Metaxalone
413b300d-4163-afef-e063-6394a90a618f
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Metaxalone is indicated as an adjunct to rest, physical therapy, and other measures for the relief of discomfort associated with acute, painful musculoskeletal conditions in adults and pediatric patients 13 years of age and older.
Dosage and Administration
The recommended dosage of metaxalone in adults and pediatric patients 13 years of age and older is 800 mg orally three to four times a day [see Use in Specific Populations ( 8 )] . Metaxalone Tablets 800 mg are not substitutable on a mg to mg basis with Metaxalone Tablets, 640 mg [see Clinical Pharmacology ( 12.3 )] . When it is appropriate to switch: Switch only in patients who have been taking Metaxalone Tablets, 640 mg on an empty stomach. Stop Metaxalone Tablets, 640 mg three times a day and start Metaxalone Tablets 800 mg three times a day on an empty stomach, OR stop Metaxalone Tablets 640 mg four times a day and start Metaxalone Tablets 800 mg four times a day on an empty stomach. Do not switch from Metaxalone Tablets, 640 mg to Metaxalone Tablets, 800 mg when the patient is taking food during administration.
Contraindications
Metaxalone is contraindicated in patients with: Known hypersensitivity to any component of metaxalone. Known tendency to drug induced, hemolytic, or other anemias. Severe renal or hepatic impairment.
Warnings and Precautions
Serotonin Syndrome : Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of metaxalone (within the recommended dosage range) and other serotonergic drugs and with the use of metaxalone as the only serotonergic drug taken at a dosage higher than the recommended dosage. If concomitant use is warranted, carefully observe the patient, particularly during treatment initiation and dosage increases. Discontinue metaxalone if serotonin syndrome is suspected or it occurs. ( 5.1 , 7.1 ) Central Nervous System (CNS) Depression : Metaxalone may impair mental and/or physical abilities required for the performance of hazardous tasks, such as operating machinery or driving a motor vehicle, especially when used with other CNS depressants including alcohol. Follow patients closely for signs and symptoms of respiratory depression and sedation. If concomitant use is warranted, closely monitor for signs of respiratory depression and sedation, particularly during treatment initiation and dosage increases. ( 5.2 , 7.2 )
Adverse Reactions
The following adverse reactions associated with the use of metaxalone were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The most frequent reactions to metaxalone were: CNS: drowsiness, dizziness, headache, and nervousness or "irritability". Digestive: nausea, vomiting, gastrointestinal upset. Other adverse reactions were: CNS: cases of serotonin syndrome have been reported during concomitant use of metaxalone (within the recommended dosage range) and other serotonergic drugs and with the use of metaxalone as the only serotonergic drug at a dosage higher than the recommended dosage [see Warnings and Precautions ( 5.1 ) , Drug Interactions ( 7.1 ) and Overdosage ( 10 )] . Hematologic: leukopenia; hemolytic anemia; Hepatobiliary: jaundice; Immune System: anaphylaxis, hypersensitivity reaction, rash with or without pruritus.
How Supplied
Metaxalone Tablets, USP are available as 800 mg pink, capsule shaped, scored tablets, debossed “LCI" on one side and "14" bisect "35" on the other side. Available in bottles of 120 (NDC 85509-1435-1) and in bottles of 30 (NDC 85509-1435-3) and in bottles of 60 (NDC 85509-1435-6) and in bottles of 90 (NDC 85509-1435-9). Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. Repackaged/Relabeled by: PHOENIX RX LLC Hatboro, PA 19040
Storage and Handling
Metaxalone Tablets, USP are available as 800 mg pink, capsule shaped, scored tablets, debossed “LCI" on one side and "14" bisect "35" on the other side. Available in bottles of 120 (NDC 85509-1435-1) and in bottles of 30 (NDC 85509-1435-3) and in bottles of 60 (NDC 85509-1435-6) and in bottles of 90 (NDC 85509-1435-9). Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. Repackaged/Relabeled by: PHOENIX RX LLC Hatboro, PA 19040
Description
Dosage and Administration ( 2 ) 06/2024
Medication Information
Warnings and Precautions
Serotonin Syndrome : Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of metaxalone (within the recommended dosage range) and other serotonergic drugs and with the use of metaxalone as the only serotonergic drug taken at a dosage higher than the recommended dosage. If concomitant use is warranted, carefully observe the patient, particularly during treatment initiation and dosage increases. Discontinue metaxalone if serotonin syndrome is suspected or it occurs. ( 5.1 , 7.1 ) Central Nervous System (CNS) Depression : Metaxalone may impair mental and/or physical abilities required for the performance of hazardous tasks, such as operating machinery or driving a motor vehicle, especially when used with other CNS depressants including alcohol. Follow patients closely for signs and symptoms of respiratory depression and sedation. If concomitant use is warranted, closely monitor for signs of respiratory depression and sedation, particularly during treatment initiation and dosage increases. ( 5.2 , 7.2 )
Indications and Usage
Metaxalone is indicated as an adjunct to rest, physical therapy, and other measures for the relief of discomfort associated with acute, painful musculoskeletal conditions in adults and pediatric patients 13 years of age and older.
