These Highlights Do Not Include All The Information Needed To Use Alecensa Safely And Effectively. See Full Prescribing Information For Alecensa.
42c49deb-713b-427a-9670-08af08adcffb
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1.1 ) 04/2024 Dosage and Administration ( 2.1 , 2.2 , 2.3 ) 04/2024 Warnings and Precautions ( 5 ) 04/2024
Indications and Usage
ALECENSA is a kinase inhibitor indicated for: adjuvant treatment in adult patients following tumor resection of anaplastic lymphoma kinase (ALK)-positive non-small cell lung cancer (NSCLC) (tumors ≥ 4 cm or node positive) as detected by an FDA-approved test. ( 1.1 ) treatment of adult patients with ALK-positive metastatic NSCLC as detected by an FDA-approved test. ( 1.2 )
Dosage and Administration
600 mg orally twice daily. Administer ALECENSA with food. ( 2.2 )
Warnings and Precautions
Hepatotoxicity: Monitor liver laboratory tests every 2 weeks during the first 3 months of treatment, then once a month and as clinically indicated, with more frequent testing in patients who develop transaminase and bilirubin elevations. In case of severe ALT, AST, or bilirubin elevations, withhold, then reduce dose, or permanently discontinue ALECENSA. ( 2.4 , 5.1 ) Interstitial Lung Disease (ILD)/Pneumonitis: Immediately withhold ALECENSA in patients diagnosed with ILD/pneumonitis and permanently discontinue if no other potential causes of ILD/pneumonitis have been identified. ( 2.4 , 5.2 ) Renal Impairment: Withhold ALECENSA for severe renal impairment, then resume ALECENSA at reduced dose upon recovery or permanently discontinue ( 2.4 , 5.3 ). Bradycardia: Monitor heart rate and blood pressure regularly. If symptomatic, withhold ALECENSA then reduce dose, or permanently discontinue. ( 2.4 , 5.4 ) Severe Myalgia and Creatine Phosphokinase (CPK) Elevation: Assess CPK every 2 weeks during the first month of treatment and in patients reporting unexplained muscle pain, tenderness, or weakness. In case of severe CPK elevations, withhold, then resume or reduce dose. ( 2.4 , 5.5 ) Hemolytic Anemia: If hemolytic anemia is suspected, withhold ALECENSA. If hemolytic anemia is confirmed, consider resuming at a reduced dose upon resolution or permanently discontinue. ( 5.6 ) Embryo-Fetal Toxicity: ALECENSA can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. ( 5.7 , 8.1 , 8.3 )
Contraindications
None.
Adverse Reactions
The dose reduction schedule for ALECENSA is provided in Table 2 . Table 2: ALECENSA Dose Reduction Schedule Dose Reduction Schedule Dose Level Starting dose 600 mg taken orally twice daily First dose reduction 450 mg taken orally twice daily Second dose reduction 300 mg taken orally twice daily Discontinue if patients are unable to tolerate the 300 mg twice daily dose. Recommendations for dose modifications of ALECENSA in case of adverse reactions are provided in Table 3 . Table 3: ALECENSA Dose Modifications for Adverse Reactions Criteria ALT = alanine transaminase; AST = aspartate transaminase; ULN = upper limit of normal; ILD = interstitial lung disease; CPK = blood creatine phosphokinase ALECENSA Dose Modification ALT or AST elevation of greater than 5 times upper limit of normal (ULN) with total bilirubin less than or equal to 2 times ULN Temporarily withhold until recovery to baseline or to less than or equal to 3 times ULN, then resume at reduced dose as per Table 2 . ALT or AST elevation greater than 3 times ULN with total bilirubin elevation greater than 2 times ULN in the absence of cholestasis or hemolysis Permanently discontinue ALECENSA. Total bilirubin elevation of greater than 3 times ULN Temporarily withhold until recovery to baseline or to less than or equal to 1.5 times ULN, then resume at reduced dose as per Table 2 . Any grade treatment-related interstitial lung disease (ILD)/pneumonitis Permanently discontinue ALECENSA. Grade 3 renal impairment Temporarily withhold until serum creatinine recovers to less than or equal to 1.5 times ULN, then resume at reduced dose. Grade 4 renal impairment Permanently discontinue ALECENSA. Symptomatic bradycardia Withhold ALECENSA until recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above. If contributing concomitant medication is identified and discontinued, or its dose is adjusted, resume ALECENSA at previous dose upon recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above. If no contributing concomitant medication is identified, or if contributing concomitant medications are not discontinued or dose modified, resume ALECENSA at reduced dose (see Table 2 ) upon recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above. Bradycardia Heart rate less than 60 beats per minute (bpm) (life-threatening consequences, urgent intervention indicated) Permanently discontinue ALECENSA if no contributing concomitant medication is identified. If contributing concomitant medication is identified and discontinued, or its dose is adjusted, resume ALECENSA at reduced dose (see Table 2 ) upon recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above, with frequent monitoring as clinically indicated. Permanently discontinue ALECENSA in case of recurrence. CPK elevation greater than 5 times ULN Temporarily withhold until recovery to baseline or to less than or equal to 2.5 times ULN, then resume at same dose. CPK elevation greater than 10 times ULN or second occurrence of CPK elevation of greater than 5 times ULN Temporarily withhold until recovery to baseline or to less than or equal to 2.5 times ULN, then resume at reduced dose as per Table 2 . Hemolytic Anemia Withhold ALECENSA if hemolytic anemia is suspected. Upon resolution, resume at reduced dose or permanently discontinue.
Storage and Handling
Hard capsules, white 150 mg capsules with "ALE" printed in black ink on the cap and "150 mg" printed in black ink on the body, available in: 240 capsules per bottle: NDC 50242-130-01
How Supplied
Hard capsules, white 150 mg capsules with "ALE" printed in black ink on the cap and "150 mg" printed in black ink on the body, available in: 240 capsules per bottle: NDC 50242-130-01
Medication Information
Warnings and Precautions
Hepatotoxicity: Monitor liver laboratory tests every 2 weeks during the first 3 months of treatment, then once a month and as clinically indicated, with more frequent testing in patients who develop transaminase and bilirubin elevations. In case of severe ALT, AST, or bilirubin elevations, withhold, then reduce dose, or permanently discontinue ALECENSA. ( 2.4 , 5.1 ) Interstitial Lung Disease (ILD)/Pneumonitis: Immediately withhold ALECENSA in patients diagnosed with ILD/pneumonitis and permanently discontinue if no other potential causes of ILD/pneumonitis have been identified. ( 2.4 , 5.2 ) Renal Impairment: Withhold ALECENSA for severe renal impairment, then resume ALECENSA at reduced dose upon recovery or permanently discontinue ( 2.4 , 5.3 ). Bradycardia: Monitor heart rate and blood pressure regularly. If symptomatic, withhold ALECENSA then reduce dose, or permanently discontinue. ( 2.4 , 5.4 ) Severe Myalgia and Creatine Phosphokinase (CPK) Elevation: Assess CPK every 2 weeks during the first month of treatment and in patients reporting unexplained muscle pain, tenderness, or weakness. In case of severe CPK elevations, withhold, then resume or reduce dose. ( 2.4 , 5.5 ) Hemolytic Anemia: If hemolytic anemia is suspected, withhold ALECENSA. If hemolytic anemia is confirmed, consider resuming at a reduced dose upon resolution or permanently discontinue. ( 5.6 ) Embryo-Fetal Toxicity: ALECENSA can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. ( 5.7 , 8.1 , 8.3 )
Indications and Usage
ALECENSA is a kinase inhibitor indicated for: adjuvant treatment in adult patients following tumor resection of anaplastic lymphoma kinase (ALK)-positive non-small cell lung cancer (NSCLC) (tumors ≥ 4 cm or node positive) as detected by an FDA-approved test. ( 1.1 ) treatment of adult patients with ALK-positive metastatic NSCLC as detected by an FDA-approved test. ( 1.2 )
Dosage and Administration
600 mg orally twice daily. Administer ALECENSA with food. ( 2.2 )
Contraindications
None.
Adverse Reactions
The dose reduction schedule for ALECENSA is provided in Table 2 . Table 2: ALECENSA Dose Reduction Schedule Dose Reduction Schedule Dose Level Starting dose 600 mg taken orally twice daily First dose reduction 450 mg taken orally twice daily Second dose reduction 300 mg taken orally twice daily Discontinue if patients are unable to tolerate the 300 mg twice daily dose. Recommendations for dose modifications of ALECENSA in case of adverse reactions are provided in Table 3 . Table 3: ALECENSA Dose Modifications for Adverse Reactions Criteria ALT = alanine transaminase; AST = aspartate transaminase; ULN = upper limit of normal; ILD = interstitial lung disease; CPK = blood creatine phosphokinase ALECENSA Dose Modification ALT or AST elevation of greater than 5 times upper limit of normal (ULN) with total bilirubin less than or equal to 2 times ULN Temporarily withhold until recovery to baseline or to less than or equal to 3 times ULN, then resume at reduced dose as per Table 2 . ALT or AST elevation greater than 3 times ULN with total bilirubin elevation greater than 2 times ULN in the absence of cholestasis or hemolysis Permanently discontinue ALECENSA. Total bilirubin elevation of greater than 3 times ULN Temporarily withhold until recovery to baseline or to less than or equal to 1.5 times ULN, then resume at reduced dose as per Table 2 . Any grade treatment-related interstitial lung disease (ILD)/pneumonitis Permanently discontinue ALECENSA. Grade 3 renal impairment Temporarily withhold until serum creatinine recovers to less than or equal to 1.5 times ULN, then resume at reduced dose. Grade 4 renal impairment Permanently discontinue ALECENSA. Symptomatic bradycardia Withhold ALECENSA until recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above. If contributing concomitant medication is identified and discontinued, or its dose is adjusted, resume ALECENSA at previous dose upon recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above. If no contributing concomitant medication is identified, or if contributing concomitant medications are not discontinued or dose modified, resume ALECENSA at reduced dose (see Table 2 ) upon recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above. Bradycardia Heart rate less than 60 beats per minute (bpm) (life-threatening consequences, urgent intervention indicated) Permanently discontinue ALECENSA if no contributing concomitant medication is identified. If contributing concomitant medication is identified and discontinued, or its dose is adjusted, resume ALECENSA at reduced dose (see Table 2 ) upon recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above, with frequent monitoring as clinically indicated. Permanently discontinue ALECENSA in case of recurrence. CPK elevation greater than 5 times ULN Temporarily withhold until recovery to baseline or to less than or equal to 2.5 times ULN, then resume at same dose. CPK elevation greater than 10 times ULN or second occurrence of CPK elevation of greater than 5 times ULN Temporarily withhold until recovery to baseline or to less than or equal to 2.5 times ULN, then resume at reduced dose as per Table 2 . Hemolytic Anemia Withhold ALECENSA if hemolytic anemia is suspected. Upon resolution, resume at reduced dose or permanently discontinue.
