These Highlights Do Not Include All The Information Needed To Use Busulfan Injection Safely And Effectively. See Full Prescribing Information For Busulfan Injection.
4049d57f-342e-4233-8292-fed6b8f128ec
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Busulfan Injection causes severe and prolonged myelosuppression at the recommended dosage. Hematopoietic progenitor cell transplantation is required to prevent potentially fatal complications of the prolonged myelosuppression [ see Warnings and Precautions (5.1) ] .
Indications and Usage
Busulfan injection is indicated for use in combination with cyclophosphamide as a conditioning regimen prior to allogeneic hematopoietic progenitor cell transplantation for chronic myelogenous leukemia.
Dosage and Administration
Pre-medicate with anticonvulsants (e.g. benzodiazepines, phenytoin, valproic acid or levetiracetam) and antiemetic. ( 2.1 , 5.2 ) Dilute and administer as intravenous infusion. Do not administer as intravenous push or bolus. ( 2.1 , 2.3 ) Recommended adult dose: 0.8 mg per kg of ideal body weight or actual body weight, whichever is lower, administered intravenously via a central venous catheter as a two-hour infusion every six hours for four consecutive days for a total of 16 doses. ( 2.1 )
Warnings and Precautions
Seizures: Initiate anticonvulsant prophylactic therapy prior to treatment with busulfan injection. Monitor patients with history of seizure disorder, head trauma or receiving epileptogenic drugs ( 5.2 ) Hepatic Veno-Occlusive Disease (HVOD): Increased risk of developing HVOD at AUC greater than 1,500 μMmin. Monitor serum transaminases, alkaline phosphatase and bilirubin daily ( 5.3 ) Embryo-fetal Toxicity: Can cause fetal harm. Advise of potential risk to a fetus and use of effective contraception ( 5.4 , 8.1 , 8.3 ) Cardiac tamponade has been reported in pediatric patients with thalassemia who received high doses of oral busulfan and cyclophosphamide. Abdominal pain and vomiting precede the tamponade in most patients ( 5.5 )
Contraindications
Busulfan is contraindicated in patients with a history of hypersensitivity to any of its components.
Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling: Myelosuppression [ see Warnings and Precautions (5.1) ] Seizures [ see Warnings and Precautions (5.2) ] Hepatic Veno-Occlusive Disease (HVOD) [ see Warnings and Precautions (5.3) ] Embryo-fetal Toxicity [ see Warnings and Precautions (5.4) ] Cardiac Tamponade [ see Warnings and Precautions (5.5) ] Bronchopulmonary Dysplasia [ see Warnings and Precautions (5.6) ] Cellular Dysplasia [ see Warnings and Precautions (5.7) ]
Drug Interactions
Drugs that Decrease busulfan Clearance: Metronidazole, itraconazole, Iron chelating agent acetaminophen. ( 7.1 ) Drugs that Increases Busulfan Clearance: Phenytoin. ( 7.2 )
Storage and Handling
Unopened vials of busulfan injection must be stored under refrigerated conditions between 2°C to 8°C (36°F to 46°F). Busulfan injection diluted in 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP is stable at room temperature (25°C) for up to 8 hours but the infusion must be completed within that time. Busulfan injection diluted in 0.9% Sodium Chloride Injection, USP is stable at refrigerated conditions (2°C to 8°C) for up to 12 hours but the infusion must be completed within that time. Busulfan injection is a cytotoxic drug. Follow applicable special handling and disposal procedures 1 .
How Supplied
Busulfan injection is packaged as a sterile solution in 10 mL single-dose clear glass vials each containing 60 mg of busulfan at a concentration of 6 mg per mL for intravenous use, NDC 72485-210-01. Busulfan injection is distributed as a unit carton of eight vials NDC 72485-210-08
Medication Information
Warnings and Precautions
Seizures: Initiate anticonvulsant prophylactic therapy prior to treatment with busulfan injection. Monitor patients with history of seizure disorder, head trauma or receiving epileptogenic drugs ( 5.2 ) Hepatic Veno-Occlusive Disease (HVOD): Increased risk of developing HVOD at AUC greater than 1,500 μMmin. Monitor serum transaminases, alkaline phosphatase and bilirubin daily ( 5.3 ) Embryo-fetal Toxicity: Can cause fetal harm. Advise of potential risk to a fetus and use of effective contraception ( 5.4 , 8.1 , 8.3 ) Cardiac tamponade has been reported in pediatric patients with thalassemia who received high doses of oral busulfan and cyclophosphamide. Abdominal pain and vomiting precede the tamponade in most patients ( 5.5 )
Indications and Usage
Busulfan injection is indicated for use in combination with cyclophosphamide as a conditioning regimen prior to allogeneic hematopoietic progenitor cell transplantation for chronic myelogenous leukemia.
Dosage and Administration
Pre-medicate with anticonvulsants (e.g. benzodiazepines, phenytoin, valproic acid or levetiracetam) and antiemetic. ( 2.1 , 5.2 ) Dilute and administer as intravenous infusion. Do not administer as intravenous push or bolus. ( 2.1 , 2.3 ) Recommended adult dose: 0.8 mg per kg of ideal body weight or actual body weight, whichever is lower, administered intravenously via a central venous catheter as a two-hour infusion every six hours for four consecutive days for a total of 16 doses. ( 2.1 )
Contraindications
Busulfan is contraindicated in patients with a history of hypersensitivity to any of its components.
Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling: Myelosuppression [ see Warnings and Precautions (5.1) ] Seizures [ see Warnings and Precautions (5.2) ] Hepatic Veno-Occlusive Disease (HVOD) [ see Warnings and Precautions (5.3) ] Embryo-fetal Toxicity [ see Warnings and Precautions (5.4) ] Cardiac Tamponade [ see Warnings and Precautions (5.5) ] Bronchopulmonary Dysplasia [ see Warnings and Precautions (5.6) ] Cellular Dysplasia [ see Warnings and Precautions (5.7) ]
Drug Interactions
Drugs that Decrease busulfan Clearance: Metronidazole, itraconazole, Iron chelating agent acetaminophen. ( 7.1 ) Drugs that Increases Busulfan Clearance: Phenytoin. ( 7.2 )
Storage and Handling
Unopened vials of busulfan injection must be stored under refrigerated conditions between 2°C to 8°C (36°F to 46°F). Busulfan injection diluted in 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP is stable at room temperature (25°C) for up to 8 hours but the infusion must be completed within that time. Busulfan injection diluted in 0.9% Sodium Chloride Injection, USP is stable at refrigerated conditions (2°C to 8°C) for up to 12 hours but the infusion must be completed within that time. Busulfan injection is a cytotoxic drug. Follow applicable special handling and disposal procedures 1 .
How Supplied
Busulfan injection is packaged as a sterile solution in 10 mL single-dose clear glass vials each containing 60 mg of busulfan at a concentration of 6 mg per mL for intravenous use, NDC 72485-210-01. Busulfan injection is distributed as a unit carton of eight vials NDC 72485-210-08
Description
Busulfan Injection causes severe and prolonged myelosuppression at the recommended dosage. Hematopoietic progenitor cell transplantation is required to prevent potentially fatal complications of the prolonged myelosuppression [ see Warnings and Precautions (5.1) ] .
Section 42229-5
Additional Adverse Reactions by Body System
Hematologic : Prolonged prothrombin time
Gastrointestinal: Esophagitis, ileus, hematemesis, pancreatitis, rectal discomfort
Hepatic: Alkaline phosphatase increases, jaundice, hepatomegaly
Graft-versus-host disease: Graft-versus-host disease. There were 3 deaths (5%) attributed to GVHD.
Edema: Hypervolemia, or documented weight increase
Infection: Infection, pneumonia (fatal in one patient and life-threatening in 3% of patients)
Cardiovascular: Arrhythmia, atrial fibrillation, ventricular extrasystoles, third degree heart block, thrombosis (all episodes were associated with the central venous catheter), hypotension, flushing and hot flashes, cardiomegaly, ECG abnormality, left-sided heart failure, and pericardial effusion
Pulmonary : Hyperventilation, alveolar hemorrhage (fatal in 3%), pharyngitis, hiccup, asthma, atelectasis, pleural effusion, hypoxia, hemoptysis, sinusitis, and interstitial fibrosis (fatal in a single case)
Neurologic: Cerebral hemorrhage, coma, delirium, agitation, encephalopathy, confusion, hallucinations, lethargy, somnolence
Renal: BUN increased, dysuria, oliguria, hematuria, hemorrhagic cystitis
Skin: Alopecia, vesicular rash, maculopapular rash, vesiculo-bullous rash, exfoliative dermatitis, erythema nodosum, acne, skin discoloration
Metabolic: Hypophosphatemia, hyponatremia
Other Events: Injection site pain, myalgia, arthralgia, ear disorder
Seizures
Advise patients of the possibility of seizures and that they will be given medication to prevent them. Patients should be asked to report a history of seizure or head trauma [see Warnings and Precautions (5.2) ].