Dosage and Administration
The recommended dosage of metaxalone in adults and pediatric patients 13 years of age and older is 800 mg orally three to four times a day [see Use in Specific Populations ( 8 )] . Metaxalone Tablets 800 mg are not substitutable on a mg to mg basis with Metaxalone Tablets, 640 mg [see Clinical Pharmacology ( 12.3 )] . When it is appropriate to switch: Switch only in patients who have been taking Metaxalone Tablets, 640 mg on an empty stomach. Stop Metaxalone Tablets, 640 mg three times a day and start Metaxalone Tablets 800 mg three times a day on an empty stomach, OR stop Metaxalone Tablets 640 mg four times a day and start Metaxalone Tablets 800 mg four times a day on an empty stomach. Do not switch from Metaxalone Tablets, 640 mg to Metaxalone Tablets, 800 mg when the patient is taking food during administration.
Contraindications
Metaxalone is contraindicated in patients with: Known hypersensitivity to any component of metaxalone. Known tendency to drug induced, hemolytic, or other anemias. Severe renal or hepatic impairment.
Adverse Reactions
The following adverse reactions associated with the use of metaxalone were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The most frequent reactions to metaxalone were: CNS: drowsiness, dizziness, headache, and nervousness or "irritability". Digestive: nausea, vomiting, gastrointestinal upset. Other adverse reactions were: CNS: cases of serotonin syndrome have been reported during concomitant use of metaxalone (within the recommended dosage range) and other serotonergic drugs and with the use of metaxalone as the only serotonergic drug at a dosage higher than the recommended dosage [see Warnings and Precautions ( 5.1 ) , Drug Interactions ( 7.1 ) and Overdosage ( 10 )] . Hematologic: leukopenia; hemolytic anemia; Hepatobiliary: jaundice; Immune System: anaphylaxis, hypersensitivity reaction, rash with or without pruritus.
Storage and Handling
Metaxalone Tablets, USP are available as 800 mg pink, capsule shaped, scored tablets, debossed “LCI" on one side and "14" bisect "35" on the other side. Available in bottles of 120 (NDC 85509-1435-1) and in bottles of 30 (NDC 85509-1435-3) and in bottles of 60 (NDC 85509-1435-6) and in bottles of 90 (NDC 85509-1435-9). Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. Repackaged/Relabeled by: PHOENIX RX LLC Hatboro, PA 19040
How Supplied
Metaxalone Tablets, USP are available as 800 mg pink, capsule shaped, scored tablets, debossed “LCI" on one side and "14" bisect "35" on the other side. Available in bottles of 120 (NDC 85509-1435-1) and in bottles of 30 (NDC 85509-1435-3) and in bottles of 60 (NDC 85509-1435-6) and in bottles of 90 (NDC 85509-1435-9). Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. Repackaged/Relabeled by: PHOENIX RX LLC Hatboro, PA 19040
Description
Dosage and Administration ( 2 ) 06/2024
Principal Display Panel
NDC 85509- 1435-1
Metaxalone
Tablets, USP
800 mg
Rx Only
100 Tablets
Lannett
Section 43683-2
Dosage and Administration ( 2) 06/2024
10 Overdosage
Clinical Presentation of Metaxalone Overdose
Deaths by deliberate or accidental overdose have occurred with metaxalone, particularly in combination with other CNS depressants (including alcohol). CNS manifestations may include CNS depression, agitation, hallucinations, delusions, seizures, respiratory depression, and coma. Cardiovascular effects may include tachycardia and hypertension; hypotension has also been reported. Serotonin syndrome, leading to muscle rigidity, tremor, and hyperthermia, has been reported
[see Warnings and Precautions (
5.1), Drug Interactions (
7.1,
7.2)]
.
Treatment of Metaxalone Overdose
The standard of treatment is supportive care. Monitor for CNS and respiratory depression and manage airway with oxygen as needed. Gastrointestinal decontamination procedures (including emesis) should generally be avoided because aspiration may result from CNS depression and seizures. Extracorporeal elimination such as hemodialysis or plasmapheresis have no proven clinical benefit.
Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
8.1 Pregnancy
Risk Summary
There are no available data on metaxalone use in pregnant patients to evaluate for a drug-associated risk of major birth defects, miscarriage or other adverse maternal or fetal outcomes despite decades of metaxalone use. Reproduction studies in rats have not revealed effects on the fetus due to metaxalone.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
8.2 Lactation
Risk Summary
There are no data available to evaluate the presence of metaxalone or its metabolite in either human or animal milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for metaxalone and any potential adverse effects on the breastfed infant from metaxalone or from the underlying maternal condition.
11 Description
Metaxalone Tablets, USP contain 800 mg of metaxalone and the following inactive ingredients: carboxymethylcellulose sodium, alginic acid, stearic acid, hydrogenated castor oil, magnesium stearate, colloidal silicon dioxide, sodium lauryl sulfate, and FD&C Red #40 Aluminum Lake. Metaxalone is a muscle relaxant for oral administration.
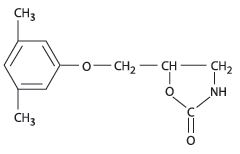
Chemically, metaxalone is 5-[(3,5- dimethylphenoxy) methyl]-2-oxazolidinone. The empirical formula is C 12H 15NO 3, which corresponds to a molecular weight of 221.25. The structural formula is:
Metaxalone is a white to almost white, odorless crystalline powder freely soluble in chloroform, soluble in methanol and in 96% ethanol, but practically insoluble in ether or water.
8.4 Pediatric Use
Metaxalone is indicated as an adjunct to rest, physical therapy, and other measures for the relief of discomforts associated with acute, painful musculoskeletal conditions in pediatric patients 13 years of age and older. The safety and effectiveness of metaxalone in pediatric patients 12 years of age or younger have not been established.
