These Highlights Do Not Include All The Information Needed To Use Pomalidomide Capsules Safely And Effectively. See Full Prescribing Information For Pomalidomide Capsules.
41400b90-b64e-447b-90f3-1cad58050eba
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: EMBRYO-FETAL TOXICITY and VENOUS AND ARTERIAL THROMBOEMBOLISM See full prescribing information for complete boxed warning EMBRYO-FETAL TOXICITY Pomalidomide capsules are contraindicated in pregnancy. Pomalidomide capsules are a thalidomide analogue. Thalidomide is a known human teratogen that causes severe life-threatening birth defects ( 4 , 5.1 , 8.1 ). For females of reproductive potential: Exclude pregnancy before start of treatment. Prevent pregnancy during treatment by the use of 2 reliable methods of contraception ( 5.1 , 8.3 ). Pomalidomide capsules are available only through a restricted program called PS-Pomalidomide REMS ( 5.2 ). VENOUS AND ARTERIAL THROMBOEMBOLISM Deep venous thrombosis (DVT), pulmonary embolism (PE), myocardial infarction, and stroke occur in patients with multiple myeloma treated with pomalidomide capsules. Antithrombotic prophylaxis is recommended ( 5.3 ).
Indications and Usage
Pomalidomide capsules are a thalidomide analogue indicated for the treatment of adult patients: in combination with dexamethasone, for patients with multiple myeloma (MM) who have received at least two prior therapies including lenalidomide and a proteasome inhibitor and have demonstrated disease progression on or within 60 days of completion of the last therapy ( 1.1 ). with AIDS-related Kaposi sarcoma (KS) after failure of highly active antiretroviral therapy (HAART) or in patients with KS who are HIV-negative. This indication is approved under accelerated approval based on overall response rate. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s) ( 1.2 ).
Dosage and Administration
MM: 4 mg per day taken orally on Days 1 through 21 of repeated 28-day cycles until disease progression ( 2.2 ). Refer to section 14.1 for dexamethasone dosing ( 14.1 ) KS: 5 mg per day taken orally on Days 1 through 21 of repeated 28-day cycles until disease progression or unacceptable toxicity ( 2.3 ). Modify the dosage for certain patients with renal impairment ( 2.7 , 8.6 ) or hepatic impairment ( 2.8 , 8.7 ).
Warnings and Precautions
Increased Mortality: Observed in patients with MM when pembrolizumab was added to dexamethasone and a thalidomide analogue ( 5.4 ). Hematologic Toxicity: Neutropenia was the most frequently reported Grade 3/4 adverse event. Monitor patients for hematologic toxicities, especially neutropenia ( 5.5 ). Hepatotoxicity: Hepatic failure including fatalities; monitor liver function tests monthly ( 5.6 ). Severe Cutaneous Reactions: Discontinue pomalidomide capsules for severe reactions ( 5.7 ). Tumor Lysis Syndrome (TLS): Monitor patients at risk of TLS (i.e., those with high tumor burden) and take appropriate precautions ( 5.11 ). Hypersensitivity: Monitor patients for potential hypersensitivity. Discontinue pomalidomide capsules for angioedema and anaphylaxis ( 5.12 ).
Contraindications
Pregnancy ( 4.1 ) Hypersensitivity ( 4.2 )
Adverse Reactions
The following clinically significant adverse reactions are described in detail in other labeling sections: Embryo-Fetal Toxicity [see Warnings and Precautions (5.1 , 5.2) ] Venous and Arterial Thromboembolism [see Warnings and Precautions (5.3) ] Increased Mortality in Patients with Multiple Myeloma When Pembrolizumab Is Added to a Thalidomide Analogue and Dexamethasone [see Warnings and Precautions (5.4) ] Hematologic Toxicity [see Warnings and Precautions (5.5) ] Hepatotoxicity [see Warnings and Precautions (5.6) ] Severe Cutaneous Reactions [see Warnings and Precautions (5.7) ] Dizziness and Confusional State [see Warnings and Precautions (5.8) ] Neuropathy [see Warnings and Precautions (5.9) ] Risk of Second Primary Malignancies [see Warnings and Precautions (5.10) ] Tumor Lysis Syndrome [see Warnings and Precautions (5.11) ] Hypersensitivity [see Warnings and Precautions (5.12) ]
Drug Interactions
Strong CYP1A2 Inhibitors: Avoid concomitant use of strong CYP1A2 inhibitors. If concommitant use of a strong CYP1A2 inhibitor is unavoidable, reduce pomalidomide capsules dose to 2 mg ( 2.6 , 7.1 , 12.3 ).
Storage and Handling
Dark blue cap and yellow body printed with "NAT'' with white ink on cap and "1mg" with black ink on body of the capsule 1 mg bottles of 21 (NDC 51991-342-21) 1 mg bottles of 100 (NDC 51991-342-01) Dark blue cap and orange body printed with "NAT" with white ink on cap and "2mg" with white ink on body of the capsule 2 mg bottles of 21 (NDC 51991-343-21) 2 mg bottles of 100 (NDC 51991-343-01) Dark blue cap and green body printed with "NAT" with white ink on cap and "3mg" with white ink on body of the capsule 3 mg bottles of 21 (NDC 51991-344-21) 3 mg bottles of 100 (NDC 51991-344-01) Dark blue cap and blue body printed with "NAT" with white ink on cap and "4mg" with white ink on body of the capsule 4 mg bottles of 21 (NDC 51991-346-21) 4 mg bottles of 100 (NDC 51991-346-01)
How Supplied
Dark blue cap and yellow body printed with "NAT'' with white ink on cap and "1mg" with black ink on body of the capsule 1 mg bottles of 21 (NDC 51991-342-21) 1 mg bottles of 100 (NDC 51991-342-01) Dark blue cap and orange body printed with "NAT" with white ink on cap and "2mg" with white ink on body of the capsule 2 mg bottles of 21 (NDC 51991-343-21) 2 mg bottles of 100 (NDC 51991-343-01) Dark blue cap and green body printed with "NAT" with white ink on cap and "3mg" with white ink on body of the capsule 3 mg bottles of 21 (NDC 51991-344-21) 3 mg bottles of 100 (NDC 51991-344-01) Dark blue cap and blue body printed with "NAT" with white ink on cap and "4mg" with white ink on body of the capsule 4 mg bottles of 21 (NDC 51991-346-21) 4 mg bottles of 100 (NDC 51991-346-01)
Medication Information
Warnings and Precautions
Increased Mortality: Observed in patients with MM when pembrolizumab was added to dexamethasone and a thalidomide analogue ( 5.4 ). Hematologic Toxicity: Neutropenia was the most frequently reported Grade 3/4 adverse event. Monitor patients for hematologic toxicities, especially neutropenia ( 5.5 ). Hepatotoxicity: Hepatic failure including fatalities; monitor liver function tests monthly ( 5.6 ). Severe Cutaneous Reactions: Discontinue pomalidomide capsules for severe reactions ( 5.7 ). Tumor Lysis Syndrome (TLS): Monitor patients at risk of TLS (i.e., those with high tumor burden) and take appropriate precautions ( 5.11 ). Hypersensitivity: Monitor patients for potential hypersensitivity. Discontinue pomalidomide capsules for angioedema and anaphylaxis ( 5.12 ).
Indications and Usage
Pomalidomide capsules are a thalidomide analogue indicated for the treatment of adult patients: in combination with dexamethasone, for patients with multiple myeloma (MM) who have received at least two prior therapies including lenalidomide and a proteasome inhibitor and have demonstrated disease progression on or within 60 days of completion of the last therapy ( 1.1 ). with AIDS-related Kaposi sarcoma (KS) after failure of highly active antiretroviral therapy (HAART) or in patients with KS who are HIV-negative. This indication is approved under accelerated approval based on overall response rate. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s) ( 1.2 ).
Dosage and Administration
MM: 4 mg per day taken orally on Days 1 through 21 of repeated 28-day cycles until disease progression ( 2.2 ). Refer to section 14.1 for dexamethasone dosing ( 14.1 ) KS: 5 mg per day taken orally on Days 1 through 21 of repeated 28-day cycles until disease progression or unacceptable toxicity ( 2.3 ). Modify the dosage for certain patients with renal impairment ( 2.7 , 8.6 ) or hepatic impairment ( 2.8 , 8.7 ).
Contraindications
Pregnancy ( 4.1 ) Hypersensitivity ( 4.2 )
Adverse Reactions
The following clinically significant adverse reactions are described in detail in other labeling sections: Embryo-Fetal Toxicity [see Warnings and Precautions (5.1 , 5.2) ] Venous and Arterial Thromboembolism [see Warnings and Precautions (5.3) ] Increased Mortality in Patients with Multiple Myeloma When Pembrolizumab Is Added to a Thalidomide Analogue and Dexamethasone [see Warnings and Precautions (5.4) ] Hematologic Toxicity [see Warnings and Precautions (5.5) ] Hepatotoxicity [see Warnings and Precautions (5.6) ] Severe Cutaneous Reactions [see Warnings and Precautions (5.7) ] Dizziness and Confusional State [see Warnings and Precautions (5.8) ] Neuropathy [see Warnings and Precautions (5.9) ] Risk of Second Primary Malignancies [see Warnings and Precautions (5.10) ] Tumor Lysis Syndrome [see Warnings and Precautions (5.11) ] Hypersensitivity [see Warnings and Precautions (5.12) ]
Drug Interactions
Strong CYP1A2 Inhibitors: Avoid concomitant use of strong CYP1A2 inhibitors. If concommitant use of a strong CYP1A2 inhibitor is unavoidable, reduce pomalidomide capsules dose to 2 mg ( 2.6 , 7.1 , 12.3 ).
Storage and Handling
Dark blue cap and yellow body printed with "NAT'' with white ink on cap and "1mg" with black ink on body of the capsule 1 mg bottles of 21 (NDC 51991-342-21) 1 mg bottles of 100 (NDC 51991-342-01) Dark blue cap and orange body printed with "NAT" with white ink on cap and "2mg" with white ink on body of the capsule 2 mg bottles of 21 (NDC 51991-343-21) 2 mg bottles of 100 (NDC 51991-343-01) Dark blue cap and green body printed with "NAT" with white ink on cap and "3mg" with white ink on body of the capsule 3 mg bottles of 21 (NDC 51991-344-21) 3 mg bottles of 100 (NDC 51991-344-01) Dark blue cap and blue body printed with "NAT" with white ink on cap and "4mg" with white ink on body of the capsule 4 mg bottles of 21 (NDC 51991-346-21) 4 mg bottles of 100 (NDC 51991-346-01)
How Supplied
Dark blue cap and yellow body printed with "NAT'' with white ink on cap and "1mg" with black ink on body of the capsule 1 mg bottles of 21 (NDC 51991-342-21) 1 mg bottles of 100 (NDC 51991-342-01) Dark blue cap and orange body printed with "NAT" with white ink on cap and "2mg" with white ink on body of the capsule 2 mg bottles of 21 (NDC 51991-343-21) 2 mg bottles of 100 (NDC 51991-343-01) Dark blue cap and green body printed with "NAT" with white ink on cap and "3mg" with white ink on body of the capsule 3 mg bottles of 21 (NDC 51991-344-21) 3 mg bottles of 100 (NDC 51991-344-01) Dark blue cap and blue body printed with "NAT" with white ink on cap and "4mg" with white ink on body of the capsule 4 mg bottles of 21 (NDC 51991-346-21) 4 mg bottles of 100 (NDC 51991-346-01)
Description
WARNING: EMBRYO-FETAL TOXICITY and VENOUS AND ARTERIAL THROMBOEMBOLISM See full prescribing information for complete boxed warning EMBRYO-FETAL TOXICITY Pomalidomide capsules are contraindicated in pregnancy. Pomalidomide capsules are a thalidomide analogue. Thalidomide is a known human teratogen that causes severe life-threatening birth defects ( 4 , 5.1 , 8.1 ). For females of reproductive potential: Exclude pregnancy before start of treatment. Prevent pregnancy during treatment by the use of 2 reliable methods of contraception ( 5.1 , 8.3 ). Pomalidomide capsules are available only through a restricted program called PS-Pomalidomide REMS ( 5.2 ). VENOUS AND ARTERIAL THROMBOEMBOLISM Deep venous thrombosis (DVT), pulmonary embolism (PE), myocardial infarction, and stroke occur in patients with multiple myeloma treated with pomalidomide capsules. Antithrombotic prophylaxis is recommended ( 5.3 ).