Lactation
Advise females to discontinue breastfeeding during treatment with busulfan injection [see Use in Specific Populations (8.2) ].
Packaging
Infertility
Advise females and males of reproductive potential that busulfan injection may cause temporary or permanent infertility [see Use in Specific Populations (8.3) ].
5.2 Seizures
Seizures have been reported in patients receiving high-dose oral busulfan at doses producing plasma drug levels similar to those achieved following the recommended dosage of busulfan. Despite prophylactic therapy with phenytoin, one seizure (1/42 patients) was reported during an autologous transplantation clinical trial of busulfan. This episode occurred during the cyclophosphamide portion of the conditioning regimen, 36 hours after the last busulfan dose. Initiate phenytoin therapy or any other alternative anti-convulsant prophylactic therapy (e.g., benzodiazepines, valproic acid or levetiracetam) prior to busulfan treatment [See Dosage and Administration (2.1) ]. Use caution when administering the recommended dose of busulfan to patients with a history of a seizure disorder or head trauma or who are receiving other potentially epileptogenic drugs.
10 Overdosage
There is no known antidote to busulfan injection other than hematopoietic progenitor cell transplantation. In the absence of hematopoietic progenitor cell transplantation, the recommended dosage for busulfan would constitute an overdose of busulfan. The principal toxic effect is profound bone marrow hypoplasia/aplasia and pancytopenia, but the central nervous system, liver, lungs, and gastrointestinal tract may be affected. Monitor hematologic status closely and institute vigorous supportive measures as medically indicated. Survival after a single 140 mg dose of Myleran Tablets in an 18 kg, 4-year old child has been reported. Inadvertent administration of a greater than normal dose of oral busulfan (2.1 mg per kg; total dose of 23.3 mg per kg) occurred in a 2-year old child prior to a scheduled bone marrow transplant without sequelae. An acute dose of 2.4 g was fatal in a 10-year old boy. There is one report that busulfan is dialyzable, thus dialysis should be considered in the case of overdose.
15 References
1. OSHA Hazardous Drugs. OSHA. [Accessed on June 18, 2014 from http://www.osha.gov/SLTC/hazardousdrugs/index.html
11 Description
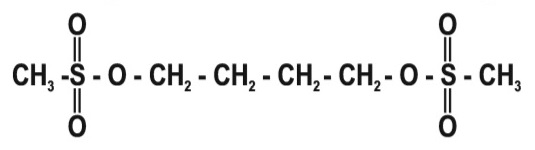
Busulfan is a bifunctional alkylating agent known chemically as 1,4-butanediol, dimethanesulfonate. Busulfan Injection is intended for intravenous administration. It is supplied as a clear, colorless, sterile, solution in 10 mL single-dose vials. Each vial of busulfan injection contains 60 mg (6 mg/mL) of busulfan, the active ingredient, a white crystalline powder with a molecular formula of CH3SO2O(CH2)4OSO2CH3 and a molecular weight of 246 g/mole. Busulfan has the following chemical structure:
Busulfan is dissolved in N,N-dimethylacetamide (DMA), 3.3 mL and Polyethylene Glycol 400, NF 6.7 mL. The solubility of busulfan in water is 0.1 g per L and the pH of busulfan injection diluted to approximately 0.5 mg per mL busulfan in 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP as recommended for infusion reflects the pH of the diluent used and ranges from 3.4 to 3.9.
Myelosuppression
Advise patients of the possibility of developing low blood cell counts and the need for hematopoietic progenitor cell infusion. Instruct patients to immediately report to their healthcare provider if fever develops [see Warnings and Precautions (5.1) ].
16.1 How Supplied
Busulfan injection is packaged as a sterile solution in 10 mL single-dose clear glass vials each containing 60 mg of busulfan at a concentration of 6 mg per mL for intravenous use, NDC 72485-210-01.
Busulfan injection is distributed as a unit carton of eight vials NDC 72485-210-08
8.4 Pediatric Use
The effectiveness of busulfan in the treatment of CML has not been specifically studied in pediatric patients. An open-label, uncontrolled study evaluated the pharmacokinetics of busulfan in 24 pediatric patients receiving busulfan as part of a conditioning regimen administered prior to hematopoietic progenitor cell transplantation for a variety of malignant hematologic (N=15) or non-malignant diseases (N=9). Patients ranged in age from 5 months to 16 years (median 3 years). Busulfan dosing was targeted to achieve an area under the plasma concentration curve (AUC) of 900-1350 μM•min with an initial dose of 0.8 mg per kg or 1.0 mg per kg (based on Actual Body Weight (ABW)) if the patient was greater than 4 or less than or equal to 4 years, respectively. The dose was adjusted based on plasma concentration after completion of dose 1.
Patients received busulfan doses every six hours as a two-hour infusion over four days for a total of 16 doses, followed by cyclophosphamide 50 mg per kg once daily for four days. After one rest day, hematopoietic progenitor cells were infused. All patients received phenytoin as seizure prophylaxis. The target AUC (900-1350±5% μM•min) for busulfan was achieved at dose 1 in 71% (17/24) of patients. Steady state pharmacokinetic testing was performed at dose 9 and 13. Busulfan levels were within the target range for 21 of 23 evaluable patients.
All 24 patients experienced neutropenia (absolute neutrophil count (ANC) less than 0.5×109/L) and thrombocytopenia (platelet transfusions or platelet count less than 20,000/mm3). Seventy-nine percent (19/24) of patients experienced lymphopenia (absolute lymphocyte count less than 0.1×109). In 23 patients, the ANC recovered to greater than 0.5×109/L (median time to recovery = BMT day +13; range = BMT day +9 to +22). One patient who died on day +20 had not recovered to an ANC >0.5×109/L.
Four (17%) patients died during the study. Two patients died within 28 days of transplant; one with pneumonia and capillary leak syndrome, and the other with pneumonia and veno-occlusive disease. Two patients died prior to day 100; one due to progressive disease and one due to multiorgan failure.
Adverse reactions were reported in all 24 patients during the study period (BMT day -10 through BMT day +28) or post-study surveillance period (day +29 through +100). These included vomiting (100%), nausea (83%), stomatitis (79%), HVOD (21%), graft-versus host disease (GVHD) (25%), and pneumonia (21%).
Based on the results of this 24-patient clinical trial, a suggested dosing regimen of busulfan in pediatric patients is shown in the following dosing nomogram:
| Patient's Actual Body Weight (ABW) | Busulfan Dosage |
|---|---|
| less than or equal to12 kg | 1.1 (mg per kg) |
| greater than 12 kg | 0.8 (mg per kg) |
Simulations based on a pediatric population pharmacokinetic model indicate that approximately 60% of pediatric patients will achieve a target busulfan exposure (AUC) between 900 to 1350 μM•min with the first dose of busulfan using this dosing nomogram. Therapeutic drug monitoring and dose adjustment following the first dose of busulfan is recommended.
8.5 Geriatric Use
Clinical studies of busulfan did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
Cardiac Tamponade
Advise patients of the risk of cardiac tamponade. Instruct patients to report to their healthcare provider symptoms of abdominal pain and vomiting [see Warnings and Precautions (5.5) ].
14 Clinical Studies
Documentation of the safety and efficacy of busulfan as a component of a conditioning regimen prior to allogeneic hematopoietic progenitor cell reconstitution is derived from two sources:
i) analysis of a prospective clinical trial of busulfan that involved 61 patients diagnosed with various hematologic malignancies, and
ii) the published reports of randomized, controlled trials that employed high-dose oral busulfan as a component of a conditioning regimen for transplantation, which were identified in a literature review of five established commercial databases.
4 Contraindications
Busulfan is contraindicated in patients with a history of hypersensitivity to any of its components.