8.5 Geriatric Use
Clinical studies of metaxalone did not include sufficient numbers of patients 65 years of age and older to determine whether they respond differently from younger adult patients.
Geriatric patients may be especially susceptible to CNS depression associated with metaxalone use [see Warnings and Precautions ( 5.2)] .
The recommended metaxalone dosage in patients 65 years of age and older is the same as in younger adult patients. Metaxalone peak plasma concentrations (C max) and area under the curve (AUC) were higher in patients 65 years of age and older in the fasted state; however, a clinically significant difference was not observed when metaxalone was administered in the fed state [see Clinical Pharmacology ( 12.3)] .
4 Contraindications
Metaxalone is contraindicated in patients with:
- Known hypersensitivity to any component of metaxalone.
- Known tendency to drug induced, hemolytic, or other anemias.
- Severe renal or hepatic impairment.
6 Adverse Reactions
The following adverse reactions associated with the use of metaxalone were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The most frequent reactions to metaxalone were:
- CNS: drowsiness, dizziness, headache, and nervousness or "irritability".
- Digestive: nausea, vomiting, gastrointestinal upset.
Other adverse reactions were:
- CNS: cases of serotonin syndrome have been reported during concomitant use of metaxalone (within the recommended dosage range) and other serotonergic drugs and with the use of metaxalone as the only serotonergic drug at a dosage higher than the recommended dosage [see Warnings and Precautions ( 5.1) , Drug Interactions ( 7.1) and Overdosage ( 10)] .
- Hematologic: leukopenia; hemolytic anemia;
- Hepatobiliary: jaundice;
- Immune System: anaphylaxis, hypersensitivity reaction, rash with or without pruritus.
7.2 Cns Depressants
If concomitant use of metaxalone and another CNS depressant is warranted, closely monitor for signs of respiratory depression and sedation, particularly during treatment initiation and dosage increases.
Due to the additive pharmacologic effect, concomitant use of metaxalone with other CNS depressants may increase the risk of sedation and respiratory depression [see Warnings and Precautions ( 5.2)] .
8.7 Renal Impairment
Metaxalone is contraindicated in patients with severe renal impairment. Metaxalone should be used with caution and additional follow-up should be considered in patients with mild to moderate renal impairment. The effect of renal impairment on metaxalone pharmacokinetics is unknown; however, metaxalone undergoes renal excretion as unidentified metabolites [see Clinical Pharmacology ( 12.3)] .
12.2 Pharmacodynamics
The exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of metaxalone not been fully characterized.
12.3 Pharmacokinetics
Metaxalone pharmacokinetics were evaluated in two groups of healthy volunteers that received a single oral dose of 800 mg of metaxalone or 400 mg of metaxalone (0.5 times the approved recommended dose) [the 400 mg strength of metaxalone is not currently marketed by Lannett Company, Inc.]. Metaxalone pharmacokinetic parameters are presented below as mean (% CV) unless otherwise specified. Observed metaxalone peak plasma concentrations (C max) and area under the curve (AUC) are shown in Table 1. Doubling the dose of metaxalone from 400 mg (0.5 times the approved recommended dose) to 800 mg resulted in a proportional increase in metaxalone C maxand AUC.
Table 1: Metaxalone Exposure after a Single-Dose of Metaxalone Under Fasting Conditions
| Dose | C max 2 | AUC ∞ 2 |
| 400 mg 1 | 983 (53) ng/mL | 7,479 (51) ng•h/mL |
| 800 mg | 1,816 (43) ng/mL | 15,044 (46) ng•h/mL |
10.5 times the approved recommended dose
2Mean (% CV)
Absorption
The absolute bioavailability of metaxalone is not known. Peak plasma metaxalone concentrations occurred at a mean T
maxof 3.3 hours (1.5 – 5 hours) of metaxalone under fasted conditions.
The single-dose pharmacokinetic parameters of metaxalone in two groups of healthy volunteers who received 400 mg or 800 mg of metaxalone are shown in Table 1.
Effect of Food:Peak plasma metaxalone concentrations were noted at a mean T maxof 4.3 hours (1.5 – 12 hours) under fed conditions. The mean T maxunder fasting and fed conditions was 3.3 and 4.3 hours, respectively. Metaxalone exposure was increased and the half-life (t 1/2) was decreased following metaxalone administration with a high fat meal as shown in Table 2. The increase in metaxalone exposure coinciding with a reduction in half-life may be attributed to more complete absorption of metaxalone in the presence of a high fat meal.
Table 2: Relative Changes in Metaxalone Exposure, T max, and t 1/2Following Metaxalone Administration with a High Fat Meal Compared to Fasting
|
Dose (mg) |
C max (ng/mL) |
AUC 0-t (ng•h/mL) |
AUC
0-INF
(ng∙h/mL) |
T
max
(hrs) |
t
1/2
(hrs) |
| 400 mg* | ↑ 78% | ↑ 24% | ↑ 15% | ↑ 30% | ↓ 73% |
| 800 mg | ↑ 94% | ↑ 46% | ↑ 42% | ↑ 63% | ↓ 48% |
* 0.5 times the approved recommended dose.
Distribution
Metaxalone apparent volume of distribution is approximately 800 Liters; however, plasma protein binding is unknown.
Elimination
Metaxalone mean ± SD terminal t
1/2is 9 ± 4.8 hours and apparent clearance is approximately 67 ± 34 L/h under fasted conditions.
