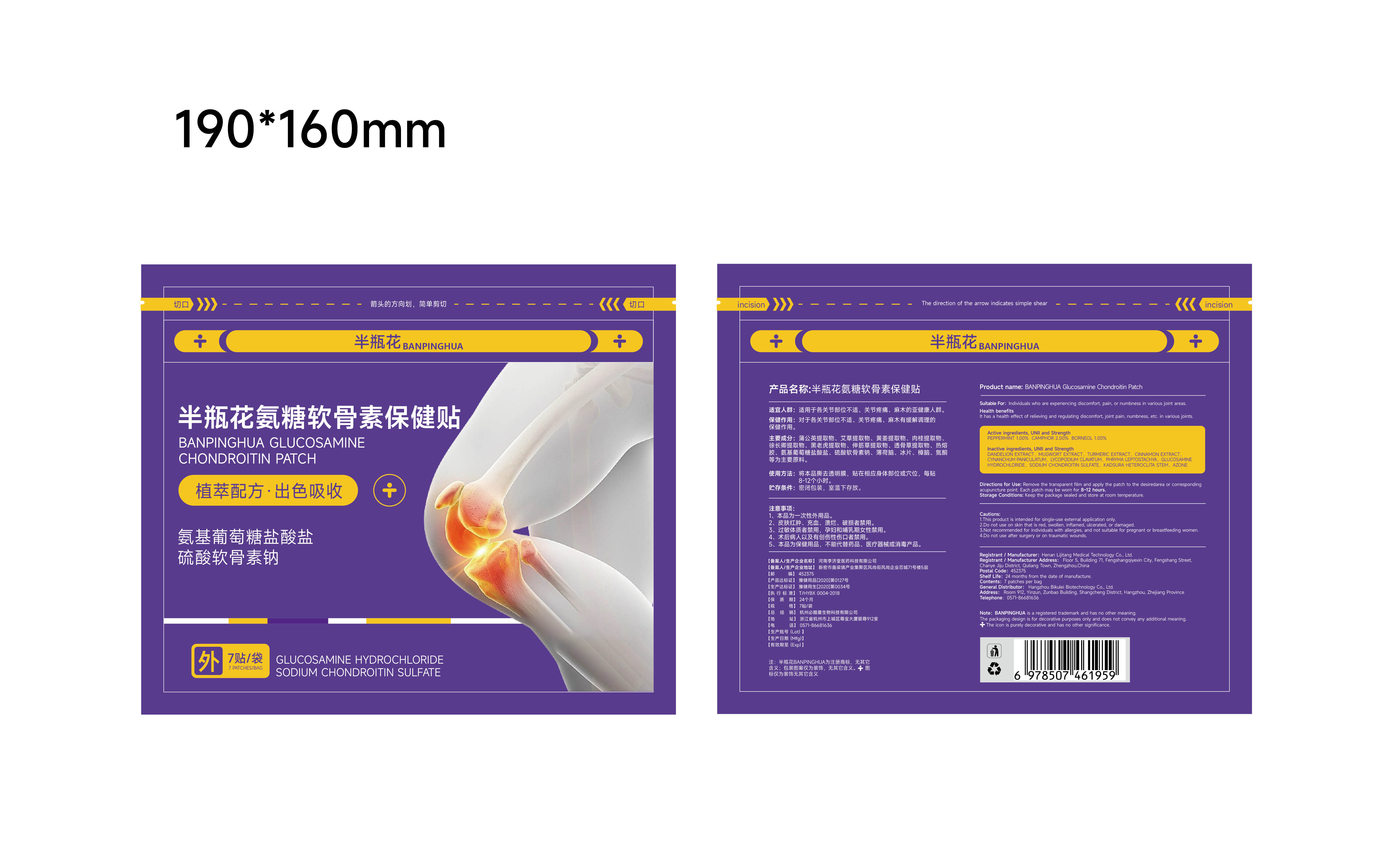
85305-004 Banpinghua Glucosamine Chondroitin Patch
42f8c2ae-50a2-2358-e063-6394a90a5979
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
PEPPERMINT 1% CAMPHOR 2% BORNEOL 1%
Medication Information
Warnings and Precautions
This product is intended for single-use external application only.
Indications and Usage
Suitable For: Individuals who are experiencing discomfort, pain, or numbness in various joint areas.
It has a health effect of relieving and requlating discomfort, joint pain, numbness, etc. in various joints.
Dosage and Administration
Directions for Use: Remove the transparent film and apply the patch to the desiredarea or corresponding acupuncture point. Each patch may be worn for 8-12 hours.
Description
PEPPERMINT 1% CAMPHOR 2% BORNEOL 1%
Section 50565-1
KEEP OUT OF REACH OF CHILDREN SECTION
Section 50567-7
Suitable For: individuals who are experiencing discomfort, pain, or numbness in various joint areas.
Section 50570-1
2.Do not use on skin that is red, swollen, inflamed, ulcerated, or damaged.
3.Not recommended for individuals with allergies, and not suitable for pregnant or breastfeeding women.
Section 51727-6
DANDELION EXTRACT, MUGWORT EXTRACT, TURMERIC EXTRACT, CINNAMON EXTRACTCYNANCHUM PANICULATUM. LYCOPODIUM CLAVATUM, PHRYMA LEPTOSTACHYA, GLUCOSAMINEHYDROCHLORIDE, SODIUM CHONDROITIN SULFATE, KADSURA HETEROCLITA STEM, AZONE
Section 51945-4
Section 55105-1
Analgesia
Section 55106-9
PEPPERMINT 1%
CAMPHOR 2%
BORNEOL 1%
Structured Label Content
Indications and Usage (34067-9)
Suitable For: Individuals who are experiencing discomfort, pain, or numbness in various joint areas.
It has a health effect of relieving and requlating discomfort, joint pain, numbness, etc. in various joints.
Dosage and Administration (34068-7)
Directions for Use: Remove the transparent film and apply the patch to the desiredarea or corresponding acupuncture point. Each patch may be worn for 8-12 hours.
Warnings and Precautions (34071-1)
This product is intended for single-use external application only.
Section 50565-1 (50565-1)
KEEP OUT OF REACH OF CHILDREN SECTION
Section 50567-7 (50567-7)
Suitable For: individuals who are experiencing discomfort, pain, or numbness in various joint areas.
Section 50570-1 (50570-1)
2.Do not use on skin that is red, swollen, inflamed, ulcerated, or damaged.
3.Not recommended for individuals with allergies, and not suitable for pregnant or breastfeeding women.
Section 51727-6 (51727-6)
DANDELION EXTRACT, MUGWORT EXTRACT, TURMERIC EXTRACT, CINNAMON EXTRACTCYNANCHUM PANICULATUM. LYCOPODIUM CLAVATUM, PHRYMA LEPTOSTACHYA, GLUCOSAMINEHYDROCHLORIDE, SODIUM CHONDROITIN SULFATE, KADSURA HETEROCLITA STEM, AZONE
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Analgesia
Section 55106-9 (55106-9)
PEPPERMINT 1%
CAMPHOR 2%
BORNEOL 1%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:57.643383 · Updated: 2026-03-14T23:13:28.374847