Anafranil™
4074b555-7635-41a9-809d-fae3b3610059
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Suicidality and Antidepressant Drugs Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of clomipramine hydrochloride or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Clomipramine hydrochloride is not approved for use in pediatric patients except for patients with obsessive compulsive disorder (OCD) ( see WARNINGS, Clinical Worsening and Suicide Risk ; PRECAUTIONS, Information for Patients ; and PRECAUTIONS, Pediatric Use ).
Indications and Usage
Anafranil ™ (clomipramine hydrochloride) Capsules USP is indicated for the treatment of obsessions and compulsions in patients with Obsessive-Compulsive Disorder (OCD). The obsessions or compulsions must cause marked distress, be time-consuming, or significantly interfere with social or occupational functioning, in order to meet the DSM-III-R (circa 1989) diagnosis of OCD. Obsessions are recurrent, persistent ideas, thoughts, images, or impulses that are ego-dystonic. Compulsions are repetitive, purposeful, and intentional behaviors performed in response to an obsession or in a stereotyped fashion, and are recognized by the person as excessive or unreasonable. The effectiveness of Anafranil for the treatment of OCD was demonstrated in multicenter, placebo-controlled, parallel-group studies, including two 10-week studies in adults and one 8-week study in children and adolescents 10 to 17 years of age. Patients in all studies had moderate-to-severe OCD (DSM-III), with mean baseline ratings on the Yale-Brown Obsessive Compulsive Scale (YBOCS) ranging from 26 to 28 and a mean baseline rating of 10 on the NIMH Clinical Global Obsessive Compulsive Scale (NIMH-OC). Patients taking CMI experienced a mean reduction of approximately 10 on the YBOCS, representing an average improvement on this scale of 35% to 42% among adults and 37% among children and adolescents. CMI-treated patients experienced a 3.5 unit decrement on the NIMH-OC. Patients on placebo showed no important clinical response on either scale. The maximum dose was 250 mg/day for most adults and 3 mg/kg/day (up to 200 mg) for all children and adolescents. The effectiveness of Anafranil for long-term use (i.e., for more than 10 weeks) has not been systematically evaluated in placebo-controlled trials. The physician who elects to use Anafranil for extended periods should periodically reevaluate the long-term usefulness of the drug for the individual patient ( see DOSAGE AND ADMINISTRATION ).
Dosage and Administration
The treatment regimens described below are based on those used in controlled clinical trials of Anafranil in 520 adults, and 91 children and adolescents with OCD. During initial titration, Anafranil should be given in divided doses with meals to reduce gastrointestinal side effects. The goal of this initial titration phase is to minimize side effects by permitting tolerance to side effects to develop or allowing the patient time to adapt if tolerance does not develop. Because both CMI and its active metabolite, DMI, have long elimination half-lives, the prescriber should take into consideration the fact that steady-state plasma levels may not be achieved until 2 to 3 weeks after dosage change ( see CLINICAL PHARMACOLOGY ). Therefore, after initial titration, it may be appropriate to wait 2 to 3 weeks between further dosage adjustments.
Contraindications
Anafranil is contraindicated in patients with a history of hypersensitivity to Anafranil or other tricyclic antidepressants. Monoamine Oxidase Inhibitors (MAOIs) The use of MAOIs intended to treat psychiatric disorders with Anafranil or within 14 days of stopping treatment with Anafranil is contraindicated because of an increased risk of serotonin syndrome. The use of Anafranil within 14 days of stopping an MAOI intended to treat psychiatric disorders is also contraindicated ( see WARNINGS and DOSAGE AND ADMINISTRATION ). Starting Anafranil in a patient who is being treated with linezolid or intravenous methylene blue is also contraindicated because of an increased risk of serotonin syndrome ( see WARNINGS and DOSAGE AND ADMINISTRATION ). Myocardial Infarction Anafranil is contraindicated during the acute recovery period after a myocardial infarction.
Drug Interactions
The risks of using Anafranil in combination with other drugs have not been systematically evaluated. Given the primary CNS effects of Anafranil, caution is advised in using it concomitantly with other CNS-active drugs ( see Information for Patients ). Anafranil should not be used with MAO inhibitors ( see CONTRAINDICATIONS ). Close supervision and careful adjustment of dosage are required when Anafranil is administered with anticholinergic or sympathomimetic drugs. Several tricyclic antidepressants have been reported to block the pharmacologic effects of guanethidine, clonidine, or similar agents, and such an effect may be anticipated with CMI because of its structural similarity to other tricyclic antidepressants. The plasma concentration of CMI has been reported to be increased by the concomitant administration of haloperidol; plasma levels of several closely related tricyclic antidepressants have been reported to be increased by the concomitant administration of methylphenidate or hepatic enzyme inhibitors (e.g., cimetidine, fluoxetine) and decreased by the concomitant administration of hepatic enzyme inducers (e.g., barbiturates, phenytoin), and such an effect may be anticipated with CMI as well. Administration of CMI has been reported to increase the plasma levels of phenobarbital, if given concomitantly ( see CLINICAL PHARMACOLOGY, Interactions ).
How Supplied
Anafranil™ (clomipramine hydrochloride) Capsules USP Capsules 25 mg – ivory body imprinted in black with " " and melon-yellow cap imprinted in black with "ANAFRANIL 25 mg" Bottles of 30………....………...……….....………… NDC 0406-9906-03 Capsules 50 mg – ivory body imprinted in black with " " and aqua blue cap imprinted in black with "ANAFRANIL 50 mg" Bottles of 30………..………....………...……….....…NDC 0406-9907-03 Capsules 75 mg – ivory body imprinted in black with " " and yellow cap imprinted in black with "ANAFRANIL 75 mg" Bottles of 30………..………...……….....………....…NDC 0406-9908-03 Storage – Store at 20º to 25ºC (68° to 77°F) [see USP Controlled Room Temperature]. Dispense in well-closed containers with a child-resistant closure. Protect from moisture.
Medication Information
Recent Major Changes
Suicidality and Antidepressant Drugs
Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of clomipramine hydrochloride or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Clomipramine hydrochloride is not approved for use in pediatric patients except for patients with obsessive compulsive disorder (OCD) (see WARNINGS, Clinical Worsening and Suicide Risk; PRECAUTIONS, Information for Patients; and PRECAUTIONS, Pediatric Use).
Indications and Usage
Anafranil™ (clomipramine hydrochloride) Capsules USP is indicated for the treatment of obsessions and compulsions in patients with Obsessive-Compulsive Disorder (OCD). The obsessions or compulsions must cause marked distress, be time-consuming, or significantly interfere with social or occupational functioning, in order to meet the DSM-III-R (circa 1989) diagnosis of OCD.
Obsessions are recurrent, persistent ideas, thoughts, images, or impulses that are ego-dystonic. Compulsions are repetitive, purposeful, and intentional behaviors performed in response to an obsession or in a stereotyped fashion, and are recognized by the person as excessive or unreasonable.
The effectiveness of Anafranil for the treatment of OCD was demonstrated in multicenter, placebo-controlled, parallel-group studies, including two 10-week studies in adults and one 8-week study in children and adolescents 10 to 17 years of age. Patients in all studies had moderate-to-severe OCD (DSM-III), with mean baseline ratings on the Yale-Brown Obsessive Compulsive Scale (YBOCS) ranging from 26 to 28 and a mean baseline rating of 10 on the NIMH Clinical Global Obsessive Compulsive Scale (NIMH-OC). Patients taking CMI experienced a mean reduction of approximately 10 on the YBOCS, representing an average improvement on this scale of 35% to 42% among adults and 37% among children and adolescents. CMI-treated patients experienced a 3.5 unit decrement on the NIMH-OC. Patients on placebo showed no important clinical response on either scale. The maximum dose was 250 mg/day for most adults and 3 mg/kg/day (up to 200 mg) for all children and adolescents.
The effectiveness of Anafranil for long-term use (i.e., for more than 10 weeks) has not been systematically evaluated in placebo-controlled trials. The physician who elects to use Anafranil for extended periods should periodically reevaluate the long-term usefulness of the drug for the individual patient (see DOSAGE AND ADMINISTRATION ).
Dosage and Administration
The treatment regimens described below are based on those used in controlled clinical trials of Anafranil in 520 adults, and 91 children and adolescents with OCD. During initial titration, Anafranil should be given in divided doses with meals to reduce gastrointestinal side effects. The goal of this initial titration phase is to minimize side effects by permitting tolerance to side effects to develop or allowing the patient time to adapt if tolerance does not develop.
Because both CMI and its active metabolite, DMI, have long elimination half-lives, the prescriber should take into consideration the fact that steady-state plasma levels may not be achieved until 2 to 3 weeks after dosage change (see CLINICAL PHARMACOLOGY ). Therefore, after initial titration, it may be appropriate to wait 2 to 3 weeks between further dosage adjustments.
Contraindications
Anafranil is contraindicated in patients with a history of hypersensitivity to Anafranil or other tricyclic antidepressants.
Monoamine Oxidase Inhibitors (MAOIs)
The use of MAOIs intended to treat psychiatric disorders with Anafranil or within 14 days of stopping treatment with Anafranil is contraindicated because of an increased risk of serotonin syndrome. The use of Anafranil within 14 days of stopping an MAOI intended to treat psychiatric disorders is also contraindicated (see WARNINGS and DOSAGE AND ADMINISTRATION ).
Starting Anafranil in a patient who is being treated with linezolid or intravenous methylene blue is also contraindicated because of an increased risk of serotonin syndrome (see WARNINGS and DOSAGE AND ADMINISTRATION ).
Myocardial Infarction
Anafranil is contraindicated during the acute recovery period after a myocardial infarction.
Drug Interactions
The risks of using Anafranil in combination with other drugs have not been systematically evaluated. Given the primary CNS effects of Anafranil, caution is advised in using it concomitantly with other CNS-active drugs (see Information for Patients ). Anafranil should not be used with MAO inhibitors (see CONTRAINDICATIONS ).
Close supervision and careful adjustment of dosage are required when Anafranil is administered with anticholinergic or sympathomimetic drugs.
Several tricyclic antidepressants have been reported to block the pharmacologic effects of guanethidine, clonidine, or similar agents, and such an effect may be anticipated with CMI because of its structural similarity to other tricyclic antidepressants.
The plasma concentration of CMI has been reported to be increased by the concomitant administration of haloperidol; plasma levels of several closely related tricyclic antidepressants have been reported to be increased by the concomitant administration of methylphenidate or hepatic enzyme inhibitors (e.g., cimetidine, fluoxetine) and decreased by the concomitant administration of hepatic enzyme inducers (e.g., barbiturates, phenytoin), and such an effect may be anticipated with CMI as well. Administration of CMI has been reported to increase the plasma levels of phenobarbital, if given concomitantly (see CLINICAL PHARMACOLOGY, Interactions ).
How Supplied
Anafranil™ (clomipramine hydrochloride) Capsules USP
Capsules 25 mg – ivory body imprinted in black with "
Bottles of 30………....………...……….....………… NDC 0406-9906-03
Capsules 50 mg – ivory body imprinted in black with "
" and aqua blue cap imprinted in black with "ANAFRANIL 50 mg"Bottles of 30………..………....………...……….....…NDC 0406-9907-03
Capsules 75 mg – ivory body imprinted in black with "
" and yellow cap imprinted in black with "ANAFRANIL 75 mg"Bottles of 30………..………...……….....………....…NDC 0406-9908-03
Storage – Store at 20º to 25ºC (68° to 77°F) [see USP Controlled Room Temperature].