Metabolism:Metaxalone is primarily metabolized by CYP1A2, CYP2D6, CYP2E1, and CYP3A4 and, to a lesser extent, CYP2C8, CYP2C9, and CYP2C19.
Excretion:Metaxalone is metabolized by the liver and excreted in the urine as unidentified metabolites.
Specific Populations
The effect of renal impairment and hepatic impairment on metaxalone pharmacokinetics is unknown
[see Use in Specific Populations (
8.6,
8.7)]
.
Geriatric Patients:The effects of age on the pharmacokinetics of metaxalone were determined following administration of 800 mg of metaxalone under fasted and fed conditions. Age had a significantly greater effect on metaxalone pharmacokinetics under fasted conditions than under fed conditions. Bioavailability under fasted conditions increased with age. Metaxalone bioavailability under fasted and fed conditions in the three groups of healthy volunteers of varying age is shown in Table 3.
Table 3: Metaxalone Pharmacokinetic Parameters under Fasted and Fed Conditions in Three Age Groups Following Oral Administration of 800 mg of Metaxalone
|
Mean Age in Years
(± SD) |
26 ± 9 Years Old |
39 ± 11 Years Old |
72 ± 5 Years Old |
|||
| Fasted vs. Fed State | Fasted | Fed | Fasted | Fed | Fasted | Fed |
| C max(ng/mL) 1 | 1816 (43) | 3510 (41) | 2719 (46) | 2915 (55) | 3168 (43) | 3680 (59) |
| T max(hours) 1 | 3 (39) | 4.9 (4.8) | 3 (40) | 8.7 (91) | 2.6 (30) | 6.5 (67) |
| AUC 0-t(ng∙h/mL) 1 | 14531 (47) | 20683 (41) | 19836 (40) | 20482 (37) | 23797 (45) | 24340 (48) |
| AUC ∞(ng∙h/mL) 1 | 15045 (46) | 20833 (41) | 20490 (39) | 20815 (37) | 24194 (44) | 24704 (47) |
1Mean values (% CV)
Male and Female Patients:The exposure of metaxalone was significantly higher in females compared to males as evidenced by C max. (2115 ng/mL versus 1335 ng/mL) and AUC ∞(17884 ng∙h/mL versus 10328 ng∙h/mL) following administration of 800 mg of metaxalone under fasted conditions. The mean half-life was 11.1 hours in females and 7.6 hours in males. The apparent volume of distribution of metaxalone was approximately 22% higher in males than in females.
Drug Interaction Studies
In Vitro Studies:Metaxalone does not inhibit CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP2E1, and CYP3A4. Metaxalone does not induce CYP1A2, CYP2B6, and CYP3A4.
5.1 Serotonin Syndrome
Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of metaxalone (within the recommended dosage range) and other serotonergic drugs [see Drug Interactions ( 7)] and with the use of metaxalone as the only serotonergic drug taken at a dosage higher than the recommended dosage [see Overdosage ( 10)] .
Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, coma), autonomic instability (e.g., tachycardia, labile blood pressure, hyperthermia), neuromuscular aberrations (e.g., hyperreflexia, incoordination, rigidity), and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). The onset of symptoms generally occurs within several hours to a few days after initiation of a serotonergic drug, but may occur later than that.
If concomitant use of metaxalone and another serotoneric drug is warranted, reassess the patient, particularly during treatment initiation and dosage increases. Discontinue metaxalone if serotonin syndrome is suspected or it occurs.
7.1 Serotonergic Drugs
If concomitant use of metaxalone and another serotoneric drug is warranted, carefully observe the patient, particularly during treatment initiation and dosage modification. Discontinue metaxalone if serotonin syndrome is suspected or if it occurs.
Serotonin syndrome has resulted from concomitant use of metaxalone (within the recommended dosage range) with other serotonergic drugs [see Warnings and Precautions ( 5.1) and Adverse Reactions ( 6)] .
Serotonergic drugs include selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, opioids (particularly fentanyl, meperidine, and methadone), drugs that affect the serotonergic neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), and drugs that impair metabolism of serotonin (including monoamine oxidase (MAO) inhibitors, both those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue).
8.6 Hepatic Impairment
Metaxalone is contraindicated in patients with severe hepatic impairment. Metaxalone should be used with caution and additional follow-up should be considered in patients with mild to moderate hepatic impairment. The effect of hepatic impairment on metaxalone pharmacokinetics is unknown; however, metaxalone undergoes expensive hepatic metabolism [see Clinical Pharmacology ( 12.3)] .
1 Indications and Usage
Metaxalone is indicated as an adjunct to rest, physical therapy, and other measures for the relief of discomfort associated with acute, painful musculoskeletal conditions in adults and pediatric patients 13 years of age and older.
12.1 Mechanism of Action
Metaxalone’s mechanism of action has not been fully characterized, but may be related to its sedative properties. Metaxalone has no direct action on the contractile mechanism of striated muscle, the motor end plate, or the nerve fiber.
5 Warnings and Precautions
- Serotonin Syndrome: Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of metaxalone (within the recommended dosage range) and other serotonergic drugs and with the use of metaxalone as the only serotonergic drug taken at a dosage higher than the recommended dosage. If concomitant use is warranted, carefully observe the patient, particularly during treatment initiation and dosage increases. Discontinue metaxalone if serotonin syndrome is suspected or it occurs. ( 5.1, 7.1)
- Central Nervous System (CNS) Depression: Metaxalone may impair mental and/or physical abilities required for the performance of hazardous tasks, such as operating machinery or driving a motor vehicle, especially when used with other CNS depressants including alcohol. Follow patients closely for signs and symptoms of respiratory depression and sedation. If concomitant use is warranted, closely monitor for signs of respiratory depression and sedation, particularly during treatment initiation and dosage increases. ( 5.2, 7.2)
2 Dosage and Administration
The recommended dosage of metaxalone in adults and pediatric patients 13 years of age and older is 800 mg orally three to four times a day [see Use in Specific Populations ( 8)] .
