These Highlights Do Not Include All The Information Needed To Use Bivalirudin For Injection Safely And Effectively. See Full Prescribing Information For Bivalirudin For Injection.
4119b710-ef30-4e3e-b99a-38e42b4b3fca
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Bivalirudin for Injection is indicated for use as an anticoagulant for use in patients undergoing percutaneous coronary intervention (PCI) including patients with heparin-induced thrombocytopenia and heparin-induced thrombocytopenia and thrombosis syndrome.
Indications and Usage
Bivalirudin for Injection is indicated for use as an anticoagulant for use in patients undergoing percutaneous coronary intervention (PCI) including patients with heparin-induced thrombocytopenia and heparin-induced thrombocytopenia and thrombosis syndrome.
Dosage and Administration
The recommended dosage is a 0.75 mg/kg intravenous bolus dose followed immediately by a 1.75 mg/kg/h intravenous infusion for the duration of the procedure. Five minutes after the bolus dose has been administered, an activated clotting time (ACT) should be performed and an additional bolus dose of 0.3 mg/kg should be given if needed. Extending duration of infusion post-procedure up to 4 hours should be considered in patients with ST segment elevation MI (STEMI). ( 2.1 )
Warnings and Precautions
Bleeding Events: Bivalirudin increases the risk of bleeding. ( 5.1 , 6.1 , 12.2 ) Acute Stent Thrombosis: Increased incidence of acute stent thrombosis in STEMI patients undergoing primary PCI. ( 2.1 , 5.2 ) Thrombotic Risk with Coronary Artery Brachytherapy: An increased risk of thrombus formation, including fatal outcomes, in gamma brachytherapy. ( 5.3 )
Contraindications
Bivalirudin is contraindicated in patients with: Active major bleeding; Hypersensitivity (e.g., anaphylaxis) to bivalirudin or its components [see Adverse Reactions ( 6.3 )].
Adverse Reactions
Most common adverse reaction (>2%) was bleeding. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Meitheal Pharmaceuticals, Inc. at 1-844-824-8426 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
In clinical trials in patients undergoing PCI/percutaneous transluminal coronary angioplasty (PTCA), co-administration of bivalirudin with heparin, warfarin, thrombolytics, or GPIs was associated with increased risks of major bleeding events compared to patients not receiving these concomitant medications.
How Supplied
Bivalirudin for Injection is supplied as follows: NDC Bivalirudin for Injection Package Factor 71288- 427 -11 250 mg Single-Dose Vial 10 vials per carton Bivalirudin for Injection is a lyophilized powder. Each vial contains 250 mg of bivalirudin equivalent to an average of 275 mg of bivalirudin trifluoroacetate*. * The range of bivalirudin trifluoroacetate is 266 to 284 mg based on a range of trifluoroacetic acid composition of 1.2 to 2.6 equivalents.
Medication Information
Warnings and Precautions
Bleeding Events: Bivalirudin increases the risk of bleeding. ( 5.1 , 6.1 , 12.2 ) Acute Stent Thrombosis: Increased incidence of acute stent thrombosis in STEMI patients undergoing primary PCI. ( 2.1 , 5.2 ) Thrombotic Risk with Coronary Artery Brachytherapy: An increased risk of thrombus formation, including fatal outcomes, in gamma brachytherapy. ( 5.3 )
Indications and Usage
Bivalirudin for Injection is indicated for use as an anticoagulant for use in patients undergoing percutaneous coronary intervention (PCI) including patients with heparin-induced thrombocytopenia and heparin-induced thrombocytopenia and thrombosis syndrome.
Dosage and Administration
The recommended dosage is a 0.75 mg/kg intravenous bolus dose followed immediately by a 1.75 mg/kg/h intravenous infusion for the duration of the procedure. Five minutes after the bolus dose has been administered, an activated clotting time (ACT) should be performed and an additional bolus dose of 0.3 mg/kg should be given if needed. Extending duration of infusion post-procedure up to 4 hours should be considered in patients with ST segment elevation MI (STEMI). ( 2.1 )
Contraindications
Bivalirudin is contraindicated in patients with: Active major bleeding; Hypersensitivity (e.g., anaphylaxis) to bivalirudin or its components [see Adverse Reactions ( 6.3 )].
Adverse Reactions
Most common adverse reaction (>2%) was bleeding. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Meitheal Pharmaceuticals, Inc. at 1-844-824-8426 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
In clinical trials in patients undergoing PCI/percutaneous transluminal coronary angioplasty (PTCA), co-administration of bivalirudin with heparin, warfarin, thrombolytics, or GPIs was associated with increased risks of major bleeding events compared to patients not receiving these concomitant medications.
How Supplied
Bivalirudin for Injection is supplied as follows: NDC Bivalirudin for Injection Package Factor 71288- 427 -11 250 mg Single-Dose Vial 10 vials per carton Bivalirudin for Injection is a lyophilized powder. Each vial contains 250 mg of bivalirudin equivalent to an average of 275 mg of bivalirudin trifluoroacetate*. * The range of bivalirudin trifluoroacetate is 266 to 284 mg based on a range of trifluoroacetic acid composition of 1.2 to 2.6 equivalents.