Section 42229-5
Embryo-Fetal Toxicity
- Pomalidomide capsules are contraindicated in pregnancy. Pomalidomide capsules are a thalidomide analogue. Thalidomide is a known human teratogen that causes severe birth defects or embryo-fetal death. In females of reproductive potential, obtain 2 negative pregnancy tests before starting pomalidomide capsules treatment.
- Females of reproductive potential must use 2 forms of contraception or continuously abstain from heterosexual sex during and for 4 weeks after stopping pomalidomide capsules treatment [see Contraindications (4), Warnings and Precautions (5.1) and Use in Specific Populations (8.1, 8.3)].
Pomalidomide capsules are only available through a restricted distribution program called PS-Pomalidomide REMS [see Warnings and Precautions (5.2)]. Information about PS-Pomalidomide REMS is available at www.PS-PomalidomideREMS.com or by calling the REMS Call Center at 1-888-423-5436.
Section 43683-2
| Boxed Warning | 2/2025 |
| Warnings and Precautions (5.1, 5.2) | 2/2025 |
Section 44425-7
Store at 20°C-25°C (68°F-77°F); excursions permitted to 15°C-30°C (59°F-86°F). [see USP Controlled Room Temperature].
Care should be exercised in handling of pomalidomide capsules. Do not open or crush pomalidomide capsules. If powder from pomalidomide capsules contacts the skin, wash the skin immediately and thoroughly with soap and water. If pomalidomide capsules contacts the mucous membranes, flush thoroughly with water.
Follow procedures for proper handling and disposal of hazardous drugs.1
10 Overdosage
Hemodialysis can remove pomalidomide from circulation.
15 References
- OSHA Hazardous Drugs. OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
4.1 Pregnancy
Pomalidomide capsules are contraindicated in females who are pregnant. Pomalidomide capsules can cause fetal harm when administered to a pregnant female [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)]. Pomalidomide is a thalidomide analogue and is teratogenic in both rats and rabbits when administered during the period of organogenesis. If the patient becomes pregnant while taking this drug, the patient should be apprised of the potential risk to a fetus.
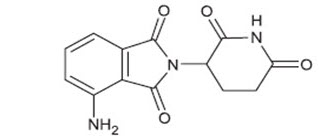
11 Description
Pomalidomide is a thalidomide analog. The chemical name is (RS)-4-Amino-2-(2,6-dioxo-piperidin-3-yl)-isoindoline-1,3-dione and it has the following chemical structure:
The empirical formula for pomalidomide is C13H11N3O4 and the gram molecular weight is 273.25.
Pomalidomide is a light yellow to yellow colored powder. It has limited to low solubility into organic solvents and it has low solubility in all pH solutions (about 0.01 mg/mL). Pomalidomide has a chiral carbon atom which exists as a racemic mixture of the R(+) and S(−) enantiomers.
Pomalidomide capsules are available in 1-mg, 2-mg, 3-mg, and 4-mg capsules for oral administration. Each capsule contains pomalidomide as the active ingredient and the following inactive ingredients: mannitol, pregelatinized starch, croscarmellose sodium and sodium stearyl fumarate.
The 1-mg capsule shell contains gelatin, titanium dioxide, FD&C blue 2, yellow iron oxide, white ink and Black ink. The 2-mg capsule shell contains gelatin, FD&C Blue #2, Titanium Dioxide, Iron Oxide Yellow, FD&C Red #3, and white ink. The 3-mg capsule shell contains gelatin, FD&C Blue #2, Titanium Dioxide, Iron Oxide Yellow, and white ink. The 4-mg capsule shell contains gelatin, FD&C Blue #2, Titanium Dioxide, and white ink.
Black ink contains Shellac, Dehydrated alcohol, Isopropyl alcohol, Butyl alcohol, Propylene Glycol, Strong Ammonia Solution, Black Iron Oxide, Potassium hydroxide, and Purified water.
White ink contains Shellac, Ethanol, Isopropyl alcohol, n-Butanol, Propylene Glycol, Ammonia Solution, Purified Water, Potassium Hydroxide, and Titanium Dioxide.
5.9 Neuropathy
In trials 1 and 2 in patients who received pomalidomide capsules + Low-dose Dex, 18% of patients experienced neuropathy, with approximately 12% of the patients experiencing peripheral neuropathy. Two percent of patients experienced Grade 3 neuropathy in trial 2. There were no cases of Grade 4 neuropathy adverse reactions reported in either trial.
Medication Guide
Pomalidomide (poe˝-ma-lid'-oh-mide) capsules
What is the most important information I should know about pomalidomide capsules?
Before you begin taking pomalidomide capsules, you must read and agree to all of the instructions in PS-Pomalidomide REMS. For more information, call 1-888-423-5436 or go to www.PS-PomalidomideREMS.com. Before prescribing pomalidomide capsules, your healthcare provider will explain PS-Pomalidomide REMS to you and have you sign the Patient-Physician Agreement Form.
Pomalidomide capsules can cause serious side effects including:
-
Possible birth defects (deformed babies) or death of an unborn baby. Females who are pregnant or who plan to become pregnant must not take pomalidomide capsules.
Pomalidomide capsules are similar to the medicine thalidomide (THALOMID). We know thalidomide can cause severe life- threatening birth defects. Pomalidomide capsules have not been tested in pregnant females. Pomalidomide capsules have harmed unborn animals in animal testing.
Females must not get pregnant:- For at least 4 weeks before starting pomalidomide capsules
- While taking pomalidomide capsules
- During any breaks (interruptions) in your treatment with pomalidomide capsules
- For at least 4 weeks after stopping pomalidomide capsules
- Will have pregnancy tests weekly for 4 weeks, then every 4 weeks if your menstrual cycle is regular, or every 2 weeks if your menstrual cycle is irregular.
If you miss your period or have unusual bleeding, you will need to have a pregnancy test and receive counseling. - Must agree to use two acceptable forms of birth control at the same time, for at least 4 weeks before, while taking, during any breaks (interruptions) in your treatment, and for at least 4 weeks after stopping pomalidomide capsules.
- Talk with your healthcare provider to find out about options for acceptable forms of birth control that you may use to prevent pregnancy before, during, and after treatment with pomalidomide capsules.
If your healthcare provider is not available, you can call the REMS Call Center at 1-888-423-5436. Healthcare providers and patients should report all cases of pregnancy to:- FDA MedWatch at 1-800-FDA-1088, and
- REMS Call Center at 1-888-423-5436
Pomalidomide can pass into human semen:- Males, including those who have had a vasectomy, must always use a latex or synthetic condom during any sexual contact with a pregnant female or a female that can become pregnant while taking pomalidomide capsules, during any breaks (interruptions) in your treatment with pomalidomide capsules, and for 4 weeks after stopping pomalidomide capsules.
- Do not have unprotected sexual contact with a female who is or could become pregnant. Tell your healthcare provider if you do have unprotected sexual contact with a female who is or could become pregnant.
Men, if your female partner becomes pregnant, you should call your healthcare provider right away. -
Blood clots in your arteries, veins, and lungs, heart attack, and stroke can happen if you take pomalidomide capsules.
Most people who take pomalidomide capsules will also take a blood thinner medicine to help prevent blood clots. Before taking pomalidomide capsules, tell your healthcare provider:- If you have had a blood clot in the past
- If you have high blood pressure, smoke, or if you have been told you have a high level of fat in your blood (hyperlipidemia)
- About all the medicines you take. Certain other medicines can also increase your risk for blood clots
Call your healthcare provider or get medical help right away if you get any of the following during treatment with pomalidomide capsules:- Signs or symptoms of a blood clot in the lung, arm, or leg may include: shortness of breath, chest pain, or arm or leg swelling
- Signs or symptoms of a heart attack may include: chest pain that may spread to the arms, neck, jaw, back, or stomach area (abdomen), feeling sweaty, shortness of breath, feeling sick or vomiting
- Signs or symptoms of stroke may include: sudden numbness or weakness, especially on one side of the body, severe headache or confusion, or problems with vision, speech, or balance.
What are pomalidomide capsules?
Pomalidomide capsules are a prescription medicine used to treat adults with:
-
Multiple myeloma. Pomalidomide capsules are taken along with the medicine dexamethasone, in people who:
- have received at least 2 prior medicines to treat multiple myeloma, including a type of medicine known as a proteasome inhibitor and lenalidomide, and
- their disease has become worse during treatment or within 60 days of finishing the last treatment.
- AIDS-related Kaposi sarcoma (KS). Pomalidomide capsules are taken when highly active antiretroviral therapy (HAART) has not worked well enough or stopped working (failed).
- KS who do not have HIV infection (HIV negative).
It is not known if pomalidomide capsules are safe and effective in children.
Who should not take pomalidomide capsules?
Do not take pomalidomide capsules if you:
- are pregnant, plan to become pregnant, or become pregnant during treatment with pomalidomide capsules. See "What is the most important information I should know about pomalidomide capsules?"
are allergic to pomalidomide or any of the ingredients in pomalidomide capsules. See the end of this Medication Guide for a complete list of ingredients in pomalidomide capsules.
What should I tell my healthcare provider before taking pomalidomide capsules?
Before you take pomalidomide capsules, tell your healthcare provider if you:
- smoke cigarettes. Pomalidomide capsules may not work as well in people who smoke
- have liver problems
- have kidney problems and are receiving hemodialysis treatment
- have any other medical conditions
- are breastfeeding. You should not breastfeed during treatment with pomalidomide capsules. It is not known if pomalidomide passes into your breast milk and can harm your baby.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Pomalidomide capsules and other medicines may affect each other, causing serious side effects. Talk with your healthcare provider before taking any new medicines.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist.
How should I take pomalidomide capsules?
- Take pomalidomide capsules exactly as prescribed and follow all the instructions of PS-Pomalidomide REMS.
- Swallow pomalidomide capsules whole with water 1 time a day. Do not break, chew, or open your capsules.
- Pomalidomide capsules may be taken with or without food.
- Take pomalidomide capsules at about the same time each day.
- If you are on hemodialysis, take pomalidomide capsules after hemodialysis, on hemodialysis days.
- Do not open the pomalidomide capsules or handle them any more than needed. If you touch a broken pomalidomide capsule or the medicine in the capsule, wash the area of your body right away with soap and water.
- If you miss a dose of pomalidomide capsules and it has been less than 12 hours since your regular time, take it as soon as you remember. If it has been more than 12 hours, just skip your missed dose. Do not take 2 doses at the same time.
