These Highlights Do Not Include All The Information Needed To Use Jynarque Safely And Effectively. See Full Prescribing Information For Jynarque.
3febc0a1-9e5a-4ce0-843d-210f21d862c4
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
JYNARQUE (tolvaptan) can cause serious and potentially fatal liver injury. Acute liver failure requiring liver transplantation has been reported [see Warnings and Precautions (5.1) ] . Measure ALT, AST and bilirubin before initiating treatment, at 2 weeks and 4 weeks after initiation, then monthly for the first 18 months and every 3 months thereafter [see Warnings and Precautions (5.1) ] . Prompt action in response to laboratory abnormalities, signs, or symptoms indicative of hepatic injury can mitigate, but not eliminate, the risk of serious hepatotoxicity. Because of the risks of serious liver injury, JYNARQUE is available only through a restricted distribution program under a Risk Evaluation and Mitigation Strategy (REMS) called the Tolvaptan for ADPKD Shared System REMS [see Warnings and Precautions (5.2) ] .
Indications and Usage
JYNARQUE is indicated to slow kidney function decline in adults at risk of rapidly progressing autosomal dominant polycystic kidney disease (ADPKD).
Dosage and Administration
Recommended dosage ( 2.1 ) Initial Dosage Titration Step Target Dosage 1st Dose 45 mg 1st Dose 60 mg 1st Dose 90 mg 2nd Dose (8 hours later) 15 mg 2nd Dose (8 hours later) 30 mg 2nd Dose (8 hours later) 30 mg Total Daily Dose 60 mg Total Daily Dose 90 mg Total Daily Dose 120 mg Dose adjustment is recommended for patients taking moderate CYP3A inhibitors ( 2.4 , 5.4 , 7.1 )
Warnings and Precautions
Hypernatremia, dehydration and hypovolemia: May require intervention ( 5.3 )
Contraindications
JYNARQUE is contraindicated in patients: With a history, signs or symptoms of significant liver impairment or injury. This contraindication does not apply to uncomplicated polycystic liver disease [see Warnings and Precautions (5.1) ] Taking strong CYP3A inhibitors With uncorrected abnormal blood sodium concentrations [see Warnings and Precautions (5.3) ] Unable to sense or respond to thirst [see Warnings and Precautions (5.3) ] Hypovolemia [see Warnings and Precautions (5.3) ] Hypersensitivity (e.g., anaphylaxis, rash) to tolvaptan or any component of the product [see Adverse Reactions (6) ] Uncorrected urinary outflow obstruction Anuria
Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling: Serious Liver Injury [see Boxed Warning and Warnings and Precautions (5.1) ] Hypernatremia, Dehydration and Hypovolemia [see Warnings and Precautions (5.3) ] Drug Interactions with Inhibitors of CYP3A [see Warnings and Precautions (5.4) ]
Drug Interactions
Avoid concomitant use with: Strong CYP3A Inducers ( 7.1 ) V 2 -Receptor Agonists ( 7.2 )
Medication Information
Warnings and Precautions
Hypernatremia, dehydration and hypovolemia: May require intervention ( 5.3 )
Indications and Usage
JYNARQUE is indicated to slow kidney function decline in adults at risk of rapidly progressing autosomal dominant polycystic kidney disease (ADPKD).