Description
Bivalirudin for Injection is indicated for use as an anticoagulant for use in patients undergoing percutaneous coronary intervention (PCI) including patients with heparin-induced thrombocytopenia and heparin-induced thrombocytopenia and thrombosis syndrome.
Section 42229-5
Bolus Dose
No reduction in the bolus dose is needed for any degree of renal impairment.
Section 51945-4
Principal Display Panel – Bivalirudin for Injection 250 mg Vial Label
NDC 71288-427-10
Rx Only
Bivalirudin for Injection
250 mg per vial
For Intravenous Use Only
Single-Dose Vial
Discard unused portion
16.2 Storage
Storage Conditions
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.]
Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
10 Overdosage
Cases of overdose of up to 10 times the recommended bolus or continuous infusion dose of bivalirudin have been reported in clinical trials and in postmarketing reports. A number of the reported overdoses were due to failure to adjust the infusion dose of bivalirudin in persons with renal dysfunction including persons on hemodialysis [see Dosage and Administration (2.2)]. Bleeding, as well as deaths due to hemorrhage, have been observed in some reports of overdose. In cases of suspected overdosage, discontinue bivalirudin immediately and monitor the patient closely for signs of bleeding. There is no known antidote to bivalirudin. Bivalirudin is hemodialyzable [see Clinical Pharmacology (12.3)].
11 Description
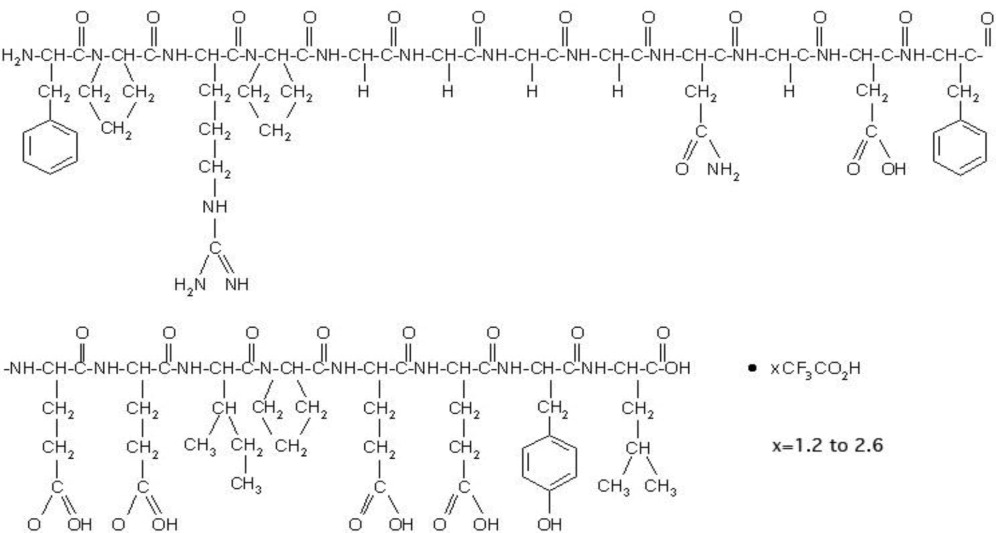
Bivalirudin for Injection contains bivalirudin which is a specific and reversible direct thrombin inhibitor. Bivalirudin is a synthetic, 20 amino acid peptide, with the chemical name of D-phenylalanyl-L-prolyl-L-arginyl-L-prolyl-glycyl-glycyl-glycyl-glycyl-L-asparagyl-glycyl-L-aspartyl-L-phenylalanyl-L-glutamyl-L-glutamyl-L-isoleucyl-L-prolyl-L-glutamyl-L-glutamyl-L-tyrosyl-L-leucine. The active pharmaceutical ingredient is in the form of bivalirudin trifluoroacetate as a white to off-white powder. The chemical name for bivalirudin trifluoroacetate is D-phenylalanyl-L-prolyl-L-arginyl-L-prolyl-glycyl-glycyl-glycyl-glycyl-L-asparagyl-glycyl-L-aspartyl-L-phenylalanyl-L-glutamyl-L-glutamyl-L-isoleucyl-L-prolyl-L-glutamyl-L-glutamyl-L-tyrosyl-L-leucine trifluoroacetate (Figure 1). The molecular weight of bivalirudin is 2180 daltons (anhydrous free base peptide).
Figure 1: Structural formula for bivalirudin
Bivalirudin for Injection is supplied as a sterile white lyophilized cake, in single-dose vials. Each vial contains 250 mg bivalirudin, equivalent to an average of 275 mg of bivalirudin trifluoroacetate*, 125 mg mannitol, and sodium hydroxide to adjust the pH to 5-6 (equivalent of approximately 12.5 mg sodium). When reconstituted with Sterile Water for Injection, the product yields a clear to opalescent, colorless to slightly yellow solution, pH 5-6.