Dispense in well-closed containers with a child-resistant closure. Protect from moisture.
Description
Anafranil™ (clomipramine hydrochloride) Capsules USP is an antiobsessional drug that belongs to the class (dibenzazepine) of pharmacologic agents known as tricyclic antidepressants. Anafranil is available as capsules of 25, 50, and 75 mg for oral administration.
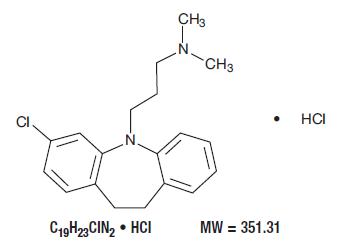
Clomipramine hydrochloride USP is 3-chloro-5-[3-(dimethylamino)propyl]-10,11-dihydro-5H-dibenz[b,f] azepine monohydrochloride, and its structural formula is:
Clomipramine hydrochloride USP is a white to off-white crystalline powder. It is freely soluble in water, in methanol, and in methylene chloride, and insoluble in ethyl ether and in hexane.
Inactive Ingredients. D&C Red No. 33 (25 mg capsules only), D&C Yellow No. 10, FD&C Blue No. 1 (50 mg capsules only), FD&C Yellow No. 6, gelatin, magnesium stearate, methylparaben, propylparaben, starch (corn), and titanium dioxide.
Dress
Rare cases of drug rash with eosinophilia and systemic symptoms (DRESS) have been reported with the use of clomipramine. In the event of severe acute reactions such as DRESS, discontinue clomipramine therapy immediately and institute appropriate treatment.
Section 42229-5
Absorption/Bioavailability – CMI from Anafranil capsules is as bioavailable as CMI from a solution. The bioavailability of CMI from capsules is not significantly affected by food.
In a dose proportionality study involving multiple CMI doses, steady-state plasma concentrations (Css) and area-under-plasma-concentration-time curves (AUC) of CMI and CMI's major active metabolite, desmethylclomipramine (DMI), were not proportional to dose over the ranges evaluated, i.e., between 25 to 100 mg/day and between 25 to 150 mg/day, although Css and AUC are approximately linearly related to dose between 100 to 150 mg/day. The relationship between dose and CMI/DMI concentrations at higher daily doses has not been systematically assessed, but if there is significant dose dependency at doses above 150 mg/day, there is the potential for dramatically higher Css and AUC even for patients dosed within the recommended range. This may pose a potential risk to some patients (see WARNINGS and PRECAUTIONS, Drug Interactions ).
After a single 50 mg oral dose, maximum plasma concentrations of CMI occur within 2 to 6 hours (mean, 4.7 hr) and range from 56 ng/mL to 154 ng/mL (mean, 92 ng/mL). After multiple daily doses of 150 mg of Anafranil, steady-state maximum plasma concentrations range from 94 ng/mL to 339 ng/mL (mean, 218 ng/mL) for CMI and from 134 ng/mL to 532 ng/mL (mean, 274 ng/mL) for DMI. Additional information from a rising dose study of doses up to 250 mg suggests that DMI may exhibit nonlinear pharmacokinetics over the usual dosing range. At a dose of Anafranil 200 mg, subjects who had a single blood sample taken approximately 9 to 22 hours, (median 16 hours), after the dose had plasma concentrations of up to 605 ng/mL for CMI, 781 ng/mL for DMI, and 1386 ng/mL for both.
Seizures
During premarket evaluation, seizure was identified as the most significant risk of Anafranil use.
The observed cumulative incidence of seizures among patients exposed to Anafranil at doses up to 300 mg/day was 0.64% at 90 days, 1.12% at 180 days, and 1.45% at 365 days. The cumulative rates correct the crude rate of 0.7% (25 of 3519 patients) for the variable duration of exposure in clinical trials.
Although dose appears to be a predictor of seizure, there is a confounding of dose and duration of exposure, making it difficult to assess independently the effect of either factor alone. The ability to predict the occurrence of seizures in subjects exposed to doses of CMI greater than 250 mg is limited, given that the plasma concentration of CMI may be dose-dependent and may vary among subjects given the same dose. Nevertheless, prescribers are advised to limit the daily dose to a maximum of 250 mg in adults and 3 mg/kg (or 200 mg) in children and adolescents (see DOSAGE AND ADMINISTRATION ).
Caution should be used in administering Anafranil to patients with a history of seizures or other predisposing factors, e.g., brain damage of varying etiology, alcoholism, and concomitant use with other drugs that lower the seizure threshold.
Rare reports of fatalities in association with seizures have been reported by foreign postmarketing surveillance, but not in U.S. clinical trials. In some of these cases, Anafranil had been administered with other epileptogenic agents; in others, the patients involved had possibly predisposing medical conditions. Thus a causal association between Anafranil treatment and these fatalities has not been established.
Physicians should discuss with patients the risk of taking Anafranil while engaging in activities in which sudden loss of consciousness could result in serious injury to the patient or others, e.g., the operation of complex machinery, driving, swimming, climbing.
Management
Obtain an ECG and immediately initiate cardiac monitoring. Protect the patient's airway, establish an intravenous line, and initiate gastric decontamination. A minimum of 6 hours of observation with cardiac monitoring and observation for signs of CNS or respiratory depression, hypotension, cardiac dysrhythmias and/or conduction blocks, and seizures is necessary.
If signs of toxicity occur at any time during this period, extended monitoring is required. There are case reports of patients succumbing to fatal dysrhythmias late after overdose; these patients had clinical evidence of significant poisoning prior to death and most received inadequate gastrointestinal decontamination. Monitoring of plasma drug levels should not guide management of the patient.
Overdosage
Deaths may occur from overdosage with this class of drugs. Multiple drug ingestion (including alcohol) is common in deliberate tricyclic overdose. As the management is complex and changing, it is recommended that the physician contact a poison control center for current information on treatment. Signs and symptoms of toxicity develop rapidly after tricyclic overdose. Therefore, hospital monitoring is required as soon as possible.
Geriatric Use
Clinical studies of Anafranil did not include sufficient numbers of subjects age 65 and over to determine whether they respond differently from younger subjects; 152 patients at least 60 years of age participating in various U.S. clinical trials received Anafranil for periods of several months to several years. No unusual age-related adverse events were identified in this population. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
Anafranil has been associated with cases of clinically significant hyponatremia. Elderly patients may be at greater risk for this adverse reaction (see PRECAUTIONS, Hyponatremia ).
Pediatric Use
Safety and effectiveness in the pediatric population other than pediatric patients with OCD have not been established (see BOX WARNING and WARNINGS, Clinical Worsening and Suicide Risk ). Anyone considering the use of Anafranil in a child or adolescent must balance the potential risks with the clinical need.
In a controlled clinical trial in children and adolescents (10 to 17 years of age), 46 outpatients received Anafranil for up to 8 weeks. In addition, 150 adolescent patients have received Anafranil in open-label protocols for periods of several months to several years. Of the 196 adolescents studied, 50 were 13 years of age or less and 146 were 14 to 17 years of age. The adverse reaction profile in this age group (see ADVERSE REACTIONS ) is similar to that observed in adults.
The risks, if any, that may be associated with Anafranil's extended use in children and adolescents with OCD have not been systematically assessed. The evidence supporting the conclusion that Anafranil is safe for use in children and adolescents is derived from relatively short term clinical studies and from extrapolation of experience gained with adult patients. In particular, there are no studies that directly evaluate the effects of long term Anafranil use on the growth, development, and maturation of children and adolescents. Although there is no evidence to suggest that Anafranil adversely affects growth, development or maturation, the absence of such findings is not adequate to rule out a potential for such effects in chronic use.
The safety and effectiveness in pediatric patients below the age of 10 have not been established. Therefore, specific recommendations cannot be made for the use of Anafranil in pediatric patients under the age of 10.
Manifestations
Signs and symptoms vary in severity depending upon factors such as the amount of drug absorbed, the age of the patient, and the time elapsed since drug ingestion. Critical manifestations of overdose include cardiac dysrhythmias, severe hypotension, convulsions, and CNS depression including coma. Changes in the electrocardiogram, particularly in QRS axis or width, are clinically significant indicators of tricyclic toxicity. Other CNS manifestations may include drowsiness, stupor, ataxia, restlessness, agitation, delirium, severe perspiration, hyperactive reflexes, muscle rigidity, and athetoid and choreiform movements. Cardiac abnormalities may include tachycardia, signs of congestive heart failure, and in very rare cases, cardiac arrest. Respiratory depression, cyanosis, shock, vomiting, hyperpyrexia, mydriasis, and oliguria or anuria may also be present.
Nursing Mothers
Anafranil has been found in human milk. Because of the potential for adverse reactions, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Human Experience
In U.S. clinical trials, 2 deaths occurred in 12 reported cases of acute overdosage with Anafranil either alone or in combination with other drugs. One death involved a patient suspected of ingesting a dose of 7000 mg. The second death involved a patient suspected of ingesting a dose of 5750 mg. The 10 nonfatal cases involved doses of up to 5000 mg, accompanied by plasma levels of up to 1010 ng/mL. All 10 patients completely recovered. Among reports from other countries of Anafranil overdose, the lowest dose associated with a fatality was 750 mg. Based upon postmarketing reports in the United Kingdom, CMI's lethality in overdose is considered to be similar to that reported for closely related tricyclic compounds marketed as antidepressants.
Pharmacodynamics
Clomipramine (CMI) is presumed to influence obsessive and compulsive behaviors through its effects on serotonergic neuronal transmission. The actual neurochemical mechanism is unknown, but CMI's capacity to inhibit the reuptake of serotonin (5-HT) is thought to be important.
Animal Toxicology
Phospholipidosis and testicular changes, commonly associated with tricyclic compounds, have been observed with Anafranil. In chronic rat studies, changes related to Anafranil consisted of systemic phospholipidosis, alterations in the testes (atrophy, mineralization) and secondary changes in other tissues. In addition cardiac thrombosis and dermatitis/keratitis were observed in rats treated for 2 years at doses which were 24 and 10 times the maximum recommended human daily dose (MRHD), respectively, on a mg/kg basis, and 4 and 1.5 times the MRHD, respectively, on a mg/m2 basis.
Mallinckrodt, the “M” brand mark, the Mallinckrodt Pharmaceuticals logo,
and other brands are trademarks of a Mallinckrodt company.© 2024 Mallinckrodt.
Product of Canada, Active Ingredient made in Ireland
Manufactured by
Patheon Inc.
Whitby, Ontario, Canada
L1N 5Z5
for SpecGx LLC
Webster Groves, MO 63119 USA
Rev 09/2024
Mallinckrodt™
Pharmaceuticals
Commonly Observed
The most commonly observed adverse events associated with the use of Anafranil and not seen at an equivalent incidence among placebo-treated patients were gastrointestinal complaints, including dry mouth, constipation, nausea, dyspepsia, and anorexia; nervous system complaints, including somnolence, tremor, dizziness, nervousness, and myoclonus; genitourinary complaints, including changed libido, ejaculatory failure, impotence, and micturition disorder; and other miscellaneous complaints, including fatigue, sweating, increased appetite, weight gain, and visual changes.
