These Highlights Do Not Include All The Information Needed To Use Isosulfan Blue Injection Safely And Effectively. See Full Prescribing Information For Isosulfan Blue Injection.
42f2d62a-c7f2-4706-87bc-64f3e72d63a9
34391-3
Human Prescription Drug Label
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Isosulfan blue injection 1% upon subcutaneous administration, delineates the lymphatic vessels draining the region of injection. It is an adjunct to lymphography in: primary and secondary lymphedema of the extremities; chyluria, chylous ascites or chylothorax; lymph node involvement by primary or secondary neoplasm; lymph node response to therapeutic modalities (1.1).
Indications and Usage
Isosulfan blue injection 1% upon subcutaneous administration, delineates the lymphatic vessels draining the region of injection. It is an adjunct to lymphography in: primary and secondary lymphedema of the extremities; chyluria, chylous ascites or chylothorax; lymph node involvement by primary or secondary neoplasm; lymph node response to therapeutic modalities (1.1).
Dosage and Administration
Isosulfan blue injection 1% is to be administered subcutaneously, one-half (1/2) mL into three (3) interdigital spaces of each extremity per study. A maximum dose of 3 mL (30 mg) isosulfan blue is, therefore, injected (2.1).
Warnings and Precautions
Life-threatening anaphylactic reactions have occurred after isosulfan blue 1% administration. Monitor patients closely for at least 60 minutes after administration of isosulfan blue 1% (5.1). The admixture of isosulfan blue 1% with local anesthetics results in an immediate precipitation of 4 to 9% drug complex. Use a separate syringe for anesthetics (5.2). Isosulfan blue 1% interferes with measurements in peripheral blood pulse oximetry. Arterial blood gas analysis may be needed (5.3).
Contraindications
Isosulfan blue injection 1% is contraindicated in those individuals with known hypersensitivity to triphenylmethane or related compounds.
Adverse Reactions
Hypersensitivity Reactions : Hypersensitivity reactions occurring approximately 2% of patients and include life-threatening anaphylactic reactions with respiratory distress, shock, angioedema, urticaria, pruritus. A death has been reported following I.V. administration of a similar compound (6) . To report SUSPECTED ADVERSE REACTIONS, contact Eugia US LLC at 1-866-850-2876 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
No drug interactions have been identified with isosulfan blue 1%.
Storage and Handling
Isosulfan blue injection 1% is a sterile, non-pyrogenic, aqueous dark blue color solution and is supplied as follows: Isosulfan blue injection 1% 50 mg per 5 mL (10 mg / mL): 5 mL Single-Dose Vials in a Carton of 6 NDC 55150-240-05 Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Avoid excessive heat. Discard unused portion. The vial stoppers are not made with natural rubber latex.
How Supplied
Isosulfan blue injection 1% is a sterile, non-pyrogenic, aqueous dark blue color solution and is supplied as follows: Isosulfan blue injection 1% 50 mg per 5 mL (10 mg / mL): 5 mL Single-Dose Vials in a Carton of 6 NDC 55150-240-05 Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Avoid excessive heat. Discard unused portion. The vial stoppers are not made with natural rubber latex.
Medication Information
Warnings and Precautions
Life-threatening anaphylactic reactions have occurred after isosulfan blue 1% administration. Monitor patients closely for at least 60 minutes after administration of isosulfan blue 1% (5.1). The admixture of isosulfan blue 1% with local anesthetics results in an immediate precipitation of 4 to 9% drug complex. Use a separate syringe for anesthetics (5.2). Isosulfan blue 1% interferes with measurements in peripheral blood pulse oximetry. Arterial blood gas analysis may be needed (5.3).
Indications and Usage
Isosulfan blue injection 1% upon subcutaneous administration, delineates the lymphatic vessels draining the region of injection. It is an adjunct to lymphography in: primary and secondary lymphedema of the extremities; chyluria, chylous ascites or chylothorax; lymph node involvement by primary or secondary neoplasm; lymph node response to therapeutic modalities (1.1).
Dosage and Administration
Isosulfan blue injection 1% is to be administered subcutaneously, one-half (1/2) mL into three (3) interdigital spaces of each extremity per study. A maximum dose of 3 mL (30 mg) isosulfan blue is, therefore, injected (2.1).
Contraindications
Isosulfan blue injection 1% is contraindicated in those individuals with known hypersensitivity to triphenylmethane or related compounds.