6 Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Myelosuppression [see Warnings and Precautions (5.1) ]
- Seizures [see Warnings and Precautions (5.2) ]
- Hepatic Veno-Occlusive Disease (HVOD) [see Warnings and Precautions (5.3) ]
- Embryo-fetal Toxicity [see Warnings and Precautions (5.4) ]
- Cardiac Tamponade [see Warnings and Precautions (5.5) ]
- Bronchopulmonary Dysplasia [see Warnings and Precautions (5.6) ]
- Cellular Dysplasia [see Warnings and Precautions (5.7) ]
7 Drug Interactions
5.1 Myelosuppression
The most frequent serious consequence of treatment with busulfan at the recommended dose and schedule is prolonged myelosuppression, occurring in all patients (100%). Severe granulocytopenia, thrombocytopenia, anemia, or any combination thereof may develop. Hematopoietic progenitor cell transplantation is required to prevent potentially fatal complications of the prolonged myelosuppression. Monitor complete blood counts, including white blood cell differentials, and quantitative platelet counts daily during treatment and until engraftment is demonstrated. Absolute neutrophil counts dropped below 0.5x109/L at a median of 4 days post-transplant in 100% of patients treated in the busulfan clinical trial. The absolute neutrophil count recovered at a median of 13 days following allogeneic transplantation when prophylactic filgrastim was used in the majority of patients. Thrombocytopenia (less than 25,000/mm3 or requiring platelet transfusion) occurred at a median of 5-6 days in 98% of patients. Anemia (hemoglobin less than 8.0 g/dL) occurred in 69% of patients. Use antibiotic therapy and platelet and red blood cell support when medically indicated.
12.3 Pharmacokinetics
The pharmacokinetics of busulfan was studied in 59 patients participating in a prospective trial of a busulfan cyclophosphamide preparatory regimen prior to allogeneic hematopoietic progenitor stem cell transplantation. Patients received 0.8 mg/kg busulfan every six hours, for a total of 16 doses over four days. Fifty-five of fifty-nine patients (93%) administered busulfan maintained AUC values below the target value (less than1500 μM•min).
| Mean | CV (%) | Range | |
| Cmax (ng per mL) | 1222 | 18 | 496 - 1684 |
| AUC (μM∙min) | 1167 | 20 | 556 - 1673 |
| CL (mL per min per kg)1 | 2.52 | 25 | 1.49 - 4.31 |
| 1.Clearance normalized to actual body weight for all patients. |
Busulfan pharmacokinetics showed consistency between dose 9 and dose 13 as demonstrated by reproducibility of steady state Cmax and a low coefficient of variation for this parameter.
5.5 Cardiac Tamponade
Cardiac tamponade has been reported in pediatric patients with thalassemia (8/400 or 2% in one series) who received high doses of oral busulfan and cyclophosphamide as the preparatory regimen for hematopoietic progenitor cell transplantation. Six of the eight children died and two were saved by rapid pericardiocentesis. Abdominal pain and vomiting preceded the tamponade in most patients. Monitor for signs and symptoms, promptly evaluate and treat if cardiac tamponade is suspected.
Embryo Fetal Toxicity
Advise females of reproductive potential of the potential risk to a fetus and to inform their healthcare provider with a known or suspected pregnancy [see Warnings and Precautions (5.4) and Use in Specific Populations (8.1) ].
5.7 Cellular Dysplasia
Busulfan may cause cellular dysplasia in many organs. Cytologic abnormalities characterized by giant, hyperchromatic nuclei have been reported in lymph nodes, pancreas, thyroid, adrenal glands, liver, lungs and bone marrow. This cytologic dysplasia may be severe enough to cause difficulty in the interpretation of exfoliative cytologic examinations of the lungs, bladder, breast and the uterine cervix.
1 Indications and Usage
Busulfan injection is indicated for use in combination with cyclophosphamide as a conditioning regimen prior to allogeneic hematopoietic progenitor cell transplantation for chronic myelogenous leukemia.
12.1 Mechanism of Action
Busulfan is a bifunctional alkylating agent in which two labile methanesulfonate groups are attached to opposite ends of a four-carbon alkyl chain. In aqueous media, busulfan hydrolyzes to release the methanesulfonate groups. This produces reactive carbonium ions that can alkylate DNA. DNA damage is thought to be responsible for much of the cytotoxicity of busulfan
16.2 Storage and Handling
Unopened vials of busulfan injection must be stored under refrigerated conditions between 2°C to 8°C (36°F to 46°F).
Busulfan injection diluted in 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP is stable at room temperature (25°C) for up to 8 hours but the infusion must be completed within that time.
Busulfan injection diluted in 0.9% Sodium Chloride Injection, USP is stable at refrigerated conditions (2°C to 8°C) for up to 12 hours but the infusion must be completed within that time.
Busulfan injection is a cytotoxic drug. Follow applicable special handling and disposal procedures 1 .
5.4 Embryo Fetal Toxicity
Busulfan can cause fetal harm when administered to a pregnant woman based on animal data. Busulfan was teratogenic in mice, rats, and rabbits. The solvent, DMA, may also cause fetal harm when administered to a pregnant woman based on findings in animals. Advise pregnant women of the potential risk to a fetus. Advise females and males of reproductive potential to use effective contraception during and after treatment with busulfan [see Use in Specific Populations (8.1) and (8.3) ].
Warning: Myelosuppression
Busulfan Injection causes severe and prolonged myelosuppression at the recommended dosage. Hematopoietic progenitor cell transplantation is required to prevent potentially fatal complications of the prolonged myelosuppression [see Warnings and Precautions (5.1) ].
5 Warnings and Precautions
- Seizures: Initiate anticonvulsant prophylactic therapy prior to treatment with busulfan injection. Monitor patients with history of seizure disorder, head trauma or receiving epileptogenic drugs (5.2)
- Hepatic Veno-Occlusive Disease (HVOD): Increased risk of developing HVOD at AUC greater than 1,500 μMmin. Monitor serum transaminases, alkaline phosphatase and bilirubin daily (5.3)
- Embryo-fetal Toxicity: Can cause fetal harm. Advise of potential risk to a fetus and use of effective contraception (5.4, 8.1, 8.3)
- Cardiac tamponade has been reported in pediatric patients with thalassemia who received high doses of oral busulfan and cyclophosphamide. Abdominal pain and vomiting precede the tamponade in most patients (5.5)
Bronchopulmonary Dysplasia
Advise patients of the possibility of bronchopulmonary dysplasia with pulmonary fibrosis with chronic busulfan therapy. Instruct patients to report symptoms of shortness of breath and cough to their healthcare provider. These symptoms could occur several months or years after therapy with busulfan injection [see Warnings and Precautions (5.6) ].
2 Dosage and Administration
- Pre-medicate with anticonvulsants (e.g. benzodiazepines, phenytoin, valproic acid or levetiracetam) and antiemetic. ( 2.1, 5.2 )
- Dilute and administer as intravenous infusion. Do not administer as intravenous push or bolus. (2.1, 2.3)
- Recommended adult dose: 0.8 mg per kg of ideal body weight or actual body weight, whichever is lower, administered intravenously via a central venous catheter as a two-hour infusion every six hours for four consecutive days for a total of 16 doses. (2.1)
3 Dosage Forms and Strengths
Busulfan injection is supplied as a clear, colorless, sterile, solution in 10 mL single-dose vial containing 60 mg of busulfan at a concentration of 6 mg per mL for intravenous use only.
6.2 Postmarketing Experience
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The following adverse reactions have been identified during post-approval use of busulfan Injection:
Blood and Lymphatic System Disorders: febrile neutropenia
Gastrointestinal Disorders: tooth hypoplasia
Metabolism and Nutrition Disorders: tumor lysis syndrome
Vascular Disorders: thrombotic microangiopathy (TMA)
Infections and Infestations: severe bacterial, viral (e.g., cytomegalovirus viremia) and fungal infections; and sepsis.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse reaction information is primarily derived from the clinical study (N=61) of busulfan and the data obtained for high-dose oral busulfan conditioning in the setting of randomized, controlled trials identified through a literature review.
In the busulfan Injection allogeneic stem cell transplantation clinical trial, all patients were treated with busulfan 0.8 mg per kg as a two-hour infusion every six hours for 16 doses over four days, combined with cyclophosphamide 60 mg per kg x2 days. Ninety-three percent (93%) of evaluable patients receiving this dose of busulfan maintained an AUC less than 1,500 μM•min for dose 9, which has generally been considered the level that minimizes the risk of HVOD.