* The range of bivalirudin trifluoroacetate is 266 to 284 mg based on a range of trifluoroacetic acid composition of 1.2 to 2.6 equivalents.
16.1 How Supplied
Bivalirudin for Injection is supplied as follows:
| NDC | Bivalirudin for Injection | Package Factor |
| 71288-427-11 | 250 mg Single-Dose Vial | 10 vials per carton |
Bivalirudin for Injection is a lyophilized powder. Each vial contains 250 mg of bivalirudin equivalent to an average of 275 mg of bivalirudin trifluoroacetate*.
* The range of bivalirudin trifluoroacetate is 266 to 284 mg based on a range of trifluoroacetic acid composition of 1.2 to 2.6 equivalents.
8.4 Pediatric Use
The safety and effectiveness of bivalirudin in pediatric patients have not been established.
8.5 Geriatric Use
In studies of patients undergoing PCI, 44% were ≥65 years of age and 12% of patients were ≥75 years old. Elderly patients experienced more bleeding events than younger patients.
6.2 Immunogenicity
As with all peptides, there is potential for immunogenicity. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to bivalirudin in the studies described below with the incidence of antibodies in other studies or to other products may be misleading.
In in vitro studies, bivalirudin exhibited no platelet aggregation response against sera from patients with a history of HIT/HITTS.
Among 494 subjects who received bivalirudin in clinical trials and were tested for antibodies, 2 subjects had treatment-emergent positive bivalirudin antibody tests. Neither subject demonstrated clinical evidence of allergic or anaphylactic reactions and repeat testing was not performed. Nine additional patients who had initial positive tests were negative on repeat testing.
4 Contraindications
Bivalirudin is contraindicated in patients with:
- Active major bleeding;
- Hypersensitivity (e.g., anaphylaxis) to bivalirudin or its components [see Adverse Reactions (6.3)].
5.1 Bleeding Events
Bivalirudin for injection increases the risk of bleeding [see Adverse Reactions (6.1)]. An unexplained fall in blood pressure or hematocrit should lead to serious consideration of a hemorrhagic event and cessation of bivalirudin for injection administration. Monitor patients receiving bivalirudin for injection for signs and symptoms of bleeding. Monitor patients with disease states associated with an increased risk of bleeding more frequently for bleeding.
6 Adverse Reactions
Most common adverse reaction (>2%) was bleeding. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Meitheal Pharmaceuticals, Inc. at 1-844-824-8426 or FDA at 1-800-FDA-1088 or
www.fda.gov/medwatch
.
7 Drug Interactions
In clinical trials in patients undergoing PCI/percutaneous transluminal coronary angioplasty (PTCA), co-administration of bivalirudin with heparin, warfarin, thrombolytics, or GPIs was associated with increased risks of major bleeding events compared to patients not receiving these concomitant medications.
8.6 Renal Impairment
The disposition of bivalirudin was studied in PTCA patients with mild, moderate and severe renal impairment. The clearance of bivalirudin was reduced approximately 21% in patients with moderate and severe renal impairment and was reduced approximately 70% in dialysis-dependent patients [see Clinical Pharmacology (12.3)]. Reduce the infusion dose of bivalirudin and monitor the anticoagulant status more frequently in patients with renal impairment creatinine clearance less than 30 mL/min (by Cockcroft Gault equation) [see Dosage and Administration (2.2)].
12.2 Pharmacodynamics
In healthy volunteers and patients (with ≥70% vessel occlusion undergoing routine PTCA), bivalirudin exhibited dose- and concentration-dependent anticoagulant activity as evidenced by prolongation of the ACT, aPTT, PT, and TT. Intravenous administration of bivalirudin produces an immediate anticoagulant effect. Coagulation times return to baseline approximately 1 hour following cessation of bivalirudin administration. Bivalirudin also increases INR. Therefore INR measurements made in bivalirudin treated patients may not be useful for determining the appropriate dose of warfarin.
In 291 patients with ≥70% vessel occlusion undergoing routine PTCA, a positive correlation was observed between the dose of bivalirudin and the proportion of patients achieving ACT values of 300 sec or 350 sec. At a bivalirudin dose of 1 mg/kg IV bolus plus 2.5 mg/kg/h IV infusion for 4 hours, followed by 0.2 mg/kg/h, all patients reached maximal ACT values >300 sec.
12.3 Pharmacokinetics
Bivalirudin exhibits linear pharmacokinetics following IV administration to patients undergoing PTCA. In these patients, a mean steady state bivalirudin concentration of 12.3 ± 1.7 mcg/mL is achieved following an IV bolus of 1 mg/kg and a 4-hour 2.5 mg/kg/h IV infusion.
2.1 Recommended Dosage
Bivalirudin for injection has been studied only in patients receiving concomitant aspirin.