Adverse Reactions
Hypersensitivity Reactions : Hypersensitivity reactions occurring approximately 2% of patients and include life-threatening anaphylactic reactions with respiratory distress, shock, angioedema, urticaria, pruritus. A death has been reported following I.V. administration of a similar compound (6) . To report SUSPECTED ADVERSE REACTIONS, contact Eugia US LLC at 1-866-850-2876 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
No drug interactions have been identified with isosulfan blue 1%.
Storage and Handling
Isosulfan blue injection 1% is a sterile, non-pyrogenic, aqueous dark blue color solution and is supplied as follows: Isosulfan blue injection 1% 50 mg per 5 mL (10 mg / mL): 5 mL Single-Dose Vials in a Carton of 6 NDC 55150-240-05 Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Avoid excessive heat. Discard unused portion. The vial stoppers are not made with natural rubber latex.
How Supplied
Isosulfan blue injection 1% is a sterile, non-pyrogenic, aqueous dark blue color solution and is supplied as follows: Isosulfan blue injection 1% 50 mg per 5 mL (10 mg / mL): 5 mL Single-Dose Vials in a Carton of 6 NDC 55150-240-05 Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Avoid excessive heat. Discard unused portion. The vial stoppers are not made with natural rubber latex.
Description
Isosulfan blue injection 1% upon subcutaneous administration, delineates the lymphatic vessels draining the region of injection. It is an adjunct to lymphography in: primary and secondary lymphedema of the extremities; chyluria, chylous ascites or chylothorax; lymph node involvement by primary or secondary neoplasm; lymph node response to therapeutic modalities (1.1).
10 Overdosage
Do not exceed indicated recommended dosage as overdosage levels have not been identified for isosulfan blue 1%.
11 Description
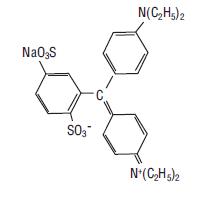
The chemical name of isosulfan blue is N-[4-[[4-(diethylamino)phenyl] (2,5-disulfophenyl) methylene]-2,5-cyclohexadien-1-ylidene]-N-ethylethanaminium hydroxide, inner salt, sodium salt. Isosulfan blue is a greenish blue color hygroscopic powder. Its structural formula is:
Isosulfan blue injection 1% is a sterile, non-pyrogenic, aqueous dark blue color solution for subcutaneous administration. Phosphate buffer in water for injection is added in sufficient quantity to yield a final pH of 6.8 to 7.4. Each mL of solution contains 10 mg isosulfan blue, 6.6 mg sodium monohydrogen phosphate and 2.7 mg potassium dihydrogen phosphate. The solution contains no preservative. Isosulfan blue injection 1% is a contrast agent for the delineation of lymphatic vessels.
8.4 Pediatric Use
Safety and effectiveness of isosulfan blue 1% in children have not been established.
4 Contraindications
Isosulfan blue injection 1% is contraindicated in those individuals with known hypersensitivity to triphenylmethane or related compounds.
6 Adverse Reactions
Hypersensitivity Reactions
: Hypersensitivity reactions occurring approximately 2% of patients and include life-threatening anaphylactic reactions with respiratory distress, shock, angioedema, urticaria, pruritus. A death has been reported following I.V. administration of a similar compound (6).
To report SUSPECTED ADVERSE REACTIONS, contact Eugia US LLC at 1-866-850-2876 or FDA at 1-800-FDA-1088 or
www.fda.gov/medwatch
.
7 Drug Interactions
No drug interactions have been identified with isosulfan blue 1%.
8.3 Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when isosulfan blue 1% is administered to a nursing mother.
12.2 Pharmacodynamics
Following subcutaneous administration, isosulfan blue 1% binds to serum proteins and is picked up by the lymphatic vessels. Thus, the lymphatic vessels are delineated by the blue dye.
12.3 Pharmacokinetics
Up to 10% of the subcutaneously administered dose of isosulfan blue 1% is excreted unchanged in the urine in 24 hours in human.
1 Indications and Usage
Isosulfan blue injection 1% upon subcutaneous administration, delineates the lymphatic vessels draining the region of injection. It is an adjunct to lymphography in: primary and secondary lymphedema of the extremities; chyluria, chylous ascites or chylothorax; lymph node involvement by primary or secondary neoplasm; lymph node response to therapeutic modalities (1.1).
13.2 Teratogenic Effects
Pregnancy Category C. Animal reproduction studies have not been conducted with isosulfan blue 1%. It is not known whether isosulfan blue 1% can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Isosulfan blue 1% should be given to a pregnant woman only if clearly needed.
5 Warnings and Precautions
- Life-threatening anaphylactic reactions have occurred after isosulfan blue 1% administration. Monitor patients closely for at least 60 minutes after administration of isosulfan blue 1% (5.1).