Storage and Handling
Hard capsules, white 150 mg capsules with "ALE" printed in black ink on the cap and "150 mg" printed in black ink on the body, available in: 240 capsules per bottle: NDC 50242-130-01
How Supplied
Hard capsules, white 150 mg capsules with "ALE" printed in black ink on the cap and "150 mg" printed in black ink on the body, available in: 240 capsules per bottle: NDC 50242-130-01
Description
Indications and Usage ( 1.1 ) 04/2024 Dosage and Administration ( 2.1 , 2.2 , 2.3 ) 04/2024 Warnings and Precautions ( 5 ) 04/2024
Section 42229-5
Adjuvant Treatment of Resected ALK-Positive NSCLC
The safety of ALECENSA was evaluated in ALINA, a multi-center, open-label, randomized trial for the adjuvant treatment of patients with resected ALK-positive NSCLC [see Clinical Studies (14.1) ]. At the time of DFS analysis, the median duration of exposure was 23.9 months for ALECENSA and 2.1 months for platinum-based chemotherapy.
Serious adverse reactions occurred in 13% of patients treated with ALECENSA; the most frequent serious adverse reactions (≥ 1%) were pneumonia (3.9%), appendicitis (3.1%), and acute myocardial infarction (1.6%). Permanent discontinuation of ALECENSA due to an adverse event occurred in 5% of patients; the most frequent adverse reactions (≥ 1%) that led to treatment discontinuation were pneumonitis and hepatotoxicity.
Dosage interruptions of ALECENSA due to an adverse reaction occurred in 27% of patients. Adverse reactions which required dosage interruption in ≥ 2% of patients included hepatotoxicity, increased blood CPK, COVID-19, myalgia, abdominal pain, and pneumonia.
Dose reductions of ALECENSA due to an adverse reaction occurred in 26% of patients. Adverse reactions which required dose reductions in ≥ 2% of patients included hepatotoxicity, increased blood CPK, rash, bradycardia and myalgia.
Table 4 and 5 summarize the common adverse reactions and laboratory abnormalities observed in ALINA.
| Adverse Reaction | ALECENSA N=128 |
Chemotherapy N=120 |
||
|---|---|---|---|---|
| All Grades (%) | Grades 3-4 (%) | All Grades (%) | Grades 3-4 (%) | |
| Based on NCI CTCAE v5.0 | ||||
| Hepatobiliary System Disorders | ||||
| Hepatotoxicity Includes increased alanine aminotransferase, increased aspartate aminotransferase, increased bile acids, increased conjugated bilirubin, increased blood bilirubin, increased unconjugated blood bilirubin, increased gamma-glutamyltransferase, hepatotoxicity, hyperbilirubinemia, increased liver function test, ocular icterus and increased transaminases.
|
61 | 4.7 All events are Grade 3
|
13 | 0 |
| Gastrointestinal Disorders | ||||
| Constipation | 42 | 0.8 | 25 | 0.8 |
| Abdominal pain Includes abdominal discomfort, abdominal pain, lower abdominal pain, upper abdominal pain, abdominal tenderness, epigastric discomfort and gastrointestinal pain.
|
13 | 0 | 10 | 1.7 |
| Diarrhea Includes colitis and diarrhea.
|
13 | 0.8 | 9 | 1.7 |
| Musculoskeletal | ||||
| Myalgia Includes muscle fatigue, muscular weakness, musculoskeletal chest pain, musculoskeletal stiffness and myalgia.
|
34 | 0.8 | 1.7 | 0 |
| Infections and Infestations | ||||
| COVID-19 | 29 | 0 | 0.8 | 0 |
| General Disorders and Administration Site Conditions | ||||
| Fatigue Includes asthenia and fatigue.
|
25 | 0.8 | 28 | 4.2 |
| Edema Includes edema, face edema, localized edema, peripheral edema, face swelling and peripheral swelling.
|
16 | 0 | 1.7 | 0 |
| Skin and Subcutaneous Tissue Disorders | ||||
| Rash Includes acneiform dermatitis, bullous dermatitis, drug eruption, eczema, rash, erythematous rash, maculo-papular rash, papular rash, seborrheic dermatitis, urticaria and xeroderma.
|
23 | 1.6 | 10 | 0 |
| Respiratory System Disorders | ||||
| Cough Includes cough and productive cough.
|
20 | 0.8 | 3.3 | 0 |
| Dyspnea Includes dyspnea and exertional dyspnea.
|
13 | 0.8 | 2.5 | 0 |
| Renal | ||||
| Renal Impairment Includes azotemia, increased blood creatinine, decreased renal creatinine clearance, decreased glomerular filtration rate, hypercreatininemia, renal impairment and renal failure.
|
16 | 0.8 | 9 | 0 |
| Nervous System Disorders | ||||
| Dysgeusia Includes dysgeusia and taste disorder.
|
13 | 0 | 3.3 | 0 |
| Headache | 11 | 0 | 7 | 0 |
| Investigations | ||||
| Increased weight | 13 | 0.8 | 0.8 | 0 |
| Cardiac Disorders | ||||
| Bradycardia Includes bradycardia and sinus bradycardia.
|
12 | 0 | 0 | 0 |
Clinically significant adverse reactions in < 10% of patients who received ALECENSA in ALINA: nausea (8%), vomiting (7%), vision disorders (4.7%; includes blurred vision, visual acuity reduced and photopsia), stomatitis (4.7%; includes stomatitis and mouth ulceration), photosensitivity reaction (3.9%) and pneumonitis (2.3%).
| Parameter | ALECENSA N=128 |
Chemotherapy N=120 |
||
|---|---|---|---|---|
| All Grades (%) | Grades 3–4 (%) | All Grades (%) | Grades 3–4 (%) | |
| Based on NCI CTCAE v5.0 | ||||
| Chemistry | ||||
| Increased CPK | 77 | 8 | 8 | 1.7 All events were Grade 3
|
| Increased AST | 75 | 0.8 | 25 | 0 |
| Increased bilirubin | 68 | 2.3 | 4.2 | 0 |
| Increased alkaline phosphatase | 64 | 0 | 14 | 0 |
| Increased ALT | 57 | 2.3 | 28 | 0 |
| Increased creatinine | 41 | 0 | 23 | 0 |
| Increased uric acid | 30 | 0 | 19 | 0 |
| Hematology | ||||
| Decreased hemoglobin | 69 | 0 | 67 | 0.8 |
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 04/2024 | |
|
PATIENT INFORMATION
ALECENSA® (a-le-sen-sah) (alectinib) capsules |
||
|
What is the most important information I should know about ALECENSA? ALECENSA may cause serious side effects, including:
|
||
|
|
|
|
||
|
What is ALECENSA?
ALECENSA is a prescription medicine used to treat adults with non-small cell lung cancer (NSCLC) that is caused by an abnormal anaplastic lymphoma kinase (ALK) gene:
It is not known if ALECENSA is safe and effective in children. |
||
Before you take ALECENSA, tell your healthcare provider about all of your medical conditions, including if you:
|
||
How should I take ALECENSA?
|
||
|
What should I avoid while taking ALECENSA?
Avoid spending time in the sunlight during treatment with ALECENSA and for 7 days after the last dose of ALECENSA. Your skin may be sensitive to the sun (photosensitivity) and you may burn more easily and get severe sunburns. Use sun protecting measures, such as sunscreen and lip balm with an SPF 50 or greater to help protect against sunburn. |
||
|
What are the possible side effects of ALECENSA? ALECENSA may cause serious side effects, including: The most common side effects of ALECENSA include: |
||
|
|
|
| These are not all of the possible side effects of ALECENSA. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store ALECENSA?
|
||
|
General information about the safe and effective use of ALECENSA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ALECENSA for a condition for which it was not prescribed. Do not give ALECENSA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ALECENSA that is written for health professionals. |
||
|
What are the ingredients in ALECENSA?
Active ingredient: alectinib Inactive ingredients: lactose monohydrate, hydroxypropylcellulose, sodium lauryl sulfate, magnesium stearate and carboxymethylcellulose calcium. Capsule shell contains: hypromellose, carrageenan, potassium chloride, titanium dioxide, corn starch, and carnauba wax. Printing ink contains: red iron oxide (E172), yellow iron oxide (E172), FD&C Blue No. 2 aluminum lake (E132), carnauba wax, white shellac, and glyceryl monooleate. Distributed by: Genentech, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990 ALECENSA® is a registered trademark of Chugai Pharmaceutical Co., Ltd., Tokyo, Japan ©2024 Genentech, Inc. For more information, go to www.ALECENSA.com or call 1-800-253-2367. |
Section 43683-2
Section 44425-7
Storage and stability: Do not store above 30°C (86°F). Store in the original container to protect from light and moisture.
10 Overdosage
No experience with overdose is available. There is no specific antidote for overdose with ALECENSA. Alectinib and its major active metabolite M4 are > 99% bound to plasma proteins; therefore, hemodialysis is likely to be ineffective in the treatment of overdose.