The recommended dose of bivalirudin for injection is an intravenous bolus dose of 0.75 mg/kg, followed immediately by an infusion of 1.75 mg/kg/h for the duration of the procedure. Five minutes after the bolus dose has been administered, an activated clotting time (ACT) should be performed and an additional bolus of 0.3 mg/kg should be given if needed.
Extended duration of infusion following PCI at 1.75 mg/kg/h for up to 4 hours post-procedure should be considered in patients with ST segment elevation MI (STEMI).
1 Indications and Usage
Bivalirudin for Injection is indicated for use as an anticoagulant for use in patients undergoing percutaneous coronary intervention (PCI) including patients with heparin-induced thrombocytopenia and heparin-induced thrombocytopenia and thrombosis syndrome.
12.1 Mechanism of Action
Bivalirudin directly inhibits thrombin by specifically binding both to the catalytic site and to the anion-binding exosite of circulating and clot-bound thrombin. Thrombin is a serine proteinase that plays a central role in the thrombotic process, acting to cleave fibrinogen into fibrin monomers and to activate Factor XIII to Factor XIIIa, allowing fibrin to develop a covalently cross-linked framework which stabilizes the thrombus; thrombin also activates Factors V and VIII, promoting further thrombin generation, and activates platelets, stimulating aggregation and granule release. The binding of bivalirudin to thrombin is reversible as thrombin slowly cleaves the bivalirudin-Arg3-Pro4 bond, resulting in recovery of thrombin active site functions.
In in vitro studies, bivalirudin inhibited both soluble (free) and clot-bound thrombin, was not neutralized by products of the platelet release reaction, and prolonged the activated partial thromboplastin time (aPTT), thrombin time (TT), and prothrombin time (PT) of normal human plasma in a concentration-dependent manner. The clinical relevance of these findings is unknown.
5 Warnings and Precautions
- Bleeding Events: Bivalirudin increases the risk of bleeding. (5.1, 6.1, 12.2)
- Acute Stent Thrombosis: Increased incidence of acute stent thrombosis in STEMI patients undergoing primary PCI. (2.1, 5.2)
- Thrombotic Risk with Coronary Artery Brachytherapy: An increased risk of thrombus formation, including fatal outcomes, in gamma brachytherapy. (5.3)
2 Dosage and Administration
- The recommended dosage is a 0.75 mg/kg intravenous bolus dose followed immediately by a 1.75 mg/kg/h intravenous infusion for the duration of the procedure. Five minutes after the bolus dose has been administered, an activated clotting time (ACT) should be performed and an additional bolus dose of 0.3 mg/kg should be given if needed.
- Extending duration of infusion post-procedure up to 4 hours should be considered in patients with ST segment elevation MI (STEMI). (2.1)
3 Dosage Forms and Strengths
Bivalirudin for Injection: 250 mg of bivalirudin as a lyophilized powder in a single-dose vial for reconstitution. Each vial contains 250 mg of bivalirudin equivalent to an average of 275 mg bivalirudin trifluoroacetate*.
* The range of bivalirudin trifluoroacetate is 266 to 284 mg based on a range of trifluoroacetic acid composition of 1.2 to 2.6 equivalents.
6.3 Postmarketing Experience
Because postmarketing adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been identified during post approval use of bivalirudin: fatal bleeding; hypersensitivity and allergic reactions including reports of anaphylaxis; lack of anticoagulant effect; thrombus formation during PCI with and without intracoronary brachytherapy, including reports of fatal outcomes; pulmonary hemorrhage; cardiac tamponade; and INR increased.
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In the BAT trials, 79 of the 2161 (3.7%) patients undergoing PCI for treatment of unstable angina and randomized to bivalirudin experienced major bleeding events which consisted of: intracranial bleeding, retroperitoneal bleeding, and clinically overt bleeding with a decrease in hemoglobin >3 g/dL or leading to a transfusion of >2 units of blood.
2.4 Storage After Reconstitution
Do not freeze reconstituted or diluted bivalirudin for injection. Reconstituted material may be stored at 2 to 8°C for up to 24 hours. Diluted bivalirudin for injection with a concentration of between 0.5 mg per mL and 5 mg per mL is stable at room temperature for up to 24 hours. Discard any unused portion of reconstituted solution remaining in the vial.
17 Patient Counseling Information
Advise patients to watch carefully for any signs of bleeding or bruising and to report these to their health care provider when they occur.
meitheal®
Mfd. for Meitheal Pharmaceuticals
Chicago, IL 60631 (USA)
©2020 Meitheal Pharmaceuticals Inc.
Revised: October 2020
810077-00
2.3 Instructions for Preparation and Administration
Bivalirudin for injection is intended for intravenous bolus injection and continuous infusion after reconstitution and dilution.
Preparation Instructions for Bolus Injection and Continuous Infusion
- To each 250 mg vial, add 5 mL of Sterile Water for Injection, USP.
- Gently swirl until all material is dissolved.
