised during use.
Description
Ethyl Alcohol(70%)
Section 42229-5
FORMULA
ETHYL ALCOHOL 70%
Aloe
Vitamin E
Formulated & Developed in Canada
Made in China
Lic.Under Lantern Enterprises Ltd.
Imported for:
Nasco Products Inc.
Richmond, BC. Canada
L.E.D. Cosmetics Inc.
Diamond Bar, CA. USA 91765
Not tested on animals
Section 44425-7
• Do not store above 43˚C. May discolor certain fabrics.
Section 50565-1
KEEP OUT OF REACH OF CHILDREN.
Packaging
Drug Facts
Active Ingredient
Ethyl Alcohol(70%)
Inactive Ingredients
Aqua (water), Carbomer, Glycerin, Propylene Glycol, Triethanolamine, Tocopheryl Acetate, Aloe Barbadensis Leaf Juice, Parfum, FD&C Yellow No.5, FD&C Blue No.1
Structured Label Content
Indications and Usage (34067-9)
Uses • to decrease bacteria on the skin that could disease
• recommended for repeated use
Dosage and Administration (34068-7)
Directions • Put a thumbnail size amount in your palm and rub hands briskly until dry.
Note : • Children under 6 years of age should be supervised during use.
Warnings and Precautions (34071-1)
Caution: Flammable. Keep away from fire or flame. For external use only. Keep out of eyes. In case of eye contact, immediately flush with water.
If swallowed, call a physician. Discontinue use if irritation or redness develop. If condition persists for more than 72 hours, consult a physician.
Section 42229-5 (42229-5)
FORMULA
ETHYL ALCOHOL 70%
Aloe
Vitamin E
Formulated & Developed in Canada
Made in China
Lic.Under Lantern Enterprises Ltd.
Imported for:
Nasco Products Inc.
Richmond, BC. Canada
L.E.D. Cosmetics Inc.
Diamond Bar, CA. USA 91765
Not tested on animals
Section 44425-7 (44425-7)
• Do not store above 43˚C. May discolor certain fabrics.
Section 50565-1 (50565-1)
KEEP OUT OF REACH OF CHILDREN.
Purpose
Antiseptic
Packaging
Drug Facts
Active Ingredient
Ethyl Alcohol(70%)
Inactive Ingredients
Aqua (water), Carbomer, Glycerin, Propylene Glycol, Triethanolamine, Tocopheryl Acetate, Aloe Barbadensis Leaf Juice, Parfum, FD&C Yellow No.5, FD&C Blue No.1
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:43.765013 · Updated: 2026-03-14T23:05:37.016425