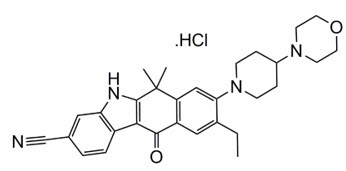
11 Description
ALECENSA (alectinib) is a kinase inhibitor for oral administration. The molecular formula for alectinib is C30H34N4O2 ∙ HCl. The molecular weight is 482.62 g/mol (free base form) and 519.08 g/mol (hydrochloride salt). Alectinib is described chemically as 9-ethyl-6, 6-dimethyl-8-[4-(morpholin-4-yl)piperidin-1-yl]-11-oxo-6, 11-dihydro-5H-benzo[b]carbazole-3-carbonitrile hydrochloride. The chemical structure of alectinib is shown below:
Alectinib HCl is a white to yellow white powder or powder with lumps with a pKa of 7.05 (base).
ALECENSA is supplied as hard capsules containing 150 mg of alectinib (equivalent to 161.33 mg alectinib HCl) and the following inactive ingredients: lactose monohydrate, hydroxypropylcellulose, sodium lauryl sulfate, magnesium stearate, and carboxymethylcellulose calcium. The capsule shell contains hypromellose, carrageenan, potassium chloride, titanium dioxide, corn starch, and carnauba wax. The printing ink contains red iron oxide (E172), yellow iron oxide (E172), FD&C Blue No. 2 aluminum lake (E132), carnauba wax, white shellac, and glyceryl monooleate.
5.4 Bradycardia
Symptomatic bradycardia occurred in patients treated with ALECENSA.
In the pooled safety population [see Adverse Reactions (6.1)], bradycardia occurred in 11% of patients treated with ALECENSA. Twenty percent of 521 patients treated with ALECENSA, for whom serial electrocardiograms (ECGs) were available, had post-dose heart rates of less than 50 beats per minute (bpm).
Monitor heart rate and blood pressure regularly. For asymptomatic bradycardia dose modification is not required. For symptomatic bradycardia that is not life-threatening, withhold ALECENSA until recovery to asymptomatic bradycardia or to a heart rate ≥ 60 bpm and evaluate concomitant medications known to cause bradycardia, as well as anti-hypertensive medications. If bradycardia is attributable to a concomitant medication, resume ALECENSA at a reduced dose (see Table 2) upon recovery to asymptomatic bradycardia or to a heart rate of ≥ 60 bpm, with frequent monitoring as clinically indicated.
Permanently discontinue ALECENSA in cases of life-threatening bradycardia if no contributing concomitant medication is identified [see Dosage and Administration (2.4)]. Permanently discontinue ALECENSA for recurrence of life-threatening bradycardia.
8.4 Pediatric Use
The safety and effectiveness of ALECENSA in pediatric patients have not been established.
8.5 Geriatric Use
Nineteen percent of the 533 patients studied in NP28761, NP28673, ALEX and ALINA were 65 years of age and older (3.2% were 75 years of age and older). No overall differences in effectiveness were observed based on age. Exploratory analysis suggests a higher incidence of serious adverse events (38% vs 25%), more frequent adverse events leading to treatment discontinuations (18% vs 6%) and dose modifications (48% vs 35%) in patients 65 years or older as compared to those younger than 65 years.
5.1 Hepatotoxicity
Severe hepatotoxicity, including drug-induced liver injury, occurred in patients treated with ALECENSA.
In the pooled safety population [see Adverse Reactions (6.1)] of patients who received ALECENSA, hepatotoxicity occurred in 41% of patients and the incidence of Grade ≥ 3 hepatotoxicity was 8%. In the ALINA study, hepatotoxicity occurred in 61% of patients treated with ALECENSA and the incidence of Grade ≥ 3 hepatotoxicity was 4.7%. The majority (72% of 136 patients) of elevated transaminases occurred during the first 3 months of treatment. Treatment discontinuation due to hepatotoxicity occurred in 3.6% of patients who received ALECENSA in the pooled safety population and 1.6% of patients treated in the ALINA study.
In the pooled safety population, concurrent elevations in ALT or AST greater than or equal to 3 times the ULN and total bilirubin greater than or equal to 2 times the ULN, with normal alkaline phosphatase, occurred in less than 1% of patients treated with ALECENSA. Three patients with Grades 3–4 AST/ALT elevations had drug-induced liver injury (documented by liver biopsy in two cases).
Monitor liver function tests including ALT, AST, and total bilirubin every 2 weeks during the first 3 months of treatment, then once a month and as clinically indicated, with more frequent testing in patients who develop transaminase and bilirubin elevations. Based on the severity of the adverse drug reaction, withhold ALECENSA and resume at a reduced dose or permanently discontinue ALECENSA as described in Table 3 [see Dosage and Administration (2.4)].
4 Contraindications
None.
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the label:
- Hepatotoxicity [see Warnings and Precautions (5.1)]
- Interstitial Lung Disease (ILD)/Pneumonitis [see Warnings and Precautions (5.2)]
- Renal Impairment [see Warnings and Precautions (5.3)]
- Bradycardia [see Warnings and Precautions (5.4)]
- Severe Myalgia and Creatine Phosphokinase (CPK) Elevation [see Warnings and Precautions (5.5)]
- Hemolytic Anemia [see Warnings and Precautions (5.6)]
- Embryo-Fetal Toxicity [see Warnings and Precautions (5.7)]
5.3 Renal Impairment
Renal impairment, including fatal cases, occurred in patients treated with ALECENSA.
In the pooled safety population [see Adverse Reactions (6.1)], renal impairment occurred in 12% of patients treated with ALECENSA, including Grade ≥ 3 in 1.7% of patients, of which 0.4% were fatal events. The median time to Grade ≥ 3 renal impairment was 3.7 months (range 0.5 to 31.8 months). Dosage modifications for renal impairment were required in 2.4% of patients.
Permanently discontinue ALECENSA for Grade 4 renal toxicity. Withhold ALECENSA for Grade 3 renal toxicity until recovery to less than or equal to 1.5 times ULN, then resume at reduced dose [see Dosage and Administration (2.4)].
5.6 Hemolytic Anemia
Hemolytic anemia occurred in patients treated with ALECENSA.
Hemolytic anemia was initially reported with ALECENSA in the postmarketing setting, including cases associated with a negative direct antiglobulin test (DAT) result. Assessments for the determination of hemolytic anemia were subsequently collected in the ALINA study, where hemolytic anemia was observed in 3.1% of patients treated with ALECENSA. If hemolytic anemia is suspected, withhold ALECENSA and initiate appropriate laboratory testing. If hemolytic anemia is confirmed, consider resuming at a reduced dose upon resolution or permanently discontinue ALECENSA [see Dosage and Administration (2.4)].
8.6 Renal Impairment
No dose adjustment is recommended for patients with mild or moderate renal impairment. The safety of ALECENSA in patients with severe renal impairment (creatinine clearance less than 30 mL/min) or end-stage renal disease has not been studied [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The pharmacokinetics of alectinib and its major active metabolite M4 have been characterized in patients with ALK-positive NSCLC and healthy subjects.
In patients with ALK-positive NSCLC, the geometric mean (coefficient of variation %) steady-state maximal concentration (Cmax,ss) for alectinib was 665 ng/mL (44%) and for M4 was 246 ng/mL (45%) with peak to trough concentration ratio of 1.2. The geometric mean steady-state area under the curve from 0 to 12 hours (AUC0-12h,ss) for alectinib was 7,430 ng*h/mL (46%) and for M4 was 2,810 ng*h/mL (46%). Alectinib exposure is dose proportional across the dose range of 460 mg to 900 mg (i.e., 0.75 to 1.5 times the approved recommended dosage) under fed conditions. Alectinib and M4 reached steady-state concentrations by day 7. The geometric mean accumulation was approximately 6-fold for both alectinib and M4.
2.1 Patient Selection
Select patients with resectable tumors for the adjuvant treatment of NSCLC with ALECENSA based on the presence of ALK positivity in tumor tissue [see Indications and Usage (1.1) and Clinical Studies (14.1)].
Select patients for the treatment of metastatic NSCLC with ALECENSA based on the presence of ALK positivity in tumor tissue or plasma specimens [see Indications and Usage (1.2) and Clinical Studies (14.2)]. If ALK rearrangements are not detected in a plasma specimen, test tumor tissue if feasible.
Information on FDA-approved tests for the detection of ALK rearrangements in NSCLC is available at http://www.fda.gov/CompanionDiagnostics.
8.7 Hepatic Impairment
No dose adjustment is recommended for patients with mild (Child-Pugh A) or moderate (Child-Pugh B) hepatic impairment. Increased exposure of alectinib occurred in patients with severe hepatic impairment (Child-Pugh C). The recommended dose of ALECENSA in patients with severe hepatic impairment (Child-Pugh C) is 450 mg orally twice daily [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
1 Indications and Usage
ALECENSA is a kinase inhibitor indicated for:
- adjuvant treatment in adult patients following tumor resection of anaplastic lymphoma kinase (ALK)-positive non-small cell lung cancer (NSCLC) (tumors ≥ 4 cm or node positive) as detected by an FDA-approved test. (1.1)
- treatment of adult patients with ALK-positive metastatic NSCLC as detected by an FDA-approved test. (1.2)
12.1 Mechanism of Action
Alectinib is a tyrosine kinase inhibitor that targets ALK and RET. In nonclinical studies, alectinib inhibited ALK phosphorylation and ALK-mediated activation of the downstream signaling proteins STAT3 and AKT, and decreased tumor cell viability in multiple cell lines harboring ALK fusions, amplifications, or activating mutations. The major active metabolite of alectinib, M4, showed similar in vitro potency and activity.
Alectinib and M4 demonstrated in vitro and in vivo activity against multiple mutant forms of the ALK enzyme, including some mutations identified in NSCLC tumors in patients who have progressed on crizotinib.