- Withdraw and discard 5 mL from a 50 mL infusion bag containing 5% Dextrose in Water or 0.9% Sodium Chloride for Injection.
- Add the contents of the reconstituted vial to the infusion bag containing 5% Dextrose in Water or 0.9% Sodium Chloride for Injection to yield a final concentration of 5 mg per mL (e.g., 1 vial in 50 mL; 2 vials in 100 mL; 5 vials in 250 mL).
- Adjust the dose to be administered according to the patient's weight (see Table 1).
|
Weight
(kg) |
Using 5 mg per mL
Concentration |
|
| Bolus | Infusion | |
| 0.75 mg/kg | 1.75 mg/kg/h | |
| (mL) | (mL/h) | |
| 43-47 | 7 | 16 |
| 48-52 | 7.5 | 17.5 |
| 53-57 | 8 | 19 |
| 58-62 | 9 | 21 |
| 63-67 | 10 | 23 |
| 68-72 | 10.5 | 24.5 |
| 73-77 | 11 | 26 |
| 78-82 | 12 | 28 |
| 83-87 | 13 | 30 |
| 88-92 | 13.5 | 31.5 |
| 93-97 | 14 | 33 |
| 98-102 | 15 | 35 |
| 103-107 | 16 | 37 |
| 108-112 | 16.5 | 38.5 |
| 113-117 | 17 | 40 |
| 118-122 | 18 | 42 |
| 123-127 | 19 | 44 |
| 128-132 | 19.5 | 45.5 |
| 133-137 | 20 | 47 |
| 138-142 | 21 | 49 |
| 143-147 | 22 | 51 |
| 148-152 | 22.5 | 52.5 |
5.3 Thrombotic Risk With Coronary Artery Brachytherapy
An increased risk of thrombus formation, including fatal outcomes, has been associated with the use of bivalirudin in gamma brachytherapy.
If a decision is made to use bivalirudin during brachytherapy procedures, maintain meticulous catheter technique, with frequent aspiration and flushing, paying special attention to minimizing conditions of stasis within the catheter or vessels [see Adverse Reactions (6.1)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term studies in animals have been performed to evaluate the carcinogenic potential of bivalirudin. Bivalirudin displayed no genotoxic potential in the in vitro bacterial cell reverse mutation assay (Ames test), the in vitro Chinese hamster ovary cell forward gene mutation test (CHO/HGPRT), the in vitro human lymphocyte chromosomal aberration assay, the in vitro rat hepatocyte unscheduled DNA synthesis (UDS) assay, and the in vivo rat micronucleus assay. Fertility and general reproductive performance in rats were unaffected by subcutaneous doses of bivalirudin up to 150 mg/kg/day, about 1.6 times the dose on a body surface area basis (mg/m2) of a 50 kg person given the maximum recommended dose of 15 mg/kg/day.
5.2 Acute Stent Thrombosis in Patients With Stemi Undergoing Pci
Acute stent thrombosis (AST) (<4 hours) has been observed at a greater frequency in bivalirudin treated patients (1.2%, 36/2889) compared to heparin treated patients (0.2%, 6/2911) with STEMI undergoing primary PCI. Among patients who experienced an AST, one fatality (0.03%) occurred in an bivalirudin treated patient and one fatality (0.03%) in a heparin treated patient. These patients have been managed by Target Vessel Revascularization (TVR). Patients should remain for at least 24 hours in a facility capable of managing ischemic complications and should be carefully monitored following primary PCI for signs and symptoms consistent with myocardial ischemia.
Structured Label Content
Section 42229-5 (42229-5)
Bolus Dose
No reduction in the bolus dose is needed for any degree of renal impairment.
Section 51945-4 (51945-4)
Principal Display Panel – Bivalirudin for Injection 250 mg Vial Label
NDC 71288-427-10
Rx Only
Bivalirudin for Injection
250 mg per vial
For Intravenous Use Only
Single-Dose Vial
Discard unused portion
16.2 Storage
Storage Conditions
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.]
Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
10 Overdosage (10 OVERDOSAGE)
Cases of overdose of up to 10 times the recommended bolus or continuous infusion dose of bivalirudin have been reported in clinical trials and in postmarketing reports. A number of the reported overdoses were due to failure to adjust the infusion dose of bivalirudin in persons with renal dysfunction including persons on hemodialysis [see Dosage and Administration (2.2)]. Bleeding, as well as deaths due to hemorrhage, have been observed in some reports of overdose. In cases of suspected overdosage, discontinue bivalirudin immediately and monitor the patient closely for signs of bleeding. There is no known antidote to bivalirudin. Bivalirudin is hemodialyzable [see Clinical Pharmacology (12.3)].