Table 1 lists the non-hematologic adverse reactions events through Bone Marrow Transplantation (BMT) Day +28 at a rate greater than or equal to 20% in patients treated with busulfan prior to allogeneic hematopoietic cell transplantation.
| Non-Hematological Adverse Reactions 1 | Percent Incidence |
| BODY AS A WHOLE | |
| Fever | 80 |
| Headache | 69 |
| Asthenia | 51 |
| Chills | 46 |
| Pain | 44 |
| Edema General | 28 |
| Allergic Reaction | 26 |
| Chest Pain | 26 |
| Inflammation at Injection Site | 25 |
| Back Pain | 23 |
| CARDIOVASCULAR SYSTEM | |
| Tachycardia | 44 |
| Hypertension | 36 |
| Thrombosis | 33 |
| Vasodilation | 25 |
| DIGESTIVE SYSTEM | |
| Nausea | 98 |
| Stomatitis (Mucositis) | 97 |
| Vomiting | 95 |
| Anorexia | 85 |
| Diarrhea | 84 |
| Abdominal Pain | 72 |
| Dyspepsia | 44 |
| Constipation | 38 |
| Dry Mouth | 26 |
| Rectal Disorder | 25 |
| Abdominal Enlargement | 23 |
| METABOLIC AND NUTRITIONAL SYSTEM | |
| Hypomagnesemia | 77 |
| Hyperglycemia | 66 |
| Hypokalemia | 64 |
| Hypocalcemia | 49 |
| Hyperbilirubinemia | 49 |
| Edema | 36 |
| SGPT Elevation | 31 |
| Creatinine Increased | 21 |
| NERVOUS SYSTEM | |
| Insomnia | 84 |
| Anxiety | 72 |
| Dizziness | 30 |
| Depression | 23 |
| RESPIRATORY SYSTEM | |
| Rhinitis | 44 |
| Lung Disorder | 34 |
| Cough | 28 |
| Epistaxis | 25 |
| Dyspnea | 25 |
| SKIN AND APPENDAGES | |
| Rash | 57 |
| Pruritus | 28 |
| 1. Includes all reported adverse reactions regardless of severity (toxicity grades 1-4) |
8 Use in Specific Populations
- Lactation: Discontinue breastfeeding (8.2)
2.1 Initial Dosing Information
- Administer busulfan injection in combination with cyclophosphamide as a conditioning regimen prior to bone marrow or peripheral blood progenitor cell replacement. For patients weighing more than 12 kg, the recommended doses are:
- Busulfan Injection 0.8 mg per kg (ideal body weight or actual body weight, whichever is lower) intravenously via a central venous catheter as a two-hour infusion every six hours for four consecutive days for a total of 16 doses (Days -7, -6, -5 and -4).
- Cyclophosphamide 60 mg per kg intravenously as a one-hour infusion on each of two days beginning no sooner than six hours following the 16th dose of busulfan injection (Days -3 and -2).
- Administer hematopoietic progenitor cells on Day 0.
- Premedicate patients with anticonvulsants (e.g., benzodiazepines, phenytoin, valproic acid or levetiracetam) to prevent seizures reported with the use of high dose busulfan injection. Administer anticonvulsants 12 hours prior to busulfan injection to 24 hours after the last dose of busulfan injection [see Warnings and Precautions (5.2) ].
- Administer antiemetics prior to the first dose of busulfan injection and continue on a fixed schedule through busulfan injection administration.
- Busulfan injection clearance is best predicted when the busulfan injection dose is administered based on adjusted ideal body weight. Dosing busulfan injection based on actual body weight, ideal body weight or other factors can produce significant differences in busulfan injection clearance among lean, normal and obese patients.
- Calculate ideal body weight (IBW) as follows (height in cm, and weight in kg):
Women: IBW (kg)=45+0.91x (height in cm -152)
- For obese or severely obese patients, base busulfan injection dosing on adjusted ideal body weight (AIBW):
5.6 Bronchopulmonary Dysplasia
Bronchopulmonary dysplasia with pulmonary fibrosis is a rare but serious complication following chronic busulfan therapy. The average onset of symptoms is 4 years after therapy (range 4 months to 10 years).
Males of Reproductive Potential
Advise males with female sexual partners of reproductive potential to use effective contraception during treatment with busulfan injection and for 3 months following cessation of therapy [see Use in Specific Populations (8.3) ].
Females of Reproductive Potential
Advise females of reproductive potential to use effective contraception during treatment with busulfan and for 6 months following cessation of therapy [see Use in Specific Populations (8.3) ].
6.3 Oral Busulfan Literature Review
A literature review identified four randomized, controlled trials that evaluated a high-dose oral busulfan-containing conditioning regimen for allogeneic bone marrow transplantation in the setting of CML [see Clinical Studies (14) ]. The safety outcomes reported in those trials are summarized in Table 2 below for a mixed population of hematological malignancies (AML, CML, and ALL).
|
Clift CML Chronic Phase |
|||||
| TRM1 | VOD2 | GVHD3 | Pulmonary | Hemorrhagic Cystitis | Seizure |
| Death ≤100d=4.1% (3/73) | No Report | Acute≥Grade 2=35% Chronic=41% (30/73) |
1 death from Idiopathic Interstitial Pneumonitis And 1 death from Pulmonary Fibrosis | No Report | No Report |
|
Devergie CML Chronic Phase |
|||||
| TRM | VOD | GVHD | Pulmonary | Hemorrhagic Cystitis | Seizure |
| 38% | 7.7% (5/65) Deaths=4.6% (3/65) |
Acute≥Grade 2=41% (24/59 at risk) | Interstitial Pneumonitis=16.9% (11/65) | 10.8% (7/65) | No report |
|
Ringden CML, AML, ALL |
|||||
| TRM | VOD | GVHD | Pulmonary | Hemorrhagic Cystitis | Seizure |
| 28% | 12% | Acute≥Grade 2 GVHD=26% Chronic GVHD=45% |
Interstitial Pneumonitis=14% | 24% | 6% |
|
Blume CML, AML, ALL |
|||||
| TRM | VOD | GVHD | Pulmonary | Hemorrhagic Cystitis | Seizure |
| No Report |
Deaths=4.9% |
Acute≥Grade 2 GVHD=22% (13/58 at risk) Chronic GVHD=31% (14/45 at risk) |
No Report |
No Report |
No Report |
|
1 TRM = Transplantation Related Mortality 2 VOD = Veno-Occlusive Diseases of the Liver 3 GVHD = Graft versus Host Disease |
Hepatic Veno Occlusive Disease (hvod)
Advise patients of the risks associated with the use of busulfan as well as the plan for regular blood monitoring during therapy. Specifically inform patients of the following: The risk of veno-occlusive liver disease [see Warnings and Precautions (5.3) ].
5.3 Hepatic Veno Occlusive Disease (hvod)
Current literature suggests that high busulfan area under the plasma concentration verses time curve (AUC) values (greater than 1,500 μM•min) may be associated with an increased risk of developing HVOD. Patients who have received prior radiation therapy, greater than or equal to three cycles of chemotherapy, or a prior progenitor cell transplant may be at an increased risk of developing HVOD with the recommended busulfan dose and regimen. Based on clinical examination and laboratory findings, HVOD was diagnosed in 8% (5/61) of patients treated with busulfan in the setting of allogeneic transplantation, was fatal in 2/5 cases (40%), and yielded an overall mortality from HVOD in the entire study population of 2/61 (3%). Three of the five patients diagnosed with HVOD were retrospectively found to meet the Jones' criteria. The incidence of HVOD reported in the literature from the randomized, controlled trials was 7.7%-12% [see Clinical Studies (14) ]. Monitor serum transaminases, alkaline phosphatase, and bilirubin daily through BMT Day +28 to detect hepatotoxicity, which may herald the onset of HVOD.
7.1 Drugs That Decrease Busulfan Clearance
Itraconazole decreases busulfan clearance by up to 25%. Metronidazole decreases the clearance of busulfan to a greater extent than does itraconazole; metronidazole coadministration has been associated with increased busulfan toxicity. Fluconazole (200 mg) has been used with busulfan.
Decreased clearance of busulfan was observed with concomitant use with deferasirox. The mechanism of this interaction is not fully elucidated. Discontinue iron chelating agents well in advance of administration of busulfan to avoid increased exposure to busulfan.
Because busulfan is eliminated from the body via conjugation with glutathione, use of acetaminophen prior to (less than 72 hours) or concurrent with busulfan may result in reduced busulfan clearance based upon the known property of acetaminophen to decrease glutathione levels in the blood and tissues.
7.2 Drugs That Increase Busulfan Clearance
Phenytoin increases the clearance of busulfan by 15% or more, possibly due to the induction of glutathione-S-transferase. Since the pharmacokinetics of busulfan was studied in patients treated with phenytoin, the clearance of busulfan at the recommended dose may be lower and exposure (AUC) higher in patients not treated with phenytoin.