In mouse models implanted with tumors carrying ALK fusions, administration of alectinib resulted in antitumor activity and prolonged survival, including in mouse models implanted intracranially with ALK-driven tumor cell lines.
5.7 Embryo Fetal Toxicity
Based on findings from animal studies and its mechanism of action, ALECENSA can cause fetal harm when administered to pregnant women. Oral administration of alectinib to pregnant rats and rabbits during the period of organogenesis resulted in embryo-fetal toxicity and abortion at maternally toxic doses with exposures approximately 2.7-fold those observed in humans with alectinib 600 mg twice daily. Advise pregnant women and females of reproductive potential of the potential risk to a fetus.
Advise females of reproductive potential to use effective contraception during treatment with ALECENSA and for 5 weeks following the last dose [see Use in Specific Populations (8.1 and 8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions
- Hepatotoxicity: Monitor liver laboratory tests every 2 weeks during the first 3 months of treatment, then once a month and as clinically indicated, with more frequent testing in patients who develop transaminase and bilirubin elevations. In case of severe ALT, AST, or bilirubin elevations, withhold, then reduce dose, or permanently discontinue ALECENSA. (2.4, 5.1)
- Interstitial Lung Disease (ILD)/Pneumonitis: Immediately withhold ALECENSA in patients diagnosed with ILD/pneumonitis and permanently discontinue if no other potential causes of ILD/pneumonitis have been identified. (2.4, 5.2)
- Renal Impairment: Withhold ALECENSA for severe renal impairment, then resume ALECENSA at reduced dose upon recovery or permanently discontinue (2.4, 5.3).
- Bradycardia: Monitor heart rate and blood pressure regularly. If symptomatic, withhold ALECENSA then reduce dose, or permanently discontinue. (2.4, 5.4)
- Severe Myalgia and Creatine Phosphokinase (CPK) Elevation: Assess CPK every 2 weeks during the first month of treatment and in patients reporting unexplained muscle pain, tenderness, or weakness. In case of severe CPK elevations, withhold, then resume or reduce dose. (2.4, 5.5)
- Hemolytic Anemia: If hemolytic anemia is suspected, withhold ALECENSA. If hemolytic anemia is confirmed, consider resuming at a reduced dose upon resolution or permanently discontinue. (5.6)
- Embryo-Fetal Toxicity: ALECENSA can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.7, 8.1, 8.3)
2 Dosage and Administration
600 mg orally twice daily. Administer ALECENSA with food. (2.2)
3 Dosage Forms and Strengths
150 mg hard capsules, white, with "ALE" printed in black ink on the cap and "150 mg" printed in black ink on the body.
2.2 Dosing and Administration
The recommended dosage information for ALECENSA is provided in Table 1.
| Indication | Recommended Dosage of ALECENSA | Duration |
|---|---|---|
| Adjuvant treatment of resected NSCLC | 600 mg orally twice daily with food [see Clinical Pharmacology (12.3)] |
For a total of 2 years or until disease recurrence or unacceptable toxicity |
| Metastatic NSCLC | Until disease progression or unacceptable toxicity | |
|
8 Use in Specific Populations
Lactation: Do not breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The pooled safety population described in the WARNINGS AND PRECAUTIONS reflect exposure to ALECENSA as a single agent at 600 mg orally twice daily in 533 patients in Studies NP28761, NP28673, ALEX and ALINA [see Clinical Studies (14)]. Among 533 patients who received ALECENSA, 75% were exposed for 6 months or longer and 64% were exposed for greater than one year. In this pooled safety population, the most common (≥ 20%) adverse reactions were hepatotoxicity (41%), constipation (39%), fatigue (36%), myalgia (31%), edema (29%), rash (23%) and cough (21%). The most common (≥ 2%) Grade 3 or 4 laboratory abnormalities were increased CPK (6%), decreased hemoglobin (4.4%), increased ALT (4.2%), increased bilirubin (4.0%) and increased AST (3.4%).
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Inform patients of the following:
16 How Supplied/storage and Handling
Hard capsules, white 150 mg capsules with "ALE" printed in black ink on the cap and "150 mg" printed in black ink on the body, available in:
| 240 capsules per bottle: | NDC 50242-130-01 |
2.4 Dose Modifications for Adverse Reactions
The dose reduction schedule for ALECENSA is provided in Table 2.
| Dose Reduction Schedule | Dose Level |
|---|---|
| Starting dose | 600 mg taken orally twice daily |
| First dose reduction | 450 mg taken orally twice daily |
| Second dose reduction | 300 mg taken orally twice daily |
Discontinue if patients are unable to tolerate the 300 mg twice daily dose.
Recommendations for dose modifications of ALECENSA in case of adverse reactions are provided in Table 3.
| Criteria ALT = alanine transaminase; AST = aspartate transaminase; ULN = upper limit of normal; ILD = interstitial lung disease; CPK = blood creatine phosphokinase
|
ALECENSA Dose Modification |
|---|---|
| ALT or AST elevation of greater than 5 times upper limit of normal (ULN) with total bilirubin less than or equal to 2 times ULN | Temporarily withhold until recovery to baseline or to less than or equal to 3 times ULN, then resume at reduced dose as per Table 2. |
| ALT or AST elevation greater than 3 times ULN with total bilirubin elevation greater than 2 times ULN in the absence of cholestasis or hemolysis | Permanently discontinue ALECENSA. |
| Total bilirubin elevation of greater than 3 times ULN | Temporarily withhold until recovery to baseline or to less than or equal to 1.5 times ULN, then resume at reduced dose as per Table 2. |
| Any grade treatment-related interstitial lung disease (ILD)/pneumonitis | Permanently discontinue ALECENSA. |
| Grade 3 renal impairment | Temporarily withhold until serum creatinine recovers to less than or equal to 1.5 times ULN, then resume at reduced dose. |
| Grade 4 renal impairment | Permanently discontinue ALECENSA. |
| Symptomatic bradycardia | Withhold ALECENSA until recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above. If contributing concomitant medication is identified and discontinued, or its dose is adjusted, resume ALECENSA at previous dose upon recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above. If no contributing concomitant medication is identified, or if contributing concomitant medications are not discontinued or dose modified, resume ALECENSA at reduced dose (see Table 2) upon recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above. |
| Bradycardia Heart rate less than 60 beats per minute (bpm) (life-threatening consequences, urgent intervention indicated) |
Permanently discontinue ALECENSA if no contributing concomitant medication is identified. If contributing concomitant medication is identified and discontinued, or its dose is adjusted, resume ALECENSA at reduced dose (see Table 2) upon recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above, with frequent monitoring as clinically indicated. Permanently discontinue ALECENSA in case of recurrence. |
| CPK elevation greater than 5 times ULN | Temporarily withhold until recovery to baseline or to less than or equal to 2.5 times ULN, then resume at same dose. |
| CPK elevation greater than 10 times ULN or second occurrence of CPK elevation of greater than 5 times ULN | Temporarily withhold until recovery to baseline or to less than or equal to 2.5 times ULN, then resume at reduced dose as per Table 2. |
| Hemolytic Anemia | Withhold ALECENSA if hemolytic anemia is suspected. Upon resolution, resume at reduced dose or permanently discontinue. |
2.3 Recommended Dosage for Hepatic Impairment
The recommended dose of ALECENSA in patients with severe hepatic impairment (Child-Pugh C) is 450 mg orally twice daily [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
1.2 Treatment of Metastatic Alk Positive Nsclc
ALECENSA is indicated for the treatment of adult patients with ALK-positive metastatic NSCLC as detected by an FDA-approved test [see Dosage & Administration (2.1)].
5.2 Interstitial Lung Disease (ild)/pneumonitis
ILD/pneumonitis occurred in patients treated with ALECENSA.
In the pooled safety population [see Adverse Reactions (6.1)], ILD/pneumonitis occurred in 1.3% of patients treated with ALECENSA with 0.4% of patients experiencing Grade 3 ILD/pneumonitis.
Five patients (0.9%) in the pooled safety population discontinued ALECENSA due to ILD/pneumonitis. The median time-to-onset of Grade 3 or higher ILD/pneumonitis was 2.1 months (range: 0.6 months to 3.6 months).
Promptly investigate for ILD/pneumonitis in any patient who presents with worsening of respiratory symptoms indicative of ILD/pneumonitis (e.g., dyspnea, cough, and fever). Immediately withhold ALECENSA treatment in patients diagnosed with ILD/pneumonitis and permanently discontinue ALECENSA if no other potential causes of ILD/pneumonitis have been identified [see Dosage and Administration (2.4) and Adverse Reactions (6)].
8.3 Females and Males of Reproductive Potential
ALECENSA can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
14.1 Adjuvant Treatment of Resected Alk Positive Nsclc
The efficacy of ALECENSA for the adjuvant treatment of patients with ALK-positive NSCLC following complete tumor resection was evaluated in a global, randomized open-label clinical trial (ALINA: NCT03456076). Eligible patients were required to have resectable ALK-positive NSCLC, Stage IB (tumors ≥ 4 cm) – IIIA per the Union for International Cancer Control/American Joint Committee on Cancer (UICC/AJCC) Staging System, 7th Edition. ALK rearrangements were identified by a locally performed FDA-approved ALK test or by a centrally performed VENTANA ALK (D5F3) CDx assay.
Randomization was stratified by race (Asian vs. other races) and stage of disease (IB vs. II vs. IIIA). Patients were randomized (1:1) to receive ALECENSA 600 mg orally twice daily or platinum-based chemotherapy following tumor resection. Treatment with ALECENSA continued for a total of 2 years, or until disease recurrence or unacceptable toxicity. Platinum-based chemotherapy was administered intravenously for 4 cycles, with each cycle lasting 21 days, according to one of the following regimens:
- Cisplatin 75 mg/m2 on Day 1 plus vinorelbine 25 mg/m2 on Days 1 and 8
- Cisplatin 75 mg/m2 on Day 1 plus gemcitabine 1250 mg/m2 on Days 1 and 8
- Cisplatin 75 mg/m2 on Day 1 plus pemetrexed 500 mg/m2 on Day 1
In the event of intolerance to a cisplatin-based regimen, carboplatin was administered instead of cisplatin in the above combinations at a dose of AUC 5 mg/mL/min or 6 mg/mL/min.