If you take too much pomalidomide capsules, call your healthcare provider right away.
What should I avoid while taking pomalidomide capsules?
- See "What is the most important information I should know about pomalidomide capsules?"
- Females: Do not get pregnant and do not breastfeed while taking pomalidomide capsules.
- Males: Do not donate sperm.
- Do not share pomalidomide capsules with other people. It may cause birth defects and other serious problems.
- Do not donate blood while you take pomalidomide capsules, during any breaks (interruptions) in your treatment, and for 4 weeks after stopping pomalidomide capsules. If someone who is pregnant gets your donated blood, her baby may be exposed to pomalidomide capsules and may be born with birth defects.
- Pomalidomide capsules can cause dizziness and confusion. Avoid taking other medicines that may cause dizziness and confusion during treatment with pomalidomide capsules. Avoid situations that require you to be alert until you know how pomalidomide capsules affect you.
What are the possible side effects of pomalidomide capsules?
Pomalidomide capsules can cause serious side effects, including:
- See "What is the most important information I should know about pomalidomide capsules?"
- Low white blood cells (neutropenia), low platelets (thrombocytopenia), and low red blood cells (anemia) are common with pomalidomide capsules, but can also be serious. You may need a blood transfusion or certain medicines if your blood counts drop too low. Your blood counts should be checked weekly for the first 8 weeks of treatment and monthly after that.
-
Severe liver problems, including liver failure and death. Your healthcare provider should do blood tests to check your liver function during your treatment with pomalidomide capsules. Tell your healthcare provider right away if you develop any of the following symptoms of liver problems:
- Yellowing of your skin or the white part of your eyes (jaundice)
- Dark or brown (tea-colored) urine
- Pain on the upper right side of your stomach area (abdomen)
- Bleeding or bruising more easily than normal
- Feeling very tired
-
Severe allergic reactions and severe skin reactions can happen with pomalidomide capsules and may cause death.
Call your healthcare provider if you develop any of the following signs or symptoms during treatment with pomalidomide capsules:- a red, itchy, skin rash
- peeling of your skin or blisters
- severe itching
- fever
Get emergency medical help right away if you develop any of the following signs or symptoms during treatment with pomalidomide capsules:
- swelling of your lips, mouth, tongue, or throat
- trouble breathing or swallowing
- raised red areas on your skin (hives)
- a very fast heartbeat
- you feel dizzy or faint
- Dizziness and confusion. See "What should I avoid while taking pomalidomide capsules?"
- Nerve damage. Stop taking pomalidomide capsules and call your healthcare provider if you develop symptoms of nerve damage including: numbness, tingling, pain, burning sensation in your hands, legs, or feet.
- Risk of new cancers (malignancies). New cancers, including certain blood cancers (acute myelogenous leukemia or AML) have been seen in people who received pomalidomide capsules. Talk with your healthcare provider about your risk of developing new cancers if you take pomalidomide capsules.
Tumor lysis syndrome (TLS). TLS is caused by the fast breakdown of cancer cells. TLS can cause kidney failure and the need for dialysis treatment, abnormal heart rhythm, seizure, and sometimes death. Your healthcare provider may do blood tests to check you for TLS.
Your healthcare provider may tell you to stop taking pomalidomide capsules if you develop certain serious side effects during treatment.
The most common side effects of pomalidomide capsules in people with Multiple Myeloma include:
|
|
|
The most common side effects of pomalidomide capsules in people with KS include:
|
|
These are not all the possible side effects of pomalidomide capsules.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store pomalidomide capsules?
- Store pomalidomide capsules at room temperature between 68°F to 77°F (20°C to 25°C).
- Return any unused pomalidomide capsules to PS-Pomalidomide REMS by calling 1-888-423-5436 or your healthcare provider.
Keep pomalidomide capsules and all medicines out of the reach of children.
General information about the safe and effective use of pomalidomide capsules.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not take pomalidomide capsules for conditions for which it was not prescribed. Do not give pomalidomide capsules to other people, even if they have the same symptoms you have. It may harm them and may cause birth defects.
If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about pomalidomide capsules that is written for health professionals.
For more information, call 1-888-423-5436 or go to www.PS-PomalidomideREMS.com.
What are the ingredients in pomalidomide capsules?
Active ingredient: pomalidomide
Inactive ingredients: mannitol, pregelatinized starch, croscarmellose sodium and sodium stearyl fumarate.
The 1-mg capsule shell contains Gelatin, titanium dioxide, FD&C blue 2, yellow iron oxide, white ink and Black ink. The 2-mg capsule shell contains Gelatin, FD&C Blue #2, Titanium Dioxide, Iron Oxide Yellow, FD&C Red #3, and white ink. The 3-mg capsule shell contains Gelatin, FD&C Blue #2, Titanium Dioxide, Iron Oxide Yellow, and white ink. The 4-mg capsule shell contains Gelatin, FD&C Blue #2, Titanium Dioxide, and white ink.
Black ink contains Shellac, Dehydrated alcohol, Isopropyl alcohol, Butyl alcohol, Propylene Glycol, Strong Ammonia Solution, Black Iron Oxide, Potassium hydroxide, and Purified water.
White ink contains Shellac, Ethanol, Isopropyl alcohol, n-Butanol, Propylene Glycol, Ammonia Solution, Purified Water, Potassium Hydroxide, and Titanium Dioxide.
Manufactured by:
Natco Pharma Limited
Parawada – 531019, India.
Distributed by:
Breckenridge Pharmaceutical, Inc.
Berkeley Heights, NJ 07922
Trademarks are the property of their respective owners.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Revised: 08/2025
8.4 Pediatric Use
The safety and effectiveness of pomalidomide capsules have not been established in pediatric patients. The safety and effectiveness were assessed but not established in two open-label studies: a dose escalation study in 25 pediatric patients aged 5 to <17 with recurrent, progressive or refractory CNS tumors [NCT02415153] and a parallel-group study conducted in 47 pediatric patients aged 4 to <17 years with recurrent or progressive high-grade glioma, medulloblastoma, ependymoma, or diffuse intrinsic pontine glioma (DIPG) [NCT03257631]. No new safety signals were observed in pediatric patients across these studies.
At the same dose by body surface area, pomalidomide exposure in 55 pediatric patients aged 4 to < 17 years old was within the range observed in adult patients with MM but higher than the exposure observed in adults with KS [see Clinical Pharmacology (12.3)].
1.2 Kaposi Sarcoma
Pomalidomide capsules are indicated for the treatment of:
- Adult patients with AIDS-related Kaposi sarcoma (KS) after failure of highly active antiretroviral therapy (HAART).
- Kaposi sarcoma (KS) in adult patients who are HIV-negative.
This indication is approved under accelerated approval based on overall response rate [see Clinical Studies (14.2)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s).
2.9 Administration
Swallow capsules whole with water. Do not break, chew, or open the capsules.
Pomalidomide capsules may be taken with or without food.
5.6 Hepatotoxicity
Hepatic failure, including fatal cases, has occurred in patients treated with pomalidomide capsules. Elevated levels of alanine aminotransferase and bilirubin have also been observed in patients treated with pomalidomide capsules. Monitor liver function tests monthly. Stop pomalidomide capsules upon elevation of liver enzymes and evaluate. After return to baseline values, treatment at a lower dose may be considered.
14.2 Kaposi Sarcoma
The clinical trial 12-C-0047 (NCT01495598), was an open label, single center, single arm clinical study that evaluated the safety and efficacy of pomalidomide capsules in patients with Kaposi sarcoma (KS). A total of 28 patients (18 HIV-positive, 10 HIV-negative) received pomalidomide capsules 5 mg orally once daily on Days 1 through 21 of each 28-day cycle until disease progression or unacceptable toxicity. All HIV-positive patients continued highly active antiretroviral therapy (HAART). The trial excluded patients with symptomatic pulmonary or visceral KS, history of venous or arterial thromboembolism, or procoagulant disorders. Patients received thromboprophylaxis with aspirin 81 mg once daily throughout therapy.
The median age was 52.5 years, all were male, 75% were White, and 14% Black or African American. Seventy- five percent of patients had advanced disease (T1) at the time of enrollment, 11% had ≥ 50 lesions, and 75% had received prior chemotherapy.
The major efficacy outcome measure was overall response rate (ORR), which included complete response (CR), clinical complete response (cCR), and partial response (PR). Response was assessed by the investigator according to the AIDS Clinical Trial Group (ACTG) Oncology Committee response criteria for KS. The median time to first response was 1.8 months (0.9 to 7.6). Efficacy results are presented in Table 11.
| All Patients N=28 |
HIV-Positive N=18 |
HIV-Negative N=10 |
|
|---|---|---|---|
| CI: confidence interval, ORR: overall response rate, CR: complete response, PR: partial response | |||
| ORR CR includes one HIV-negative patient who achieved a cCR. , n (%) |
20 (71) | 12 (67) | 8 (80) |
| [95% CI] | [51, 87] | (41, 87) | (44, 98) |
| CR, n (%) | 4 (14) | 3 (17) | 1 (10) |
| PR, n (%) | 16 (57) | 9 (50) | 7 (70) |
| Duration of Response, KS Calculated as date of first documented response to date of first documented disease progression, receipt of new treatment or second course of treatment, or death due to any cause, whichever occurs first. Median estimate is from Kaplan-Meier analysis. , |
12.1 | 12.5 | 10.5 |
| Median in months [95% CI] From Kaplan-Meier analysis.
|
[7.6, 16.8] | [6.5, 24.9] | [3.9, 24.2] |
| Duration of Response, KS (%) | |||
| Percent greater than 12 months | 50 | 58 | 38 |
| Percent greater than 24 months | 20 | 17 | 25 |
6 Adverse Reactions
The following clinically significant adverse reactions are described in detail in other labeling sections:
- Embryo-Fetal Toxicity [see Warnings and Precautions (5.1, 5.2)]
- Venous and Arterial Thromboembolism [see Warnings and Precautions (5.3)]
- Increased Mortality in Patients with Multiple Myeloma When Pembrolizumab Is Added to a Thalidomide Analogue and Dexamethasone [see Warnings and Precautions (5.4)]
- Hematologic Toxicity [see Warnings and Precautions (5.5)]
- Hepatotoxicity [see Warnings and Precautions (5.6)]
- Severe Cutaneous Reactions [see Warnings and Precautions (5.7)]
- Dizziness and Confusional State [see Warnings and Precautions (5.8)]
- Neuropathy [see Warnings and Precautions (5.9)]
- Risk of Second Primary Malignancies [see Warnings and Precautions (5.10)]
- Tumor Lysis Syndrome [see Warnings and Precautions (5.11)]
- Hypersensitivity [see Warnings and Precautions (5.12)]
7 Drug Interactions
8.8 Smoking Tobacco
Cigarette smoking reduces pomalidomide AUC due to CYP1A2 induction. Advise patients that smoking may reduce the efficacy of pomalidomide [see Clinical Pharmacology (12.3)].
1.1 Multiple Myeloma
Pomalidomide capsules, in combination with dexamethasone, are indicated for adult patients with multiple myeloma (MM) who have received at least two prior therapies including lenalidomide and a proteasome inhibitor and have demonstrated disease progression on or within 60 days of completion of the last therapy.
4.2 Hypersensitivity
Pomalidomide Capsules are contraindicated in patients who have demonstrated severe hypersensitivity (e.g., angioedema, anaphylaxis) to pomalidomide or any of the excipients [see Warnings and Precautions (5.7), Description (11)].