Dosage and Administration
Recommended dosage ( 2.1 ) Initial Dosage Titration Step Target Dosage 1st Dose 45 mg 1st Dose 60 mg 1st Dose 90 mg 2nd Dose (8 hours later) 15 mg 2nd Dose (8 hours later) 30 mg 2nd Dose (8 hours later) 30 mg Total Daily Dose 60 mg Total Daily Dose 90 mg Total Daily Dose 120 mg Dose adjustment is recommended for patients taking moderate CYP3A inhibitors ( 2.4 , 5.4 , 7.1 )
Contraindications
JYNARQUE is contraindicated in patients: With a history, signs or symptoms of significant liver impairment or injury. This contraindication does not apply to uncomplicated polycystic liver disease [see Warnings and Precautions (5.1) ] Taking strong CYP3A inhibitors With uncorrected abnormal blood sodium concentrations [see Warnings and Precautions (5.3) ] Unable to sense or respond to thirst [see Warnings and Precautions (5.3) ] Hypovolemia [see Warnings and Precautions (5.3) ] Hypersensitivity (e.g., anaphylaxis, rash) to tolvaptan or any component of the product [see Adverse Reactions (6) ] Uncorrected urinary outflow obstruction Anuria
Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling: Serious Liver Injury [see Boxed Warning and Warnings and Precautions (5.1) ] Hypernatremia, Dehydration and Hypovolemia [see Warnings and Precautions (5.3) ] Drug Interactions with Inhibitors of CYP3A [see Warnings and Precautions (5.4) ]
Drug Interactions
Avoid concomitant use with: Strong CYP3A Inducers ( 7.1 ) V 2 -Receptor Agonists ( 7.2 )
Description
JYNARQUE (tolvaptan) can cause serious and potentially fatal liver injury. Acute liver failure requiring liver transplantation has been reported [see Warnings and Precautions (5.1) ] . Measure ALT, AST and bilirubin before initiating treatment, at 2 weeks and 4 weeks after initiation, then monthly for the first 18 months and every 3 months thereafter [see Warnings and Precautions (5.1) ] . Prompt action in response to laboratory abnormalities, signs, or symptoms indicative of hepatic injury can mitigate, but not eliminate, the risk of serious hepatotoxicity. Because of the risks of serious liver injury, JYNARQUE is available only through a restricted distribution program under a Risk Evaluation and Mitigation Strategy (REMS) called the Tolvaptan for ADPKD Shared System REMS [see Warnings and Precautions (5.2) ] .
7.2 V2
As a V2-receptor antagonist, tolvaptan will interfere with the V2-agonist activity of desmopressin (dDAVP). Avoid concomitant use of JYNARQUE with a V2-agonist.
Section 42229-5
CYP3A Inhibitors
Concomitant use of strong CYP3A inhibitors is contraindicated [see Contraindications (4) and Warnings and Precautions (5.4)].
In patients taking concomitant moderate CYP3A inhibitors, reduce the dose of JYNARQUE per Table 1. Consider further reductions if patients cannot tolerate the reduced dose [see Warnings and Precautions (5.4) and Drug Interactions (7.1)]. Interrupt JYNARQUE temporarily for short term therapy with moderate CYP3A inhibitors if the recommended reduced doses are not available.
| Standard Morning and Afternoon Dose (mg) | Dose (mg) with Moderate CYP3A Inhibitors |
|---|---|
| 90 mg and 30 mg | 45 mg and 15 mg |
| 60 mg and 30 mg | 30 mg and 15 mg |
| 45 mg and 15 mg | 15 mg and 15 mg |
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 3/2025 | ||
|
MEDICATION GUIDE JYNARQUE® (jin-AR-kew) (tolvaptan) Tablets |
|||
|
What is the most important information I should know about JYNARQUE? JYNARQUE can cause serious side effects, including:
|
|||
|
|
||
| To help reduce your risk of liver problems, your healthcare provider will do a blood test to check your liver: | |||
|
|||
| It is important to stay under the care of your healthcare provider during treatment with JYNARQUE. Because of the risk of serious liver problems JYNARQUE is only available through a restricted distribution program called the Tolvaptan for ADPKD Shared System REMS.
|
|||
|
What is JYNARQUE?
JYNARQUE is a prescription medicine used to slow kidney function decline in adults who are at risk of rapidly progressing autosomal dominant polycystic kidney disease (ADPKD). It is not known if JYNARQUE is safe and effective in children. |
|||
Do not take JYNARQUE if you:
|
|||
Before taking JYNARQUE, tell your healthcare provider about all your medical conditions, including if you:
|
|||
How should I take JYNARQUE?
|
|||
|
What are the possible side effects of JYNARQUE? JYNARQUE may cause serious side effects, including: See "What is the most important information I should know about JYNARQUE?"
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
How should I store JYNARQUE?
JYNARQUE comes in a child-resistant package. Store JYNARQUE between 68°F to 77°F (20°C to 25°C). Keep JYNARQUE and all medicines out of the reach of children. |
|||
|
General information about the safe and effective use of JYNARQUE.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use JYNARQUE for a condition for which it was not prescribed. Do not give JYNARQUE to other people, even if they have the same symptoms you have. It may harm them. |
|||
|
What are the ingredients in JYNARQUE? Active ingredient: tolvaptan Inactive ingredients: corn starch, hydroxypropyl cellulose, lactose monohydrate, low-substituted hydroxypropyl cellulose, magnesium stearate and microcrystalline cellulose, and FD&C Blue no. 2 Aluminum Lake as colorant. |
|||
| Manufactured by Otsuka Pharmaceutical Co., Ltd., Tokyo, 101-8535 Japan Distributed and marketed by Otsuka America Pharmaceutical, Inc., Rockville, MD 20850 USA JYNARQUE is a registered trademark of Otsuka Pharmaceutical Co., Ltd., Tokyo, 101-8535 Japan ©2025, Otsuka Pharmaceutical Co., Ltd., Tokyo, 101-8535 Japan For more information about JYNARQUE, go to www.TolvaptanADPKDSharedREMS.com or call 1-866-244-9446. |
Section 43683-2
|
Warnings and Precautions
Tolvaptan for ADPKD Shared System REMS (5.2) |
3/2025 |
Section 44425-7
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (59°F to 86°F) [see USP controlled Room Temperature].