The major efficacy outcome measures were disease-free survival (DFS) in patients with stage II-IIIA NSCLC and DFS in patients with stage IB-IIIA NSCLC (intent-to-treat [ITT] population) as assessed by investigator. DFS was defined as the time from date of randomization to the date of occurrence of any of the following: first documented recurrence of disease, new primary NSCLC, or death due to any cause, whichever occurred first. An additional efficacy outcome measure was overall survival (OS) in the ITT population.
A total of 257 patients were randomized to ALECENSA (N=130) or to chemotherapy (N=127). The median age was 56 years (range: 26 to 87), 24% were ≥ 65 years old; 52% were female; 56% were Asian, 42% were White, 0.4% were Black or African American, 2.3% were race unknown; 0.4% were Hispanic or Latino; 60% were never smokers; 53% had an ECOG PS of 0; 10% of patients had Stage IB, 35% had Stage II and 55% had Stage IIIA disease.
ALINA demonstrated a statistically significant improvement in DFS for patients treated with ALECENSA compared to patients treated with chemotherapy. OS data were not mature at the time of DFS analysis with 2.3% of deaths reported in the ITT population.
The efficacy results from ALINA are summarized in Table 10 and Figure 1.
| Efficacy Parameter | Stage II-IIIA Population | ITT Population | ||
|---|---|---|---|---|
| ALECENSA N=116 |
Chemotherapy N=115 |
ALECENSA N=130 |
Chemotherapy N=127 |
|
| DFS = Disease-Free Survival; ITT = Intent-to-Treat; CI = Confidence Interval; NR = Not Reached; NE = Not Estimable. | ||||
| DFS events (%) | 14 (12) | 45 (39) | 15 (12) | 50 (39) |
| Disease recurrence (%) | 14 (12) | 44 (38) | 15 (12) | 49 (38) |
| Death | 0 | 1 (0.9) | 0 | 1 (0.8) |
| Median DFS, months (95% CI) Kaplan-Meier method.
|
NR (NE, NE) |
44.4 (27.8, NE) |
NR (NE, NE) |
41.3 (28.5, NE) |
| Hazard Ratio (95% CI) Stratified Cox model, stratified by race (Asian vs. other races) in Stage II-IIIA and stratified by race (Asian vs. other races) and tumor stage (Stage IB vs. II vs. IIIA) in ITT.
|
0.24 (0.13, 0.45) | 0.24 (0.13, 0.43) | ||
| p-value Stratified log-rank test stratified by race (Asian vs. other races) in Stage II-IIIA and stratified by race (Asian vs. other races) and tumor stage (Stage IB vs. II vs. IIIA) in ITT.
|
<0.0001 | <0.0001 |
Figure 1: Kaplan-Meier Curves of Investigator-Assessed DFS (ITT Population) in ALINA
In an exploratory analysis of site(s) of relapse, the proportion of patients with brain involvement at the time of disease recurrence was 4 patients (3.1%) in the ALECENSA arm and 14 patients (11%) in the chemotherapy arm.
Principal Display Panel 150 Mg Capsule Bottle Carton
NDC 50242-130-01
Alecensa®
(alectinib)
capsules
150 mg
Each capsule contains 150 mg of alectinib
(equivalent to 161.33 mg alectinib HCl).
Rx only
240 capsules
Genentech
11008914
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with alectinib have not been conducted.
Alectinib was not mutagenic in vitro in the bacterial reverse mutation (Ames) assay, but was positive with an increased number of micronuclei in a rat bone marrow micronucleus test. The mechanism of micronucleus induction was abnormal chromosome segregation (aneugenicity) and not a clastogenic effect on chromosomes.
No studies in animals have been performed to evaluate the effect of alectinib on fertility. No adverse effects on male and female reproductive organs were observed in general toxicology studies conducted in rats and monkeys.
5.5 Severe Myalgia and Creatine Phosphokinase (cpk) Elevation
Severe myalgia and creatine phosphokinase (CPK) elevation occurred in patients treated with ALECENSA.
In the pooled safety population [see Adverse Reactions (6.1)], myalgia (including muscle- and musculoskeletal-related reactions) occurred in 31% of patients treated with ALECENSA, including Grade ≥ 3 in 0.8% of patients. Dosage modifications for myalgia events were required in 2.1% of patients.
In the pooled safety population, of the 491 patients with CPK laboratory data available, elevated CPK occurred in 56% of patients treated with ALECENSA, including 6% Grade ≥ 3. The median time to Grade ≥ 3 CPK elevation was 15 days (interquartile range - 15 –337 days). Dosage modifications for elevation of CPK occurred in 5% of patients.
In the ALINA study, elevated CPK occurred in 77% of 128 patients with CPK laboratory data, including 6% Grade ≥ 3 elevations.
Advise patients to report any unexplained muscle pain, tenderness, or weakness. Assess CPK levels every 2 weeks for the first month of treatment and as clinically indicated in patients reporting symptoms. Based on the severity of the CPK elevation, withhold ALECENSA, then resume or reduce dose [see Dosage and Administration (2.4)].
1.1 Adjuvant Treatment of Resected Alk Positive Non Small Cell Lung Cancer (nsclc)
ALECENSA is indicated as adjuvant treatment in adult patients following tumor resection of anaplastic lymphoma kinase (ALK)-positive non-small cell lung cancer (NSCLC) (tumors ≥ 4 cm or node positive), as detected by an FDA-approved test [see Dosage & Administration (2.1)].
Structured Label Content
Section 42229-5 (42229-5)
Adjuvant Treatment of Resected ALK-Positive NSCLC
The safety of ALECENSA was evaluated in ALINA, a multi-center, open-label, randomized trial for the adjuvant treatment of patients with resected ALK-positive NSCLC [see Clinical Studies (14.1) ]. At the time of DFS analysis, the median duration of exposure was 23.9 months for ALECENSA and 2.1 months for platinum-based chemotherapy.
Serious adverse reactions occurred in 13% of patients treated with ALECENSA; the most frequent serious adverse reactions (≥ 1%) were pneumonia (3.9%), appendicitis (3.1%), and acute myocardial infarction (1.6%). Permanent discontinuation of ALECENSA due to an adverse event occurred in 5% of patients; the most frequent adverse reactions (≥ 1%) that led to treatment discontinuation were pneumonitis and hepatotoxicity.
Dosage interruptions of ALECENSA due to an adverse reaction occurred in 27% of patients. Adverse reactions which required dosage interruption in ≥ 2% of patients included hepatotoxicity, increased blood CPK, COVID-19, myalgia, abdominal pain, and pneumonia.
Dose reductions of ALECENSA due to an adverse reaction occurred in 26% of patients. Adverse reactions which required dose reductions in ≥ 2% of patients included hepatotoxicity, increased blood CPK, rash, bradycardia and myalgia.
Table 4 and 5 summarize the common adverse reactions and laboratory abnormalities observed in ALINA.
| Adverse Reaction | ALECENSA N=128 |
Chemotherapy N=120 |
||
|---|---|---|---|---|
| All Grades (%) | Grades 3-4 (%) | All Grades (%) | Grades 3-4 (%) | |
| Based on NCI CTCAE v5.0 | ||||
| Hepatobiliary System Disorders | ||||
| Hepatotoxicity Includes increased alanine aminotransferase, increased aspartate aminotransferase, increased bile acids, increased conjugated bilirubin, increased blood bilirubin, increased unconjugated blood bilirubin, increased gamma-glutamyltransferase, hepatotoxicity, hyperbilirubinemia, increased liver function test, ocular icterus and increased transaminases.
|
61 | 4.7 All events are Grade 3
|
13 | 0 |
| Gastrointestinal Disorders | ||||
| Constipation | 42 | 0.8 | 25 | 0.8 |
| Abdominal pain Includes abdominal discomfort, abdominal pain, lower abdominal pain, upper abdominal pain, abdominal tenderness, epigastric discomfort and gastrointestinal pain.
|
13 | 0 | 10 | 1.7 |
| Diarrhea Includes colitis and diarrhea.
|
13 | 0.8 | 9 | 1.7 |
| Musculoskeletal | ||||
| Myalgia Includes muscle fatigue, muscular weakness, musculoskeletal chest pain, musculoskeletal stiffness and myalgia.
|
34 | 0.8 | 1.7 | 0 |
| Infections and Infestations | ||||
| COVID-19 | 29 | 0 | 0.8 | 0 |
| General Disorders and Administration Site Conditions | ||||
| Fatigue Includes asthenia and fatigue.
|
25 | 0.8 | 28 | 4.2 |
| Edema Includes edema, face edema, localized edema, peripheral edema, face swelling and peripheral swelling.
|
16 | 0 | 1.7 | 0 |
| Skin and Subcutaneous Tissue Disorders | ||||
| Rash Includes acneiform dermatitis, bullous dermatitis, drug eruption, eczema, rash, erythematous rash, maculo-papular rash, papular rash, seborrheic dermatitis, urticaria and xeroderma.
|
23 | 1.6 | 10 | 0 |
| Respiratory System Disorders | ||||
| Cough Includes cough and productive cough.
|
20 | 0.8 | 3.3 | 0 |
| Dyspnea Includes dyspnea and exertional dyspnea.
|
13 | 0.8 | 2.5 | 0 |
| Renal | ||||
| Renal Impairment Includes azotemia, increased blood creatinine, decreased renal creatinine clearance, decreased glomerular filtration rate, hypercreatininemia, renal impairment and renal failure.
|
16 | 0.8 | 9 | 0 |
| Nervous System Disorders | ||||
| Dysgeusia Includes dysgeusia and taste disorder.