2.2 Preparation and Administration Precautions
Busulfan injection is incompatible with polycarbonate. Do not use any infusion components (syringes, filter needles, intravenous tubing, etc.) containing polycarbonate with busulfan injection.
Use an administration set with minimal residual hold-up volume (2mL - 5mL) for product administration.
Busulfan injection is a cytotoxic drug. Follow applicable special handling and disposal procedures. Skin reactions may occur with accidental exposure. Use gloves when preparing busulfan injection. If busulfan or diluted busulfan solution contacts the skin or mucosa, wash the skin or mucosa thoroughly with water.
Visually inspect parenteral drug products for particulate matter and discoloration prior to administration whenever the solution and container permit. Do not use if particulate matter is seen in the busulfan injection vial.
2.3 Preparation for Intravenous Administration
Busulfan injection must be diluted prior to intravenous infusion with either 0.9% Sodium Chloride Injection, USP (normal saline) or 5% Dextrose Injection, USP (D5W). The diluent quantity should be 10 times the volume of busulfan injection, so that the final concentration of busulfan is approximately 0.5 mg per mL. Calculation of the dose for a 70 kg patient would be performed as follows:
(70 kg patient) x (0.8 mg per kg) ÷ (6 mg per mL) =9.3 mL busulfan injection (56 mg total dose).
To prepare the final solution for infusion, add 9.3 mL of busulfan injection to 93 mL of diluent (normal saline or D5W) as calculated below:
(9.3 mL busulfan injection) x (10) =93 mL of either diluent plus the 9.3 mL of busulfan injection to yield a final concentration of busulfan of 0.54 mg per mL (9.3 mL x 6 mg per mL ÷ 102.3 mL =0.54 mg per mL).
All transfer procedures require strict adherence to aseptic techniques, preferably employing a vertical laminar flow safety hood while wearing gloves and protective clothing.
DO NOT put the busulfan injection into an intravenous bag or large-volume syringe that does not contain normal saline or D5W. Always add the busulfan injection to the diluent, not the diluent to the busulfan injection. Mix thoroughly by inverting several times.
Infusion pumps should be used to administer the diluted busulfan injection solution. Set the flow rate of the pump to deliver the entire prescribed busulfan injection dose over two hours. Prior to and following each infusion, flush the indwelling catheter line with approximately 5 mL of 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP. DO NOT infuse concomitantly with another intravenous solution of unknown compatibility. WARNING: RAPID INFUSION OF BUSULFAN INJECTION HAS NOT BEEN TESTED AND IS NOT RECOMMENDED.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Busulfan is a mutagen and a clastogen. In in vitro tests it caused mutations in Salmonella typhimurium Drosophila melanogaster. Chromosomal aberrations induced by busulfan have been reported in vivo (rats, mice, hamsters, and humans) and in vitro (rodent and human cells). The intravenous administration of busulfan (48 mg/kg given as biweekly doses of 12 mg/kg, or 30% of the total busulfan dose on a mg/m² basis) has been shown to increase the incidence of thymic and ovarian tumors in mice.
Busulfan depleted oocytes of female rats and induced sterility in male rats and hamsters. The solvent DMA may also impair fertility. A DMA daily dose of 0.45 g/kg/day given to rats for nine days (equivalent to 44% of the daily dose of DMA contained in the recommended dose of busulfan on a mg/m² basis) significantly decreased spermatogenesis in rats. A single subcutaneous dose of 2.2 g/kg (27% of the total DMA dose contained in busulfan on a mg/m² basis) four days after insemination terminated pregnancy in 100% of tested hamsters [see Use in Specific Populations (8.3) ].
Structured Label Content
Section 42229-5 (42229-5)
Additional Adverse Reactions by Body System
Hematologic : Prolonged prothrombin time
Gastrointestinal: Esophagitis, ileus, hematemesis, pancreatitis, rectal discomfort
Hepatic: Alkaline phosphatase increases, jaundice, hepatomegaly
Graft-versus-host disease: Graft-versus-host disease. There were 3 deaths (5%) attributed to GVHD.
Edema: Hypervolemia, or documented weight increase
Infection: Infection, pneumonia (fatal in one patient and life-threatening in 3% of patients)
Cardiovascular: Arrhythmia, atrial fibrillation, ventricular extrasystoles, third degree heart block, thrombosis (all episodes were associated with the central venous catheter), hypotension, flushing and hot flashes, cardiomegaly, ECG abnormality, left-sided heart failure, and pericardial effusion
Pulmonary : Hyperventilation, alveolar hemorrhage (fatal in 3%), pharyngitis, hiccup, asthma, atelectasis, pleural effusion, hypoxia, hemoptysis, sinusitis, and interstitial fibrosis (fatal in a single case)
Neurologic: Cerebral hemorrhage, coma, delirium, agitation, encephalopathy, confusion, hallucinations, lethargy, somnolence
Renal: BUN increased, dysuria, oliguria, hematuria, hemorrhagic cystitis
Skin: Alopecia, vesicular rash, maculopapular rash, vesiculo-bullous rash, exfoliative dermatitis, erythema nodosum, acne, skin discoloration
Metabolic: Hypophosphatemia, hyponatremia
Other Events: Injection site pain, myalgia, arthralgia, ear disorder
Seizures
Advise patients of the possibility of seizures and that they will be given medication to prevent them. Patients should be asked to report a history of seizure or head trauma [see Warnings and Precautions (5.2) ].
Lactation
Advise females to discontinue breastfeeding during treatment with busulfan injection [see Use in Specific Populations (8.2) ].
Packaging
Infertility
Advise females and males of reproductive potential that busulfan injection may cause temporary or permanent infertility [see Use in Specific Populations (8.3) ].
5.2 Seizures
Seizures have been reported in patients receiving high-dose oral busulfan at doses producing plasma drug levels similar to those achieved following the recommended dosage of busulfan. Despite prophylactic therapy with phenytoin, one seizure (1/42 patients) was reported during an autologous transplantation clinical trial of busulfan. This episode occurred during the cyclophosphamide portion of the conditioning regimen, 36 hours after the last busulfan dose. Initiate phenytoin therapy or any other alternative anti-convulsant prophylactic therapy (e.g., benzodiazepines, valproic acid or levetiracetam) prior to busulfan treatment [See Dosage and Administration (2.1) ]. Use caution when administering the recommended dose of busulfan to patients with a history of a seizure disorder or head trauma or who are receiving other potentially epileptogenic drugs.
10 Overdosage (10 OVERDOSAGE)
There is no known antidote to busulfan injection other than hematopoietic progenitor cell transplantation. In the absence of hematopoietic progenitor cell transplantation, the recommended dosage for busulfan would constitute an overdose of busulfan. The principal toxic effect is profound bone marrow hypoplasia/aplasia and pancytopenia, but the central nervous system, liver, lungs, and gastrointestinal tract may be affected. Monitor hematologic status closely and institute vigorous supportive measures as medically indicated. Survival after a single 140 mg dose of Myleran Tablets in an 18 kg, 4-year old child has been reported. Inadvertent administration of a greater than normal dose of oral busulfan (2.1 mg per kg; total dose of 23.3 mg per kg) occurred in a 2-year old child prior to a scheduled bone marrow transplant without sequelae. An acute dose of 2.4 g was fatal in a 10-year old boy. There is one report that busulfan is dialyzable, thus dialysis should be considered in the case of overdose.
15 References (15 REFERENCES)
1. OSHA Hazardous Drugs. OSHA. [Accessed on June 18, 2014 from http://www.osha.gov/SLTC/hazardousdrugs/index.html
11 Description (11 DESCRIPTION)
Busulfan is a bifunctional alkylating agent known chemically as 1,4-butanediol, dimethanesulfonate. Busulfan Injection is intended for intravenous administration. It is supplied as a clear, colorless, sterile, solution in 10 mL single-dose vials. Each vial of busulfan injection contains 60 mg (6 mg/mL) of busulfan, the active ingredient, a white crystalline powder with a molecular formula of CH3SO2O(CH2)4OSO2CH3 and a molecular weight of 246 g/mole. Busulfan has the following chemical structure:
Busulfan is dissolved in N,N-dimethylacetamide (DMA), 3.3 mL and Polyethylene Glycol 400, NF 6.7 mL. The solubility of busulfan in water is 0.1 g per L and the pH of busulfan injection diluted to approximately 0.5 mg per mL busulfan in 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP as recommended for infusion reflects the pH of the diluent used and ranges from 3.4 to 3.9.
Myelosuppression
Advise patients of the possibility of developing low blood cell counts and the need for hematopoietic progenitor cell infusion. Instruct patients to immediately report to their healthcare provider if fever develops [see Warnings and Precautions (5.1) ].