- The admixture of isosulfan blue 1% with local anesthetics results in an immediate precipitation of 4 to 9% drug complex. Use a separate syringe for anesthetics (5.2).
- Isosulfan blue 1% interferes with measurements in peripheral blood pulse oximetry. Arterial blood gas analysis may be needed (5.3).
2 Dosage and Administration
Isosulfan blue injection 1% is to be administered subcutaneously, one-half (1/2) mL into three (3) interdigital spaces of each extremity per study. A maximum dose of 3 mL (30 mg) isosulfan blue is, therefore, injected (2.1).
3 Dosage Forms and Strengths
1% aqueous solution (isosulfan blue)
6.1 Postmarketing Experience
Hypersensitivity Reactions: Case series report an overall incidence of hypersensitivity reactions in approximately 2% of patients. Life-threatening anaphylactic reactions have occurred. Manifestations include respiratory distress, shock, angioedema, urticaria, pruritus. A death has been reported following administration of a similar compound employed to estimate the depth of a severe burn. Reactions are more likely to occur in patients with a personal or family history of bronchial asthma, significant allergies, drug reactions or previous reactions to triphenylmethane dyes
[see Warnings and Precautions (5)].
Laboratory Tests: Isosulfan blue 1% interferes with measurements of oxygen saturation by pulse oximetry and of methemoglobin by gas analyzer
[see Warnings and Precautions (5)].
Skin: transient or long-term (tattooing) blue coloration.
8 Use in Specific Populations
5.1 Hypersensitivity Reactions
Life-threatening anaphylactic reactions (respiratory distress, shock, angioedema) have occurred after isosulfan blue 1% administration. Reactions are more likely to occur in patients with a history of bronchial asthma, allergies, drug reactions or previous reactions to triphenylmethane dyes. Monitor patients closely for at least 60 minutes after administration of isosulfan blue 1%. Trained personnel should be available to administer emergency care including resuscitation.
2.1 Subcutaneous Administration
Isosulfan blue injection 1% is to be administered subcutaneously, one-half (1/2) mL into three (3) interdigital spaces of each extremity per study. A maximum dose of 3 mL (30 mg) isosulfan blue is, therefore, injected.
1.1 Lymphatic Vessel Delineation
Isosulfan blue injection 1% upon subcutaneous administration, delineates lymphatic vessels draining the region of injection. It is an adjunct to lymphography in: primary and secondary lymphedema of the extremities; chyluria, chylous ascites or chylothorax; lymph node involvement by primary or secondary neoplasm; and lymph node response to therapeutic modalities.
17 Patient Counseling Information
Inform patients that urine color may be blue for 24 hours following administration of isosulfan blue injection 1%.
Distributed by:
Eugia US LLC
279 Princeton-Hightstown Rd.
E. Windsor, NJ 08520
Manufactured by:
Eugia Pharma Specialities Limited
Hyderabad - 500032
India
16 How Supplied/storage and Handling
Isosulfan blue injection 1% is a sterile, non-pyrogenic, aqueous dark blue color solution and is supplied as follows:
Isosulfan blue injection 1%
50 mg per 5 mL (10 mg / mL):
5 mL Single-Dose Vials
in a Carton of 6 NDC 55150-240-05
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Avoid excessive heat.
Discard unused portion.
The vial stoppers are not made with natural rubber latex.
5.2 Precipitation of Isosulfan Blue 1% By Lidocaine
The admixture of isosulfan blue 1% (with local anesthetics (i.e. lidocaine)) in the same syringe results in an immediate precipitation of 4 to 9% drug complex. Use a separate syringe to administer a local anesthetic.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate the carcinogenic potential of isosulfan blue 1%. Reproduction studies in animals have not been conducted and, therefore, it is unknown if a problem concerning mutagenesis or impairment of fertility in either males or females exists.
5.3 Interference With Oxygen Saturation and Methemoglobin Measurements
Isosulfan blue 1% interferes with measurements of oxygen saturation in peripheral blood by pulse oximetry and can cause falsely low readings. The interference effect is maximal at 30 minutes and minimal generally by four hours after administration. Arterial blood gas analysis may be needed to verify decreased arterial partial pressure of oxygen.
Isosulfan blue 1% may also cause falsely elevated readings of methemoglobin by arterial blood gas analyzer. Therefore, co-oximetry may be needed to verify methemoglobin level.