Serotonergic Drugs
(See CONTRAINDICATIONS, WARNINGS, and DOSAGE AND ADMINISTRATION .)
Pregnancy Category C
No teratogenic effects were observed in studies performed in rats and mice at doses up to 100 mg/kg, which is 24 times the maximum recommended human daily dose (MRHD) on a mg/kg basis and 4 times (rats) and 2 times (mice) the MRHD on a mg/m2 basis. Slight nonspecific embryo/fetotoxic effects were seen in the offspring of treated rats given 50 and 100 mg/kg and of treated mice given 100 mg/kg.
There are no adequate or well-controlled studies in pregnant women. Withdrawal symptoms, including jitteriness, tremor, and seizures, have been reported in neonates whose mothers had taken Anafranil until delivery. Anafranil should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Information for Patients
Prescribers or other health professionals should inform patients, their families, and their caregivers about the benefits and risks associated with treatment with clomipramine hydrochloride and should counsel them in its appropriate use. A patient Medication Guide about “Antidepressant Medicines, Depression and other Serious Mental Illness, and Suicidal Thoughts or Actions” is available for clomipramine hydrochloride. The prescriber or health professional should instruct patients, their families, and their caregivers to read the Medication Guide and should assist them in understanding its contents. Patients should be given the opportunity to discuss the contents of the Medication Guide and to obtain answers to any questions they may have. The complete text of the Medication Guide is reprinted at the end of this document.
Patients should be advised of the following issues and asked to alert their prescriber if these occur while taking clomipramine hydrochloride.
Drug Abuse and Dependence
Anafranil has not been systematically studied in animals or humans for its potential for abuse, tolerance, or physical dependence. While a variety of withdrawal symptoms have been described in association with Anafranil discontinuation (see PRECAUTIONS, Withdrawal Symptoms ), there is no evidence for drug-seeking behavior, except for a single report of potential Anafranil abuse by a patient with a history of dependence on codeine, benzodiazepines, and multiple psychoactive drugs. The patient received Anafranil for depression and panic attacks and appeared to become dependent after hospital discharge.
Despite the lack of evidence suggesting an abuse liability for Anafranil in foreign marketing, it is not possible to predict the extent to which Anafranil might be misused or abused once marketed in the U.S. Consequently, physicians should carefully evaluate patients for a history of drug abuse and follow such patients closely.
Clinical Worsening and Suicide Risk
Patients with major depressive disorder (MDD), both adult and pediatric, may experience worsening of their depression and/or the emergence of suicidal ideation and behavior (suicidality) or unusual changes in behavior, whether or not they are taking antidepressant medications, and this risk may persist until significant remission occurs. Suicide is a known risk of depression and certain other psychiatric disorders, and these disorders themselves are the strongest predictors of suicide. There has been a long-standing concern, however, that antidepressants may have a role in inducing worsening of depression and the emergence of suicidality in certain patients during the early phases of treatment. Pooled analyses of short-term placebo-controlled trials of antidepressant drugs (SSRIs and others) showed that these drugs increase the risk of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults (ages 18 to 24) with major depressive disorder (MDD) and other psychiatric disorders. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction with antidepressants compared to placebo in adults aged 65 and older.
The pooled analyses of placebo-controlled trials in children and adolescents with MDD, obsessive compulsive disorder (OCD), or other psychiatric disorders included a total of 24 short-term trials of 9 antidepressant drugs in over 4400 patients. The pooled analyses of placebo-controlled trials in adults with MDD or other psychiatric disorders included a total of 295 short-term trials (median duration of 2 months) of 11 antidepressant drugs in over 77,000 patients. There was considerable variation in risk of suicidality among drugs, but a tendency toward an increase in the younger patients for almost all drugs studied. There were differences in absolute risk of suicidality across the different indications, with the highest incidence in MDD. The risk differences (drug vs placebo), however, were relatively stable within age strata and across indications. These risk differences (drug-placebo difference in the number of cases of suicidality per 1000 patients treated) are provided in Table 1.
| Age Range | Drug-Placebo Difference in
Number of Cases of Suicidality per 1000 Patients Treated |
| Increases Compared to Placebo | |
| <18 | 14 additional cases |
| 18-24 | 5 additional cases |
| Decreases Compared to Placebo | |
| 25-64 | 1 fewer case |
| ≥65 | 6 fewer cases |
No suicides occurred in any of the pediatric trials. There were suicides in the adult trials, but the number was not sufficient to reach any conclusion about drug effect on suicide.
It is unknown whether the suicidality risk extends to longer-term use, i.e., beyond several months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with depression that the use of antidepressants can delay the recurrence of depression.
All patients being treated with antidepressants for any indication should be monitored appropriately and observed closely for clinical worsening, suicidality, and unusual changes in behavior, especially during the initial few months of a course of drug therapy, or at times of dose changes, either increases or decreases.
The following symptoms, anxiety, agitation, panic attacks, insomnia, irritability, hostility, aggressiveness, impulsivity, akathisia (psychomotor restlessness), hypomania, and mania, have been reported in adult and pediatric patients being treated with antidepressants for major depressive disorder as well as for other indications, both psychiatric and nonpsychiatric. Although a causal link between the emergence of such symptoms and either the worsening of depression and/or the emergence of suicidal impulses has not been established, there is concern that such symptoms may represent precursors to emerging suicidality.
Consideration should be given to changing the therapeutic regimen, including possibly discontinuing the medication, in patients whose depression is persistently worse, or who are experiencing emergent suicidality or symptoms that might be precursors to worsening depression or suicidality, especially if these symptoms are severe, abrupt in onset, or were not part of the patient's presenting symptoms.
Families and caregivers of patients being treated with antidepressants for major depressive disorder or other indications, both psychiatric and nonpsychiatric, should be alerted about the need to monitor patients for the emergence of agitation, irritability, unusual changes in behavior, and the other symptoms described above, as well as the emergence of suicidality, and to report such symptoms immediately to healthcare providers. Such monitoring should include daily observation by families and caregivers. Prescriptions for clomipramine hydrochloride should be written for the smallest quantity of capsules consistent with good patient management, in order to reduce the risk of overdose.
Monoamine Oxidase Inhibitors (maois)
(See CONTRAINDICATIONS, WARNINGS, and DOSAGE AND ADMINISTRATION .)
Incidence in Controlled Clinical Trials
The following table enumerates adverse events that occurred at an incidence of 1% or greater among patients with OCD who received Anafranil in adult or pediatric placebo-controlled clinical trials. The frequencies were obtained from pooled data of clinical trials involving either adults receiving Anafranil (N=322) or placebo (N=319) or children treated with Anafranil (N=46) or placebo (N=44). The prescriber should be aware that these figures cannot be used to predict the incidence of side effects in the course of usual medical practice, in which patient characteristics and other factors differ from those that prevailed in the clinical trials. Similarly, the cited frequencies cannot be compared with figures obtained from other clinical investigations involving different treatments, uses, and investigators. The cited figures, however, provide the physician with a basis for estimating the relative contribution of drug and nondrug factors to the incidence of side effects in the populations studied.
|
*Events reported by at least 1% of Anafranil patients are included. |
|||||
| Adults | Children and Adolescents | ||||
| Body System/ Adverse Event* |
Anafranil (N=322) |
Placebo (N=319) |
Anafranil (N=46) |
Placebo (N=44) |
|
| Nervous System | |||||
| Somnolence | 54 | 16 | 46 | 11 | |
| Tremor | 54 | 2 | 33 | 2 | |
| Dizziness | 54 | 14 | 41 | 14 | |
| Headache | 52 | 41 | 28 | 34 | |
| Insomnia | 25 | 15 | 11 | 7 | |
| Libido change | 21 | 3 | - | - | |
| Nervousness | 18 | 2 | 4 | 2 | |
| Myoclonus | 13 | - | 2 | - | |
| Increased appetite | 11 | 2 | - | 2 | |
| Paresthesia | 9 | 3 | 2 | 2 | |
| Memory impairment | 9 | 1 | 7 | 2 | |
| Anxiety | 9 | 4 | 2 | - | |
| Twitching | 7 | 1 | 4 | 5 | |
| Impaired concentration | 5 | 2 | - | - | |
| Depression | 5 | 1 | - | - | |
| Hypertonia | 4 | 1 | 2 | - | |
| Sleep disorder | 4 | - | 9 | 5 | |
| Psychosomatic disorder | 3 | - | - | - | |
| Yawning | 3 | - | - | - | |
| Confusion | 3 | - | 2 | - | |
| Speech disorder | 3 | - | - | - | |
| Abnormal dreaming | 3 | - | - | 2 | |
| Agitation | 3 | - | - | - | |
| Migraine | 3 | - | - | - | |
| Depersonalization | 2 | - | 2 | - | |
| Irritability | 2 | 2 | 2 | - | |
| Emotional lability | 2 | - | - | 2 | |
| Panic reaction | 1 | - | 2 | - | |
| Aggressive reaction | - | - | 2 | - | |
| Paresis | - | - | 2 | - | |
| Skin and Appendages | |||||
| Increased sweating | 29 | 3 | 9 | - | |
| Rash | 8 | 1 | 4 | 2 | |
| Pruritus | 6 | - | 2 | 2 | |
| Dermatitis | 2 | - | - | 2 | |
| Acne | 2 | 2 | - | 5 | |
| Dry skin | 2 | - | - | 5 | |
| Urticaria | 1 | - | - | - | |
| Abnormal skin odor | - | - | 2 | - | |
| Digestive System | |||||
| Dry mouth | 84 | 17 | 63 | 16 | |
| Constipation | 47 | 11 | 22 | 9 | |
| Nausea | 33 | 14 | 9 | 11 | |
| Dyspepsia | 22 | 10 | 13 | 2 | |
| Diarrhea | 13 | 9 | 7 | 5 | |
| Anorexia | 12 | - | 22 | 2 | |
| Abdominal pain | 11 | 9 | 13 | 16 | |
| Vomiting | 7 | 2 | 7 | - | |
| Flatulence | 6 | 3 | - | 2 | |
| Tooth disorder | 5 | - | - | - | |
| Gastrointestinal disorder | 2 | - | - | 2 | |
| Dysphagia | 2 | - | - | - | |
| Esophagitis | 1 | - | - | - | |
| Eructation | - | - | 2 | 2 | |
| Ulcerative stomatitis | - | - | 2 | - | |
| Body as a Whole | |||||
| Fatigue | 39 | 18 | 35 | 9 | |
| Weight increase | 18 | 1 | 2 | - | |
| Flushing | 8 | - | 7 | - | |
| Hot flushes | 5 | - | 2 | - | |
| Chest pain | 4 | 4 | 7 | - | |
| Fever | 4 | - | 2 | 7 | |
