87111-001 Hand Sanitizer
40b6c96c-e36f-6588-e063-6394a90adb46
34390-5
HUMAN OTC DRUG LABEL
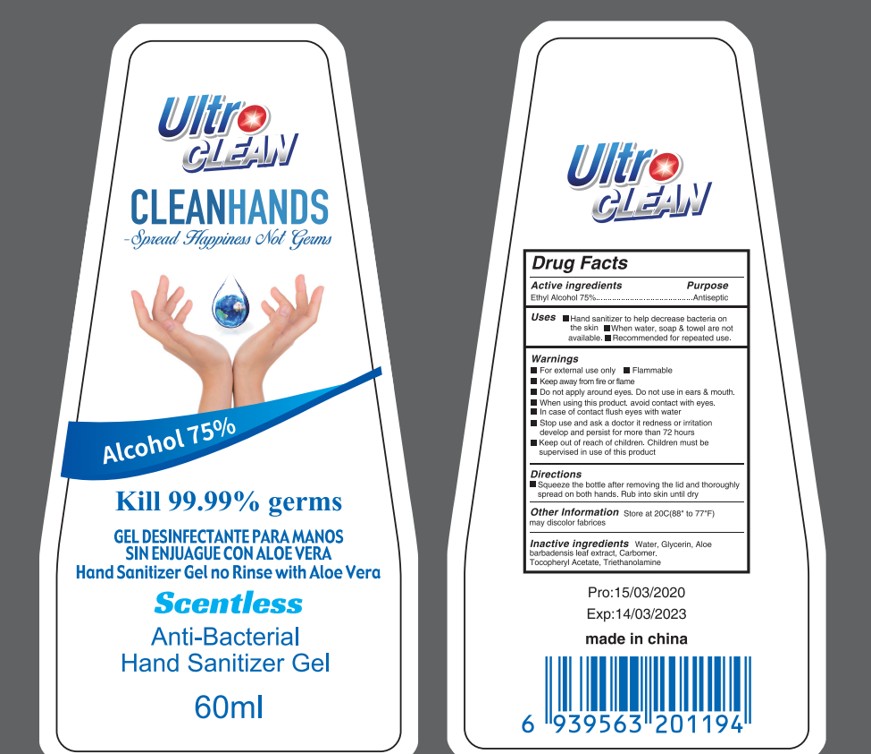
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ethyil Alcohol 75%
Medication Information
Warnings and Precautions
For external use only
Flammable
Keep away from fire or flame
Indications and Usage
Uses
Hand sanitizer to help decrease bacteria on the skin
When water, soap & towel are not available.
Recommended for repeated use.
Dosage and Administration
Pump as needed into your palms and thoroughly spread on both hands. Rub into skin until dry
Description
Ethyil Alcohol 75%
Section 50565-1
Keep out of reach of children. Children must be supervised in use of this product
Section 50566-9
Stop use and ask a doctor it redness or irritation develop and persist for more than 72 hours
Section 50567-7
When using this product. avoid contact with eyes.
In case of contact flush eyes with water
Section 50570-1
Do not apply around eyes.
Do not use in ears & mouth.
Section 51727-6
WATER
Tocopheryl Acetate
GLYERIN
Section 51945-4
Section 55105-1
Antiseptic
Section 55106-9
Ethyil Alcohol 75%
Structured Label Content
Indications and Usage (34067-9)
Uses
Hand sanitizer to help decrease bacteria on the skin
When water, soap & towel are not available.
Recommended for repeated use.
Dosage and Administration (34068-7)
Pump as needed into your palms and thoroughly spread on both hands. Rub into skin until dry
Warnings and Precautions (34071-1)
For external use only
Flammable
Keep away from fire or flame
Section 50565-1 (50565-1)
Keep out of reach of children. Children must be supervised in use of this product
Section 50566-9 (50566-9)
Stop use and ask a doctor it redness or irritation develop and persist for more than 72 hours
Section 50567-7 (50567-7)
When using this product. avoid contact with eyes.
In case of contact flush eyes with water
Section 50570-1 (50570-1)
Do not apply around eyes.
Do not use in ears & mouth.
Section 51727-6 (51727-6)
WATER
Tocopheryl Acetate
GLYERIN
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Antiseptic
Section 55106-9 (55106-9)
Ethyil Alcohol 75%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:39.687844 · Updated: 2026-03-14T23:12:58.166895