8.6 Renal Impairment
In patients with severe renal impairment requiring dialysis, the AUC of pomalidomide increased by 38% and the rate of SAE increased by 64% relative to patients with normal renal function; therefore, starting dose adjustment is recommended. For patients with severe renal impairment requiring dialysis, administer pomalidomide capsules after the completion of hemodialysis on dialysis days because exposure of pomalidomide could be significantly decreased during dialysis [see Dosage and Administration (2.7) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Pomalidomide exposure-response analyses showed that there was no relationship between systemic pomalidomide exposure level and efficacy or safety following pomalidomide dose of 4 mg.
12.3 Pharmacokinetics
In patients with MM who received pomalidomide capsules 4 mg daily alone or in combination with dexamethasone, pomalidomide steady-state drug exposure was characterized by AUC (CV%) of 860 (37%) ng∙h/mL and Cmax (CV%) of 75 (32%) ng/mL. In patients with Kaposi sarcoma (KS) who received pomalidomide capsules 5 mg daily, pomalidomide steady-state drug exposure was characterized by AUC of 462.3 ng∙h/mL (82%) and Cmax of 53.1 ng/mL (50%).
5.12 Hypersensitivity
Hypersensitivity, including angioedema, anaphylaxis, and anaphylactic reactions to pomalidomide capsules have been reported. Permanently discontinue pomalidomide capsules for angioedema or anaphylaxis [see Dosage and Administration (2.5)].
8.7 Hepatic Impairment
Pomalidomide is metabolized primarily by the liver. Following single dose administration, the AUC of pomalidomide increased 51%, 58%, and 72% in subjects with mild (Child-Pugh class A), moderate (Child-Pugh class B), and severe (Child-Pugh class C) hepatic impairment, respectively compared to subjects with normal liver function. Dose adjustment is recommended in patients with hepatic impairment [see Dosage and Administration (2.8) and Clinical Pharmacology (12.3)].
1 Indications and Usage
Pomalidomide capsules are a thalidomide analogue indicated for the treatment of adult patients:
- in combination with dexamethasone, for patients with multiple myeloma (MM) who have received at least two prior therapies including lenalidomide and a proteasome inhibitor and have demonstrated disease progression on or within 60 days of completion of the last therapy (1.1).
- with AIDS-related Kaposi sarcoma (KS) after failure of highly active antiretroviral therapy (HAART) or in patients with KS who are HIV-negative. This indication is approved under accelerated approval based on overall response rate. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s) (1.2).
12.1 Mechanism of Action
Pomalidomide is an analogue of thalidomide with immunomodulatory, antiangiogenic, and antineoplastic properties. Cellular activities of pomalidomide are mediated through its target cereblon, a component of a cullin ring E3 ubiquitin ligase enzyme complex. In vitro, in the presence of drug, substrate proteins (including Aiolos and Ikaros) are targeted for ubiquitination and subsequent degradation leading to direct cytotoxic and immunomodulatory effects. In in vitro cellular assays, pomalidomide inhibited proliferation and induced apoptosis of hematopoietic tumor cells. Additionally, pomalidomide inhibited the proliferation of lenalidomide-resistant multiple myeloma (MM) cell lines and synergized with dexamethasone in both lenalidomide-sensitive and lenalidomide-resistant cell lines to induce tumor cell apoptosis. Pomalidomide enhanced T cell- and natural killer (NK) cell-mediated immunity and inhibited production of pro-inflammatory cytokines (e.g., TNF-α and IL-6) by monocytes. Pomalidomide demonstrated anti-angiogenic activity in a mouse tumor model and in the in vitro umbilical cord model.
5.2 Ps Pomalidomide Rems
Because of the embryo-fetal risk [see Warnings and Precautions (5.1)], pomalidomide capsules are available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS), "PS-Pomalidomide REMS".
Required components of PS-Pomalidomide REMS include the following:
- Prescribers must be certified with PS-Pomalidomide REMS by enrolling and complying with the REMS requirements.
- Patients must sign a Patient-Physician Agreement Form and comply with the REMS requirements. In particular, female patients of reproductive potential who are not pregnant must comply with the pregnancy testing and contraception requirements [see Use in Specific Populations (8.3)] and males must comply with contraception requirements [see Use in Specific Populations (8.3)].
- Pharmacies must be certified with PS-Pomalidomide REMS, must only dispense to patients who are authorized to receive pomalidomide capsules and comply with REMS requirements.
Further information about PS-Pomalidomide REMS is available at www.PS-PomalidomideREMS.com or by telephone at 1-888-423-5436.
5.1 Embryo Fetal Toxicity
Pomalidomide capsules are a thalidomide analogue and are contraindicated for use during pregnancy. Thalidomide is a known human teratogen that causes severe birth defects or embryo-fetal death [see Use in Specific Populations (8.1)]. Pomalidomide capsules are only available through PS-Pomalidomide REMS [see Warnings and Precautions (5.2)].
5.11 Tumor Lysis Syndrome
Tumor lysis syndrome (TLS) may occur in patients treated with pomalidomide. Patients at risk for TLS are those with high tumor burden prior to treatment. These patients should be monitored closely and appropriate precautions taken.
5 Warnings and Precautions
- Increased Mortality: Observed in patients with MM when pembrolizumab was added to dexamethasone and a thalidomide analogue (5.4).
- Hematologic Toxicity: Neutropenia was the most frequently reported Grade 3/4 adverse event. Monitor patients for hematologic toxicities, especially neutropenia (5.5).
- Hepatotoxicity: Hepatic failure including fatalities; monitor liver function tests monthly (5.6).
- Severe Cutaneous Reactions: Discontinue pomalidomide capsules for severe reactions (5.7).
- Tumor Lysis Syndrome (TLS): Monitor patients at risk of TLS (i.e., those with high tumor burden) and take appropriate precautions (5.11).
- Hypersensitivity: Monitor patients for potential hypersensitivity. Discontinue pomalidomide capsules for angioedema and anaphylaxis (5.12).
2 Dosage and Administration
- MM: 4 mg per day taken orally on Days 1 through 21 of repeated 28-day cycles until disease progression (2.2). Refer to section 14.1 for dexamethasone dosing (14.1)
- KS: 5 mg per day taken orally on Days 1 through 21 of repeated 28-day cycles until disease progression or unacceptable toxicity (2.3).
- Modify the dosage for certain patients with renal impairment (2.7, 8.6) or hepatic impairment (2.8, 8.7).
3 Dosage Forms and Strengths
- Capsules: 1 mg, dark blue cap and yellow body printed with "NAT'' with white ink on cap and "1 mg" with black ink on body of the capsule
- 2 mg, dark blue cap and orange body printed with "NAT" with white ink on cap and "2 mg" with white ink on body of the capsule
- 3 mg, dark blue cap and green body printed with "NAT" with white ink on cap and "3 mg" with white ink on body of the capsule
- 4 mg, dark blue cap and blue body printed with "NAT" with white ink on cap and "4 mg" with white ink on body of the capsule
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of pomalidomide capsules. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders : Pancytopenia
Endocrine Disorders: Hypothyroidism, hyperthyroidism
Gastrointestinal Disorders : Gastrointestinal hemorrhage
Hepatobiliary Disorders : Hepatic failure (including fatal cases), elevated liver enzymes
Immune System Disorders : Allergic reactions (e.g., angioedema, anaphylaxis, urticaria), solid organ transplant rejection
Infections and Infestations : Hepatitis B virus reactivation, Herpes zoster, progressive multifocal leukoencephalopathy (PML)
Neoplasms benign, malignant and unspecified (incl cysts and polyps): Tumor lysis syndrome, basal cell carcinoma, and squamous cell carcinoma of the skin
Skin and Subcutaneous Tissue Disorders : Stevens-Johnson Syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms (DRESS)
8 Use in Specific Populations
- Lactation: Advise women not to breastfeed (8.2).
5.7 Severe Cutaneous Reactions
Severe cutaneous reactions including Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS) have been reported. DRESS may present with a cutaneous reaction (such as rash or exfoliative dermatitis), eosinophilia, fever, and/or lymphadenopathy with systemic complications such as hepatitis, nephritis, pneumonitis, myocarditis, and/or pericarditis. These reactions can be fatal. Consider pomalidomide capsules interruption or discontinuation for Grade 2 or 3 skin rash. Permanently discontinue pomalidomide capsules for Grade 4 rash, exfoliative or bullous rash, or for other severe cutaneous reactions such as SJS, TEN or DRESS [see Dosage and Administration (2.5)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.8 Dizziness and Confusional State
In trials 1 and 2 in patients who received pomalidomide capsules + Low-dose Dex, 14% of patients experienced dizziness and 7% of patients experienced a confusional state; 1% of patients experienced Grade 3 or 4 dizziness, and 3% of patients experienced Grade 3 or 4 confusional state. Instruct patients to avoid situations where dizziness or confusional state may be a problem and not to take other medications that may cause dizziness or confusional state without adequate medical advice.
16 How Supplied/storage and Handling
Dark blue cap and yellow body printed with "NAT'' with white ink on cap and "1mg" with black ink on body of the capsule
1 mg bottles of 21 (NDC 51991-342-21)
1 mg bottles of 100 (NDC 51991-342-01)
Dark blue cap and orange body printed with "NAT" with white ink on cap and "2mg" with white ink on body of the capsule
2 mg bottles of 21 (NDC 51991-343-21)
2 mg bottles of 100 (NDC 51991-343-01)
Dark blue cap and green body printed with "NAT" with white ink on cap and "3mg" with white ink on body of the capsule
3 mg bottles of 21 (NDC 51991-344-21)
3 mg bottles of 100 (NDC 51991-344-01)
Dark blue cap and blue body printed with "NAT" with white ink on cap and "4mg" with white ink on body of the capsule
4 mg bottles of 21 (NDC 51991-346-21)
4 mg bottles of 100 (NDC 51991-346-01)
5.3 Venous and Arterial Thromboembolism
Venous thromboembolic events (deep venous thrombosis and pulmonary embolism) and arterial thromboembolic events (myocardial infarction and stroke) have been observed in patients treated with pomalidomide capsules. In Trial 2, where anticoagulant therapies were mandated, thromboembolic events occurred in 8.0% of patients treated with pomalidomide capsules and low dose-dexamethasone (Low-dose Dex), and 3.3% of patients treated with high-dose dexamethasone. Venous thromboembolic events (VTE) occurred in 4.7% of patients treated with pomalidomide capsules and Low-dose Dex, and 1.3% of patients treated with high-dose dexamethasone. Arterial thromboembolic events include terms for arterial thromboembolic events, ischemic cerebrovascular conditions, and ischemic heart disease. Arterial thromboembolic events occurred in 3.0% of patients treated with pomalidomide capsules and Low-dose Dex, and 1.3% of patients treated with high-dose dexamethasone.
Patients with known risk factors, including prior thrombosis, may be at greater risk, and actions should be taken to try to minimize all modifiable factors (e.g., hyperlipidemia, hypertension, smoking). Thromboprophylaxis is recommended, and the choice of regimen should be based on assessment of the patient's underlying risk factors.
5.10 Risk of Second Primary Malignancies
Cases of acute myelogenous leukemia have been reported in patients receiving pomalidomide capsules as an investigational therapy outside of MM.