| Allergy | 3 | 3 | 7 | 5 | |
| Pain | 3 | 2 | 4 | 2 | |
| Local edema | 2 | 4 | - | - | |
| Chills | 2 | 1 | - | - | |
| Weight decrease | - | - | 7 | - | |
| Otitis media | - | - | 4 | 5 | |
| Asthenia | - | - | 2 | - | |
| Halitosis | - | - | 2 | - | |
| Cardiovascular System | |||||
| Postural hypotension | 6 | - | 4 | - | |
| Palpitation | 4 | 2 | 4 | - | |
| Tachycardia | 4 | - | 2 | - | |
| Syncope | - | - | 2 | - | |
| Respiratory System | |||||
| Pharyngitis | 14 | 9 | - | 5 | |
| Rhinitis | 12 | 10 | 7 | 9 | |
| Sinusitis | 6 | 4 | 2 | 5 | |
| Coughing | 6 | 6 | 4 | 5 | |
| Bronchospasm | 2 | - | 7 | 2 | |
| Epistaxis | 2 | - | - | 2 | |
| Dyspnea | - | - | 2 | - | |
| Laryngitis | - | 1 | 2 | - | |
| Urogenital System | |||||
| Male and Female Patients Combined | |||||
| Micturition disorder | 14 | 2 | 4 | 2 | |
| Urinary tract infection | 6 | 1 | - | - | |
| Micturition frequency | 5 | 3 | - | - | |
| Urinary retention | 2 | - | 7 | - | |
| Dysuria | 2 | 2 | - | - | |
| Cystitis | 2 | - | - | - | |
| Female Patients Only | (N=182) | (N=167) | (N=10) | (N=21) | |
| Dysmenorrhea | 12 | 14 | 10 | 10 | |
| Lactation (nonpuerperal) | 4 | - | - | - | |
| Menstrual disorder | 4 | 2 | - | - | |
| Vaginitis | 2 | - | - | - | |
| Leukorrhea | 2 | - | - | - | |
| Breast enlargement | 2 | - | - | - | |
| Breast pain | 1 | - | - | - | |
| Amenorrhea | 1 | - | - | - | |
| Male Patients Only | (N=140) | (N=152) | (N=36) | (N=23) | |
| Ejaculation failure | 42 | 2 | 6 | - | |
| Impotence | 20 | 3 | - | - | |
| Special Senses | |||||
| Abnormal vision | 18 | 4 | 7 | 2 | |
| Taste perversion | 8 | - | 4 | - | |
| Tinnitus | 6 | - | 4 | - | |
| Abnormal lacrimation | 3 | 2 | - | - | |
| Mydriasis | 2 | - | - | - | |
| Conjunctivitis | 1 | - | - | - | |
| Anisocoria | - | - | 2 | - | |
| Blepharospasm | - | - | 2 | - | |
| Ocular allergy | - | - | 2 | - | |
| Vestibular disorder | - | - | 2 | 2 | |
| Musculoskeletal | |||||
| Myalgia | 13 | 9 | - | - | |
| Back pain | 6 | 6 | - | - | |
| Arthralgia | 3 | 5 | - | - | |
| Muscle weakness | 1 | - | 2 | - | |
| Hemic and Lymphatic | |||||
| Purpura | 3 | - | - | - | |
| Anemia | - | - | 2 | 2 | |
| Metabolic and Nutritional | |||||
| Thirst | 2 | 2 | - | 2 | |
Leading to Discontinuation of Treatment
Approximately 20% of 3616 patients who received Anafranil in U.S. premarketing clinical trials discontinued treatment because of an adverse event. Approximately one-half of the patients who discontinued (9% of the total) had multiple complaints, none of which could be classified as primary. Where a primary reason for discontinuation could be identified, most patients discontinued because of nervous system complaints (5.4%), primarily somnolence. The second-most-frequent reason for discontinuation was digestive system complaints (1.3%), primarily vomiting and nausea.
There was no apparent relationship between the adverse events and elevated plasma drug concentrations.
Initial Treatment/dose Adjustment (adults)
Treatment with Anafranil should be initiated at a dosage of 25 mg daily and gradually increased, as tolerated, to approximately 100 mg during the first 2 weeks. During initial titration, Anafranil should be given in divided doses with meals to reduce gastrointestinal side effects. Thereafter, the dosage may be increased gradually over the next several weeks, up to a maximum of 250 mg daily. After titration, the total daily dose may be given once daily at bedtime to minimize daytime sedation.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No evidence of carcinogenicity was found in two 2-year bioassays in rats at doses up to 100 mg/kg, which is 24 and 4 times the maximum recommended human daily dose (MRHD) on a mg/kg and mg/m2 basis, respectively, or in a 2-year bioassay in mice at doses up to 80 mg/kg, which is 20 and 1.5 times the MRHD on a mg/kg and mg/m2 basis, respectively.
In reproduction studies, no effects on fertility were found in rats given up to 24 mg/kg, which is 6 times, and approximately equal to, the MRHD on a mg/kg and mg/m2 basis, respectively.
Package Label Principal Display Panel 25 Mg Bottle
NDC 0406-9906-03
30 Capsules
Anafranil™
(clomiPRAMINE Hydrochloride)
Capsules USP
25 mg
Rx only
Each capsule contains:
Clomipramine Hydrochloride USP ... 25 mg
PHARMACIST: PLEASE DISPENSE WITH ATTACHED MEDICATION GUIDE
MALLINCKRODT™
Rev 09/2024
2000017741
Package Label Principal Display Panel 50 Mg Bottle
NDC 0406-9907-03
30 Capsules
Anafranil™
(clomiPRAMINE Hydrochloride)
Capsules USP
50 mg
Rx only
Each capsule contains:
Clomipramine Hydrochloride USP ... 50 mg
PHARMACIST: PLEASE DISPENSE WITH ATTACHED MEDICATION GUIDE
MALLINCKRODT™
Rev 9/2024
2000017742
Package Label Principal Display Panel 75 Mg Bottle
NDC 0406-9908-03
30 Capsules
Anafranil™
(clomiPRAMINE Hydrochloride)
Capsules USP
75 mg
Rx only
Each capsule contains:
Clomipramine Hydrochloride USP ... 75 mg
PHARMACIST: PLEASE DISPENSE WITH ATTACHED MEDICATION GUIDE
MALLINCKRODT™
Rev 09/2024
2000017743
Initial Treatment/dose Adjustment (children and Adolescents)
As with adults, the starting dose is 25 mg daily and should be gradually increased (also given in divided doses with meals to reduce gastrointestinal side effects) during the first 2 weeks, as tolerated, up to a daily maximum of 3 mg/kg or 100 mg, whichever is smaller. Thereafter, the dosage may be increased gradually over the next several weeks up to a daily maximum of 3 mg/kg or 200 mg, whichever is smaller (see PRECAUTIONS, Pediatric Use ). As with adults, after titration, the total daily dose may be given once daily at bedtime to minimize daytime sedation.
Other Events Observed During the Premarketing Evaluation of Anafranil
During clinical testing in the U.S., multiple doses of Anafranil were administered to approximately 3600 subjects. Untoward events associated with this exposure were recorded by clinical investigators using terminology of their own choosing. Consequently, it is not possible to provide a meaningful estimate of the proportion of individuals experiencing adverse events without first grouping similar types of untoward events into a smaller number of standardized event categories.
In the tabulations that follow, a modified World Health Organization dictionary of terminology has been used to classify reported adverse events. The frequencies presented, therefore, represent the proportion of the 3525 individuals exposed to Anafranil who experienced an event of the type cited on at least one occasion while receiving Anafranil. All events are included except those already listed in the previous table, those reported in terms so general as to be uninformative, and those in which an association with the drug was remote. It is important to emphasize that although the events reported occurred during treatment with Anafranil, they were not necessarily caused by it.
Events are further categorized by body system and listed in order of decreasing frequency according to the following definitions: frequent adverse events are those occurring on one or more occasions in at least 1/100 patients; infrequent adverse events are those occurring in 1/100 to 1/1000 patients; rare events are those occurring in less than 1/1000 patients.
Maintenance/continuation Treatment (adults, Children, and Adolescents)
While there are no systematic studies that answer the question of how long to continue Anafranil, OCD is a chronic condition and it is reasonable to consider continuation for a responding patient. Although the efficacy of Anafranil after 10 weeks has not been documented in controlled trials, patients have been continued in therapy under double-blind conditions for up to 1 year without loss of benefit. However, dosage adjustments should be made to maintain the patient on the lowest effective dosage, and patients should be periodically reassessed to determine the need for treatment. During maintenance, the total daily dose may be given once daily at bedtime.
Use of Anafranil With other Maois, Such As Linezolid Or Methylene Blue
Do not start Anafranil in a patient who is being treated with linezolid or intravenous methylene blue because there is increased risk of serotonin syndrome. In a patient who requires more urgent treatment of a psychiatric condition, other interventions, including hospitalization, should be considered (see CONTRAINDICATIONS ).
In some cases, a patient already receiving Anafranil therapy may require urgent treatment with linezolid or intravenous methylene blue. If acceptable alternatives to linezolid or intravenous methylene blue treatment are not available and the potential benefits of linezolid or intravenous methylene blue treatment are judged to outweigh the risks of serotonin syndrome in a particular patient, Anafranil should be stopped promptly, and linezolid or intravenous methylene blue can be administered. The patient should be monitored for symptoms of serotonin syndrome for two weeks or until
24 hours after the last dose of linezolid or intravenous methylene blue, whichever comes first. Therapy with Anafranil may be resumed 24 hours after the last dose of linezolid or intravenous methylene blue (see
WARNINGS
).
The risk of administering methylene blue by non-intravenous routes (such as oral tablets or by local injection) or in intravenous doses much lower than 1 mg/kg with Anafranil is unclear. The clinician should, nevertheless, be aware of the possibility of emergent symptoms of serotonin syndrome with such use (see WARNINGS ).
Medication Guide Anafranil™ (clomipramine Hydrochloride) Capsules Usp
Read the Medication Guide that comes with you or your family member's antidepressant medicine. This Medication Guide is only about the risk of suicidal thoughts and actions with antidepressant medicines. Talk to your, or your family member's, healthcare provider about:
- all risks and benefits of treatment with antidepressant medicines
- all treatment choices for depression or other serious mental illness
What is the most important information I should know about antidepressant medicines, depression and other serious mental illnesses, and suicidal thoughts or actions?
-
Antidepressant medicines may increase suicidal thoughts or actions in some children, teenagers, and young adults within the first few months of treatment.
- Depression and other serious mental illnesses are the most important causes of suicidal thoughts and actions. Some people may have a particularly high risk of having suicidal thoughts or actions. These include people who have (or have a family history of) bipolar illness (also called manic-depressive illness) or suicidal thoughts or actions.
-
How can I watch for and try to prevent suicidal thoughts and actions in myself or a family member?
- Pay close attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings. This is very important when an antidepressant medicine is started or when the dose is changed.
- Call the healthcare provider right away to report new or sudden changes in mood, behavior, thoughts, or feelings.
- Keep all follow-up visits with the healthcare provider as scheduled. Call the healthcare provider between visits as needed, especially if you have concerns about symptoms.
