42af8bf8-80b4-e1a1-e063-6394a90a306c
42af8bf8-80b4-e1a1-e063-6394a90a306c
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
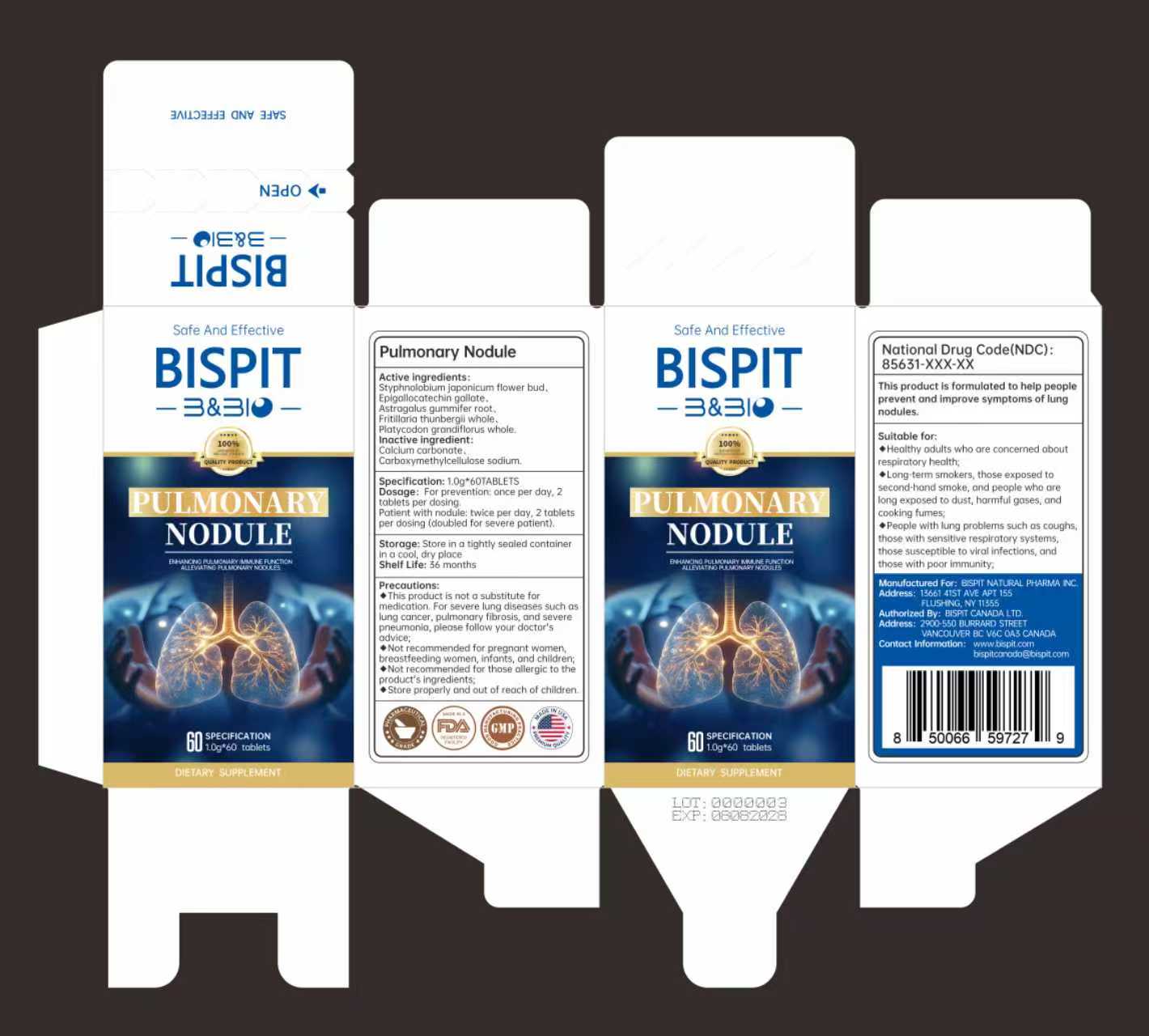
STYPHNOLOBIUM JAPONICUM FLOWER BUD EPIGALLOCATECHIN GALLATE ASTRAGALUS GUMMIFER ROOT FRITILLARIA THUNBERGII WHOLE PLATYCODON GRANDIFLORUS WHOLE
Medication Information
Warnings and Precautions
This product is not a substitute formedication.For severe lung diseases such aslung cancer, pulmonary fibrosis, and severepneumonia, please follow your doctor'sadvice:Not recommended for pregnant women,breastfeeding women, infants, and children;Not recommended for those allergic to theproduct's ingredients;
Active Ingredient
STYPHNOLOBIUM JAPONICUM FLOWER BUD
EPIGALLOCATECHIN GALLATE
ASTRAGALUS GUMMIFER ROOT
FRITILLARIA THUNBERGII WHOLE
PLATYCODON GRANDIFLORUS WHOLE
Indications and Usage
Suitable for:◆Healthy adults who are concerned aboutrespiratory health;◆Long-term smokers, those exposed tosecond-hand smoke, and people who arelong exposed to dust, harmful gases, andcooking fumes;People with lung problems such as coughsthose with sensitive respiratory systemsthose susceptible to viral infections, andthose with poor immunity
Dosage and Administration
Dosage: For prevention: once per day,2tablets per dosing.Patient with nodule: twice per day, 2 tabletsper dosing (doubled for severe patient).
Description
STYPHNOLOBIUM JAPONICUM FLOWER BUD EPIGALLOCATECHIN GALLATE ASTRAGALUS GUMMIFER ROOT FRITILLARIA THUNBERGII WHOLE PLATYCODON GRANDIFLORUS WHOLE
Section 50565-1
Store properly and out of reach of children.
Section 51727-6
CALCIUM CARBONATE
CARBOXYMETHYLCELLULOSE SODIUM
Section 51945-4
Section 55105-1
This product is formulated to help peopleprevent and improve symptoms of lungnodules.
Structured Label Content
Warnings and Precautions (34071-1)
This product is not a substitute formedication.For severe lung diseases such aslung cancer, pulmonary fibrosis, and severepneumonia, please follow your doctor'sadvice:Not recommended for pregnant women,breastfeeding women, infants, and children;Not recommended for those allergic to theproduct's ingredients;
Active Ingredient (55106-9)
STYPHNOLOBIUM JAPONICUM FLOWER BUD
EPIGALLOCATECHIN GALLATE
ASTRAGALUS GUMMIFER ROOT
FRITILLARIA THUNBERGII WHOLE
PLATYCODON GRANDIFLORUS WHOLE
Indications and Usage (34067-9)
Suitable for:◆Healthy adults who are concerned aboutrespiratory health;◆Long-term smokers, those exposed tosecond-hand smoke, and people who arelong exposed to dust, harmful gases, andcooking fumes;People with lung problems such as coughsthose with sensitive respiratory systemsthose susceptible to viral infections, andthose with poor immunity
Dosage and Administration (34068-7)
Dosage: For prevention: once per day,2tablets per dosing.Patient with nodule: twice per day, 2 tabletsper dosing (doubled for severe patient).
Section 50565-1 (50565-1)
Store properly and out of reach of children.
Section 51727-6 (51727-6)
CALCIUM CARBONATE
CARBOXYMETHYLCELLULOSE SODIUM
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
This product is formulated to help peopleprevent and improve symptoms of lungnodules.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:12:14.015418 · Updated: 2026-03-14T23:17:40.306527