|
13 | 0 | 3.3 | 0 |
| Headache | 11 | 0 | 7 | 0 |
| Investigations | ||||
| Increased weight | 13 | 0.8 | 0.8 | 0 |
| Cardiac Disorders | ||||
| Bradycardia Includes bradycardia and sinus bradycardia.
|
12 | 0 | 0 | 0 |
Clinically significant adverse reactions in < 10% of patients who received ALECENSA in ALINA: nausea (8%), vomiting (7%), vision disorders (4.7%; includes blurred vision, visual acuity reduced and photopsia), stomatitis (4.7%; includes stomatitis and mouth ulceration), photosensitivity reaction (3.9%) and pneumonitis (2.3%).
| Parameter | ALECENSA N=128 |
Chemotherapy N=120 |
||
|---|---|---|---|---|
| All Grades (%) | Grades 3–4 (%) | All Grades (%) | Grades 3–4 (%) | |
| Based on NCI CTCAE v5.0 | ||||
| Chemistry | ||||
| Increased CPK | 77 | 8 | 8 | 1.7 All events were Grade 3
|
| Increased AST | 75 | 0.8 | 25 | 0 |
| Increased bilirubin | 68 | 2.3 | 4.2 | 0 |
| Increased alkaline phosphatase | 64 | 0 | 14 | 0 |
| Increased ALT | 57 | 2.3 | 28 | 0 |
| Increased creatinine | 41 | 0 | 23 | 0 |
| Increased uric acid | 30 | 0 | 19 | 0 |
| Hematology | ||||
| Decreased hemoglobin | 69 | 0 | 67 | 0.8 |
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 04/2024 | |
|
PATIENT INFORMATION
ALECENSA® (a-le-sen-sah) (alectinib) capsules |
||
|
What is the most important information I should know about ALECENSA? ALECENSA may cause serious side effects, including:
|
||
|
|
|
|
||
|
What is ALECENSA?
ALECENSA is a prescription medicine used to treat adults with non-small cell lung cancer (NSCLC) that is caused by an abnormal anaplastic lymphoma kinase (ALK) gene:
It is not known if ALECENSA is safe and effective in children. |
||
Before you take ALECENSA, tell your healthcare provider about all of your medical conditions, including if you:
|
||
How should I take ALECENSA?
|
||
|
What should I avoid while taking ALECENSA?
Avoid spending time in the sunlight during treatment with ALECENSA and for 7 days after the last dose of ALECENSA. Your skin may be sensitive to the sun (photosensitivity) and you may burn more easily and get severe sunburns. Use sun protecting measures, such as sunscreen and lip balm with an SPF 50 or greater to help protect against sunburn. |
||
|
What are the possible side effects of ALECENSA? ALECENSA may cause serious side effects, including: The most common side effects of ALECENSA include: |
||
|
|
|
| These are not all of the possible side effects of ALECENSA. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store ALECENSA?
|
||
|
General information about the safe and effective use of ALECENSA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ALECENSA for a condition for which it was not prescribed. Do not give ALECENSA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ALECENSA that is written for health professionals. |
||
|
What are the ingredients in ALECENSA?
Active ingredient: alectinib Inactive ingredients: lactose monohydrate, hydroxypropylcellulose, sodium lauryl sulfate, magnesium stearate and carboxymethylcellulose calcium. Capsule shell contains: hypromellose, carrageenan, potassium chloride, titanium dioxide, corn starch, and carnauba wax. Printing ink contains: red iron oxide (E172), yellow iron oxide (E172), FD&C Blue No. 2 aluminum lake (E132), carnauba wax, white shellac, and glyceryl monooleate. Distributed by: Genentech, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990 ALECENSA® is a registered trademark of Chugai Pharmaceutical Co., Ltd., Tokyo, Japan ©2024 Genentech, Inc. For more information, go to www.ALECENSA.com or call 1-800-253-2367. |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage and stability: Do not store above 30°C (86°F). Store in the original container to protect from light and moisture.
10 Overdosage (10 OVERDOSAGE)
No experience with overdose is available. There is no specific antidote for overdose with ALECENSA. Alectinib and its major active metabolite M4 are > 99% bound to plasma proteins; therefore, hemodialysis is likely to be ineffective in the treatment of overdose.
11 Description (11 DESCRIPTION)
ALECENSA (alectinib) is a kinase inhibitor for oral administration. The molecular formula for alectinib is C30H34N4O2 ∙ HCl. The molecular weight is 482.62 g/mol (free base form) and 519.08 g/mol (hydrochloride salt). Alectinib is described chemically as 9-ethyl-6, 6-dimethyl-8-[4-(morpholin-4-yl)piperidin-1-yl]-11-oxo-6, 11-dihydro-5H-benzo[b]carbazole-3-carbonitrile hydrochloride. The chemical structure of alectinib is shown below:
Alectinib HCl is a white to yellow white powder or powder with lumps with a pKa of 7.05 (base).
ALECENSA is supplied as hard capsules containing 150 mg of alectinib (equivalent to 161.33 mg alectinib HCl) and the following inactive ingredients: lactose monohydrate, hydroxypropylcellulose, sodium lauryl sulfate, magnesium stearate, and carboxymethylcellulose calcium. The capsule shell contains hypromellose, carrageenan, potassium chloride, titanium dioxide, corn starch, and carnauba wax. The printing ink contains red iron oxide (E172), yellow iron oxide (E172), FD&C Blue No. 2 aluminum lake (E132), carnauba wax, white shellac, and glyceryl monooleate.
5.4 Bradycardia
Symptomatic bradycardia occurred in patients treated with ALECENSA.
In the pooled safety population [see Adverse Reactions (6.1)], bradycardia occurred in 11% of patients treated with ALECENSA. Twenty percent of 521 patients treated with ALECENSA, for whom serial electrocardiograms (ECGs) were available, had post-dose heart rates of less than 50 beats per minute (bpm).
Monitor heart rate and blood pressure regularly. For asymptomatic bradycardia dose modification is not required. For symptomatic bradycardia that is not life-threatening, withhold ALECENSA until recovery to asymptomatic bradycardia or to a heart rate ≥ 60 bpm and evaluate concomitant medications known to cause bradycardia, as well as anti-hypertensive medications. If bradycardia is attributable to a concomitant medication, resume ALECENSA at a reduced dose (see Table 2) upon recovery to asymptomatic bradycardia or to a heart rate of ≥ 60 bpm, with frequent monitoring as clinically indicated.
Permanently discontinue ALECENSA in cases of life-threatening bradycardia if no contributing concomitant medication is identified [see Dosage and Administration (2.4)]. Permanently discontinue ALECENSA for recurrence of life-threatening bradycardia.
8.4 Pediatric Use
The safety and effectiveness of ALECENSA in pediatric patients have not been established.
8.5 Geriatric Use
Nineteen percent of the 533 patients studied in NP28761, NP28673, ALEX and ALINA were 65 years of age and older (3.2% were 75 years of age and older). No overall differences in effectiveness were observed based on age. Exploratory analysis suggests a higher incidence of serious adverse events (38% vs 25%), more frequent adverse events leading to treatment discontinuations (18% vs 6%) and dose modifications (48% vs 35%) in patients 65 years or older as compared to those younger than 65 years.
5.1 Hepatotoxicity
Severe hepatotoxicity, including drug-induced liver injury, occurred in patients treated with ALECENSA.
In the pooled safety population [see Adverse Reactions (6.1)] of patients who received ALECENSA, hepatotoxicity occurred in 41% of patients and the incidence of Grade ≥ 3 hepatotoxicity was 8%. In the ALINA study, hepatotoxicity occurred in 61% of patients treated with ALECENSA and the incidence of Grade ≥ 3 hepatotoxicity was 4.7%. The majority (72% of 136 patients) of elevated transaminases occurred during the first 3 months of treatment. Treatment discontinuation due to hepatotoxicity occurred in 3.6% of patients who received ALECENSA in the pooled safety population and 1.6% of patients treated in the ALINA study.
In the pooled safety population, concurrent elevations in ALT or AST greater than or equal to 3 times the ULN and total bilirubin greater than or equal to 2 times the ULN, with normal alkaline phosphatase, occurred in less than 1% of patients treated with ALECENSA. Three patients with Grades 3–4 AST/ALT elevations had drug-induced liver injury (documented by liver biopsy in two cases).
Monitor liver function tests including ALT, AST, and total bilirubin every 2 weeks during the first 3 months of treatment, then once a month and as clinically indicated, with more frequent testing in patients who develop transaminase and bilirubin elevations. Based on the severity of the adverse drug reaction, withhold ALECENSA and resume at a reduced dose or permanently discontinue ALECENSA as described in Table 3 [see Dosage and Administration (2.4)].
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the label:
- Hepatotoxicity [see Warnings and Precautions (5.1)]
- Interstitial Lung Disease (ILD)/Pneumonitis [see Warnings and Precautions (5.2)]
- Renal Impairment [see Warnings and Precautions (5.3)]
- Bradycardia [see Warnings and Precautions (5.4)]
- Severe Myalgia and Creatine Phosphokinase (CPK) Elevation [see Warnings and Precautions (5.5)]
- Hemolytic Anemia [see Warnings and Precautions (5.6)]
- Embryo-Fetal Toxicity [see Warnings and Precautions (5.7)]
5.3 Renal Impairment
Renal impairment, including fatal cases, occurred in patients treated with ALECENSA.
In the pooled safety population [see Adverse Reactions (6.1)], renal impairment occurred in 12% of patients treated with ALECENSA, including Grade ≥ 3 in 1.7% of patients, of which 0.4% were fatal events. The median time to Grade ≥ 3 renal impairment was 3.7 months (range 0.5 to 31.8 months). Dosage modifications for renal impairment were required in 2.4% of patients.
Permanently discontinue ALECENSA for Grade 4 renal toxicity. Withhold ALECENSA for Grade 3 renal toxicity until recovery to less than or equal to 1.5 times ULN, then resume at reduced dose [see Dosage and Administration (2.4)].
5.6 Hemolytic Anemia
Hemolytic anemia occurred in patients treated with ALECENSA.