Call a healthcare provider right away if you or your family member has any of the following symptoms, especially if they are new, worse, or worry you:
- thoughts about suicide or dying
- attempts to commit suicide
- new or worse depression
- new or worse anxiety
- feeling very agitated or restless
- panic attacks
- trouble sleeping (insomnia)
- new or worse irritability
- acting aggressive, being angry, or violent
- acting on dangerous impulses
- an extreme increase in activity and talking (mania)
- other unusual changes in behavior or mood
Low salt (sodium) levels in the blood. Elderly people may be at greater risk for this. Symptoms may include:
- headache
- weakness or feeling unsteady
- confusion, problems concentrating or thinking or memory problems
Visual problems
- eye pain
- changes in vision
- swelling or redness in or around the eye
Only some people are at risk for these problems. You may want to undergo an eye examination to see if you are at risk and receive preventative treatment if you are.
Who should not take Anafranil?
Do not take Anafranil if you:
- take a monoamine oxidase inhibitor (MAOI). Ask your healthcare provider or pharmacist if you are not sure if you take an MAOI, including the antibiotic linezolid.
- Do not take an MAOI within 2 weeks of stopping Anafranil unless directed to do so by your physician.
- Do not start Anafranil if you stopped taking an MAOI in the last 2 weeks unless directed to do so by your physician.
What else do I need to know about antidepressant medicines?
- Never stop an antidepressant medicine without first talking to a healthcare provider. Stopping an antidepressant medicine suddenly can cause other symptoms.
- Antidepressants are medicines used to treat depression and other illnesses. It is important to discuss all the risks of treating depression and also the risks of not treating it. Patients and their families or other caregivers should discuss all treatment choices with the healthcare provider, not just the use of antidepressants.
- Antidepressant medicines have other side effects. Talk to the healthcare provider about the side effects of the medicine prescribed for you or your family member.
- Antidepressant medicines can interact with other medicines. Know all of the medicines that you or your family member takes. Keep a list of all medicines to show the healthcare provider. Do not start new medicines without first checking with your healthcare provider.
- Not all antidepressant medicines prescribed for children are FDA approved for use in children. Talk to your child's healthcare provider for more information.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Anafranil is a trademark of Mallinckrodt LLC.
Product of Canada, Active Ingredient made in Ireland
Manufactured by
Patheon Inc.
Whitby, Ontario, Canada
L1N 5Z5
for SpecGx LLC
Webster Groves, MO 63119 USA
Rev 09/2024
Mallinckrodt™
Switching A Patient to Or From A Monoamine Oxidase Inhibitor (maoi) Intended to Treat Psychiatric Disorders
At least 14 days should elapse between discontinuation of an MAOI intended to treat psychiatric disorders and initiation of therapy with Anafranil. Conversely, at least 14 days should be allowed after stopping Anafranil before starting an MAOI intended to treat psychiatric disorders (see CONTRAINDICATIONS ).
Structured Label Content
Dress (DRESS)
Rare cases of drug rash with eosinophilia and systemic symptoms (DRESS) have been reported with the use of clomipramine. In the event of severe acute reactions such as DRESS, discontinue clomipramine therapy immediately and institute appropriate treatment.
Recent Major Changes (34066-1)
Suicidality and Antidepressant Drugs
Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of clomipramine hydrochloride or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Clomipramine hydrochloride is not approved for use in pediatric patients except for patients with obsessive compulsive disorder (OCD) (see WARNINGS, Clinical Worsening and Suicide Risk; PRECAUTIONS, Information for Patients; and PRECAUTIONS, Pediatric Use).
Section 42229-5 (42229-5)
Absorption/Bioavailability – CMI from Anafranil capsules is as bioavailable as CMI from a solution. The bioavailability of CMI from capsules is not significantly affected by food.
In a dose proportionality study involving multiple CMI doses, steady-state plasma concentrations (Css) and area-under-plasma-concentration-time curves (AUC) of CMI and CMI's major active metabolite, desmethylclomipramine (DMI), were not proportional to dose over the ranges evaluated, i.e., between 25 to 100 mg/day and between 25 to 150 mg/day, although Css and AUC are approximately linearly related to dose between 100 to 150 mg/day. The relationship between dose and CMI/DMI concentrations at higher daily doses has not been systematically assessed, but if there is significant dose dependency at doses above 150 mg/day, there is the potential for dramatically higher Css and AUC even for patients dosed within the recommended range. This may pose a potential risk to some patients (see WARNINGS and PRECAUTIONS, Drug Interactions ).
After a single 50 mg oral dose, maximum plasma concentrations of CMI occur within 2 to 6 hours (mean, 4.7 hr) and range from 56 ng/mL to 154 ng/mL (mean, 92 ng/mL). After multiple daily doses of 150 mg of Anafranil, steady-state maximum plasma concentrations range from 94 ng/mL to 339 ng/mL (mean, 218 ng/mL) for CMI and from 134 ng/mL to 532 ng/mL (mean, 274 ng/mL) for DMI. Additional information from a rising dose study of doses up to 250 mg suggests that DMI may exhibit nonlinear pharmacokinetics over the usual dosing range. At a dose of Anafranil 200 mg, subjects who had a single blood sample taken approximately 9 to 22 hours, (median 16 hours), after the dose had plasma concentrations of up to 605 ng/mL for CMI, 781 ng/mL for DMI, and 1386 ng/mL for both.
Seizures
During premarket evaluation, seizure was identified as the most significant risk of Anafranil use.
The observed cumulative incidence of seizures among patients exposed to Anafranil at doses up to 300 mg/day was 0.64% at 90 days, 1.12% at 180 days, and 1.45% at 365 days. The cumulative rates correct the crude rate of 0.7% (25 of 3519 patients) for the variable duration of exposure in clinical trials.
Although dose appears to be a predictor of seizure, there is a confounding of dose and duration of exposure, making it difficult to assess independently the effect of either factor alone. The ability to predict the occurrence of seizures in subjects exposed to doses of CMI greater than 250 mg is limited, given that the plasma concentration of CMI may be dose-dependent and may vary among subjects given the same dose. Nevertheless, prescribers are advised to limit the daily dose to a maximum of 250 mg in adults and 3 mg/kg (or 200 mg) in children and adolescents (see DOSAGE AND ADMINISTRATION ).
Caution should be used in administering Anafranil to patients with a history of seizures or other predisposing factors, e.g., brain damage of varying etiology, alcoholism, and concomitant use with other drugs that lower the seizure threshold.
Rare reports of fatalities in association with seizures have been reported by foreign postmarketing surveillance, but not in U.S. clinical trials. In some of these cases, Anafranil had been administered with other epileptogenic agents; in others, the patients involved had possibly predisposing medical conditions. Thus a causal association between Anafranil treatment and these fatalities has not been established.
Physicians should discuss with patients the risk of taking Anafranil while engaging in activities in which sudden loss of consciousness could result in serious injury to the patient or others, e.g., the operation of complex machinery, driving, swimming, climbing.
Management
Obtain an ECG and immediately initiate cardiac monitoring. Protect the patient's airway, establish an intravenous line, and initiate gastric decontamination. A minimum of 6 hours of observation with cardiac monitoring and observation for signs of CNS or respiratory depression, hypotension, cardiac dysrhythmias and/or conduction blocks, and seizures is necessary.
If signs of toxicity occur at any time during this period, extended monitoring is required. There are case reports of patients succumbing to fatal dysrhythmias late after overdose; these patients had clinical evidence of significant poisoning prior to death and most received inadequate gastrointestinal decontamination. Monitoring of plasma drug levels should not guide management of the patient.
Overdosage (OVERDOSAGE)
Deaths may occur from overdosage with this class of drugs. Multiple drug ingestion (including alcohol) is common in deliberate tricyclic overdose. As the management is complex and changing, it is recommended that the physician contact a poison control center for current information on treatment. Signs and symptoms of toxicity develop rapidly after tricyclic overdose. Therefore, hospital monitoring is required as soon as possible.
Description (DESCRIPTION)
Anafranil™ (clomipramine hydrochloride) Capsules USP is an antiobsessional drug that belongs to the class (dibenzazepine) of pharmacologic agents known as tricyclic antidepressants. Anafranil is available as capsules of 25, 50, and 75 mg for oral administration.
Clomipramine hydrochloride USP is 3-chloro-5-[3-(dimethylamino)propyl]-10,11-dihydro-5H-dibenz[b,f] azepine monohydrochloride, and its structural formula is:
Clomipramine hydrochloride USP is a white to off-white crystalline powder. It is freely soluble in water, in methanol, and in methylene chloride, and insoluble in ethyl ether and in hexane.
Inactive Ingredients. D&C Red No. 33 (25 mg capsules only), D&C Yellow No. 10, FD&C Blue No. 1 (50 mg capsules only), FD&C Yellow No. 6, gelatin, magnesium stearate, methylparaben, propylparaben, starch (corn), and titanium dioxide.
How Supplied (HOW SUPPLIED)
Anafranil™ (clomipramine hydrochloride) Capsules USP
Capsules 25 mg – ivory body imprinted in black with "
Bottles of 30………....………...……….....………… NDC 0406-9906-03
Capsules 50 mg – ivory body imprinted in black with "
" and aqua blue cap imprinted in black with "ANAFRANIL 50 mg"Bottles of 30………..………....………...……….....…NDC 0406-9907-03
Capsules 75 mg – ivory body imprinted in black with "
" and yellow cap imprinted in black with "ANAFRANIL 75 mg"Bottles of 30………..………...……….....………....…NDC 0406-9908-03
Storage – Store at 20º to 25ºC (68° to 77°F) [see USP Controlled Room Temperature].
Dispense in well-closed containers with a child-resistant closure. Protect from moisture.
Geriatric Use
Clinical studies of Anafranil did not include sufficient numbers of subjects age 65 and over to determine whether they respond differently from younger subjects; 152 patients at least 60 years of age participating in various U.S. clinical trials received Anafranil for periods of several months to several years. No unusual age-related adverse events were identified in this population. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
Anafranil has been associated with cases of clinically significant hyponatremia. Elderly patients may be at greater risk for this adverse reaction (see PRECAUTIONS, Hyponatremia ).
Pediatric Use
Safety and effectiveness in the pediatric population other than pediatric patients with OCD have not been established (see BOX WARNING and WARNINGS, Clinical Worsening and Suicide Risk ). Anyone considering the use of Anafranil in a child or adolescent must balance the potential risks with the clinical need.
In a controlled clinical trial in children and adolescents (10 to 17 years of age), 46 outpatients received Anafranil for up to 8 weeks. In addition, 150 adolescent patients have received Anafranil in open-label protocols for periods of several months to several years. Of the 196 adolescents studied, 50 were 13 years of age or less and 146 were 14 to 17 years of age. The adverse reaction profile in this age group (see ADVERSE REACTIONS ) is similar to that observed in adults.
The risks, if any, that may be associated with Anafranil's extended use in children and adolescents with OCD have not been systematically assessed. The evidence supporting the conclusion that Anafranil is safe for use in children and adolescents is derived from relatively short term clinical studies and from extrapolation of experience gained with adult patients. In particular, there are no studies that directly evaluate the effects of long term Anafranil use on the growth, development, and maturation of children and adolescents. Although there is no evidence to suggest that Anafranil adversely affects growth, development or maturation, the absence of such findings is not adequate to rule out a potential for such effects in chronic use.
The safety and effectiveness in pediatric patients below the age of 10 have not been established. Therefore, specific recommendations cannot be made for the use of Anafranil in pediatric patients under the age of 10.