11 Description (11 DESCRIPTION)
Bivalirudin for Injection contains bivalirudin which is a specific and reversible direct thrombin inhibitor. Bivalirudin is a synthetic, 20 amino acid peptide, with the chemical name of D-phenylalanyl-L-prolyl-L-arginyl-L-prolyl-glycyl-glycyl-glycyl-glycyl-L-asparagyl-glycyl-L-aspartyl-L-phenylalanyl-L-glutamyl-L-glutamyl-L-isoleucyl-L-prolyl-L-glutamyl-L-glutamyl-L-tyrosyl-L-leucine. The active pharmaceutical ingredient is in the form of bivalirudin trifluoroacetate as a white to off-white powder. The chemical name for bivalirudin trifluoroacetate is D-phenylalanyl-L-prolyl-L-arginyl-L-prolyl-glycyl-glycyl-glycyl-glycyl-L-asparagyl-glycyl-L-aspartyl-L-phenylalanyl-L-glutamyl-L-glutamyl-L-isoleucyl-L-prolyl-L-glutamyl-L-glutamyl-L-tyrosyl-L-leucine trifluoroacetate (Figure 1). The molecular weight of bivalirudin is 2180 daltons (anhydrous free base peptide).
Figure 1: Structural formula for bivalirudin
Bivalirudin for Injection is supplied as a sterile white lyophilized cake, in single-dose vials. Each vial contains 250 mg bivalirudin, equivalent to an average of 275 mg of bivalirudin trifluoroacetate*, 125 mg mannitol, and sodium hydroxide to adjust the pH to 5-6 (equivalent of approximately 12.5 mg sodium). When reconstituted with Sterile Water for Injection, the product yields a clear to opalescent, colorless to slightly yellow solution, pH 5-6.
* The range of bivalirudin trifluoroacetate is 266 to 284 mg based on a range of trifluoroacetic acid composition of 1.2 to 2.6 equivalents.
16.1 How Supplied
Bivalirudin for Injection is supplied as follows:
| NDC | Bivalirudin for Injection | Package Factor |
| 71288-427-11 | 250 mg Single-Dose Vial | 10 vials per carton |
Bivalirudin for Injection is a lyophilized powder. Each vial contains 250 mg of bivalirudin equivalent to an average of 275 mg of bivalirudin trifluoroacetate*.
* The range of bivalirudin trifluoroacetate is 266 to 284 mg based on a range of trifluoroacetic acid composition of 1.2 to 2.6 equivalents.
8.4 Pediatric Use
The safety and effectiveness of bivalirudin in pediatric patients have not been established.
8.5 Geriatric Use
In studies of patients undergoing PCI, 44% were ≥65 years of age and 12% of patients were ≥75 years old. Elderly patients experienced more bleeding events than younger patients.
6.2 Immunogenicity
As with all peptides, there is potential for immunogenicity. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to bivalirudin in the studies described below with the incidence of antibodies in other studies or to other products may be misleading.
In in vitro studies, bivalirudin exhibited no platelet aggregation response against sera from patients with a history of HIT/HITTS.
Among 494 subjects who received bivalirudin in clinical trials and were tested for antibodies, 2 subjects had treatment-emergent positive bivalirudin antibody tests. Neither subject demonstrated clinical evidence of allergic or anaphylactic reactions and repeat testing was not performed. Nine additional patients who had initial positive tests were negative on repeat testing.
4 Contraindications (4 CONTRAINDICATIONS)
Bivalirudin is contraindicated in patients with:
- Active major bleeding;
- Hypersensitivity (e.g., anaphylaxis) to bivalirudin or its components [see Adverse Reactions (6.3)].
5.1 Bleeding Events
Bivalirudin for injection increases the risk of bleeding [see Adverse Reactions (6.1)]. An unexplained fall in blood pressure or hematocrit should lead to serious consideration of a hemorrhagic event and cessation of bivalirudin for injection administration. Monitor patients receiving bivalirudin for injection for signs and symptoms of bleeding. Monitor patients with disease states associated with an increased risk of bleeding more frequently for bleeding.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reaction (>2%) was bleeding. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Meitheal Pharmaceuticals, Inc. at 1-844-824-8426 or FDA at 1-800-FDA-1088 or
www.fda.gov/medwatch
.
7 Drug Interactions (7 DRUG INTERACTIONS)
In clinical trials in patients undergoing PCI/percutaneous transluminal coronary angioplasty (PTCA), co-administration of bivalirudin with heparin, warfarin, thrombolytics, or GPIs was associated with increased risks of major bleeding events compared to patients not receiving these concomitant medications.
8.6 Renal Impairment
The disposition of bivalirudin was studied in PTCA patients with mild, moderate and severe renal impairment. The clearance of bivalirudin was reduced approximately 21% in patients with moderate and severe renal impairment and was reduced approximately 70% in dialysis-dependent patients [see Clinical Pharmacology (12.3)]. Reduce the infusion dose of bivalirudin and monitor the anticoagulant status more frequently in patients with renal impairment creatinine clearance less than 30 mL/min (by Cockcroft Gault equation) [see Dosage and Administration (2.2)].