16.1 How Supplied
Busulfan injection is packaged as a sterile solution in 10 mL single-dose clear glass vials each containing 60 mg of busulfan at a concentration of 6 mg per mL for intravenous use, NDC 72485-210-01.
Busulfan injection is distributed as a unit carton of eight vials NDC 72485-210-08
8.4 Pediatric Use
The effectiveness of busulfan in the treatment of CML has not been specifically studied in pediatric patients. An open-label, uncontrolled study evaluated the pharmacokinetics of busulfan in 24 pediatric patients receiving busulfan as part of a conditioning regimen administered prior to hematopoietic progenitor cell transplantation for a variety of malignant hematologic (N=15) or non-malignant diseases (N=9). Patients ranged in age from 5 months to 16 years (median 3 years). Busulfan dosing was targeted to achieve an area under the plasma concentration curve (AUC) of 900-1350 μM•min with an initial dose of 0.8 mg per kg or 1.0 mg per kg (based on Actual Body Weight (ABW)) if the patient was greater than 4 or less than or equal to 4 years, respectively. The dose was adjusted based on plasma concentration after completion of dose 1.
Patients received busulfan doses every six hours as a two-hour infusion over four days for a total of 16 doses, followed by cyclophosphamide 50 mg per kg once daily for four days. After one rest day, hematopoietic progenitor cells were infused. All patients received phenytoin as seizure prophylaxis. The target AUC (900-1350±5% μM•min) for busulfan was achieved at dose 1 in 71% (17/24) of patients. Steady state pharmacokinetic testing was performed at dose 9 and 13. Busulfan levels were within the target range for 21 of 23 evaluable patients.
All 24 patients experienced neutropenia (absolute neutrophil count (ANC) less than 0.5×109/L) and thrombocytopenia (platelet transfusions or platelet count less than 20,000/mm3). Seventy-nine percent (19/24) of patients experienced lymphopenia (absolute lymphocyte count less than 0.1×109). In 23 patients, the ANC recovered to greater than 0.5×109/L (median time to recovery = BMT day +13; range = BMT day +9 to +22). One patient who died on day +20 had not recovered to an ANC >0.5×109/L.
Four (17%) patients died during the study. Two patients died within 28 days of transplant; one with pneumonia and capillary leak syndrome, and the other with pneumonia and veno-occlusive disease. Two patients died prior to day 100; one due to progressive disease and one due to multiorgan failure.
Adverse reactions were reported in all 24 patients during the study period (BMT day -10 through BMT day +28) or post-study surveillance period (day +29 through +100). These included vomiting (100%), nausea (83%), stomatitis (79%), HVOD (21%), graft-versus host disease (GVHD) (25%), and pneumonia (21%).
Based on the results of this 24-patient clinical trial, a suggested dosing regimen of busulfan in pediatric patients is shown in the following dosing nomogram:
| Patient's Actual Body Weight (ABW) | Busulfan Dosage |
|---|---|
| less than or equal to12 kg | 1.1 (mg per kg) |
| greater than 12 kg | 0.8 (mg per kg) |
Simulations based on a pediatric population pharmacokinetic model indicate that approximately 60% of pediatric patients will achieve a target busulfan exposure (AUC) between 900 to 1350 μM•min with the first dose of busulfan using this dosing nomogram. Therapeutic drug monitoring and dose adjustment following the first dose of busulfan is recommended.
8.5 Geriatric Use
Clinical studies of busulfan did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
Cardiac Tamponade
Advise patients of the risk of cardiac tamponade. Instruct patients to report to their healthcare provider symptoms of abdominal pain and vomiting [see Warnings and Precautions (5.5) ].
14 Clinical Studies (14 CLINICAL STUDIES)
Documentation of the safety and efficacy of busulfan as a component of a conditioning regimen prior to allogeneic hematopoietic progenitor cell reconstitution is derived from two sources:
i) analysis of a prospective clinical trial of busulfan that involved 61 patients diagnosed with various hematologic malignancies, and
ii) the published reports of randomized, controlled trials that employed high-dose oral busulfan as a component of a conditioning regimen for transplantation, which were identified in a literature review of five established commercial databases.
4 Contraindications (4 CONTRAINDICATIONS)
Busulfan is contraindicated in patients with a history of hypersensitivity to any of its components.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Myelosuppression [see Warnings and Precautions (5.1) ]
- Seizures [see Warnings and Precautions (5.2) ]
- Hepatic Veno-Occlusive Disease (HVOD) [see Warnings and Precautions (5.3) ]
- Embryo-fetal Toxicity [see Warnings and Precautions (5.4) ]
- Cardiac Tamponade [see Warnings and Precautions (5.5) ]
- Bronchopulmonary Dysplasia [see Warnings and Precautions (5.6) ]
- Cellular Dysplasia [see Warnings and Precautions (5.7) ]
7 Drug Interactions (7 DRUG INTERACTIONS)
5.1 Myelosuppression
The most frequent serious consequence of treatment with busulfan at the recommended dose and schedule is prolonged myelosuppression, occurring in all patients (100%). Severe granulocytopenia, thrombocytopenia, anemia, or any combination thereof may develop. Hematopoietic progenitor cell transplantation is required to prevent potentially fatal complications of the prolonged myelosuppression. Monitor complete blood counts, including white blood cell differentials, and quantitative platelet counts daily during treatment and until engraftment is demonstrated. Absolute neutrophil counts dropped below 0.5x109/L at a median of 4 days post-transplant in 100% of patients treated in the busulfan clinical trial. The absolute neutrophil count recovered at a median of 13 days following allogeneic transplantation when prophylactic filgrastim was used in the majority of patients. Thrombocytopenia (less than 25,000/mm3 or requiring platelet transfusion) occurred at a median of 5-6 days in 98% of patients. Anemia (hemoglobin less than 8.0 g/dL) occurred in 69% of patients. Use antibiotic therapy and platelet and red blood cell support when medically indicated.
12.3 Pharmacokinetics
The pharmacokinetics of busulfan was studied in 59 patients participating in a prospective trial of a busulfan cyclophosphamide preparatory regimen prior to allogeneic hematopoietic progenitor stem cell transplantation. Patients received 0.8 mg/kg busulfan every six hours, for a total of 16 doses over four days. Fifty-five of fifty-nine patients (93%) administered busulfan maintained AUC values below the target value (less than1500 μM•min).
| Mean | CV (%) | Range | |
| Cmax (ng per mL) | 1222 | 18 | 496 - 1684 |
| AUC (μM∙min) | 1167 | 20 | 556 - 1673 |
| CL (mL per min per kg)1 | 2.52 | 25 | 1.49 - 4.31 |
| 1.Clearance normalized to actual body weight for all patients. |
Busulfan pharmacokinetics showed consistency between dose 9 and dose 13 as demonstrated by reproducibility of steady state Cmax and a low coefficient of variation for this parameter.
5.5 Cardiac Tamponade
Cardiac tamponade has been reported in pediatric patients with thalassemia (8/400 or 2% in one series) who received high doses of oral busulfan and cyclophosphamide as the preparatory regimen for hematopoietic progenitor cell transplantation. Six of the eight children died and two were saved by rapid pericardiocentesis. Abdominal pain and vomiting preceded the tamponade in most patients. Monitor for signs and symptoms, promptly evaluate and treat if cardiac tamponade is suspected.
Embryo Fetal Toxicity (Embryo-fetal Toxicity)
Advise females of reproductive potential of the potential risk to a fetus and to inform their healthcare provider with a known or suspected pregnancy [see Warnings and Precautions (5.4) and Use in Specific Populations (8.1) ].
5.7 Cellular Dysplasia
Busulfan may cause cellular dysplasia in many organs. Cytologic abnormalities characterized by giant, hyperchromatic nuclei have been reported in lymph nodes, pancreas, thyroid, adrenal glands, liver, lungs and bone marrow. This cytologic dysplasia may be severe enough to cause difficulty in the interpretation of exfoliative cytologic examinations of the lungs, bladder, breast and the uterine cervix.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Busulfan injection is indicated for use in combination with cyclophosphamide as a conditioning regimen prior to allogeneic hematopoietic progenitor cell transplantation for chronic myelogenous leukemia.
12.1 Mechanism of Action
Busulfan is a bifunctional alkylating agent in which two labile methanesulfonate groups are attached to opposite ends of a four-carbon alkyl chain. In aqueous media, busulfan hydrolyzes to release the methanesulfonate groups. This produces reactive carbonium ions that can alkylate DNA. DNA damage is thought to be responsible for much of the cytotoxicity of busulfan
16.2 Storage and Handling
Unopened vials of busulfan injection must be stored under refrigerated conditions between 2°C to 8°C (36°F to 46°F).