2.3 Recommended Dosage for Kaposi Sarcoma
The recommended dosage of pomalidomide capsules is 5 mg once daily taken orally with or without food on Days 1 through 21 of each 28-day cycle until disease progression or unacceptable toxicity. Continue HAART as HIV treatment in patients with AIDS-related Kaposi sarcoma (KS) [see Clinical Studies (14.2)].
2.2 Recommended Dosage for Multiple Myeloma
The recommended dosage of pomalidomide capsules is 4 mg once daily orally with or without food on Days 1 through 21 of each 28-day cycle until disease progression. Give pomalidomide capsules in combination with dexamethasone [see Clinical Studies (14.1)].
2.1 Pregnancy Testing Prior to Administration
Females of reproductive potential must have negative pregnancy testing and use contraception methods before initiating pomalidomide capsules [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1, 8.3)].
Principal Display Panel 1 Mg Capsule Bottle Label
NDC 51991-342-21
Pomalidomide
Capsules
1 mg
WARNING: POTENTIAL FOR
HUMAN BIRTH DEFECTS
PHARMACIST: Dispense the enclosed
Medication Guide to each patient.
21 Capsules
Rx Only
breckenridge
A Towa
Company
Principal Display Panel 2 Mg Capsule Bottle Label
NDC 51991-343-21
Pomalidomide
Capsules
2 mg
WARNING: POTENTIAL FOR
HUMAN BIRTH DEFECTS
PHARMACIST: Dispense the enclosed
Medication Guide to each patient.
21 Capsules
Rx Only
breckenridge
A Towa
Company
Principal Display Panel 3 Mg Capsule Bottle Label
NDC 51991-344-21
Pomalidomide
Capsules
3 mg
WARNING: POTENTIAL FOR
HUMAN BIRTH DEFECTS
PHARMACIST: Dispense the enclosed
Medication Guide to each patient.
21 Capsules
Rx Only
breckenridge
A Towa
Company
Principal Display Panel 4 Mg Capsule Bottle Label
NDC 51991-346-21
Pomalidomide
Capsules
4 mg
WARNING: POTENTIAL FOR
HUMAN BIRTH DEFECTS
PHARMACIST: Dispense the enclosed
Medication Guide to each patient.
21 Capsules
Rx Only
breckenridge
A Towa
Company
2.6 Dosage Modifications for Strong Cyp1a2 Inhibitors
Avoid concomitant use of pomalidomide capsules with strong CYP1A2 inhibitors. If concomitant use of a strong CYP1A2 inhibitor is unavoidable, reduce pomalidomide capsules dose to 2 mg [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies examining the carcinogenic potential of pomalidomide have not been conducted. One of 12 monkeys dosed with 1 mg/kg of pomalidomide (an exposure approximately 15-fold of the exposure in patients at the recommended dose of 4 mg/day) developed acute myeloid leukemia in a 9-month repeat-dose toxicology study.
Pomalidomide was not mutagenic or clastogenic in a battery of tests, including the bacteria reverse mutation assay (Ames test), the in vitro assay using human peripheral blood lymphocytes, and the micronucleus test in orally treated rats administered doses up to 2000 mg/kg/day.
In a fertility and early embryonic development study in rats, drug-treated males were mated with untreated or treated females.
Pomalidomide was administered to males and females at doses of 25 to 1000 mg/kg/day. When treated males were mated with treated females, there was an increase in post-implantation loss and a decrease in mean number of viable embryos at all dose levels. There were no other effects on reproductive functions or the number of pregnancies. The lowest dose tested in animals resulted in an exposure (AUC) approximately 100-fold of the exposure in patients at the recommended dose of 4 mg/day. When treated males in this study were mated with untreated females, all uterine parameters were comparable to the controls. Based on these results, the observed effects were attributed to the treatment of females.
2.5 Dosage Modifications for Non Hematologic Adverse Reactions
Permanently discontinue pomalidomide capsules for angioedema, anaphylaxis, Grade 4 rash, skin exfoliation, bullae, or any other severe dermatologic reaction [See Warnings and Precautions (5.7, 5.12)].
For other Grade 3 or 4 toxicities, hold treatment and restart treatment at 1 mg less than the previous dose when toxicity has resolved to less than or equal to Grade 2 at the physician's discretion.
2.7 Dosage Modification for Severe Renal Impairment On Hemodialysis
Take pomalidomide capsules after completion of dialysis procedure on hemodialysis days [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
- For patients with MM with severe renal impairment requiring dialysis, reduce the recommended dosage to 3 mg orally daily.
- For patients with KS with severe renal impairment requiring dialysis, reduce the recommended dosage to 4 mg orally daily.
Warning: Embryo Fetal Toxicity and Venous and Arterial Thromboembolism
WARNING: EMBRYO-FETAL TOXICITY and VENOUS AND ARTERIAL THROMBOEMBOLISM
See full prescribing information for complete boxed warning
EMBRYO-FETAL TOXICITY
- Pomalidomide capsules are contraindicated in pregnancy. Pomalidomide capsules are a thalidomide analogue. Thalidomide is a known human teratogen that causes severe life-threatening birth defects (4, 5.1, 8.1).
- For females of reproductive potential: Exclude pregnancy before start of treatment. Prevent pregnancy during treatment by the use of 2 reliable methods of contraception (5.1, 8.3).
Pomalidomide capsules are available only through a restricted program called PS-Pomalidomide REMS (5.2).
VENOUS AND ARTERIAL THROMBOEMBOLISM
- Deep venous thrombosis (DVT), pulmonary embolism (PE), myocardial infarction, and stroke occur in patients with multiple myeloma treated with pomalidomide capsules. Antithrombotic prophylaxis is recommended (5.3).
5.4 Increased Mortality in Patients With Multiple Myeloma When Pembrolizumab Is Added to A Thalidomide Analogue and Dexamethasone
In two randomized clinical trials in patients with MM, the addition of pembrolizumab to a thalidomide analogue plus dexamethasone, a use for which no PD-1 or PD-L1 blocking antibody is indicated, resulted in increased mortality. Treatment of patients with MM with a PD-1 or PD-L1 blocking antibody in combination with a thalidomide analogue plus dexamethasone is not recommended outside of controlled clinical trials.
Structured Label Content
Section 42229-5 (42229-5)
Embryo-Fetal Toxicity
- Pomalidomide capsules are contraindicated in pregnancy. Pomalidomide capsules are a thalidomide analogue. Thalidomide is a known human teratogen that causes severe birth defects or embryo-fetal death. In females of reproductive potential, obtain 2 negative pregnancy tests before starting pomalidomide capsules treatment.
- Females of reproductive potential must use 2 forms of contraception or continuously abstain from heterosexual sex during and for 4 weeks after stopping pomalidomide capsules treatment [see Contraindications (4), Warnings and Precautions (5.1) and Use in Specific Populations (8.1, 8.3)].
Pomalidomide capsules are only available through a restricted distribution program called PS-Pomalidomide REMS [see Warnings and Precautions (5.2)]. Information about PS-Pomalidomide REMS is available at www.PS-PomalidomideREMS.com or by calling the REMS Call Center at 1-888-423-5436.
Section 43683-2 (43683-2)
| Boxed Warning | 2/2025 |
| Warnings and Precautions (5.1, 5.2) | 2/2025 |
Section 44425-7 (44425-7)
Store at 20°C-25°C (68°F-77°F); excursions permitted to 15°C-30°C (59°F-86°F). [see USP Controlled Room Temperature].
Care should be exercised in handling of pomalidomide capsules. Do not open or crush pomalidomide capsules. If powder from pomalidomide capsules contacts the skin, wash the skin immediately and thoroughly with soap and water. If pomalidomide capsules contacts the mucous membranes, flush thoroughly with water.
Follow procedures for proper handling and disposal of hazardous drugs.1
10 Overdosage (10 OVERDOSAGE)
Hemodialysis can remove pomalidomide from circulation.
15 References (15 REFERENCES)
- OSHA Hazardous Drugs. OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
4.1 Pregnancy
Pomalidomide capsules are contraindicated in females who are pregnant. Pomalidomide capsules can cause fetal harm when administered to a pregnant female [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)]. Pomalidomide is a thalidomide analogue and is teratogenic in both rats and rabbits when administered during the period of organogenesis. If the patient becomes pregnant while taking this drug, the patient should be apprised of the potential risk to a fetus.
11 Description (11 DESCRIPTION)
Pomalidomide is a thalidomide analog. The chemical name is (RS)-4-Amino-2-(2,6-dioxo-piperidin-3-yl)-isoindoline-1,3-dione and it has the following chemical structure:
The empirical formula for pomalidomide is C13H11N3O4 and the gram molecular weight is 273.25.
Pomalidomide is a light yellow to yellow colored powder. It has limited to low solubility into organic solvents and it has low solubility in all pH solutions (about 0.01 mg/mL). Pomalidomide has a chiral carbon atom which exists as a racemic mixture of the R(+) and S(−) enantiomers.
Pomalidomide capsules are available in 1-mg, 2-mg, 3-mg, and 4-mg capsules for oral administration. Each capsule contains pomalidomide as the active ingredient and the following inactive ingredients: mannitol, pregelatinized starch, croscarmellose sodium and sodium stearyl fumarate.
The 1-mg capsule shell contains gelatin, titanium dioxide, FD&C blue 2, yellow iron oxide, white ink and Black ink. The 2-mg capsule shell contains gelatin, FD&C Blue #2, Titanium Dioxide, Iron Oxide Yellow, FD&C Red #3, and white ink. The 3-mg capsule shell contains gelatin, FD&C Blue #2, Titanium Dioxide, Iron Oxide Yellow, and white ink. The 4-mg capsule shell contains gelatin, FD&C Blue #2, Titanium Dioxide, and white ink.
Black ink contains Shellac, Dehydrated alcohol, Isopropyl alcohol, Butyl alcohol, Propylene Glycol, Strong Ammonia Solution, Black Iron Oxide, Potassium hydroxide, and Purified water.
White ink contains Shellac, Ethanol, Isopropyl alcohol, n-Butanol, Propylene Glycol, Ammonia Solution, Purified Water, Potassium Hydroxide, and Titanium Dioxide.
5.9 Neuropathy
In trials 1 and 2 in patients who received pomalidomide capsules + Low-dose Dex, 18% of patients experienced neuropathy, with approximately 12% of the patients experiencing peripheral neuropathy. Two percent of patients experienced Grade 3 neuropathy in trial 2. There were no cases of Grade 4 neuropathy adverse reactions reported in either trial.
Medication Guide (MEDICATION GUIDE)
Pomalidomide (poe˝-ma-lid'-oh-mide) capsules
What is the most important information I should know about pomalidomide capsules?
Before you begin taking pomalidomide capsules, you must read and agree to all of the instructions in PS-Pomalidomide REMS. For more information, call 1-888-423-5436 or go to www.PS-PomalidomideREMS.com. Before prescribing pomalidomide capsules, your healthcare provider will explain PS-Pomalidomide REMS to you and have you sign the Patient-Physician Agreement Form.
Pomalidomide capsules can cause serious side effects including:
-
Possible birth defects (deformed babies) or death of an unborn baby. Females who are pregnant or who plan to become pregnant must not take pomalidomide capsules.
Pomalidomide capsules are similar to the medicine thalidomide (THALOMID). We know thalidomide can cause severe life- threatening birth defects. Pomalidomide capsules have not been tested in pregnant females. Pomalidomide capsules have harmed unborn animals in animal testing.