Manifestations
Signs and symptoms vary in severity depending upon factors such as the amount of drug absorbed, the age of the patient, and the time elapsed since drug ingestion. Critical manifestations of overdose include cardiac dysrhythmias, severe hypotension, convulsions, and CNS depression including coma. Changes in the electrocardiogram, particularly in QRS axis or width, are clinically significant indicators of tricyclic toxicity. Other CNS manifestations may include drowsiness, stupor, ataxia, restlessness, agitation, delirium, severe perspiration, hyperactive reflexes, muscle rigidity, and athetoid and choreiform movements. Cardiac abnormalities may include tachycardia, signs of congestive heart failure, and in very rare cases, cardiac arrest. Respiratory depression, cyanosis, shock, vomiting, hyperpyrexia, mydriasis, and oliguria or anuria may also be present.
Nursing Mothers
Anafranil has been found in human milk. Because of the potential for adverse reactions, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Human Experience
In U.S. clinical trials, 2 deaths occurred in 12 reported cases of acute overdosage with Anafranil either alone or in combination with other drugs. One death involved a patient suspected of ingesting a dose of 7000 mg. The second death involved a patient suspected of ingesting a dose of 5750 mg. The 10 nonfatal cases involved doses of up to 5000 mg, accompanied by plasma levels of up to 1010 ng/mL. All 10 patients completely recovered. Among reports from other countries of Anafranil overdose, the lowest dose associated with a fatality was 750 mg. Based upon postmarketing reports in the United Kingdom, CMI's lethality in overdose is considered to be similar to that reported for closely related tricyclic compounds marketed as antidepressants.
Pharmacodynamics
Clomipramine (CMI) is presumed to influence obsessive and compulsive behaviors through its effects on serotonergic neuronal transmission. The actual neurochemical mechanism is unknown, but CMI's capacity to inhibit the reuptake of serotonin (5-HT) is thought to be important.
Animal Toxicology (ANIMAL TOXICOLOGY)
Phospholipidosis and testicular changes, commonly associated with tricyclic compounds, have been observed with Anafranil. In chronic rat studies, changes related to Anafranil consisted of systemic phospholipidosis, alterations in the testes (atrophy, mineralization) and secondary changes in other tissues. In addition cardiac thrombosis and dermatitis/keratitis were observed in rats treated for 2 years at doses which were 24 and 10 times the maximum recommended human daily dose (MRHD), respectively, on a mg/kg basis, and 4 and 1.5 times the MRHD, respectively, on a mg/m2 basis.
Mallinckrodt, the “M” brand mark, the Mallinckrodt Pharmaceuticals logo,
and other brands are trademarks of a Mallinckrodt company.© 2024 Mallinckrodt.
Product of Canada, Active Ingredient made in Ireland
Manufactured by
Patheon Inc.
Whitby, Ontario, Canada
L1N 5Z5
for SpecGx LLC
Webster Groves, MO 63119 USA
Rev 09/2024
Mallinckrodt™
Pharmaceuticals
Contraindications (CONTRAINDICATIONS)
Anafranil is contraindicated in patients with a history of hypersensitivity to Anafranil or other tricyclic antidepressants.
Monoamine Oxidase Inhibitors (MAOIs)
The use of MAOIs intended to treat psychiatric disorders with Anafranil or within 14 days of stopping treatment with Anafranil is contraindicated because of an increased risk of serotonin syndrome. The use of Anafranil within 14 days of stopping an MAOI intended to treat psychiatric disorders is also contraindicated (see WARNINGS and DOSAGE AND ADMINISTRATION ).
Starting Anafranil in a patient who is being treated with linezolid or intravenous methylene blue is also contraindicated because of an increased risk of serotonin syndrome (see WARNINGS and DOSAGE AND ADMINISTRATION ).
Myocardial Infarction
Anafranil is contraindicated during the acute recovery period after a myocardial infarction.
Commonly Observed
The most commonly observed adverse events associated with the use of Anafranil and not seen at an equivalent incidence among placebo-treated patients were gastrointestinal complaints, including dry mouth, constipation, nausea, dyspepsia, and anorexia; nervous system complaints, including somnolence, tremor, dizziness, nervousness, and myoclonus; genitourinary complaints, including changed libido, ejaculatory failure, impotence, and micturition disorder; and other miscellaneous complaints, including fatigue, sweating, increased appetite, weight gain, and visual changes.
Drug Interactions
The risks of using Anafranil in combination with other drugs have not been systematically evaluated. Given the primary CNS effects of Anafranil, caution is advised in using it concomitantly with other CNS-active drugs (see Information for Patients ). Anafranil should not be used with MAO inhibitors (see CONTRAINDICATIONS ).
Close supervision and careful adjustment of dosage are required when Anafranil is administered with anticholinergic or sympathomimetic drugs.
Several tricyclic antidepressants have been reported to block the pharmacologic effects of guanethidine, clonidine, or similar agents, and such an effect may be anticipated with CMI because of its structural similarity to other tricyclic antidepressants.
The plasma concentration of CMI has been reported to be increased by the concomitant administration of haloperidol; plasma levels of several closely related tricyclic antidepressants have been reported to be increased by the concomitant administration of methylphenidate or hepatic enzyme inhibitors (e.g., cimetidine, fluoxetine) and decreased by the concomitant administration of hepatic enzyme inducers (e.g., barbiturates, phenytoin), and such an effect may be anticipated with CMI as well. Administration of CMI has been reported to increase the plasma levels of phenobarbital, if given concomitantly (see CLINICAL PHARMACOLOGY, Interactions ).
Serotonergic Drugs
(See CONTRAINDICATIONS, WARNINGS, and DOSAGE AND ADMINISTRATION .)
Pregnancy Category C
No teratogenic effects were observed in studies performed in rats and mice at doses up to 100 mg/kg, which is 24 times the maximum recommended human daily dose (MRHD) on a mg/kg basis and 4 times (rats) and 2 times (mice) the MRHD on a mg/m2 basis. Slight nonspecific embryo/fetotoxic effects were seen in the offspring of treated rats given 50 and 100 mg/kg and of treated mice given 100 mg/kg.
There are no adequate or well-controlled studies in pregnant women. Withdrawal symptoms, including jitteriness, tremor, and seizures, have been reported in neonates whose mothers had taken Anafranil until delivery. Anafranil should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Indications and Usage (INDICATIONS AND USAGE)
Anafranil™ (clomipramine hydrochloride) Capsules USP is indicated for the treatment of obsessions and compulsions in patients with Obsessive-Compulsive Disorder (OCD). The obsessions or compulsions must cause marked distress, be time-consuming, or significantly interfere with social or occupational functioning, in order to meet the DSM-III-R (circa 1989) diagnosis of OCD.
Obsessions are recurrent, persistent ideas, thoughts, images, or impulses that are ego-dystonic. Compulsions are repetitive, purposeful, and intentional behaviors performed in response to an obsession or in a stereotyped fashion, and are recognized by the person as excessive or unreasonable.
The effectiveness of Anafranil for the treatment of OCD was demonstrated in multicenter, placebo-controlled, parallel-group studies, including two 10-week studies in adults and one 8-week study in children and adolescents 10 to 17 years of age. Patients in all studies had moderate-to-severe OCD (DSM-III), with mean baseline ratings on the Yale-Brown Obsessive Compulsive Scale (YBOCS) ranging from 26 to 28 and a mean baseline rating of 10 on the NIMH Clinical Global Obsessive Compulsive Scale (NIMH-OC). Patients taking CMI experienced a mean reduction of approximately 10 on the YBOCS, representing an average improvement on this scale of 35% to 42% among adults and 37% among children and adolescents. CMI-treated patients experienced a 3.5 unit decrement on the NIMH-OC. Patients on placebo showed no important clinical response on either scale. The maximum dose was 250 mg/day for most adults and 3 mg/kg/day (up to 200 mg) for all children and adolescents.
The effectiveness of Anafranil for long-term use (i.e., for more than 10 weeks) has not been systematically evaluated in placebo-controlled trials. The physician who elects to use Anafranil for extended periods should periodically reevaluate the long-term usefulness of the drug for the individual patient (see DOSAGE AND ADMINISTRATION ).
Information for Patients
Prescribers or other health professionals should inform patients, their families, and their caregivers about the benefits and risks associated with treatment with clomipramine hydrochloride and should counsel them in its appropriate use. A patient Medication Guide about “Antidepressant Medicines, Depression and other Serious Mental Illness, and Suicidal Thoughts or Actions” is available for clomipramine hydrochloride. The prescriber or health professional should instruct patients, their families, and their caregivers to read the Medication Guide and should assist them in understanding its contents. Patients should be given the opportunity to discuss the contents of the Medication Guide and to obtain answers to any questions they may have. The complete text of the Medication Guide is reprinted at the end of this document.
Patients should be advised of the following issues and asked to alert their prescriber if these occur while taking clomipramine hydrochloride.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
The treatment regimens described below are based on those used in controlled clinical trials of Anafranil in 520 adults, and 91 children and adolescents with OCD. During initial titration, Anafranil should be given in divided doses with meals to reduce gastrointestinal side effects. The goal of this initial titration phase is to minimize side effects by permitting tolerance to side effects to develop or allowing the patient time to adapt if tolerance does not develop.
Because both CMI and its active metabolite, DMI, have long elimination half-lives, the prescriber should take into consideration the fact that steady-state plasma levels may not be achieved until 2 to 3 weeks after dosage change (see CLINICAL PHARMACOLOGY ). Therefore, after initial titration, it may be appropriate to wait 2 to 3 weeks between further dosage adjustments.
Drug Abuse and Dependence (DRUG ABUSE AND DEPENDENCE)
Anafranil has not been systematically studied in animals or humans for its potential for abuse, tolerance, or physical dependence. While a variety of withdrawal symptoms have been described in association with Anafranil discontinuation (see PRECAUTIONS, Withdrawal Symptoms ), there is no evidence for drug-seeking behavior, except for a single report of potential Anafranil abuse by a patient with a history of dependence on codeine, benzodiazepines, and multiple psychoactive drugs. The patient received Anafranil for depression and panic attacks and appeared to become dependent after hospital discharge.
Despite the lack of evidence suggesting an abuse liability for Anafranil in foreign marketing, it is not possible to predict the extent to which Anafranil might be misused or abused once marketed in the U.S. Consequently, physicians should carefully evaluate patients for a history of drug abuse and follow such patients closely.
Clinical Worsening and Suicide Risk
Patients with major depressive disorder (MDD), both adult and pediatric, may experience worsening of their depression and/or the emergence of suicidal ideation and behavior (suicidality) or unusual changes in behavior, whether or not they are taking antidepressant medications, and this risk may persist until significant remission occurs. Suicide is a known risk of depression and certain other psychiatric disorders, and these disorders themselves are the strongest predictors of suicide. There has been a long-standing concern, however, that antidepressants may have a role in inducing worsening of depression and the emergence of suicidality in certain patients during the early phases of treatment. Pooled analyses of short-term placebo-controlled trials of antidepressant drugs (SSRIs and others) showed that these drugs increase the risk of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults (ages 18 to 24) with major depressive disorder (MDD) and other psychiatric disorders. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction with antidepressants compared to placebo in adults aged 65 and older.