12.2 Pharmacodynamics
In healthy volunteers and patients (with ≥70% vessel occlusion undergoing routine PTCA), bivalirudin exhibited dose- and concentration-dependent anticoagulant activity as evidenced by prolongation of the ACT, aPTT, PT, and TT. Intravenous administration of bivalirudin produces an immediate anticoagulant effect. Coagulation times return to baseline approximately 1 hour following cessation of bivalirudin administration. Bivalirudin also increases INR. Therefore INR measurements made in bivalirudin treated patients may not be useful for determining the appropriate dose of warfarin.
In 291 patients with ≥70% vessel occlusion undergoing routine PTCA, a positive correlation was observed between the dose of bivalirudin and the proportion of patients achieving ACT values of 300 sec or 350 sec. At a bivalirudin dose of 1 mg/kg IV bolus plus 2.5 mg/kg/h IV infusion for 4 hours, followed by 0.2 mg/kg/h, all patients reached maximal ACT values >300 sec.
12.3 Pharmacokinetics
Bivalirudin exhibits linear pharmacokinetics following IV administration to patients undergoing PTCA. In these patients, a mean steady state bivalirudin concentration of 12.3 ± 1.7 mcg/mL is achieved following an IV bolus of 1 mg/kg and a 4-hour 2.5 mg/kg/h IV infusion.
2.1 Recommended Dosage
Bivalirudin for injection has been studied only in patients receiving concomitant aspirin.
The recommended dose of bivalirudin for injection is an intravenous bolus dose of 0.75 mg/kg, followed immediately by an infusion of 1.75 mg/kg/h for the duration of the procedure. Five minutes after the bolus dose has been administered, an activated clotting time (ACT) should be performed and an additional bolus of 0.3 mg/kg should be given if needed.
Extended duration of infusion following PCI at 1.75 mg/kg/h for up to 4 hours post-procedure should be considered in patients with ST segment elevation MI (STEMI).
1 Indications and Usage (1 INDICATIONS AND USAGE)
Bivalirudin for Injection is indicated for use as an anticoagulant for use in patients undergoing percutaneous coronary intervention (PCI) including patients with heparin-induced thrombocytopenia and heparin-induced thrombocytopenia and thrombosis syndrome.
12.1 Mechanism of Action
Bivalirudin directly inhibits thrombin by specifically binding both to the catalytic site and to the anion-binding exosite of circulating and clot-bound thrombin. Thrombin is a serine proteinase that plays a central role in the thrombotic process, acting to cleave fibrinogen into fibrin monomers and to activate Factor XIII to Factor XIIIa, allowing fibrin to develop a covalently cross-linked framework which stabilizes the thrombus; thrombin also activates Factors V and VIII, promoting further thrombin generation, and activates platelets, stimulating aggregation and granule release. The binding of bivalirudin to thrombin is reversible as thrombin slowly cleaves the bivalirudin-Arg3-Pro4 bond, resulting in recovery of thrombin active site functions.
In in vitro studies, bivalirudin inhibited both soluble (free) and clot-bound thrombin, was not neutralized by products of the platelet release reaction, and prolonged the activated partial thromboplastin time (aPTT), thrombin time (TT), and prothrombin time (PT) of normal human plasma in a concentration-dependent manner. The clinical relevance of these findings is unknown.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Bleeding Events: Bivalirudin increases the risk of bleeding. (5.1, 6.1, 12.2)
- Acute Stent Thrombosis: Increased incidence of acute stent thrombosis in STEMI patients undergoing primary PCI. (2.1, 5.2)
- Thrombotic Risk with Coronary Artery Brachytherapy: An increased risk of thrombus formation, including fatal outcomes, in gamma brachytherapy. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- The recommended dosage is a 0.75 mg/kg intravenous bolus dose followed immediately by a 1.75 mg/kg/h intravenous infusion for the duration of the procedure. Five minutes after the bolus dose has been administered, an activated clotting time (ACT) should be performed and an additional bolus dose of 0.3 mg/kg should be given if needed.
- Extending duration of infusion post-procedure up to 4 hours should be considered in patients with ST segment elevation MI (STEMI). (2.1)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Bivalirudin for Injection: 250 mg of bivalirudin as a lyophilized powder in a single-dose vial for reconstitution. Each vial contains 250 mg of bivalirudin equivalent to an average of 275 mg bivalirudin trifluoroacetate*.
* The range of bivalirudin trifluoroacetate is 266 to 284 mg based on a range of trifluoroacetic acid composition of 1.2 to 2.6 equivalents.
6.3 Postmarketing Experience
Because postmarketing adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been identified during post approval use of bivalirudin: fatal bleeding; hypersensitivity and allergic reactions including reports of anaphylaxis; lack of anticoagulant effect; thrombus formation during PCI with and without intracoronary brachytherapy, including reports of fatal outcomes; pulmonary hemorrhage; cardiac tamponade; and INR increased.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In the BAT trials, 79 of the 2161 (3.7%) patients undergoing PCI for treatment of unstable angina and randomized to bivalirudin experienced major bleeding events which consisted of: intracranial bleeding, retroperitoneal bleeding, and clinically overt bleeding with a decrease in hemoglobin >3 g/dL or leading to a transfusion of >2 units of blood.