Hemolytic anemia was initially reported with ALECENSA in the postmarketing setting, including cases associated with a negative direct antiglobulin test (DAT) result. Assessments for the determination of hemolytic anemia were subsequently collected in the ALINA study, where hemolytic anemia was observed in 3.1% of patients treated with ALECENSA. If hemolytic anemia is suspected, withhold ALECENSA and initiate appropriate laboratory testing. If hemolytic anemia is confirmed, consider resuming at a reduced dose upon resolution or permanently discontinue ALECENSA [see Dosage and Administration (2.4)].
8.6 Renal Impairment
No dose adjustment is recommended for patients with mild or moderate renal impairment. The safety of ALECENSA in patients with severe renal impairment (creatinine clearance less than 30 mL/min) or end-stage renal disease has not been studied [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The pharmacokinetics of alectinib and its major active metabolite M4 have been characterized in patients with ALK-positive NSCLC and healthy subjects.
In patients with ALK-positive NSCLC, the geometric mean (coefficient of variation %) steady-state maximal concentration (Cmax,ss) for alectinib was 665 ng/mL (44%) and for M4 was 246 ng/mL (45%) with peak to trough concentration ratio of 1.2. The geometric mean steady-state area under the curve from 0 to 12 hours (AUC0-12h,ss) for alectinib was 7,430 ng*h/mL (46%) and for M4 was 2,810 ng*h/mL (46%). Alectinib exposure is dose proportional across the dose range of 460 mg to 900 mg (i.e., 0.75 to 1.5 times the approved recommended dosage) under fed conditions. Alectinib and M4 reached steady-state concentrations by day 7. The geometric mean accumulation was approximately 6-fold for both alectinib and M4.
2.1 Patient Selection
Select patients with resectable tumors for the adjuvant treatment of NSCLC with ALECENSA based on the presence of ALK positivity in tumor tissue [see Indications and Usage (1.1) and Clinical Studies (14.1)].
Select patients for the treatment of metastatic NSCLC with ALECENSA based on the presence of ALK positivity in tumor tissue or plasma specimens [see Indications and Usage (1.2) and Clinical Studies (14.2)]. If ALK rearrangements are not detected in a plasma specimen, test tumor tissue if feasible.
Information on FDA-approved tests for the detection of ALK rearrangements in NSCLC is available at http://www.fda.gov/CompanionDiagnostics.
8.7 Hepatic Impairment
No dose adjustment is recommended for patients with mild (Child-Pugh A) or moderate (Child-Pugh B) hepatic impairment. Increased exposure of alectinib occurred in patients with severe hepatic impairment (Child-Pugh C). The recommended dose of ALECENSA in patients with severe hepatic impairment (Child-Pugh C) is 450 mg orally twice daily [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
ALECENSA is a kinase inhibitor indicated for:
- adjuvant treatment in adult patients following tumor resection of anaplastic lymphoma kinase (ALK)-positive non-small cell lung cancer (NSCLC) (tumors ≥ 4 cm or node positive) as detected by an FDA-approved test. (1.1)
- treatment of adult patients with ALK-positive metastatic NSCLC as detected by an FDA-approved test. (1.2)
12.1 Mechanism of Action
Alectinib is a tyrosine kinase inhibitor that targets ALK and RET. In nonclinical studies, alectinib inhibited ALK phosphorylation and ALK-mediated activation of the downstream signaling proteins STAT3 and AKT, and decreased tumor cell viability in multiple cell lines harboring ALK fusions, amplifications, or activating mutations. The major active metabolite of alectinib, M4, showed similar in vitro potency and activity.
Alectinib and M4 demonstrated in vitro and in vivo activity against multiple mutant forms of the ALK enzyme, including some mutations identified in NSCLC tumors in patients who have progressed on crizotinib.
In mouse models implanted with tumors carrying ALK fusions, administration of alectinib resulted in antitumor activity and prolonged survival, including in mouse models implanted intracranially with ALK-driven tumor cell lines.
5.7 Embryo Fetal Toxicity (5.7 Embryo-Fetal Toxicity)
Based on findings from animal studies and its mechanism of action, ALECENSA can cause fetal harm when administered to pregnant women. Oral administration of alectinib to pregnant rats and rabbits during the period of organogenesis resulted in embryo-fetal toxicity and abortion at maternally toxic doses with exposures approximately 2.7-fold those observed in humans with alectinib 600 mg twice daily. Advise pregnant women and females of reproductive potential of the potential risk to a fetus.
Advise females of reproductive potential to use effective contraception during treatment with ALECENSA and for 5 weeks following the last dose [see Use in Specific Populations (8.1 and 8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hepatotoxicity: Monitor liver laboratory tests every 2 weeks during the first 3 months of treatment, then once a month and as clinically indicated, with more frequent testing in patients who develop transaminase and bilirubin elevations. In case of severe ALT, AST, or bilirubin elevations, withhold, then reduce dose, or permanently discontinue ALECENSA. (2.4, 5.1)
- Interstitial Lung Disease (ILD)/Pneumonitis: Immediately withhold ALECENSA in patients diagnosed with ILD/pneumonitis and permanently discontinue if no other potential causes of ILD/pneumonitis have been identified. (2.4, 5.2)
- Renal Impairment: Withhold ALECENSA for severe renal impairment, then resume ALECENSA at reduced dose upon recovery or permanently discontinue (2.4, 5.3).
- Bradycardia: Monitor heart rate and blood pressure regularly. If symptomatic, withhold ALECENSA then reduce dose, or permanently discontinue. (2.4, 5.4)
- Severe Myalgia and Creatine Phosphokinase (CPK) Elevation: Assess CPK every 2 weeks during the first month of treatment and in patients reporting unexplained muscle pain, tenderness, or weakness. In case of severe CPK elevations, withhold, then resume or reduce dose. (2.4, 5.5)
- Hemolytic Anemia: If hemolytic anemia is suspected, withhold ALECENSA. If hemolytic anemia is confirmed, consider resuming at a reduced dose upon resolution or permanently discontinue. (5.6)
- Embryo-Fetal Toxicity: ALECENSA can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.7, 8.1, 8.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
600 mg orally twice daily. Administer ALECENSA with food. (2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
150 mg hard capsules, white, with "ALE" printed in black ink on the cap and "150 mg" printed in black ink on the body.
2.2 Dosing and Administration
The recommended dosage information for ALECENSA is provided in Table 1.
| Indication | Recommended Dosage of ALECENSA | Duration |
|---|---|---|
| Adjuvant treatment of resected NSCLC | 600 mg orally twice daily with food [see Clinical Pharmacology (12.3)] |
For a total of 2 years or until disease recurrence or unacceptable toxicity |
| Metastatic NSCLC | Until disease progression or unacceptable toxicity | |
|
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Do not breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The pooled safety population described in the WARNINGS AND PRECAUTIONS reflect exposure to ALECENSA as a single agent at 600 mg orally twice daily in 533 patients in Studies NP28761, NP28673, ALEX and ALINA [see Clinical Studies (14)]. Among 533 patients who received ALECENSA, 75% were exposed for 6 months or longer and 64% were exposed for greater than one year. In this pooled safety population, the most common (≥ 20%) adverse reactions were hepatotoxicity (41%), constipation (39%), fatigue (36%), myalgia (31%), edema (29%), rash (23%) and cough (21%). The most common (≥ 2%) Grade 3 or 4 laboratory abnormalities were increased CPK (6%), decreased hemoglobin (4.4%), increased ALT (4.2%), increased bilirubin (4.0%) and increased AST (3.4%).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Inform patients of the following:
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Hard capsules, white 150 mg capsules with "ALE" printed in black ink on the cap and "150 mg" printed in black ink on the body, available in:
| 240 capsules per bottle: | NDC 50242-130-01 |
2.4 Dose Modifications for Adverse Reactions
The dose reduction schedule for ALECENSA is provided in Table 2.
| Dose Reduction Schedule | Dose Level |
|---|---|
| Starting dose | 600 mg taken orally twice daily |
| First dose reduction | 450 mg taken orally twice daily |
| Second dose reduction | 300 mg taken orally twice daily |
Discontinue if patients are unable to tolerate the 300 mg twice daily dose.
Recommendations for dose modifications of ALECENSA in case of adverse reactions are provided in Table 3.
| Criteria ALT = alanine transaminase; AST = aspartate transaminase; ULN = upper limit of normal; ILD = interstitial lung disease; CPK = blood creatine phosphokinase
|
ALECENSA Dose Modification |
|---|---|
| ALT or AST elevation of greater than 5 times upper limit of normal (ULN) with total bilirubin less than or equal to 2 times ULN | Temporarily withhold until recovery to baseline or to less than or equal to 3 times ULN, then resume at reduced dose as per Table 2. |
| ALT or AST elevation greater than 3 times ULN with total bilirubin elevation greater than 2 times ULN in the absence of cholestasis or hemolysis | Permanently discontinue ALECENSA. |
| Total bilirubin elevation of greater than 3 times ULN | Temporarily withhold until recovery to baseline or to less than or equal to 1.5 times ULN, then resume at reduced dose as per Table 2. |
| Any grade treatment-related interstitial lung disease (ILD)/pneumonitis | Permanently discontinue ALECENSA. |
| Grade 3 renal impairment | Temporarily withhold until serum creatinine recovers to less than or equal to 1.5 times ULN, then resume at reduced dose. |
| Grade 4 renal impairment | Permanently discontinue ALECENSA. |
| Symptomatic bradycardia | Withhold ALECENSA until recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above. If contributing concomitant medication is identified and discontinued, or its dose is adjusted, resume ALECENSA at previous dose upon recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above. If no contributing concomitant medication is identified, or if contributing concomitant medications are not discontinued or dose modified, resume ALECENSA at reduced dose (see Table 2) upon recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above. |
| Bradycardia Heart rate less than 60 beats per minute (bpm) (life-threatening consequences, urgent intervention indicated) |
Permanently discontinue ALECENSA if no contributing concomitant medication is identified. If contributing concomitant medication is identified and discontinued, or its dose is adjusted, resume ALECENSA at reduced dose (see Table 2) upon recovery to asymptomatic bradycardia or to a heart rate of 60 bpm or above, with frequent monitoring as clinically indicated. Permanently discontinue ALECENSA in case of recurrence. |
| CPK elevation greater than 5 times ULN | Temporarily withhold until recovery to baseline or to less than or equal to 2.5 times ULN, then resume at same dose. |
| CPK elevation greater than 10 times ULN or second occurrence of CPK elevation of greater than 5 times ULN | Temporarily withhold until recovery to baseline or to less than or equal to 2.5 times ULN, then resume at reduced dose as per Table 2. |
| Hemolytic Anemia | Withhold ALECENSA if hemolytic anemia is suspected. Upon resolution, resume at reduced dose or permanently discontinue. |
2.3 Recommended Dosage for Hepatic Impairment
The recommended dose of ALECENSA in patients with severe hepatic impairment (Child-Pugh C) is 450 mg orally twice daily [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
1.2 Treatment of Metastatic Alk Positive Nsclc (1.2 Treatment of Metastatic ALK-Positive NSCLC)
ALECENSA is indicated for the treatment of adult patients with ALK-positive metastatic NSCLC as detected by an FDA-approved test [see Dosage & Administration (2.1)].