The pooled analyses of placebo-controlled trials in children and adolescents with MDD, obsessive compulsive disorder (OCD), or other psychiatric disorders included a total of 24 short-term trials of 9 antidepressant drugs in over 4400 patients. The pooled analyses of placebo-controlled trials in adults with MDD or other psychiatric disorders included a total of 295 short-term trials (median duration of 2 months) of 11 antidepressant drugs in over 77,000 patients. There was considerable variation in risk of suicidality among drugs, but a tendency toward an increase in the younger patients for almost all drugs studied. There were differences in absolute risk of suicidality across the different indications, with the highest incidence in MDD. The risk differences (drug vs placebo), however, were relatively stable within age strata and across indications. These risk differences (drug-placebo difference in the number of cases of suicidality per 1000 patients treated) are provided in Table 1.
| Age Range | Drug-Placebo Difference in
Number of Cases of Suicidality per 1000 Patients Treated |
| Increases Compared to Placebo | |
| <18 | 14 additional cases |
| 18-24 | 5 additional cases |
| Decreases Compared to Placebo | |
| 25-64 | 1 fewer case |
| ≥65 | 6 fewer cases |
No suicides occurred in any of the pediatric trials. There were suicides in the adult trials, but the number was not sufficient to reach any conclusion about drug effect on suicide.
It is unknown whether the suicidality risk extends to longer-term use, i.e., beyond several months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with depression that the use of antidepressants can delay the recurrence of depression.
All patients being treated with antidepressants for any indication should be monitored appropriately and observed closely for clinical worsening, suicidality, and unusual changes in behavior, especially during the initial few months of a course of drug therapy, or at times of dose changes, either increases or decreases.
The following symptoms, anxiety, agitation, panic attacks, insomnia, irritability, hostility, aggressiveness, impulsivity, akathisia (psychomotor restlessness), hypomania, and mania, have been reported in adult and pediatric patients being treated with antidepressants for major depressive disorder as well as for other indications, both psychiatric and nonpsychiatric. Although a causal link between the emergence of such symptoms and either the worsening of depression and/or the emergence of suicidal impulses has not been established, there is concern that such symptoms may represent precursors to emerging suicidality.
Consideration should be given to changing the therapeutic regimen, including possibly discontinuing the medication, in patients whose depression is persistently worse, or who are experiencing emergent suicidality or symptoms that might be precursors to worsening depression or suicidality, especially if these symptoms are severe, abrupt in onset, or were not part of the patient's presenting symptoms.
Families and caregivers of patients being treated with antidepressants for major depressive disorder or other indications, both psychiatric and nonpsychiatric, should be alerted about the need to monitor patients for the emergence of agitation, irritability, unusual changes in behavior, and the other symptoms described above, as well as the emergence of suicidality, and to report such symptoms immediately to healthcare providers. Such monitoring should include daily observation by families and caregivers. Prescriptions for clomipramine hydrochloride should be written for the smallest quantity of capsules consistent with good patient management, in order to reduce the risk of overdose.
Monoamine Oxidase Inhibitors (maois) (Monoamine Oxidase Inhibitors (MAOIs))
(See CONTRAINDICATIONS, WARNINGS, and DOSAGE AND ADMINISTRATION .)
Incidence in Controlled Clinical Trials
The following table enumerates adverse events that occurred at an incidence of 1% or greater among patients with OCD who received Anafranil in adult or pediatric placebo-controlled clinical trials. The frequencies were obtained from pooled data of clinical trials involving either adults receiving Anafranil (N=322) or placebo (N=319) or children treated with Anafranil (N=46) or placebo (N=44). The prescriber should be aware that these figures cannot be used to predict the incidence of side effects in the course of usual medical practice, in which patient characteristics and other factors differ from those that prevailed in the clinical trials. Similarly, the cited frequencies cannot be compared with figures obtained from other clinical investigations involving different treatments, uses, and investigators. The cited figures, however, provide the physician with a basis for estimating the relative contribution of drug and nondrug factors to the incidence of side effects in the populations studied.
|
*Events reported by at least 1% of Anafranil patients are included. |
|||||
| Adults | Children and Adolescents | ||||
| Body System/ Adverse Event* |
Anafranil (N=322) |
Placebo (N=319) |
Anafranil (N=46) |
Placebo (N=44) |
|
| Nervous System | |||||
| Somnolence | 54 | 16 | 46 | 11 | |
| Tremor | 54 | 2 | 33 | 2 | |
| Dizziness | 54 | 14 | 41 | 14 | |
| Headache | 52 | 41 | 28 | 34 | |
| Insomnia | 25 | 15 | 11 | 7 | |
| Libido change | 21 | 3 | - | - | |
| Nervousness | 18 | 2 | 4 | 2 | |
| Myoclonus | 13 | - | 2 | - | |
| Increased appetite | 11 | 2 | - | 2 | |
| Paresthesia | 9 | 3 | 2 | 2 | |
| Memory impairment | 9 | 1 | 7 | 2 | |
| Anxiety | 9 | 4 | 2 | - | |
| Twitching | 7 | 1 | 4 | 5 | |
| Impaired concentration | 5 | 2 | - | - | |
| Depression | 5 | 1 | - | - | |
| Hypertonia | 4 | 1 | 2 | - | |
| Sleep disorder | 4 | - | 9 | 5 | |
| Psychosomatic disorder | 3 | - | - | - | |
| Yawning | 3 | - | - | - | |
| Confusion | 3 | - | 2 | - | |
| Speech disorder | 3 | - | - | - | |
| Abnormal dreaming | 3 | - | - | 2 | |
| Agitation | 3 | - | - | - | |
| Migraine | 3 | - | - | - | |
| Depersonalization | 2 | - | 2 | - | |
| Irritability | 2 | 2 | 2 | - | |
| Emotional lability | 2 | - | - | 2 | |
| Panic reaction | 1 | - | 2 | - | |
| Aggressive reaction | - | - | 2 | - | |
| Paresis | - | - | 2 | - | |
| Skin and Appendages | |||||
| Increased sweating | 29 | 3 | 9 | - | |
| Rash | 8 | 1 | 4 | 2 | |
| Pruritus | 6 | - | 2 | 2 | |
| Dermatitis | 2 | - | - | 2 | |
| Acne | 2 | 2 | - | 5 | |
| Dry skin | 2 | - | - | 5 | |
| Urticaria | 1 | - | - | - | |
| Abnormal skin odor | - | - | 2 | - | |
| Digestive System | |||||
| Dry mouth | 84 | 17 | 63 | 16 | |
| Constipation | 47 | 11 | 22 | 9 | |
| Nausea | 33 | 14 | 9 | 11 | |
| Dyspepsia | 22 | 10 | 13 | 2 | |
| Diarrhea | 13 | 9 | 7 | 5 | |
| Anorexia | 12 | - | 22 | 2 | |
| Abdominal pain | 11 | 9 | 13 | 16 | |
| Vomiting | 7 | 2 | 7 | - | |
| Flatulence | 6 | 3 | - | 2 | |
| Tooth disorder | 5 | - | - | - | |
| Gastrointestinal disorder | 2 | - | - | 2 | |
| Dysphagia | 2 | - | - | - | |
| Esophagitis | 1 | - | - | - | |
| Eructation | - | - | 2 | 2 | |
| Ulcerative stomatitis | - | - | 2 | - | |
| Body as a Whole | |||||
| Fatigue | 39 | 18 | 35 | 9 | |
| Weight increase | 18 | 1 | 2 | - | |
| Flushing | 8 | - | 7 | - | |
| Hot flushes | 5 | - | 2 | - | |
| Chest pain | 4 | 4 | 7 | - | |
| Fever | 4 | - | 2 | 7 | |
| Allergy | 3 | 3 | 7 | 5 | |
| Pain | 3 | 2 | 4 | 2 | |
| Local edema | 2 | 4 | - | - | |
| Chills | 2 | 1 | - | - | |
| Weight decrease | - | - | 7 | - | |
| Otitis media | - | - | 4 | 5 | |
| Asthenia | - | - | 2 | - | |
| Halitosis | - | - | 2 | - | |
| Cardiovascular System | |||||
| Postural hypotension | 6 | - | 4 | - | |
| Palpitation | 4 | 2 | 4 | - | |
| Tachycardia | 4 | - | 2 | - | |
| Syncope | - | - | 2 | - | |
| Respiratory System | |||||
| Pharyngitis | 14 | 9 | - | 5 | |
| Rhinitis | 12 | 10 | 7 | 9 | |
| Sinusitis | 6 | 4 | 2 | 5 | |
| Coughing | 6 | 6 | 4 | 5 | |
| Bronchospasm | 2 | - | 7 | 2 | |
| Epistaxis | 2 | - | - | 2 | |
| Dyspnea | - | - | 2 | - | |
| Laryngitis | - | 1 | 2 | - | |
| Urogenital System | |||||
| Male and Female Patients Combined | |||||
| Micturition disorder | 14 | 2 | 4 | 2 | |
| Urinary tract infection | 6 | 1 | - | - | |
| Micturition frequency | 5 | 3 | - | - | |
| Urinary retention | 2 | - | 7 | - | |
| Dysuria | 2 | 2 | - | - | |
| Cystitis | 2 | - | - | - | |
| Female Patients Only | (N=182) | (N=167) | (N=10) | (N=21) | |
| Dysmenorrhea | 12 | 14 | 10 | 10 | |
| Lactation (nonpuerperal) | 4 | - | - | - | |
| Menstrual disorder | 4 | 2 | - | - | |
| Vaginitis | 2 | - | - | - | |
| Leukorrhea | 2 | - | - | - | |
| Breast enlargement | 2 | - | - | - | |
| Breast pain | 1 | - | - | - | |
| Amenorrhea | 1 | - | - | - | |
| Male Patients Only | (N=140) | (N=152) | (N=36) | (N=23) | |
| Ejaculation failure | 42 | 2 | 6 | - | |
| Impotence | 20 | 3 | - | - | |
| Special Senses | |||||
| Abnormal vision | 18 | 4 | 7 | 2 | |
| Taste perversion | 8 | - | 4 | - | |
| Tinnitus | 6 | - | 4 | - | |
| Abnormal lacrimation | 3 | 2 | - | - | |
| Mydriasis | 2 | - | - | - | |
| Conjunctivitis | 1 | - | - | - | |
| Anisocoria | - | - | 2 | - | |
| Blepharospasm | - | - | 2 | - | |
| Ocular allergy | - | - | 2 | - | |
| Vestibular disorder | - | - | 2 | 2 | |
| Musculoskeletal | |||||
| Myalgia | 13 | 9 | - | - | |
| Back pain | 6 | 6 | - | - | |
| Arthralgia | 3 | 5 | - | - | |
| Muscle weakness | 1 | - | 2 | - | |
| Hemic and Lymphatic | |||||
| Purpura | 3 | - | - | - | |
| Anemia | - | - | 2 | 2 | |
| Metabolic and Nutritional | |||||
| Thirst | 2 | 2 | - | 2 | |
Leading to Discontinuation of Treatment
Approximately 20% of 3616 patients who received Anafranil in U.S. premarketing clinical trials discontinued treatment because of an adverse event. Approximately one-half of the patients who discontinued (9% of the total) had multiple complaints, none of which could be classified as primary. Where a primary reason for discontinuation could be identified, most patients discontinued because of nervous system complaints (5.4%), primarily somnolence. The second-most-frequent reason for discontinuation was digestive system complaints (1.3%), primarily vomiting and nausea.
