42634fc7-d0c7-4813-e063-6294a90a702d
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
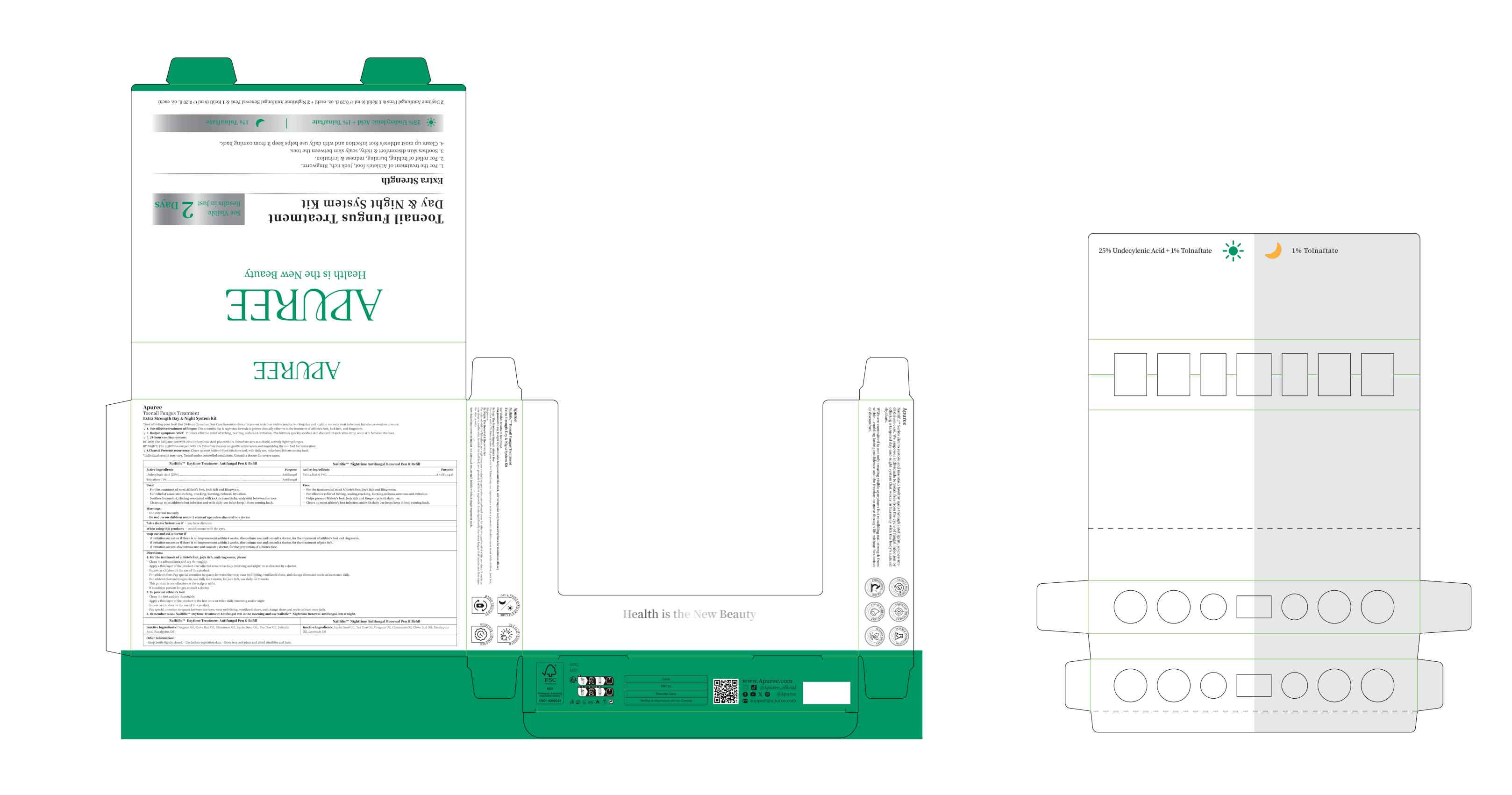
TOLNAFTATE
Medication Information
Warnings and Precautions
warnings section
Indications and Usage
Apply to the affected area twice daily.
Dosage and Administration
6ml 0.23fl
Description
TOLNAFTATE
Section 50565-1
keep out of reach of children section
Section 51727-6
JOJOBA SEED OIL,TEA TREE OIL,OREGANO OIL,CINNAMON OIL,CLOVE BUD OIL,EUCALYPTUS OIL,
LAVENDER OIL
Section 51945-4
Section 55105-1
Treatment for nail and skin fungal infections
Section 55106-9
TOLNAFTATE
Structured Label Content
Indications and Usage (34067-9)
Apply to the affected area twice daily.
Dosage and Administration (34068-7)
6ml 0.23fl
Warnings and Precautions (34071-1)
warnings section
Section 50565-1 (50565-1)
keep out of reach of children section
Section 51727-6 (51727-6)
JOJOBA SEED OIL,TEA TREE OIL,OREGANO OIL,CINNAMON OIL,CLOVE BUD OIL,EUCALYPTUS OIL,
LAVENDER OIL
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Treatment for nail and skin fungal infections
Section 55106-9 (55106-9)
TOLNAFTATE
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:26.386986 · Updated: 2026-03-14T23:12:39.597935