5.2 Interstitial Lung Disease (ild)/pneumonitis (5.2 Interstitial Lung Disease (ILD)/Pneumonitis)
ILD/pneumonitis occurred in patients treated with ALECENSA.
In the pooled safety population [see Adverse Reactions (6.1)], ILD/pneumonitis occurred in 1.3% of patients treated with ALECENSA with 0.4% of patients experiencing Grade 3 ILD/pneumonitis.
Five patients (0.9%) in the pooled safety population discontinued ALECENSA due to ILD/pneumonitis. The median time-to-onset of Grade 3 or higher ILD/pneumonitis was 2.1 months (range: 0.6 months to 3.6 months).
Promptly investigate for ILD/pneumonitis in any patient who presents with worsening of respiratory symptoms indicative of ILD/pneumonitis (e.g., dyspnea, cough, and fever). Immediately withhold ALECENSA treatment in patients diagnosed with ILD/pneumonitis and permanently discontinue ALECENSA if no other potential causes of ILD/pneumonitis have been identified [see Dosage and Administration (2.4) and Adverse Reactions (6)].
8.3 Females and Males of Reproductive Potential
ALECENSA can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
14.1 Adjuvant Treatment of Resected Alk Positive Nsclc (14.1 Adjuvant Treatment of Resected ALK-Positive NSCLC)
The efficacy of ALECENSA for the adjuvant treatment of patients with ALK-positive NSCLC following complete tumor resection was evaluated in a global, randomized open-label clinical trial (ALINA: NCT03456076). Eligible patients were required to have resectable ALK-positive NSCLC, Stage IB (tumors ≥ 4 cm) – IIIA per the Union for International Cancer Control/American Joint Committee on Cancer (UICC/AJCC) Staging System, 7th Edition. ALK rearrangements were identified by a locally performed FDA-approved ALK test or by a centrally performed VENTANA ALK (D5F3) CDx assay.
Randomization was stratified by race (Asian vs. other races) and stage of disease (IB vs. II vs. IIIA). Patients were randomized (1:1) to receive ALECENSA 600 mg orally twice daily or platinum-based chemotherapy following tumor resection. Treatment with ALECENSA continued for a total of 2 years, or until disease recurrence or unacceptable toxicity. Platinum-based chemotherapy was administered intravenously for 4 cycles, with each cycle lasting 21 days, according to one of the following regimens:
- Cisplatin 75 mg/m2 on Day 1 plus vinorelbine 25 mg/m2 on Days 1 and 8
- Cisplatin 75 mg/m2 on Day 1 plus gemcitabine 1250 mg/m2 on Days 1 and 8
- Cisplatin 75 mg/m2 on Day 1 plus pemetrexed 500 mg/m2 on Day 1
In the event of intolerance to a cisplatin-based regimen, carboplatin was administered instead of cisplatin in the above combinations at a dose of AUC 5 mg/mL/min or 6 mg/mL/min.
The major efficacy outcome measures were disease-free survival (DFS) in patients with stage II-IIIA NSCLC and DFS in patients with stage IB-IIIA NSCLC (intent-to-treat [ITT] population) as assessed by investigator. DFS was defined as the time from date of randomization to the date of occurrence of any of the following: first documented recurrence of disease, new primary NSCLC, or death due to any cause, whichever occurred first. An additional efficacy outcome measure was overall survival (OS) in the ITT population.
A total of 257 patients were randomized to ALECENSA (N=130) or to chemotherapy (N=127). The median age was 56 years (range: 26 to 87), 24% were ≥ 65 years old; 52% were female; 56% were Asian, 42% were White, 0.4% were Black or African American, 2.3% were race unknown; 0.4% were Hispanic or Latino; 60% were never smokers; 53% had an ECOG PS of 0; 10% of patients had Stage IB, 35% had Stage II and 55% had Stage IIIA disease.
ALINA demonstrated a statistically significant improvement in DFS for patients treated with ALECENSA compared to patients treated with chemotherapy. OS data were not mature at the time of DFS analysis with 2.3% of deaths reported in the ITT population.
The efficacy results from ALINA are summarized in Table 10 and Figure 1.
| Efficacy Parameter | Stage II-IIIA Population | ITT Population | ||
|---|---|---|---|---|
| ALECENSA N=116 |
Chemotherapy N=115 |
ALECENSA N=130 |
Chemotherapy N=127 |
|
| DFS = Disease-Free Survival; ITT = Intent-to-Treat; CI = Confidence Interval; NR = Not Reached; NE = Not Estimable. | ||||
| DFS events (%) | 14 (12) | 45 (39) | 15 (12) | 50 (39) |
| Disease recurrence (%) | 14 (12) | 44 (38) | 15 (12) | 49 (38) |
| Death | 0 | 1 (0.9) | 0 | 1 (0.8) |
| Median DFS, months (95% CI) Kaplan-Meier method.
|
NR (NE, NE) |
44.4 (27.8, NE) |
NR (NE, NE) |
41.3 (28.5, NE) |
| Hazard Ratio (95% CI) Stratified Cox model, stratified by race (Asian vs. other races) in Stage II-IIIA and stratified by race (Asian vs. other races) and tumor stage (Stage IB vs. II vs. IIIA) in ITT.
|
0.24 (0.13, 0.45) | 0.24 (0.13, 0.43) | ||
| p-value Stratified log-rank test stratified by race (Asian vs. other races) in Stage II-IIIA and stratified by race (Asian vs. other races) and tumor stage (Stage IB vs. II vs. IIIA) in ITT.
|
<0.0001 | <0.0001 |
Figure 1: Kaplan-Meier Curves of Investigator-Assessed DFS (ITT Population) in ALINA
In an exploratory analysis of site(s) of relapse, the proportion of patients with brain involvement at the time of disease recurrence was 4 patients (3.1%) in the ALECENSA arm and 14 patients (11%) in the chemotherapy arm.
Principal Display Panel 150 Mg Capsule Bottle Carton (PRINCIPAL DISPLAY PANEL - 150 mg Capsule Bottle Carton)
NDC 50242-130-01
Alecensa®
(alectinib)
capsules
150 mg
Each capsule contains 150 mg of alectinib
(equivalent to 161.33 mg alectinib HCl).
Rx only
240 capsules
Genentech
11008914
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with alectinib have not been conducted.
Alectinib was not mutagenic in vitro in the bacterial reverse mutation (Ames) assay, but was positive with an increased number of micronuclei in a rat bone marrow micronucleus test. The mechanism of micronucleus induction was abnormal chromosome segregation (aneugenicity) and not a clastogenic effect on chromosomes.
No studies in animals have been performed to evaluate the effect of alectinib on fertility. No adverse effects on male and female reproductive organs were observed in general toxicology studies conducted in rats and monkeys.
5.5 Severe Myalgia and Creatine Phosphokinase (cpk) Elevation (5.5 Severe Myalgia and Creatine Phosphokinase (CPK) Elevation)
Severe myalgia and creatine phosphokinase (CPK) elevation occurred in patients treated with ALECENSA.
In the pooled safety population [see Adverse Reactions (6.1)], myalgia (including muscle- and musculoskeletal-related reactions) occurred in 31% of patients treated with ALECENSA, including Grade ≥ 3 in 0.8% of patients. Dosage modifications for myalgia events were required in 2.1% of patients.
In the pooled safety population, of the 491 patients with CPK laboratory data available, elevated CPK occurred in 56% of patients treated with ALECENSA, including 6% Grade ≥ 3. The median time to Grade ≥ 3 CPK elevation was 15 days (interquartile range - 15 –337 days). Dosage modifications for elevation of CPK occurred in 5% of patients.
In the ALINA study, elevated CPK occurred in 77% of 128 patients with CPK laboratory data, including 6% Grade ≥ 3 elevations.
Advise patients to report any unexplained muscle pain, tenderness, or weakness. Assess CPK levels every 2 weeks for the first month of treatment and as clinically indicated in patients reporting symptoms. Based on the severity of the CPK elevation, withhold ALECENSA, then resume or reduce dose [see Dosage and Administration (2.4)].
1.1 Adjuvant Treatment of Resected Alk Positive Non Small Cell Lung Cancer (nsclc) (1.1 Adjuvant Treatment of Resected ALK-Positive Non-Small Cell Lung Cancer (NSCLC))
ALECENSA is indicated as adjuvant treatment in adult patients following tumor resection of anaplastic lymphoma kinase (ALK)-positive non-small cell lung cancer (NSCLC) (tumors ≥ 4 cm or node positive), as detected by an FDA-approved test [see Dosage & Administration (2.1)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:15.418659 · Updated: 2026-03-14T22:39:42.922945