Metaxalone Tablets 800 mg are not substitutable on a mg to mg basis with Metaxalone Tablets, 640 mg [see Clinical Pharmacology ( 12.3)] . When it is appropriate to switch:
- Switch only in patients who have been taking Metaxalone Tablets, 640 mg on an empty stomach.
- Stop Metaxalone Tablets, 640 mg three times a day and start Metaxalone Tablets 800 mg three times a day on an empty stomach, OR stop Metaxalone Tablets 640 mg four times a day and start Metaxalone Tablets 800 mg four times a day on an empty stomach.
Do not switch from Metaxalone Tablets, 640 mg to Metaxalone Tablets, 800 mg when the patient is taking food during administration.
3 Dosage Forms and Strengths
Tablets: 800 mg pink, capsule shaped, scored tablets, debossed “LCI" on one side and "14" bisect "35" on the other side. The 400 mg strength of metaxalone is not currently marketed by Lannett Company, Inc.
8 Use in Specific Populations
Geriatric Use: Metaxalone-treated geriatric patients may be especially susceptible to CNS depression. ( 8.5)
17 Patient Counseling Information
Serotonin Syndrome
Inform patients that metaxalone could cause a rare but potentially life-threatening condition called serotonin syndrome. Warn patients of the symptoms of serotonin syndrome and to seek medical attention right away if symptoms develop. Instruct patients to inform their healthcare providers if they are taking, or plan to take, serotonergic drugs
[see Warnings and Precautions (
5.1) and Drug Interactions (
7.1)]
.
Central Nervous System Depression
Advise patients that metaxalone may impair mental and/or physical abilities required for performance of hazardous tasks, such as operating machinery or driving a motor vehicle, especially when used with alcohol and other CNS depressants
[see Drug Interactions (
7.2)]
.
Distributed by:
Lannett Company, Inc.
Philadelphia, PA 19136
CIB71144F
Rev. 06/2024
16 How Supplied/storage and Handling
Metaxalone Tablets, USP are available as 800 mg pink, capsule shaped, scored tablets, debossed “LCI" on one side and "14" bisect "35" on the other side. Available in bottles of 120 (NDC 85509-1435-1) and in bottles of 30 (NDC 85509-1435-3) and in bottles of 60 (NDC 85509-1435-6) and in bottles of 90 (NDC 85509-1435-9).
Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature].
Repackaged/Relabeled by:
PHOENIX RX LLC
Hatboro, PA 19040
5.2 Central Nervous System Depression
Because of its central nervous system (CNS) depressant effects, metaxalone may impair mental and/or physical abilities required for performance of hazardous tasks, such as operating machinery or driving a motor vehicle, especially when used with other CNS depressants including alcohol. Geriatric patients may be especially susceptible to CNS depression associated with metaxalone use. When used concomitantly, the sedative effects of metaxalone and other CNS depressants (e.g., alcohol, benzodiazepines, opioids, tricyclic antidepressants) may be additive [see Drug Interactions ( 7)] .
Follow metaxalone-treated patients closely for signs and symptoms of respiratory depression and sedation. If concomitant use of metaxalone and another CNS depressant is warranted, closely monitor for signs of respiratory depression and sedation, particularly during treatment initiation and dosage increases.
7.3 Interaction of Metaxalone With Benedict’s Tests
False-positive Benedict's tests, due to an unknown reducing substance, have been noted in metaxalone-treated patients. A glucose-specific test will differentiate findings.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies to evaluate the carcinogenic potential of metaxalone have not been conducted. Studies to evaluate the mutagenic potential of metaxalone have not been conducted. No effects on fertility were observed in rats administered metaxalone.
Structured Label Content
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
NDC 85509- 1435-1
Metaxalone
Tablets, USP
800 mg
Rx Only
100 Tablets
Lannett
Section 43683-2 (43683-2)
Dosage and Administration ( 2) 06/2024
10 Overdosage (10 OVERDOSAGE)
Clinical Presentation of Metaxalone Overdose
Deaths by deliberate or accidental overdose have occurred with metaxalone, particularly in combination with other CNS depressants (including alcohol). CNS manifestations may include CNS depression, agitation, hallucinations, delusions, seizures, respiratory depression, and coma. Cardiovascular effects may include tachycardia and hypertension; hypotension has also been reported. Serotonin syndrome, leading to muscle rigidity, tremor, and hyperthermia, has been reported
[see Warnings and Precautions (
5.1), Drug Interactions (
7.1,
7.2)]
.
Treatment of Metaxalone Overdose
The standard of treatment is supportive care. Monitor for CNS and respiratory depression and manage airway with oxygen as needed. Gastrointestinal decontamination procedures (including emesis) should generally be avoided because aspiration may result from CNS depression and seizures. Extracorporeal elimination such as hemodialysis or plasmapheresis have no proven clinical benefit.
Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
8.1 Pregnancy
Risk Summary
There are no available data on metaxalone use in pregnant patients to evaluate for a drug-associated risk of major birth defects, miscarriage or other adverse maternal or fetal outcomes despite decades of metaxalone use. Reproduction studies in rats have not revealed effects on the fetus due to metaxalone.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
8.2 Lactation
Risk Summary
There are no data available to evaluate the presence of metaxalone or its metabolite in either human or animal milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for metaxalone and any potential adverse effects on the breastfed infant from metaxalone or from the underlying maternal condition.
11 Description (11 DESCRIPTION)
Metaxalone Tablets, USP contain 800 mg of metaxalone and the following inactive ingredients: carboxymethylcellulose sodium, alginic acid, stearic acid, hydrogenated castor oil, magnesium stearate, colloidal silicon dioxide, sodium lauryl sulfate, and FD&C Red #40 Aluminum Lake. Metaxalone is a muscle relaxant for oral administration.
Chemically, metaxalone is 5-[(3,5- dimethylphenoxy) methyl]-2-oxazolidinone. The empirical formula is C 12H 15NO 3, which corresponds to a molecular weight of 221.25. The structural formula is:
Metaxalone is a white to almost white, odorless crystalline powder freely soluble in chloroform, soluble in methanol and in 96% ethanol, but practically insoluble in ether or water.
8.4 Pediatric Use
Metaxalone is indicated as an adjunct to rest, physical therapy, and other measures for the relief of discomforts associated with acute, painful musculoskeletal conditions in pediatric patients 13 years of age and older. The safety and effectiveness of metaxalone in pediatric patients 12 years of age or younger have not been established.
8.5 Geriatric Use
Clinical studies of metaxalone did not include sufficient numbers of patients 65 years of age and older to determine whether they respond differently from younger adult patients.
Geriatric patients may be especially susceptible to CNS depression associated with metaxalone use [see Warnings and Precautions ( 5.2)] .
The recommended metaxalone dosage in patients 65 years of age and older is the same as in younger adult patients. Metaxalone peak plasma concentrations (C max) and area under the curve (AUC) were higher in patients 65 years of age and older in the fasted state; however, a clinically significant difference was not observed when metaxalone was administered in the fed state [see Clinical Pharmacology ( 12.3)] .
4 Contraindications (4 CONTRAINDICATIONS)
Metaxalone is contraindicated in patients with:
- Known hypersensitivity to any component of metaxalone.
- Known tendency to drug induced, hemolytic, or other anemias.
- Severe renal or hepatic impairment.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions associated with the use of metaxalone were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The most frequent reactions to metaxalone were:
- CNS: drowsiness, dizziness, headache, and nervousness or "irritability".
- Digestive: nausea, vomiting, gastrointestinal upset.
Other adverse reactions were:
- CNS: cases of serotonin syndrome have been reported during concomitant use of metaxalone (within the recommended dosage range) and other serotonergic drugs and with the use of metaxalone as the only serotonergic drug at a dosage higher than the recommended dosage [see Warnings and Precautions ( 5.1) , Drug Interactions ( 7.1) and Overdosage ( 10)] .
- Hematologic: leukopenia; hemolytic anemia;
- Hepatobiliary: jaundice;
- Immune System: anaphylaxis, hypersensitivity reaction, rash with or without pruritus.
7.2 Cns Depressants (7.2 CNS Depressants)
If concomitant use of metaxalone and another CNS depressant is warranted, closely monitor for signs of respiratory depression and sedation, particularly during treatment initiation and dosage increases.
Due to the additive pharmacologic effect, concomitant use of metaxalone with other CNS depressants may increase the risk of sedation and respiratory depression [see Warnings and Precautions ( 5.2)] .
8.7 Renal Impairment
Metaxalone is contraindicated in patients with severe renal impairment. Metaxalone should be used with caution and additional follow-up should be considered in patients with mild to moderate renal impairment. The effect of renal impairment on metaxalone pharmacokinetics is unknown; however, metaxalone undergoes renal excretion as unidentified metabolites [see Clinical Pharmacology ( 12.3)] .
12.2 Pharmacodynamics
The exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of metaxalone not been fully characterized.
12.3 Pharmacokinetics
Metaxalone pharmacokinetics were evaluated in two groups of healthy volunteers that received a single oral dose of 800 mg of metaxalone or 400 mg of metaxalone (0.5 times the approved recommended dose) [the 400 mg strength of metaxalone is not currently marketed by Lannett Company, Inc.]. Metaxalone pharmacokinetic parameters are presented below as mean (% CV) unless otherwise specified. Observed metaxalone peak plasma concentrations (C max) and area under the curve (AUC) are shown in Table 1. Doubling the dose of metaxalone from 400 mg (0.5 times the approved recommended dose) to 800 mg resulted in a proportional increase in metaxalone C maxand AUC.
Table 1: Metaxalone Exposure after a Single-Dose of Metaxalone Under Fasting Conditions
| Dose | C max 2 | AUC ∞ 2 |
| 400 mg 1 | 983 (53) ng/mL | 7,479 (51) ng•h/mL |
| 800 mg | 1,816 (43) ng/mL | 15,044 (46) ng•h/mL |
10.5 times the approved recommended dose
2Mean (% CV)
Absorption
The absolute bioavailability of metaxalone is not known. Peak plasma metaxalone concentrations occurred at a mean T
maxof 3.3 hours (1.5 – 5 hours) of metaxalone under fasted conditions.
The single-dose pharmacokinetic parameters of metaxalone in two groups of healthy volunteers who received 400 mg or 800 mg of metaxalone are shown in Table 1.