Females must not get pregnant:- For at least 4 weeks before starting pomalidomide capsules
- While taking pomalidomide capsules
- During any breaks (interruptions) in your treatment with pomalidomide capsules
- For at least 4 weeks after stopping pomalidomide capsules
- Will have pregnancy tests weekly for 4 weeks, then every 4 weeks if your menstrual cycle is regular, or every 2 weeks if your menstrual cycle is irregular.
If you miss your period or have unusual bleeding, you will need to have a pregnancy test and receive counseling. - Must agree to use two acceptable forms of birth control at the same time, for at least 4 weeks before, while taking, during any breaks (interruptions) in your treatment, and for at least 4 weeks after stopping pomalidomide capsules.
- Talk with your healthcare provider to find out about options for acceptable forms of birth control that you may use to prevent pregnancy before, during, and after treatment with pomalidomide capsules.
If your healthcare provider is not available, you can call the REMS Call Center at 1-888-423-5436. Healthcare providers and patients should report all cases of pregnancy to:- FDA MedWatch at 1-800-FDA-1088, and
- REMS Call Center at 1-888-423-5436
Pomalidomide can pass into human semen:- Males, including those who have had a vasectomy, must always use a latex or synthetic condom during any sexual contact with a pregnant female or a female that can become pregnant while taking pomalidomide capsules, during any breaks (interruptions) in your treatment with pomalidomide capsules, and for 4 weeks after stopping pomalidomide capsules.
- Do not have unprotected sexual contact with a female who is or could become pregnant. Tell your healthcare provider if you do have unprotected sexual contact with a female who is or could become pregnant.
Men, if your female partner becomes pregnant, you should call your healthcare provider right away. -
Blood clots in your arteries, veins, and lungs, heart attack, and stroke can happen if you take pomalidomide capsules.
Most people who take pomalidomide capsules will also take a blood thinner medicine to help prevent blood clots. Before taking pomalidomide capsules, tell your healthcare provider:- If you have had a blood clot in the past
- If you have high blood pressure, smoke, or if you have been told you have a high level of fat in your blood (hyperlipidemia)
- About all the medicines you take. Certain other medicines can also increase your risk for blood clots
Call your healthcare provider or get medical help right away if you get any of the following during treatment with pomalidomide capsules:- Signs or symptoms of a blood clot in the lung, arm, or leg may include: shortness of breath, chest pain, or arm or leg swelling
- Signs or symptoms of a heart attack may include: chest pain that may spread to the arms, neck, jaw, back, or stomach area (abdomen), feeling sweaty, shortness of breath, feeling sick or vomiting
- Signs or symptoms of stroke may include: sudden numbness or weakness, especially on one side of the body, severe headache or confusion, or problems with vision, speech, or balance.
What are pomalidomide capsules?
Pomalidomide capsules are a prescription medicine used to treat adults with:
-
Multiple myeloma. Pomalidomide capsules are taken along with the medicine dexamethasone, in people who:
- have received at least 2 prior medicines to treat multiple myeloma, including a type of medicine known as a proteasome inhibitor and lenalidomide, and
- their disease has become worse during treatment or within 60 days of finishing the last treatment.
- AIDS-related Kaposi sarcoma (KS). Pomalidomide capsules are taken when highly active antiretroviral therapy (HAART) has not worked well enough or stopped working (failed).
- KS who do not have HIV infection (HIV negative).
It is not known if pomalidomide capsules are safe and effective in children.
Who should not take pomalidomide capsules?
Do not take pomalidomide capsules if you:
- are pregnant, plan to become pregnant, or become pregnant during treatment with pomalidomide capsules. See "What is the most important information I should know about pomalidomide capsules?"
are allergic to pomalidomide or any of the ingredients in pomalidomide capsules. See the end of this Medication Guide for a complete list of ingredients in pomalidomide capsules.
What should I tell my healthcare provider before taking pomalidomide capsules?
Before you take pomalidomide capsules, tell your healthcare provider if you:
- smoke cigarettes. Pomalidomide capsules may not work as well in people who smoke
- have liver problems
- have kidney problems and are receiving hemodialysis treatment
- have any other medical conditions
- are breastfeeding. You should not breastfeed during treatment with pomalidomide capsules. It is not known if pomalidomide passes into your breast milk and can harm your baby.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Pomalidomide capsules and other medicines may affect each other, causing serious side effects. Talk with your healthcare provider before taking any new medicines.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist.
How should I take pomalidomide capsules?
- Take pomalidomide capsules exactly as prescribed and follow all the instructions of PS-Pomalidomide REMS.
- Swallow pomalidomide capsules whole with water 1 time a day. Do not break, chew, or open your capsules.
- Pomalidomide capsules may be taken with or without food.
- Take pomalidomide capsules at about the same time each day.
- If you are on hemodialysis, take pomalidomide capsules after hemodialysis, on hemodialysis days.
- Do not open the pomalidomide capsules or handle them any more than needed. If you touch a broken pomalidomide capsule or the medicine in the capsule, wash the area of your body right away with soap and water.
- If you miss a dose of pomalidomide capsules and it has been less than 12 hours since your regular time, take it as soon as you remember. If it has been more than 12 hours, just skip your missed dose. Do not take 2 doses at the same time.
If you take too much pomalidomide capsules, call your healthcare provider right away.
What should I avoid while taking pomalidomide capsules?
- See "What is the most important information I should know about pomalidomide capsules?"
- Females: Do not get pregnant and do not breastfeed while taking pomalidomide capsules.
- Males: Do not donate sperm.
- Do not share pomalidomide capsules with other people. It may cause birth defects and other serious problems.
- Do not donate blood while you take pomalidomide capsules, during any breaks (interruptions) in your treatment, and for 4 weeks after stopping pomalidomide capsules. If someone who is pregnant gets your donated blood, her baby may be exposed to pomalidomide capsules and may be born with birth defects.
- Pomalidomide capsules can cause dizziness and confusion. Avoid taking other medicines that may cause dizziness and confusion during treatment with pomalidomide capsules. Avoid situations that require you to be alert until you know how pomalidomide capsules affect you.
What are the possible side effects of pomalidomide capsules?
Pomalidomide capsules can cause serious side effects, including:
- See "What is the most important information I should know about pomalidomide capsules?"
- Low white blood cells (neutropenia), low platelets (thrombocytopenia), and low red blood cells (anemia) are common with pomalidomide capsules, but can also be serious. You may need a blood transfusion or certain medicines if your blood counts drop too low. Your blood counts should be checked weekly for the first 8 weeks of treatment and monthly after that.
-
Severe liver problems, including liver failure and death. Your healthcare provider should do blood tests to check your liver function during your treatment with pomalidomide capsules. Tell your healthcare provider right away if you develop any of the following symptoms of liver problems:
- Yellowing of your skin or the white part of your eyes (jaundice)
- Dark or brown (tea-colored) urine
- Pain on the upper right side of your stomach area (abdomen)
- Bleeding or bruising more easily than normal
- Feeling very tired
-
Severe allergic reactions and severe skin reactions can happen with pomalidomide capsules and may cause death.
Call your healthcare provider if you develop any of the following signs or symptoms during treatment with pomalidomide capsules:- a red, itchy, skin rash
- peeling of your skin or blisters
- severe itching
- fever
Get emergency medical help right away if you develop any of the following signs or symptoms during treatment with pomalidomide capsules:
- swelling of your lips, mouth, tongue, or throat
- trouble breathing or swallowing
- raised red areas on your skin (hives)
- a very fast heartbeat
- you feel dizzy or faint
- Dizziness and confusion. See "What should I avoid while taking pomalidomide capsules?"
- Nerve damage. Stop taking pomalidomide capsules and call your healthcare provider if you develop symptoms of nerve damage including: numbness, tingling, pain, burning sensation in your hands, legs, or feet.
- Risk of new cancers (malignancies). New cancers, including certain blood cancers (acute myelogenous leukemia or AML) have been seen in people who received pomalidomide capsules. Talk with your healthcare provider about your risk of developing new cancers if you take pomalidomide capsules.
Tumor lysis syndrome (TLS). TLS is caused by the fast breakdown of cancer cells. TLS can cause kidney failure and the need for dialysis treatment, abnormal heart rhythm, seizure, and sometimes death. Your healthcare provider may do blood tests to check you for TLS.
Your healthcare provider may tell you to stop taking pomalidomide capsules if you develop certain serious side effects during treatment.
The most common side effects of pomalidomide capsules in people with Multiple Myeloma include:
|
|
|
The most common side effects of pomalidomide capsules in people with KS include:
|
|
These are not all the possible side effects of pomalidomide capsules.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store pomalidomide capsules?
- Store pomalidomide capsules at room temperature between 68°F to 77°F (20°C to 25°C).
- Return any unused pomalidomide capsules to PS-Pomalidomide REMS by calling 1-888-423-5436 or your healthcare provider.
Keep pomalidomide capsules and all medicines out of the reach of children.
General information about the safe and effective use of pomalidomide capsules.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not take pomalidomide capsules for conditions for which it was not prescribed. Do not give pomalidomide capsules to other people, even if they have the same symptoms you have. It may harm them and may cause birth defects.
If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about pomalidomide capsules that is written for health professionals.
For more information, call 1-888-423-5436 or go to www.PS-PomalidomideREMS.com.
What are the ingredients in pomalidomide capsules?
Active ingredient: pomalidomide
Inactive ingredients: mannitol, pregelatinized starch, croscarmellose sodium and sodium stearyl fumarate.
The 1-mg capsule shell contains Gelatin, titanium dioxide, FD&C blue 2, yellow iron oxide, white ink and Black ink. The 2-mg capsule shell contains Gelatin, FD&C Blue #2, Titanium Dioxide, Iron Oxide Yellow, FD&C Red #3, and white ink. The 3-mg capsule shell contains Gelatin, FD&C Blue #2, Titanium Dioxide, Iron Oxide Yellow, and white ink. The 4-mg capsule shell contains Gelatin, FD&C Blue #2, Titanium Dioxide, and white ink.
Black ink contains Shellac, Dehydrated alcohol, Isopropyl alcohol, Butyl alcohol, Propylene Glycol, Strong Ammonia Solution, Black Iron Oxide, Potassium hydroxide, and Purified water.
White ink contains Shellac, Ethanol, Isopropyl alcohol, n-Butanol, Propylene Glycol, Ammonia Solution, Purified Water, Potassium Hydroxide, and Titanium Dioxide.
Manufactured by:
Natco Pharma Limited
Parawada – 531019, India.
Distributed by:
Breckenridge Pharmaceutical, Inc.
Berkeley Heights, NJ 07922
Trademarks are the property of their respective owners.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Revised: 08/2025
8.4 Pediatric Use
The safety and effectiveness of pomalidomide capsules have not been established in pediatric patients. The safety and effectiveness were assessed but not established in two open-label studies: a dose escalation study in 25 pediatric patients aged 5 to <17 with recurrent, progressive or refractory CNS tumors [NCT02415153] and a parallel-group study conducted in 47 pediatric patients aged 4 to <17 years with recurrent or progressive high-grade glioma, medulloblastoma, ependymoma, or diffuse intrinsic pontine glioma (DIPG) [NCT03257631]. No new safety signals were observed in pediatric patients across these studies.
At the same dose by body surface area, pomalidomide exposure in 55 pediatric patients aged 4 to < 17 years old was within the range observed in adult patients with MM but higher than the exposure observed in adults with KS [see Clinical Pharmacology (12.3)].
1.2 Kaposi Sarcoma
Pomalidomide capsules are indicated for the treatment of:
- Adult patients with AIDS-related Kaposi sarcoma (KS) after failure of highly active antiretroviral therapy (HAART).