There was no apparent relationship between the adverse events and elevated plasma drug concentrations.
Initial Treatment/dose Adjustment (adults) (Initial Treatment/Dose Adjustment (Adults))
Treatment with Anafranil should be initiated at a dosage of 25 mg daily and gradually increased, as tolerated, to approximately 100 mg during the first 2 weeks. During initial titration, Anafranil should be given in divided doses with meals to reduce gastrointestinal side effects. Thereafter, the dosage may be increased gradually over the next several weeks, up to a maximum of 250 mg daily. After titration, the total daily dose may be given once daily at bedtime to minimize daytime sedation.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No evidence of carcinogenicity was found in two 2-year bioassays in rats at doses up to 100 mg/kg, which is 24 and 4 times the maximum recommended human daily dose (MRHD) on a mg/kg and mg/m2 basis, respectively, or in a 2-year bioassay in mice at doses up to 80 mg/kg, which is 20 and 1.5 times the MRHD on a mg/kg and mg/m2 basis, respectively.
In reproduction studies, no effects on fertility were found in rats given up to 24 mg/kg, which is 6 times, and approximately equal to, the MRHD on a mg/kg and mg/m2 basis, respectively.
Package Label Principal Display Panel 25 Mg Bottle (PACKAGE LABEL - PRINCIPAL DISPLAY PANEL - 25 mg Bottle)
NDC 0406-9906-03
30 Capsules
Anafranil™
(clomiPRAMINE Hydrochloride)
Capsules USP
25 mg
Rx only
Each capsule contains:
Clomipramine Hydrochloride USP ... 25 mg
PHARMACIST: PLEASE DISPENSE WITH ATTACHED MEDICATION GUIDE
MALLINCKRODT™
Rev 09/2024
2000017741
Package Label Principal Display Panel 50 Mg Bottle (PACKAGE LABEL - PRINCIPAL DISPLAY PANEL - 50 mg Bottle)
NDC 0406-9907-03
30 Capsules
Anafranil™
(clomiPRAMINE Hydrochloride)
Capsules USP
50 mg
Rx only
Each capsule contains:
Clomipramine Hydrochloride USP ... 50 mg
PHARMACIST: PLEASE DISPENSE WITH ATTACHED MEDICATION GUIDE
MALLINCKRODT™
Rev 9/2024
2000017742
Package Label Principal Display Panel 75 Mg Bottle (PACKAGE LABEL - PRINCIPAL DISPLAY PANEL - 75 mg Bottle)
NDC 0406-9908-03
30 Capsules
Anafranil™
(clomiPRAMINE Hydrochloride)
Capsules USP
75 mg
Rx only
Each capsule contains:
Clomipramine Hydrochloride USP ... 75 mg
PHARMACIST: PLEASE DISPENSE WITH ATTACHED MEDICATION GUIDE
MALLINCKRODT™
Rev 09/2024
2000017743
Initial Treatment/dose Adjustment (children and Adolescents) (Initial Treatment/Dose Adjustment (Children and Adolescents))
As with adults, the starting dose is 25 mg daily and should be gradually increased (also given in divided doses with meals to reduce gastrointestinal side effects) during the first 2 weeks, as tolerated, up to a daily maximum of 3 mg/kg or 100 mg, whichever is smaller. Thereafter, the dosage may be increased gradually over the next several weeks up to a daily maximum of 3 mg/kg or 200 mg, whichever is smaller (see PRECAUTIONS, Pediatric Use ). As with adults, after titration, the total daily dose may be given once daily at bedtime to minimize daytime sedation.
Other Events Observed During the Premarketing Evaluation of Anafranil
During clinical testing in the U.S., multiple doses of Anafranil were administered to approximately 3600 subjects. Untoward events associated with this exposure were recorded by clinical investigators using terminology of their own choosing. Consequently, it is not possible to provide a meaningful estimate of the proportion of individuals experiencing adverse events without first grouping similar types of untoward events into a smaller number of standardized event categories.
In the tabulations that follow, a modified World Health Organization dictionary of terminology has been used to classify reported adverse events. The frequencies presented, therefore, represent the proportion of the 3525 individuals exposed to Anafranil who experienced an event of the type cited on at least one occasion while receiving Anafranil. All events are included except those already listed in the previous table, those reported in terms so general as to be uninformative, and those in which an association with the drug was remote. It is important to emphasize that although the events reported occurred during treatment with Anafranil, they were not necessarily caused by it.
Events are further categorized by body system and listed in order of decreasing frequency according to the following definitions: frequent adverse events are those occurring on one or more occasions in at least 1/100 patients; infrequent adverse events are those occurring in 1/100 to 1/1000 patients; rare events are those occurring in less than 1/1000 patients.
Maintenance/continuation Treatment (adults, Children, and Adolescents) (Maintenance/Continuation Treatment (Adults, Children, and Adolescents))
While there are no systematic studies that answer the question of how long to continue Anafranil, OCD is a chronic condition and it is reasonable to consider continuation for a responding patient. Although the efficacy of Anafranil after 10 weeks has not been documented in controlled trials, patients have been continued in therapy under double-blind conditions for up to 1 year without loss of benefit. However, dosage adjustments should be made to maintain the patient on the lowest effective dosage, and patients should be periodically reassessed to determine the need for treatment. During maintenance, the total daily dose may be given once daily at bedtime.
Use of Anafranil With other Maois, Such As Linezolid Or Methylene Blue (Use of Anafranil With Other MAOIs, Such as Linezolid or Methylene Blue)
Do not start Anafranil in a patient who is being treated with linezolid or intravenous methylene blue because there is increased risk of serotonin syndrome. In a patient who requires more urgent treatment of a psychiatric condition, other interventions, including hospitalization, should be considered (see CONTRAINDICATIONS ).
In some cases, a patient already receiving Anafranil therapy may require urgent treatment with linezolid or intravenous methylene blue. If acceptable alternatives to linezolid or intravenous methylene blue treatment are not available and the potential benefits of linezolid or intravenous methylene blue treatment are judged to outweigh the risks of serotonin syndrome in a particular patient, Anafranil should be stopped promptly, and linezolid or intravenous methylene blue can be administered. The patient should be monitored for symptoms of serotonin syndrome for two weeks or until
24 hours after the last dose of linezolid or intravenous methylene blue, whichever comes first. Therapy with Anafranil may be resumed 24 hours after the last dose of linezolid or intravenous methylene blue (see
WARNINGS
).
The risk of administering methylene blue by non-intravenous routes (such as oral tablets or by local injection) or in intravenous doses much lower than 1 mg/kg with Anafranil is unclear. The clinician should, nevertheless, be aware of the possibility of emergent symptoms of serotonin syndrome with such use (see WARNINGS ).
Medication Guide Anafranil™ (clomipramine Hydrochloride) Capsules Usp (Medication Guide - Anafranil™ (clomipramine hydrochloride) Capsules USP)
Read the Medication Guide that comes with you or your family member's antidepressant medicine. This Medication Guide is only about the risk of suicidal thoughts and actions with antidepressant medicines. Talk to your, or your family member's, healthcare provider about:
- all risks and benefits of treatment with antidepressant medicines
- all treatment choices for depression or other serious mental illness
What is the most important information I should know about antidepressant medicines, depression and other serious mental illnesses, and suicidal thoughts or actions?
-
Antidepressant medicines may increase suicidal thoughts or actions in some children, teenagers, and young adults within the first few months of treatment.
- Depression and other serious mental illnesses are the most important causes of suicidal thoughts and actions. Some people may have a particularly high risk of having suicidal thoughts or actions. These include people who have (or have a family history of) bipolar illness (also called manic-depressive illness) or suicidal thoughts or actions.
-
How can I watch for and try to prevent suicidal thoughts and actions in myself or a family member?
- Pay close attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings. This is very important when an antidepressant medicine is started or when the dose is changed.
- Call the healthcare provider right away to report new or sudden changes in mood, behavior, thoughts, or feelings.
- Keep all follow-up visits with the healthcare provider as scheduled. Call the healthcare provider between visits as needed, especially if you have concerns about symptoms.
Call a healthcare provider right away if you or your family member has any of the following symptoms, especially if they are new, worse, or worry you:
- thoughts about suicide or dying
- attempts to commit suicide
- new or worse depression
- new or worse anxiety
- feeling very agitated or restless
- panic attacks
- trouble sleeping (insomnia)
- new or worse irritability
- acting aggressive, being angry, or violent
- acting on dangerous impulses
- an extreme increase in activity and talking (mania)
- other unusual changes in behavior or mood
Low salt (sodium) levels in the blood. Elderly people may be at greater risk for this. Symptoms may include:
- headache
- weakness or feeling unsteady
- confusion, problems concentrating or thinking or memory problems
Visual problems
- eye pain
- changes in vision
- swelling or redness in or around the eye
Only some people are at risk for these problems. You may want to undergo an eye examination to see if you are at risk and receive preventative treatment if you are.
Who should not take Anafranil?
Do not take Anafranil if you:
- take a monoamine oxidase inhibitor (MAOI). Ask your healthcare provider or pharmacist if you are not sure if you take an MAOI, including the antibiotic linezolid.
- Do not take an MAOI within 2 weeks of stopping Anafranil unless directed to do so by your physician.
- Do not start Anafranil if you stopped taking an MAOI in the last 2 weeks unless directed to do so by your physician.
What else do I need to know about antidepressant medicines?
- Never stop an antidepressant medicine without first talking to a healthcare provider. Stopping an antidepressant medicine suddenly can cause other symptoms.
- Antidepressants are medicines used to treat depression and other illnesses. It is important to discuss all the risks of treating depression and also the risks of not treating it. Patients and their families or other caregivers should discuss all treatment choices with the healthcare provider, not just the use of antidepressants.
- Antidepressant medicines have other side effects. Talk to the healthcare provider about the side effects of the medicine prescribed for you or your family member.
- Antidepressant medicines can interact with other medicines. Know all of the medicines that you or your family member takes. Keep a list of all medicines to show the healthcare provider. Do not start new medicines without first checking with your healthcare provider.
- Not all antidepressant medicines prescribed for children are FDA approved for use in children. Talk to your child's healthcare provider for more information.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Anafranil is a trademark of Mallinckrodt LLC.
Product of Canada, Active Ingredient made in Ireland
Manufactured by
Patheon Inc.
Whitby, Ontario, Canada
L1N 5Z5
for SpecGx LLC
Webster Groves, MO 63119 USA
Rev 09/2024
Mallinckrodt™
Switching A Patient to Or From A Monoamine Oxidase Inhibitor (maoi) Intended to Treat Psychiatric Disorders (Switching a Patient To or From a Monoamine Oxidase Inhibitor (MAOI) Intended to Treat Psychiatric Disorders)
At least 14 days should elapse between discontinuation of an MAOI intended to treat psychiatric disorders and initiation of therapy with Anafranil. Conversely, at least 14 days should be allowed after stopping Anafranil before starting an MAOI intended to treat psychiatric disorders (see CONTRAINDICATIONS ).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:59.487737 · Updated: 2026-03-14T22:18:26.158923