Effect of Food:Peak plasma metaxalone concentrations were noted at a mean T maxof 4.3 hours (1.5 – 12 hours) under fed conditions. The mean T maxunder fasting and fed conditions was 3.3 and 4.3 hours, respectively. Metaxalone exposure was increased and the half-life (t 1/2) was decreased following metaxalone administration with a high fat meal as shown in Table 2. The increase in metaxalone exposure coinciding with a reduction in half-life may be attributed to more complete absorption of metaxalone in the presence of a high fat meal.
Table 2: Relative Changes in Metaxalone Exposure, T max, and t 1/2Following Metaxalone Administration with a High Fat Meal Compared to Fasting
|
Dose (mg) |
C max (ng/mL) |
AUC 0-t (ng•h/mL) |
AUC
0-INF
(ng∙h/mL) |
T
max
(hrs) |
t
1/2
(hrs) |
| 400 mg* | ↑ 78% | ↑ 24% | ↑ 15% | ↑ 30% | ↓ 73% |
| 800 mg | ↑ 94% | ↑ 46% | ↑ 42% | ↑ 63% | ↓ 48% |
* 0.5 times the approved recommended dose.
Distribution
Metaxalone apparent volume of distribution is approximately 800 Liters; however, plasma protein binding is unknown.
Elimination
Metaxalone mean ± SD terminal t
1/2is 9 ± 4.8 hours and apparent clearance is approximately 67 ± 34 L/h under fasted conditions.
Metabolism:Metaxalone is primarily metabolized by CYP1A2, CYP2D6, CYP2E1, and CYP3A4 and, to a lesser extent, CYP2C8, CYP2C9, and CYP2C19.
Excretion:Metaxalone is metabolized by the liver and excreted in the urine as unidentified metabolites.
Specific Populations
The effect of renal impairment and hepatic impairment on metaxalone pharmacokinetics is unknown
[see Use in Specific Populations (
8.6,
8.7)]
.
Geriatric Patients:The effects of age on the pharmacokinetics of metaxalone were determined following administration of 800 mg of metaxalone under fasted and fed conditions. Age had a significantly greater effect on metaxalone pharmacokinetics under fasted conditions than under fed conditions. Bioavailability under fasted conditions increased with age. Metaxalone bioavailability under fasted and fed conditions in the three groups of healthy volunteers of varying age is shown in Table 3.
Table 3: Metaxalone Pharmacokinetic Parameters under Fasted and Fed Conditions in Three Age Groups Following Oral Administration of 800 mg of Metaxalone
|
Mean Age in Years
(± SD) |
26 ± 9 Years Old |
39 ± 11 Years Old |
72 ± 5 Years Old |
|||
| Fasted vs. Fed State | Fasted | Fed | Fasted | Fed | Fasted | Fed |
| C max(ng/mL) 1 | 1816 (43) | 3510 (41) | 2719 (46) | 2915 (55) | 3168 (43) | 3680 (59) |
| T max(hours) 1 | 3 (39) | 4.9 (4.8) | 3 (40) | 8.7 (91) | 2.6 (30) | 6.5 (67) |
| AUC 0-t(ng∙h/mL) 1 | 14531 (47) | 20683 (41) | 19836 (40) | 20482 (37) | 23797 (45) | 24340 (48) |
| AUC ∞(ng∙h/mL) 1 | 15045 (46) | 20833 (41) | 20490 (39) | 20815 (37) | 24194 (44) | 24704 (47) |
1Mean values (% CV)
Male and Female Patients:The exposure of metaxalone was significantly higher in females compared to males as evidenced by C max. (2115 ng/mL versus 1335 ng/mL) and AUC ∞(17884 ng∙h/mL versus 10328 ng∙h/mL) following administration of 800 mg of metaxalone under fasted conditions. The mean half-life was 11.1 hours in females and 7.6 hours in males. The apparent volume of distribution of metaxalone was approximately 22% higher in males than in females.
Drug Interaction Studies
In Vitro Studies:Metaxalone does not inhibit CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP2E1, and CYP3A4. Metaxalone does not induce CYP1A2, CYP2B6, and CYP3A4.
5.1 Serotonin Syndrome
Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of metaxalone (within the recommended dosage range) and other serotonergic drugs [see Drug Interactions ( 7)] and with the use of metaxalone as the only serotonergic drug taken at a dosage higher than the recommended dosage [see Overdosage ( 10)] .
Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, coma), autonomic instability (e.g., tachycardia, labile blood pressure, hyperthermia), neuromuscular aberrations (e.g., hyperreflexia, incoordination, rigidity), and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). The onset of symptoms generally occurs within several hours to a few days after initiation of a serotonergic drug, but may occur later than that.
If concomitant use of metaxalone and another serotoneric drug is warranted, reassess the patient, particularly during treatment initiation and dosage increases. Discontinue metaxalone if serotonin syndrome is suspected or it occurs.
7.1 Serotonergic Drugs
If concomitant use of metaxalone and another serotoneric drug is warranted, carefully observe the patient, particularly during treatment initiation and dosage modification. Discontinue metaxalone if serotonin syndrome is suspected or if it occurs.
Serotonin syndrome has resulted from concomitant use of metaxalone (within the recommended dosage range) with other serotonergic drugs [see Warnings and Precautions ( 5.1) and Adverse Reactions ( 6)] .
Serotonergic drugs include selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, opioids (particularly fentanyl, meperidine, and methadone), drugs that affect the serotonergic neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), and drugs that impair metabolism of serotonin (including monoamine oxidase (MAO) inhibitors, both those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue).