Package Label Principal Display Panel 1% [50 Mg Per 5 Ml (10 Mg / Ml)] Container Label
Rx only NDC 55150-240-05
Isosulfan Blue
Injection 1%
50 mg per 5 mL
(10 mg / mL)
For Lymphography
For Subcutaneous Use Only
5 mL Single-Dose Vial
Package Label Principal Display Panel 1% [50 Mg Per 5 Ml (10 Mg / Ml)] Container Carton (6 Vials)
Rx only NDC 55150-240-05
Isosulfan Blue
Injection 1%
50 mg per 5 mL
(10 mg / mL)
For Lymphography
For Subcutaneous Use Only
Sterile 6 X 5 mL
Non-Pyrogenic Single-Dose Vials
eugia
Structured Label Content
10 Overdosage (10 OVERDOSAGE)
Do not exceed indicated recommended dosage as overdosage levels have not been identified for isosulfan blue 1%.
11 Description (11 DESCRIPTION)
The chemical name of isosulfan blue is N-[4-[[4-(diethylamino)phenyl] (2,5-disulfophenyl) methylene]-2,5-cyclohexadien-1-ylidene]-N-ethylethanaminium hydroxide, inner salt, sodium salt. Isosulfan blue is a greenish blue color hygroscopic powder. Its structural formula is:
Isosulfan blue injection 1% is a sterile, non-pyrogenic, aqueous dark blue color solution for subcutaneous administration. Phosphate buffer in water for injection is added in sufficient quantity to yield a final pH of 6.8 to 7.4. Each mL of solution contains 10 mg isosulfan blue, 6.6 mg sodium monohydrogen phosphate and 2.7 mg potassium dihydrogen phosphate. The solution contains no preservative. Isosulfan blue injection 1% is a contrast agent for the delineation of lymphatic vessels.
8.4 Pediatric Use
Safety and effectiveness of isosulfan blue 1% in children have not been established.
4 Contraindications (4 CONTRAINDICATIONS)
Isosulfan blue injection 1% is contraindicated in those individuals with known hypersensitivity to triphenylmethane or related compounds.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Hypersensitivity Reactions
: Hypersensitivity reactions occurring approximately 2% of patients and include life-threatening anaphylactic reactions with respiratory distress, shock, angioedema, urticaria, pruritus. A death has been reported following I.V. administration of a similar compound (6).
To report SUSPECTED ADVERSE REACTIONS, contact Eugia US LLC at 1-866-850-2876 or FDA at 1-800-FDA-1088 or
www.fda.gov/medwatch
.
7 Drug Interactions (7 DRUG INTERACTIONS)
No drug interactions have been identified with isosulfan blue 1%.
8.3 Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when isosulfan blue 1% is administered to a nursing mother.
12.2 Pharmacodynamics
Following subcutaneous administration, isosulfan blue 1% binds to serum proteins and is picked up by the lymphatic vessels. Thus, the lymphatic vessels are delineated by the blue dye.
12.3 Pharmacokinetics
Up to 10% of the subcutaneously administered dose of isosulfan blue 1% is excreted unchanged in the urine in 24 hours in human.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Isosulfan blue injection 1% upon subcutaneous administration, delineates the lymphatic vessels draining the region of injection. It is an adjunct to lymphography in: primary and secondary lymphedema of the extremities; chyluria, chylous ascites or chylothorax; lymph node involvement by primary or secondary neoplasm; lymph node response to therapeutic modalities (1.1).
13.2 Teratogenic Effects
Pregnancy Category C. Animal reproduction studies have not been conducted with isosulfan blue 1%. It is not known whether isosulfan blue 1% can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Isosulfan blue 1% should be given to a pregnant woman only if clearly needed.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Life-threatening anaphylactic reactions have occurred after isosulfan blue 1% administration. Monitor patients closely for at least 60 minutes after administration of isosulfan blue 1% (5.1).
- The admixture of isosulfan blue 1% with local anesthetics results in an immediate precipitation of 4 to 9% drug complex. Use a separate syringe for anesthetics (5.2).
- Isosulfan blue 1% interferes with measurements in peripheral blood pulse oximetry. Arterial blood gas analysis may be needed (5.3).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Isosulfan blue injection 1% is to be administered subcutaneously, one-half (1/2) mL into three (3) interdigital spaces of each extremity per study. A maximum dose of 3 mL (30 mg) isosulfan blue is, therefore, injected (2.1).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
1% aqueous solution (isosulfan blue)
6.1 Postmarketing Experience
Hypersensitivity Reactions: Case series report an overall incidence of hypersensitivity reactions in approximately 2% of patients. Life-threatening anaphylactic reactions have occurred. Manifestations include respiratory distress, shock, angioedema, urticaria, pruritus. A death has been reported following administration of a similar compound employed to estimate the depth of a severe burn. Reactions are more likely to occur in patients with a personal or family history of bronchial asthma, significant allergies, drug reactions or previous reactions to triphenylmethane dyes
[see Warnings and Precautions (5)].