Busulfan injection diluted in 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP is stable at room temperature (25°C) for up to 8 hours but the infusion must be completed within that time.
Busulfan injection diluted in 0.9% Sodium Chloride Injection, USP is stable at refrigerated conditions (2°C to 8°C) for up to 12 hours but the infusion must be completed within that time.
Busulfan injection is a cytotoxic drug. Follow applicable special handling and disposal procedures 1 .
5.4 Embryo Fetal Toxicity (5.4 Embryo-fetal Toxicity)
Busulfan can cause fetal harm when administered to a pregnant woman based on animal data. Busulfan was teratogenic in mice, rats, and rabbits. The solvent, DMA, may also cause fetal harm when administered to a pregnant woman based on findings in animals. Advise pregnant women of the potential risk to a fetus. Advise females and males of reproductive potential to use effective contraception during and after treatment with busulfan [see Use in Specific Populations (8.1) and (8.3) ].
Warning: Myelosuppression (WARNING: MYELOSUPPRESSION)
Busulfan Injection causes severe and prolonged myelosuppression at the recommended dosage. Hematopoietic progenitor cell transplantation is required to prevent potentially fatal complications of the prolonged myelosuppression [see Warnings and Precautions (5.1) ].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Seizures: Initiate anticonvulsant prophylactic therapy prior to treatment with busulfan injection. Monitor patients with history of seizure disorder, head trauma or receiving epileptogenic drugs (5.2)
- Hepatic Veno-Occlusive Disease (HVOD): Increased risk of developing HVOD at AUC greater than 1,500 μMmin. Monitor serum transaminases, alkaline phosphatase and bilirubin daily (5.3)
- Embryo-fetal Toxicity: Can cause fetal harm. Advise of potential risk to a fetus and use of effective contraception (5.4, 8.1, 8.3)
- Cardiac tamponade has been reported in pediatric patients with thalassemia who received high doses of oral busulfan and cyclophosphamide. Abdominal pain and vomiting precede the tamponade in most patients (5.5)
Bronchopulmonary Dysplasia
Advise patients of the possibility of bronchopulmonary dysplasia with pulmonary fibrosis with chronic busulfan therapy. Instruct patients to report symptoms of shortness of breath and cough to their healthcare provider. These symptoms could occur several months or years after therapy with busulfan injection [see Warnings and Precautions (5.6) ].
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Pre-medicate with anticonvulsants (e.g. benzodiazepines, phenytoin, valproic acid or levetiracetam) and antiemetic. ( 2.1, 5.2 )
- Dilute and administer as intravenous infusion. Do not administer as intravenous push or bolus. (2.1, 2.3)
- Recommended adult dose: 0.8 mg per kg of ideal body weight or actual body weight, whichever is lower, administered intravenously via a central venous catheter as a two-hour infusion every six hours for four consecutive days for a total of 16 doses. (2.1)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Busulfan injection is supplied as a clear, colorless, sterile, solution in 10 mL single-dose vial containing 60 mg of busulfan at a concentration of 6 mg per mL for intravenous use only.
6.2 Postmarketing Experience
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The following adverse reactions have been identified during post-approval use of busulfan Injection:
Blood and Lymphatic System Disorders: febrile neutropenia
Gastrointestinal Disorders: tooth hypoplasia
Metabolism and Nutrition Disorders: tumor lysis syndrome
Vascular Disorders: thrombotic microangiopathy (TMA)
Infections and Infestations: severe bacterial, viral (e.g., cytomegalovirus viremia) and fungal infections; and sepsis.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse reaction information is primarily derived from the clinical study (N=61) of busulfan and the data obtained for high-dose oral busulfan conditioning in the setting of randomized, controlled trials identified through a literature review.
In the busulfan Injection allogeneic stem cell transplantation clinical trial, all patients were treated with busulfan 0.8 mg per kg as a two-hour infusion every six hours for 16 doses over four days, combined with cyclophosphamide 60 mg per kg x2 days. Ninety-three percent (93%) of evaluable patients receiving this dose of busulfan maintained an AUC less than 1,500 μM•min for dose 9, which has generally been considered the level that minimizes the risk of HVOD.
Table 1 lists the non-hematologic adverse reactions events through Bone Marrow Transplantation (BMT) Day +28 at a rate greater than or equal to 20% in patients treated with busulfan prior to allogeneic hematopoietic cell transplantation.
| Non-Hematological Adverse Reactions 1 | Percent Incidence |
| BODY AS A WHOLE | |
| Fever | 80 |
| Headache | 69 |
| Asthenia | 51 |
| Chills | 46 |
| Pain | 44 |
| Edema General | 28 |
| Allergic Reaction | 26 |
| Chest Pain | 26 |
| Inflammation at Injection Site | 25 |
| Back Pain | 23 |
| CARDIOVASCULAR SYSTEM | |
| Tachycardia | 44 |
| Hypertension | 36 |
| Thrombosis | 33 |
| Vasodilation | 25 |
| DIGESTIVE SYSTEM | |
| Nausea | 98 |
| Stomatitis (Mucositis) | 97 |
| Vomiting | 95 |
| Anorexia | 85 |
| Diarrhea | 84 |
| Abdominal Pain | 72 |
| Dyspepsia | 44 |
| Constipation | 38 |
| Dry Mouth | 26 |
| Rectal Disorder | 25 |
| Abdominal Enlargement | 23 |
| METABOLIC AND NUTRITIONAL SYSTEM | |
| Hypomagnesemia | 77 |
| Hyperglycemia | 66 |
| Hypokalemia | 64 |
| Hypocalcemia | 49 |
| Hyperbilirubinemia | 49 |
| Edema | 36 |
| SGPT Elevation | 31 |
| Creatinine Increased | 21 |
| NERVOUS SYSTEM | |
| Insomnia | 84 |
| Anxiety | 72 |
| Dizziness | 30 |
| Depression | 23 |
| RESPIRATORY SYSTEM | |
| Rhinitis | 44 |
| Lung Disorder | 34 |
| Cough | 28 |
| Epistaxis | 25 |
| Dyspnea | 25 |
| SKIN AND APPENDAGES | |
| Rash | 57 |
| Pruritus | 28 |
| 1. Includes all reported adverse reactions regardless of severity (toxicity grades 1-4) |
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Lactation: Discontinue breastfeeding (8.2)
2.1 Initial Dosing Information
- Administer busulfan injection in combination with cyclophosphamide as a conditioning regimen prior to bone marrow or peripheral blood progenitor cell replacement. For patients weighing more than 12 kg, the recommended doses are:
- Busulfan Injection 0.8 mg per kg (ideal body weight or actual body weight, whichever is lower) intravenously via a central venous catheter as a two-hour infusion every six hours for four consecutive days for a total of 16 doses (Days -7, -6, -5 and -4).
- Cyclophosphamide 60 mg per kg intravenously as a one-hour infusion on each of two days beginning no sooner than six hours following the 16th dose of busulfan injection (Days -3 and -2).
- Administer hematopoietic progenitor cells on Day 0.
- Premedicate patients with anticonvulsants (e.g., benzodiazepines, phenytoin, valproic acid or levetiracetam) to prevent seizures reported with the use of high dose busulfan injection. Administer anticonvulsants 12 hours prior to busulfan injection to 24 hours after the last dose of busulfan injection [see Warnings and Precautions (5.2) ].
- Administer antiemetics prior to the first dose of busulfan injection and continue on a fixed schedule through busulfan injection administration.
- Busulfan injection clearance is best predicted when the busulfan injection dose is administered based on adjusted ideal body weight. Dosing busulfan injection based on actual body weight, ideal body weight or other factors can produce significant differences in busulfan injection clearance among lean, normal and obese patients.
- Calculate ideal body weight (IBW) as follows (height in cm, and weight in kg):
Women: IBW (kg)=45+0.91x (height in cm -152)
- For obese or severely obese patients, base busulfan injection dosing on adjusted ideal body weight (AIBW):
5.6 Bronchopulmonary Dysplasia
Bronchopulmonary dysplasia with pulmonary fibrosis is a rare but serious complication following chronic busulfan therapy. The average onset of symptoms is 4 years after therapy (range 4 months to 10 years).
Males of Reproductive Potential
Advise males with female sexual partners of reproductive potential to use effective contraception during treatment with busulfan injection and for 3 months following cessation of therapy [see Use in Specific Populations (8.3) ].