8.6 Hepatic Impairment
Metaxalone is contraindicated in patients with severe hepatic impairment. Metaxalone should be used with caution and additional follow-up should be considered in patients with mild to moderate hepatic impairment. The effect of hepatic impairment on metaxalone pharmacokinetics is unknown; however, metaxalone undergoes expensive hepatic metabolism [see Clinical Pharmacology ( 12.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
Metaxalone is indicated as an adjunct to rest, physical therapy, and other measures for the relief of discomfort associated with acute, painful musculoskeletal conditions in adults and pediatric patients 13 years of age and older.
12.1 Mechanism of Action
Metaxalone’s mechanism of action has not been fully characterized, but may be related to its sedative properties. Metaxalone has no direct action on the contractile mechanism of striated muscle, the motor end plate, or the nerve fiber.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Serotonin Syndrome: Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of metaxalone (within the recommended dosage range) and other serotonergic drugs and with the use of metaxalone as the only serotonergic drug taken at a dosage higher than the recommended dosage. If concomitant use is warranted, carefully observe the patient, particularly during treatment initiation and dosage increases. Discontinue metaxalone if serotonin syndrome is suspected or it occurs. ( 5.1, 7.1)
- Central Nervous System (CNS) Depression: Metaxalone may impair mental and/or physical abilities required for the performance of hazardous tasks, such as operating machinery or driving a motor vehicle, especially when used with other CNS depressants including alcohol. Follow patients closely for signs and symptoms of respiratory depression and sedation. If concomitant use is warranted, closely monitor for signs of respiratory depression and sedation, particularly during treatment initiation and dosage increases. ( 5.2, 7.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended dosage of metaxalone in adults and pediatric patients 13 years of age and older is 800 mg orally three to four times a day [see Use in Specific Populations ( 8)] .
Metaxalone Tablets 800 mg are not substitutable on a mg to mg basis with Metaxalone Tablets, 640 mg [see Clinical Pharmacology ( 12.3)] . When it is appropriate to switch:
- Switch only in patients who have been taking Metaxalone Tablets, 640 mg on an empty stomach.
- Stop Metaxalone Tablets, 640 mg three times a day and start Metaxalone Tablets 800 mg three times a day on an empty stomach, OR stop Metaxalone Tablets 640 mg four times a day and start Metaxalone Tablets 800 mg four times a day on an empty stomach.
Do not switch from Metaxalone Tablets, 640 mg to Metaxalone Tablets, 800 mg when the patient is taking food during administration.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 800 mg pink, capsule shaped, scored tablets, debossed “LCI" on one side and "14" bisect "35" on the other side. The 400 mg strength of metaxalone is not currently marketed by Lannett Company, Inc.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Geriatric Use: Metaxalone-treated geriatric patients may be especially susceptible to CNS depression. ( 8.5)
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Serotonin Syndrome
Inform patients that metaxalone could cause a rare but potentially life-threatening condition called serotonin syndrome. Warn patients of the symptoms of serotonin syndrome and to seek medical attention right away if symptoms develop. Instruct patients to inform their healthcare providers if they are taking, or plan to take, serotonergic drugs
[see Warnings and Precautions (
5.1) and Drug Interactions (
7.1)]
.
Central Nervous System Depression
Advise patients that metaxalone may impair mental and/or physical abilities required for performance of hazardous tasks, such as operating machinery or driving a motor vehicle, especially when used with alcohol and other CNS depressants
[see Drug Interactions (
7.2)]
.
Distributed by:
Lannett Company, Inc.
Philadelphia, PA 19136
CIB71144F
Rev. 06/2024
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Metaxalone Tablets, USP are available as 800 mg pink, capsule shaped, scored tablets, debossed “LCI" on one side and "14" bisect "35" on the other side. Available in bottles of 120 (NDC 85509-1435-1) and in bottles of 30 (NDC 85509-1435-3) and in bottles of 60 (NDC 85509-1435-6) and in bottles of 90 (NDC 85509-1435-9).
Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature].
Repackaged/Relabeled by:
PHOENIX RX LLC
Hatboro, PA 19040
5.2 Central Nervous System Depression
Because of its central nervous system (CNS) depressant effects, metaxalone may impair mental and/or physical abilities required for performance of hazardous tasks, such as operating machinery or driving a motor vehicle, especially when used with other CNS depressants including alcohol. Geriatric patients may be especially susceptible to CNS depression associated with metaxalone use. When used concomitantly, the sedative effects of metaxalone and other CNS depressants (e.g., alcohol, benzodiazepines, opioids, tricyclic antidepressants) may be additive [see Drug Interactions ( 7)] .
Follow metaxalone-treated patients closely for signs and symptoms of respiratory depression and sedation. If concomitant use of metaxalone and another CNS depressant is warranted, closely monitor for signs of respiratory depression and sedation, particularly during treatment initiation and dosage increases.
7.3 Interaction of Metaxalone With Benedict’s Tests (7.3 Interaction of Metaxalone with Benedict’s Tests)
False-positive Benedict's tests, due to an unknown reducing substance, have been noted in metaxalone-treated patients. A glucose-specific test will differentiate findings.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies to evaluate the carcinogenic potential of metaxalone have not been conducted. Studies to evaluate the mutagenic potential of metaxalone have not been conducted. No effects on fertility were observed in rats administered metaxalone.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:54:31.599894 · Updated: 2026-03-14T22:49:04.413893