Laboratory Tests: Isosulfan blue 1% interferes with measurements of oxygen saturation by pulse oximetry and of methemoglobin by gas analyzer
[see Warnings and Precautions (5)].
Skin: transient or long-term (tattooing) blue coloration.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
5.1 Hypersensitivity Reactions
Life-threatening anaphylactic reactions (respiratory distress, shock, angioedema) have occurred after isosulfan blue 1% administration. Reactions are more likely to occur in patients with a history of bronchial asthma, allergies, drug reactions or previous reactions to triphenylmethane dyes. Monitor patients closely for at least 60 minutes after administration of isosulfan blue 1%. Trained personnel should be available to administer emergency care including resuscitation.
2.1 Subcutaneous Administration (2.1 Subcutaneous administration)
Isosulfan blue injection 1% is to be administered subcutaneously, one-half (1/2) mL into three (3) interdigital spaces of each extremity per study. A maximum dose of 3 mL (30 mg) isosulfan blue is, therefore, injected.
1.1 Lymphatic Vessel Delineation
Isosulfan blue injection 1% upon subcutaneous administration, delineates lymphatic vessels draining the region of injection. It is an adjunct to lymphography in: primary and secondary lymphedema of the extremities; chyluria, chylous ascites or chylothorax; lymph node involvement by primary or secondary neoplasm; and lymph node response to therapeutic modalities.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Inform patients that urine color may be blue for 24 hours following administration of isosulfan blue injection 1%.
Distributed by:
Eugia US LLC
279 Princeton-Hightstown Rd.
E. Windsor, NJ 08520
Manufactured by:
Eugia Pharma Specialities Limited
Hyderabad - 500032
India
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Isosulfan blue injection 1% is a sterile, non-pyrogenic, aqueous dark blue color solution and is supplied as follows:
Isosulfan blue injection 1%
50 mg per 5 mL (10 mg / mL):
5 mL Single-Dose Vials
in a Carton of 6 NDC 55150-240-05
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Avoid excessive heat.
Discard unused portion.
The vial stoppers are not made with natural rubber latex.
5.2 Precipitation of Isosulfan Blue 1% By Lidocaine (5.2 Precipitation of Isosulfan Blue 1% by Lidocaine)
The admixture of isosulfan blue 1% (with local anesthetics (i.e. lidocaine)) in the same syringe results in an immediate precipitation of 4 to 9% drug complex. Use a separate syringe to administer a local anesthetic.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate the carcinogenic potential of isosulfan blue 1%. Reproduction studies in animals have not been conducted and, therefore, it is unknown if a problem concerning mutagenesis or impairment of fertility in either males or females exists.
5.3 Interference With Oxygen Saturation and Methemoglobin Measurements (5.3 Interference with Oxygen Saturation and Methemoglobin Measurements)
Isosulfan blue 1% interferes with measurements of oxygen saturation in peripheral blood by pulse oximetry and can cause falsely low readings. The interference effect is maximal at 30 minutes and minimal generally by four hours after administration. Arterial blood gas analysis may be needed to verify decreased arterial partial pressure of oxygen.
Isosulfan blue 1% may also cause falsely elevated readings of methemoglobin by arterial blood gas analyzer. Therefore, co-oximetry may be needed to verify methemoglobin level.
Package Label Principal Display Panel 1% [50 Mg Per 5 Ml (10 Mg / Ml)] Container Label (PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 1% [50 mg per 5 mL (10 mg / mL)] - Container Label)
Rx only NDC 55150-240-05
Isosulfan Blue
Injection 1%
50 mg per 5 mL
(10 mg / mL)
For Lymphography
For Subcutaneous Use Only
5 mL Single-Dose Vial
Package Label Principal Display Panel 1% [50 Mg Per 5 Ml (10 Mg / Ml)] Container Carton (6 Vials) (PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 1% [50 mg per 5 mL (10 mg / mL)] - Container-Carton (6 Vials))
Rx only NDC 55150-240-05
Isosulfan Blue
Injection 1%
50 mg per 5 mL
(10 mg / mL)
For Lymphography
For Subcutaneous Use Only
Sterile 6 X 5 mL
Non-Pyrogenic Single-Dose Vials
eugia
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:40:25.642052 · Updated: 2026-03-14T22:50:42.037952