Females of Reproductive Potential
Advise females of reproductive potential to use effective contraception during treatment with busulfan and for 6 months following cessation of therapy [see Use in Specific Populations (8.3) ].
6.3 Oral Busulfan Literature Review
A literature review identified four randomized, controlled trials that evaluated a high-dose oral busulfan-containing conditioning regimen for allogeneic bone marrow transplantation in the setting of CML [see Clinical Studies (14) ]. The safety outcomes reported in those trials are summarized in Table 2 below for a mixed population of hematological malignancies (AML, CML, and ALL).
|
Clift CML Chronic Phase |
|||||
| TRM1 | VOD2 | GVHD3 | Pulmonary | Hemorrhagic Cystitis | Seizure |
| Death ≤100d=4.1% (3/73) | No Report | Acute≥Grade 2=35% Chronic=41% (30/73) |
1 death from Idiopathic Interstitial Pneumonitis And 1 death from Pulmonary Fibrosis | No Report | No Report |
|
Devergie CML Chronic Phase |
|||||
| TRM | VOD | GVHD | Pulmonary | Hemorrhagic Cystitis | Seizure |
| 38% | 7.7% (5/65) Deaths=4.6% (3/65) |
Acute≥Grade 2=41% (24/59 at risk) | Interstitial Pneumonitis=16.9% (11/65) | 10.8% (7/65) | No report |
|
Ringden CML, AML, ALL |
|||||
| TRM | VOD | GVHD | Pulmonary | Hemorrhagic Cystitis | Seizure |
| 28% | 12% | Acute≥Grade 2 GVHD=26% Chronic GVHD=45% |
Interstitial Pneumonitis=14% | 24% | 6% |
|
Blume CML, AML, ALL |
|||||
| TRM | VOD | GVHD | Pulmonary | Hemorrhagic Cystitis | Seizure |
| No Report |
Deaths=4.9% |
Acute≥Grade 2 GVHD=22% (13/58 at risk) Chronic GVHD=31% (14/45 at risk) |
No Report |
No Report |
No Report |
|
1 TRM = Transplantation Related Mortality 2 VOD = Veno-Occlusive Diseases of the Liver 3 GVHD = Graft versus Host Disease |
Hepatic Veno Occlusive Disease (hvod) (Hepatic Veno-Occlusive Disease (HVOD))
Advise patients of the risks associated with the use of busulfan as well as the plan for regular blood monitoring during therapy. Specifically inform patients of the following: The risk of veno-occlusive liver disease [see Warnings and Precautions (5.3) ].
5.3 Hepatic Veno Occlusive Disease (hvod) (5.3 Hepatic Veno-Occlusive Disease (HVOD))
Current literature suggests that high busulfan area under the plasma concentration verses time curve (AUC) values (greater than 1,500 μM•min) may be associated with an increased risk of developing HVOD. Patients who have received prior radiation therapy, greater than or equal to three cycles of chemotherapy, or a prior progenitor cell transplant may be at an increased risk of developing HVOD with the recommended busulfan dose and regimen. Based on clinical examination and laboratory findings, HVOD was diagnosed in 8% (5/61) of patients treated with busulfan in the setting of allogeneic transplantation, was fatal in 2/5 cases (40%), and yielded an overall mortality from HVOD in the entire study population of 2/61 (3%). Three of the five patients diagnosed with HVOD were retrospectively found to meet the Jones' criteria. The incidence of HVOD reported in the literature from the randomized, controlled trials was 7.7%-12% [see Clinical Studies (14) ]. Monitor serum transaminases, alkaline phosphatase, and bilirubin daily through BMT Day +28 to detect hepatotoxicity, which may herald the onset of HVOD.
7.1 Drugs That Decrease Busulfan Clearance (7.1 Drugs that Decrease Busulfan Clearance)
Itraconazole decreases busulfan clearance by up to 25%. Metronidazole decreases the clearance of busulfan to a greater extent than does itraconazole; metronidazole coadministration has been associated with increased busulfan toxicity. Fluconazole (200 mg) has been used with busulfan.
Decreased clearance of busulfan was observed with concomitant use with deferasirox. The mechanism of this interaction is not fully elucidated. Discontinue iron chelating agents well in advance of administration of busulfan to avoid increased exposure to busulfan.
Because busulfan is eliminated from the body via conjugation with glutathione, use of acetaminophen prior to (less than 72 hours) or concurrent with busulfan may result in reduced busulfan clearance based upon the known property of acetaminophen to decrease glutathione levels in the blood and tissues.
7.2 Drugs That Increase Busulfan Clearance (7.2 Drugs that Increase Busulfan Clearance)
Phenytoin increases the clearance of busulfan by 15% or more, possibly due to the induction of glutathione-S-transferase. Since the pharmacokinetics of busulfan was studied in patients treated with phenytoin, the clearance of busulfan at the recommended dose may be lower and exposure (AUC) higher in patients not treated with phenytoin.
2.2 Preparation and Administration Precautions
Busulfan injection is incompatible with polycarbonate. Do not use any infusion components (syringes, filter needles, intravenous tubing, etc.) containing polycarbonate with busulfan injection.
Use an administration set with minimal residual hold-up volume (2mL - 5mL) for product administration.
Busulfan injection is a cytotoxic drug. Follow applicable special handling and disposal procedures. Skin reactions may occur with accidental exposure. Use gloves when preparing busulfan injection. If busulfan or diluted busulfan solution contacts the skin or mucosa, wash the skin or mucosa thoroughly with water.
Visually inspect parenteral drug products for particulate matter and discoloration prior to administration whenever the solution and container permit. Do not use if particulate matter is seen in the busulfan injection vial.
2.3 Preparation for Intravenous Administration
Busulfan injection must be diluted prior to intravenous infusion with either 0.9% Sodium Chloride Injection, USP (normal saline) or 5% Dextrose Injection, USP (D5W). The diluent quantity should be 10 times the volume of busulfan injection, so that the final concentration of busulfan is approximately 0.5 mg per mL. Calculation of the dose for a 70 kg patient would be performed as follows:
(70 kg patient) x (0.8 mg per kg) ÷ (6 mg per mL) =9.3 mL busulfan injection (56 mg total dose).
To prepare the final solution for infusion, add 9.3 mL of busulfan injection to 93 mL of diluent (normal saline or D5W) as calculated below:
(9.3 mL busulfan injection) x (10) =93 mL of either diluent plus the 9.3 mL of busulfan injection to yield a final concentration of busulfan of 0.54 mg per mL (9.3 mL x 6 mg per mL ÷ 102.3 mL =0.54 mg per mL).
All transfer procedures require strict adherence to aseptic techniques, preferably employing a vertical laminar flow safety hood while wearing gloves and protective clothing.
DO NOT put the busulfan injection into an intravenous bag or large-volume syringe that does not contain normal saline or D5W. Always add the busulfan injection to the diluent, not the diluent to the busulfan injection. Mix thoroughly by inverting several times.
Infusion pumps should be used to administer the diluted busulfan injection solution. Set the flow rate of the pump to deliver the entire prescribed busulfan injection dose over two hours. Prior to and following each infusion, flush the indwelling catheter line with approximately 5 mL of 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP. DO NOT infuse concomitantly with another intravenous solution of unknown compatibility. WARNING: RAPID INFUSION OF BUSULFAN INJECTION HAS NOT BEEN TESTED AND IS NOT RECOMMENDED.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Busulfan is a mutagen and a clastogen. In in vitro tests it caused mutations in Salmonella typhimurium Drosophila melanogaster. Chromosomal aberrations induced by busulfan have been reported in vivo (rats, mice, hamsters, and humans) and in vitro (rodent and human cells). The intravenous administration of busulfan (48 mg/kg given as biweekly doses of 12 mg/kg, or 30% of the total busulfan dose on a mg/m² basis) has been shown to increase the incidence of thymic and ovarian tumors in mice.
Busulfan depleted oocytes of female rats and induced sterility in male rats and hamsters. The solvent DMA may also impair fertility. A DMA daily dose of 0.45 g/kg/day given to rats for nine days (equivalent to 44% of the daily dose of DMA contained in the recommended dose of busulfan on a mg/m² basis) significantly decreased spermatogenesis in rats. A single subcutaneous dose of 2.2 g/kg (27% of the total DMA dose contained in busulfan on a mg/m² basis) four days after insemination terminated pregnancy in 100% of tested hamsters [see Use in Specific Populations (8.3) ].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:34:43.996337 · Updated: 2026-03-14T21:41:24.246163