2.4 Storage After Reconstitution (2.4 Storage after Reconstitution)
Do not freeze reconstituted or diluted bivalirudin for injection. Reconstituted material may be stored at 2 to 8°C for up to 24 hours. Diluted bivalirudin for injection with a concentration of between 0.5 mg per mL and 5 mg per mL is stable at room temperature for up to 24 hours. Discard any unused portion of reconstituted solution remaining in the vial.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients to watch carefully for any signs of bleeding or bruising and to report these to their health care provider when they occur.
meitheal®
Mfd. for Meitheal Pharmaceuticals
Chicago, IL 60631 (USA)
©2020 Meitheal Pharmaceuticals Inc.
Revised: October 2020
810077-00
2.3 Instructions for Preparation and Administration
Bivalirudin for injection is intended for intravenous bolus injection and continuous infusion after reconstitution and dilution.
Preparation Instructions for Bolus Injection and Continuous Infusion
- To each 250 mg vial, add 5 mL of Sterile Water for Injection, USP.
- Gently swirl until all material is dissolved.
- Withdraw and discard 5 mL from a 50 mL infusion bag containing 5% Dextrose in Water or 0.9% Sodium Chloride for Injection.
- Add the contents of the reconstituted vial to the infusion bag containing 5% Dextrose in Water or 0.9% Sodium Chloride for Injection to yield a final concentration of 5 mg per mL (e.g., 1 vial in 50 mL; 2 vials in 100 mL; 5 vials in 250 mL).
- Adjust the dose to be administered according to the patient's weight (see Table 1).
|
Weight
(kg) |
Using 5 mg per mL
Concentration |
|
| Bolus | Infusion | |
| 0.75 mg/kg | 1.75 mg/kg/h | |
| (mL) | (mL/h) | |
| 43-47 | 7 | 16 |
| 48-52 | 7.5 | 17.5 |
| 53-57 | 8 | 19 |
| 58-62 | 9 | 21 |
| 63-67 | 10 | 23 |
| 68-72 | 10.5 | 24.5 |
| 73-77 | 11 | 26 |
| 78-82 | 12 | 28 |
| 83-87 | 13 | 30 |
| 88-92 | 13.5 | 31.5 |
| 93-97 | 14 | 33 |
| 98-102 | 15 | 35 |
| 103-107 | 16 | 37 |
| 108-112 | 16.5 | 38.5 |
| 113-117 | 17 | 40 |
| 118-122 | 18 | 42 |
| 123-127 | 19 | 44 |
| 128-132 | 19.5 | 45.5 |
| 133-137 | 20 | 47 |
| 138-142 | 21 | 49 |
| 143-147 | 22 | 51 |
| 148-152 | 22.5 | 52.5 |
5.3 Thrombotic Risk With Coronary Artery Brachytherapy (5.3 Thrombotic Risk with Coronary Artery Brachytherapy)
An increased risk of thrombus formation, including fatal outcomes, has been associated with the use of bivalirudin in gamma brachytherapy.
If a decision is made to use bivalirudin during brachytherapy procedures, maintain meticulous catheter technique, with frequent aspiration and flushing, paying special attention to minimizing conditions of stasis within the catheter or vessels [see Adverse Reactions (6.1)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term studies in animals have been performed to evaluate the carcinogenic potential of bivalirudin. Bivalirudin displayed no genotoxic potential in the in vitro bacterial cell reverse mutation assay (Ames test), the in vitro Chinese hamster ovary cell forward gene mutation test (CHO/HGPRT), the in vitro human lymphocyte chromosomal aberration assay, the in vitro rat hepatocyte unscheduled DNA synthesis (UDS) assay, and the in vivo rat micronucleus assay. Fertility and general reproductive performance in rats were unaffected by subcutaneous doses of bivalirudin up to 150 mg/kg/day, about 1.6 times the dose on a body surface area basis (mg/m2) of a 50 kg person given the maximum recommended dose of 15 mg/kg/day.
5.2 Acute Stent Thrombosis in Patients With Stemi Undergoing Pci (5.2 Acute Stent Thrombosis in Patients with STEMI Undergoing PCI)
Acute stent thrombosis (AST) (<4 hours) has been observed at a greater frequency in bivalirudin treated patients (1.2%, 36/2889) compared to heparin treated patients (0.2%, 6/2911) with STEMI undergoing primary PCI. Among patients who experienced an AST, one fatality (0.03%) occurred in an bivalirudin treated patient and one fatality (0.03%) in a heparin treated patient. These patients have been managed by Target Vessel Revascularization (TVR). Patients should remain for at least 24 hours in a facility capable of managing ischemic complications and should be carefully monitored following primary PCI for signs and symptoms consistent with myocardial ischemia.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:36:08.430900 · Updated: 2026-03-14T21:45:28.175902