- Kaposi sarcoma (KS) in adult patients who are HIV-negative.
This indication is approved under accelerated approval based on overall response rate [see Clinical Studies (14.2)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s).
2.9 Administration
Swallow capsules whole with water. Do not break, chew, or open the capsules.
Pomalidomide capsules may be taken with or without food.
5.6 Hepatotoxicity
Hepatic failure, including fatal cases, has occurred in patients treated with pomalidomide capsules. Elevated levels of alanine aminotransferase and bilirubin have also been observed in patients treated with pomalidomide capsules. Monitor liver function tests monthly. Stop pomalidomide capsules upon elevation of liver enzymes and evaluate. After return to baseline values, treatment at a lower dose may be considered.
14.2 Kaposi Sarcoma
The clinical trial 12-C-0047 (NCT01495598), was an open label, single center, single arm clinical study that evaluated the safety and efficacy of pomalidomide capsules in patients with Kaposi sarcoma (KS). A total of 28 patients (18 HIV-positive, 10 HIV-negative) received pomalidomide capsules 5 mg orally once daily on Days 1 through 21 of each 28-day cycle until disease progression or unacceptable toxicity. All HIV-positive patients continued highly active antiretroviral therapy (HAART). The trial excluded patients with symptomatic pulmonary or visceral KS, history of venous or arterial thromboembolism, or procoagulant disorders. Patients received thromboprophylaxis with aspirin 81 mg once daily throughout therapy.
The median age was 52.5 years, all were male, 75% were White, and 14% Black or African American. Seventy- five percent of patients had advanced disease (T1) at the time of enrollment, 11% had ≥ 50 lesions, and 75% had received prior chemotherapy.
The major efficacy outcome measure was overall response rate (ORR), which included complete response (CR), clinical complete response (cCR), and partial response (PR). Response was assessed by the investigator according to the AIDS Clinical Trial Group (ACTG) Oncology Committee response criteria for KS. The median time to first response was 1.8 months (0.9 to 7.6). Efficacy results are presented in Table 11.
| All Patients N=28 |
HIV-Positive N=18 |
HIV-Negative N=10 |
|
|---|---|---|---|
| CI: confidence interval, ORR: overall response rate, CR: complete response, PR: partial response | |||
| ORR CR includes one HIV-negative patient who achieved a cCR. , n (%) |
20 (71) | 12 (67) | 8 (80) |
| [95% CI] | [51, 87] | (41, 87) | (44, 98) |
| CR, n (%) | 4 (14) | 3 (17) | 1 (10) |
| PR, n (%) | 16 (57) | 9 (50) | 7 (70) |
| Duration of Response, KS Calculated as date of first documented response to date of first documented disease progression, receipt of new treatment or second course of treatment, or death due to any cause, whichever occurs first. Median estimate is from Kaplan-Meier analysis. , |
12.1 | 12.5 | 10.5 |
| Median in months [95% CI] From Kaplan-Meier analysis.
|
[7.6, 16.8] | [6.5, 24.9] | [3.9, 24.2] |
| Duration of Response, KS (%) | |||
| Percent greater than 12 months | 50 | 58 | 38 |
| Percent greater than 24 months | 20 | 17 | 25 |
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described in detail in other labeling sections:
- Embryo-Fetal Toxicity [see Warnings and Precautions (5.1, 5.2)]
- Venous and Arterial Thromboembolism [see Warnings and Precautions (5.3)]
- Increased Mortality in Patients with Multiple Myeloma When Pembrolizumab Is Added to a Thalidomide Analogue and Dexamethasone [see Warnings and Precautions (5.4)]
- Hematologic Toxicity [see Warnings and Precautions (5.5)]
- Hepatotoxicity [see Warnings and Precautions (5.6)]
- Severe Cutaneous Reactions [see Warnings and Precautions (5.7)]
- Dizziness and Confusional State [see Warnings and Precautions (5.8)]
- Neuropathy [see Warnings and Precautions (5.9)]
- Risk of Second Primary Malignancies [see Warnings and Precautions (5.10)]
- Tumor Lysis Syndrome [see Warnings and Precautions (5.11)]
- Hypersensitivity [see Warnings and Precautions (5.12)]
7 Drug Interactions (7 DRUG INTERACTIONS)
8.8 Smoking Tobacco
Cigarette smoking reduces pomalidomide AUC due to CYP1A2 induction. Advise patients that smoking may reduce the efficacy of pomalidomide [see Clinical Pharmacology (12.3)].
1.1 Multiple Myeloma
Pomalidomide capsules, in combination with dexamethasone, are indicated for adult patients with multiple myeloma (MM) who have received at least two prior therapies including lenalidomide and a proteasome inhibitor and have demonstrated disease progression on or within 60 days of completion of the last therapy.
4.2 Hypersensitivity
Pomalidomide Capsules are contraindicated in patients who have demonstrated severe hypersensitivity (e.g., angioedema, anaphylaxis) to pomalidomide or any of the excipients [see Warnings and Precautions (5.7), Description (11)].
8.6 Renal Impairment
In patients with severe renal impairment requiring dialysis, the AUC of pomalidomide increased by 38% and the rate of SAE increased by 64% relative to patients with normal renal function; therefore, starting dose adjustment is recommended. For patients with severe renal impairment requiring dialysis, administer pomalidomide capsules after the completion of hemodialysis on dialysis days because exposure of pomalidomide could be significantly decreased during dialysis [see Dosage and Administration (2.7) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Pomalidomide exposure-response analyses showed that there was no relationship between systemic pomalidomide exposure level and efficacy or safety following pomalidomide dose of 4 mg.
12.3 Pharmacokinetics
In patients with MM who received pomalidomide capsules 4 mg daily alone or in combination with dexamethasone, pomalidomide steady-state drug exposure was characterized by AUC (CV%) of 860 (37%) ng∙h/mL and Cmax (CV%) of 75 (32%) ng/mL. In patients with Kaposi sarcoma (KS) who received pomalidomide capsules 5 mg daily, pomalidomide steady-state drug exposure was characterized by AUC of 462.3 ng∙h/mL (82%) and Cmax of 53.1 ng/mL (50%).
5.12 Hypersensitivity
Hypersensitivity, including angioedema, anaphylaxis, and anaphylactic reactions to pomalidomide capsules have been reported. Permanently discontinue pomalidomide capsules for angioedema or anaphylaxis [see Dosage and Administration (2.5)].
8.7 Hepatic Impairment
Pomalidomide is metabolized primarily by the liver. Following single dose administration, the AUC of pomalidomide increased 51%, 58%, and 72% in subjects with mild (Child-Pugh class A), moderate (Child-Pugh class B), and severe (Child-Pugh class C) hepatic impairment, respectively compared to subjects with normal liver function. Dose adjustment is recommended in patients with hepatic impairment [see Dosage and Administration (2.8) and Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Pomalidomide capsules are a thalidomide analogue indicated for the treatment of adult patients:
- in combination with dexamethasone, for patients with multiple myeloma (MM) who have received at least two prior therapies including lenalidomide and a proteasome inhibitor and have demonstrated disease progression on or within 60 days of completion of the last therapy (1.1).
- with AIDS-related Kaposi sarcoma (KS) after failure of highly active antiretroviral therapy (HAART) or in patients with KS who are HIV-negative. This indication is approved under accelerated approval based on overall response rate. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s) (1.2).
12.1 Mechanism of Action
Pomalidomide is an analogue of thalidomide with immunomodulatory, antiangiogenic, and antineoplastic properties. Cellular activities of pomalidomide are mediated through its target cereblon, a component of a cullin ring E3 ubiquitin ligase enzyme complex. In vitro, in the presence of drug, substrate proteins (including Aiolos and Ikaros) are targeted for ubiquitination and subsequent degradation leading to direct cytotoxic and immunomodulatory effects. In in vitro cellular assays, pomalidomide inhibited proliferation and induced apoptosis of hematopoietic tumor cells. Additionally, pomalidomide inhibited the proliferation of lenalidomide-resistant multiple myeloma (MM) cell lines and synergized with dexamethasone in both lenalidomide-sensitive and lenalidomide-resistant cell lines to induce tumor cell apoptosis. Pomalidomide enhanced T cell- and natural killer (NK) cell-mediated immunity and inhibited production of pro-inflammatory cytokines (e.g., TNF-α and IL-6) by monocytes. Pomalidomide demonstrated anti-angiogenic activity in a mouse tumor model and in the in vitro umbilical cord model.
5.2 Ps Pomalidomide Rems (5.2 PS-Pomalidomide REMS)
Because of the embryo-fetal risk [see Warnings and Precautions (5.1)], pomalidomide capsules are available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS), "PS-Pomalidomide REMS".
Required components of PS-Pomalidomide REMS include the following:
- Prescribers must be certified with PS-Pomalidomide REMS by enrolling and complying with the REMS requirements.
- Patients must sign a Patient-Physician Agreement Form and comply with the REMS requirements. In particular, female patients of reproductive potential who are not pregnant must comply with the pregnancy testing and contraception requirements [see Use in Specific Populations (8.3)] and males must comply with contraception requirements [see Use in Specific Populations (8.3)].
- Pharmacies must be certified with PS-Pomalidomide REMS, must only dispense to patients who are authorized to receive pomalidomide capsules and comply with REMS requirements.
Further information about PS-Pomalidomide REMS is available at www.PS-PomalidomideREMS.com or by telephone at 1-888-423-5436.
5.1 Embryo Fetal Toxicity (5.1 Embryo-Fetal Toxicity)
Pomalidomide capsules are a thalidomide analogue and are contraindicated for use during pregnancy. Thalidomide is a known human teratogen that causes severe birth defects or embryo-fetal death [see Use in Specific Populations (8.1)]. Pomalidomide capsules are only available through PS-Pomalidomide REMS [see Warnings and Precautions (5.2)].
5.11 Tumor Lysis Syndrome
Tumor lysis syndrome (TLS) may occur in patients treated with pomalidomide. Patients at risk for TLS are those with high tumor burden prior to treatment. These patients should be monitored closely and appropriate precautions taken.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Increased Mortality: Observed in patients with MM when pembrolizumab was added to dexamethasone and a thalidomide analogue (5.4).
- Hematologic Toxicity: Neutropenia was the most frequently reported Grade 3/4 adverse event. Monitor patients for hematologic toxicities, especially neutropenia (5.5).
- Hepatotoxicity: Hepatic failure including fatalities; monitor liver function tests monthly (5.6).
- Severe Cutaneous Reactions: Discontinue pomalidomide capsules for severe reactions (5.7).
- Tumor Lysis Syndrome (TLS): Monitor patients at risk of TLS (i.e., those with high tumor burden) and take appropriate precautions (5.11).
- Hypersensitivity: Monitor patients for potential hypersensitivity. Discontinue pomalidomide capsules for angioedema and anaphylaxis (5.12).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- MM: 4 mg per day taken orally on Days 1 through 21 of repeated 28-day cycles until disease progression (2.2). Refer to section 14.1 for dexamethasone dosing (14.1)
- KS: 5 mg per day taken orally on Days 1 through 21 of repeated 28-day cycles until disease progression or unacceptable toxicity (2.3).
- Modify the dosage for certain patients with renal impairment (2.7, 8.6) or hepatic impairment (2.8, 8.7).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Capsules: 1 mg, dark blue cap and yellow body printed with "NAT'' with white ink on cap and "1 mg" with black ink on body of the capsule
- 2 mg, dark blue cap and orange body printed with "NAT" with white ink on cap and "2 mg" with white ink on body of the capsule
- 3 mg, dark blue cap and green body printed with "NAT" with white ink on cap and "3 mg" with white ink on body of the capsule
- 4 mg, dark blue cap and blue body printed with "NAT" with white ink on cap and "4 mg" with white ink on body of the capsule
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of pomalidomide capsules. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders : Pancytopenia
Endocrine Disorders: Hypothyroidism, hyperthyroidism
Gastrointestinal Disorders : Gastrointestinal hemorrhage
Hepatobiliary Disorders : Hepatic failure (including fatal cases), elevated liver enzymes
Immune System Disorders : Allergic reactions (e.g., angioedema, anaphylaxis, urticaria), solid organ transplant rejection
Infections and Infestations : Hepatitis B virus reactivation, Herpes zoster, progressive multifocal leukoencephalopathy (PML)
Neoplasms benign, malignant and unspecified (incl cysts and polyps): Tumor lysis syndrome, basal cell carcinoma, and squamous cell carcinoma of the skin
Skin and Subcutaneous Tissue Disorders : Stevens-Johnson Syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms (DRESS)
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Lactation: Advise women not to breastfeed (8.2).
5.7 Severe Cutaneous Reactions
Severe cutaneous reactions including Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS) have been reported. DRESS may present with a cutaneous reaction (such as rash or exfoliative dermatitis), eosinophilia, fever, and/or lymphadenopathy with systemic complications such as hepatitis, nephritis, pneumonitis, myocarditis, and/or pericarditis. These reactions can be fatal. Consider pomalidomide capsules interruption or discontinuation for Grade 2 or 3 skin rash. Permanently discontinue pomalidomide capsules for Grade 4 rash, exfoliative or bullous rash, or for other severe cutaneous reactions such as SJS, TEN or DRESS [see Dosage and Administration (2.5)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.8 Dizziness and Confusional State
In trials 1 and 2 in patients who received pomalidomide capsules + Low-dose Dex, 14% of patients experienced dizziness and 7% of patients experienced a confusional state; 1% of patients experienced Grade 3 or 4 dizziness, and 3% of patients experienced Grade 3 or 4 confusional state. Instruct patients to avoid situations where dizziness or confusional state may be a problem and not to take other medications that may cause dizziness or confusional state without adequate medical advice.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Dark blue cap and yellow body printed with "NAT'' with white ink on cap and "1mg" with black ink on body of the capsule
1 mg bottles of 21 (NDC 51991-342-21)
1 mg bottles of 100 (NDC 51991-342-01)
Dark blue cap and orange body printed with "NAT" with white ink on cap and "2mg" with white ink on body of the capsule
2 mg bottles of 21 (NDC 51991-343-21)
2 mg bottles of 100 (NDC 51991-343-01)
Dark blue cap and green body printed with "NAT" with white ink on cap and "3mg" with white ink on body of the capsule
3 mg bottles of 21 (NDC 51991-344-21)
3 mg bottles of 100 (NDC 51991-344-01)
Dark blue cap and blue body printed with "NAT" with white ink on cap and "4mg" with white ink on body of the capsule
4 mg bottles of 21 (NDC 51991-346-21)
4 mg bottles of 100 (NDC 51991-346-01)
5.3 Venous and Arterial Thromboembolism
Venous thromboembolic events (deep venous thrombosis and pulmonary embolism) and arterial thromboembolic events (myocardial infarction and stroke) have been observed in patients treated with pomalidomide capsules. In Trial 2, where anticoagulant therapies were mandated, thromboembolic events occurred in 8.0% of patients treated with pomalidomide capsules and low dose-dexamethasone (Low-dose Dex), and 3.3% of patients treated with high-dose dexamethasone. Venous thromboembolic events (VTE) occurred in 4.7% of patients treated with pomalidomide capsules and Low-dose Dex, and 1.3% of patients treated with high-dose dexamethasone. Arterial thromboembolic events include terms for arterial thromboembolic events, ischemic cerebrovascular conditions, and ischemic heart disease. Arterial thromboembolic events occurred in 3.0% of patients treated with pomalidomide capsules and Low-dose Dex, and 1.3% of patients treated with high-dose dexamethasone.
Patients with known risk factors, including prior thrombosis, may be at greater risk, and actions should be taken to try to minimize all modifiable factors (e.g., hyperlipidemia, hypertension, smoking). Thromboprophylaxis is recommended, and the choice of regimen should be based on assessment of the patient's underlying risk factors.
5.10 Risk of Second Primary Malignancies
Cases of acute myelogenous leukemia have been reported in patients receiving pomalidomide capsules as an investigational therapy outside of MM.
2.3 Recommended Dosage for Kaposi Sarcoma
The recommended dosage of pomalidomide capsules is 5 mg once daily taken orally with or without food on Days 1 through 21 of each 28-day cycle until disease progression or unacceptable toxicity. Continue HAART as HIV treatment in patients with AIDS-related Kaposi sarcoma (KS) [see Clinical Studies (14.2)].
2.2 Recommended Dosage for Multiple Myeloma
The recommended dosage of pomalidomide capsules is 4 mg once daily orally with or without food on Days 1 through 21 of each 28-day cycle until disease progression. Give pomalidomide capsules in combination with dexamethasone [see Clinical Studies (14.1)].
2.1 Pregnancy Testing Prior to Administration
Females of reproductive potential must have negative pregnancy testing and use contraception methods before initiating pomalidomide capsules [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1, 8.3)].
Principal Display Panel 1 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 1 mg Capsule Bottle Label)
NDC 51991-342-21
Pomalidomide
Capsules
1 mg
WARNING: POTENTIAL FOR
HUMAN BIRTH DEFECTS
PHARMACIST: Dispense the enclosed
Medication Guide to each patient.
21 Capsules
Rx Only
breckenridge
A Towa
Company
Principal Display Panel 2 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 2 mg Capsule Bottle Label)
NDC 51991-343-21
Pomalidomide
Capsules
2 mg
WARNING: POTENTIAL FOR
HUMAN BIRTH DEFECTS
PHARMACIST: Dispense the enclosed
Medication Guide to each patient.
21 Capsules
Rx Only
breckenridge
A Towa
Company
Principal Display Panel 3 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 3 mg Capsule Bottle Label)
NDC 51991-344-21
Pomalidomide
Capsules
3 mg
WARNING: POTENTIAL FOR
HUMAN BIRTH DEFECTS
PHARMACIST: Dispense the enclosed
Medication Guide to each patient.
21 Capsules
Rx Only
breckenridge
A Towa
Company
Principal Display Panel 4 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 4 mg Capsule Bottle Label)
NDC 51991-346-21
Pomalidomide
Capsules
4 mg
WARNING: POTENTIAL FOR
HUMAN BIRTH DEFECTS
PHARMACIST: Dispense the enclosed
Medication Guide to each patient.
21 Capsules
Rx Only
breckenridge
A Towa
Company
2.6 Dosage Modifications for Strong Cyp1a2 Inhibitors (2.6 Dosage Modifications for Strong CYP1A2 Inhibitors)
Avoid concomitant use of pomalidomide capsules with strong CYP1A2 inhibitors. If concomitant use of a strong CYP1A2 inhibitor is unavoidable, reduce pomalidomide capsules dose to 2 mg [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies examining the carcinogenic potential of pomalidomide have not been conducted. One of 12 monkeys dosed with 1 mg/kg of pomalidomide (an exposure approximately 15-fold of the exposure in patients at the recommended dose of 4 mg/day) developed acute myeloid leukemia in a 9-month repeat-dose toxicology study.
Pomalidomide was not mutagenic or clastogenic in a battery of tests, including the bacteria reverse mutation assay (Ames test), the in vitro assay using human peripheral blood lymphocytes, and the micronucleus test in orally treated rats administered doses up to 2000 mg/kg/day.
In a fertility and early embryonic development study in rats, drug-treated males were mated with untreated or treated females.
Pomalidomide was administered to males and females at doses of 25 to 1000 mg/kg/day. When treated males were mated with treated females, there was an increase in post-implantation loss and a decrease in mean number of viable embryos at all dose levels. There were no other effects on reproductive functions or the number of pregnancies. The lowest dose tested in animals resulted in an exposure (AUC) approximately 100-fold of the exposure in patients at the recommended dose of 4 mg/day. When treated males in this study were mated with untreated females, all uterine parameters were comparable to the controls. Based on these results, the observed effects were attributed to the treatment of females.
2.5 Dosage Modifications for Non Hematologic Adverse Reactions (2.5 Dosage Modifications for Non-Hematologic Adverse Reactions)
Permanently discontinue pomalidomide capsules for angioedema, anaphylaxis, Grade 4 rash, skin exfoliation, bullae, or any other severe dermatologic reaction [See Warnings and Precautions (5.7, 5.12)].
For other Grade 3 or 4 toxicities, hold treatment and restart treatment at 1 mg less than the previous dose when toxicity has resolved to less than or equal to Grade 2 at the physician's discretion.
2.7 Dosage Modification for Severe Renal Impairment On Hemodialysis (2.7 Dosage Modification for Severe Renal Impairment on Hemodialysis)
Take pomalidomide capsules after completion of dialysis procedure on hemodialysis days [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
- For patients with MM with severe renal impairment requiring dialysis, reduce the recommended dosage to 3 mg orally daily.
- For patients with KS with severe renal impairment requiring dialysis, reduce the recommended dosage to 4 mg orally daily.
Warning: Embryo Fetal Toxicity and Venous and Arterial Thromboembolism (WARNING: EMBRYO-FETAL TOXICITY and VENOUS AND ARTERIAL THROMBOEMBOLISM)
WARNING: EMBRYO-FETAL TOXICITY and VENOUS AND ARTERIAL THROMBOEMBOLISM
See full prescribing information for complete boxed warning
EMBRYO-FETAL TOXICITY
- Pomalidomide capsules are contraindicated in pregnancy. Pomalidomide capsules are a thalidomide analogue. Thalidomide is a known human teratogen that causes severe life-threatening birth defects (4, 5.1, 8.1).
- For females of reproductive potential: Exclude pregnancy before start of treatment. Prevent pregnancy during treatment by the use of 2 reliable methods of contraception (5.1, 8.3).
Pomalidomide capsules are available only through a restricted program called PS-Pomalidomide REMS (5.2).
VENOUS AND ARTERIAL THROMBOEMBOLISM
- Deep venous thrombosis (DVT), pulmonary embolism (PE), myocardial infarction, and stroke occur in patients with multiple myeloma treated with pomalidomide capsules. Antithrombotic prophylaxis is recommended (5.3).
5.4 Increased Mortality in Patients With Multiple Myeloma When Pembrolizumab Is Added to A Thalidomide Analogue and Dexamethasone (5.4 Increased Mortality in Patients with Multiple Myeloma When Pembrolizumab Is Added to a Thalidomide Analogue and Dexamethasone)
In two randomized clinical trials in patients with MM, the addition of pembrolizumab to a thalidomide analogue plus dexamethasone, a use for which no PD-1 or PD-L1 blocking antibody is indicated, resulted in increased mortality. Treatment of patients with MM with a PD-1 or PD-L1 blocking antibody in combination with a thalidomide analogue plus dexamethasone is not recommended outside of controlled clinical trials.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:32.325492 · Updated: 2026-03-14T22:51:04.846555