10 Overdosage
Single oral doses up to 480 mg (4 times the maximum recommended daily dose) and multiple doses up to 300 mg once daily for 5 days have been well tolerated in trials in healthy subjects. There is no specific antidote for tolvaptan intoxication. The signs and symptoms of an acute overdose can be anticipated to be those of excessive pharmacologic effect: a rise in serum sodium concentration, polyuria, thirst, and dehydration/hypovolemia.
No mortality was observed in rats or dogs following single oral doses of 2000 mg/kg (maximum feasible dose). A single oral dose of 2000 mg/kg was lethal in mice, and symptoms of toxicity in affected mice included decreased locomotor activity, staggering gait, tremor and hypothermia.
In patients with suspected JYNARQUE overdosage, assessment of vital signs, electrolyte concentrations, ECG and fluid status is recommended. Continue replacement of water and electrolytes until aquaresis abates. Dialysis may not be effective in removing JYNARQUE because of its high binding affinity for human plasma protein (>98%).
11 Description
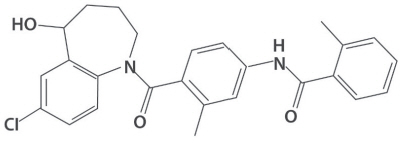
JYNARQUE contains tolvaptan, a selective vasopressin V2-receptor antagonist in immediate release tablets for oral administration available in 15 mg, 30 mg, 45 mg, 60 mg and 90 mg strengths. Tolvaptan is (±)-4'-[(7-chloro-2,3,4,5-tetrahydro-5-hydroxy-1H-1-benzazepin-1-yl) carbonyl]-o-tolu-m-toluidide. The empirical formula is C26H25ClN2O3. Molecular weight is 448.94. The chemical structure is:
Inactive ingredients include corn starch, hydroxypropyl cellulose, lactose monohydrate, low-substituted hydroxypropyl cellulose, magnesium stearate and microcrystalline cellulose and FD&C Blue No. 2 Aluminum Lake as colorant.
2.2 Monitoring
To mitigate the risk of significant or irreversible liver injury, perform blood testing for ALT, AST and bilirubin prior to initiation of JYNARQUE, at 2 and 4 weeks after initiation, monthly for 18 months and every 3 months thereafter. Monitor for concurrent symptoms that may indicate liver injury [see Warnings and Precautions (5.1)].
2.3 Missed Doses
If a dose of JYNARQUE is not taken at the scheduled time, take the next dose at its scheduled time.
8.4 Pediatric Use
Safety and effectiveness of JYNARQUE in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of tolvaptan did not include sufficient numbers of subjects aged 65 years old and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
14 Clinical Studies
JYNARQUE was shown to slow the rate of decline in renal function in adult patients at risk of rapidly progressing ADPKD in two trials: TEMPO 3:4 in patients at earlier stages of disease and REPRISE in patients at later stages. The findings from these trials, when taken together, suggest that JYNARQUE slows the loss of renal function progressively through the course of the disease.
4 Contraindications
JYNARQUE is contraindicated in patients:
- With a history, signs or symptoms of significant liver impairment or injury. This contraindication does not apply to uncomplicated polycystic liver disease [see Warnings and Precautions (5.1)]
- Taking strong CYP3A inhibitors
- With uncorrected abnormal blood sodium concentrations [see Warnings and Precautions (5.3)]
- Unable to sense or respond to thirst [see Warnings and Precautions (5.3)]
- Hypovolemia [see Warnings and Precautions (5.3)]
- Hypersensitivity (e.g., anaphylaxis, rash) to tolvaptan or any component of the product [see Adverse Reactions (6)]
- Uncorrected urinary outflow obstruction
- Anuria
6 Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Serious Liver Injury [see Boxed Warning and Warnings and Precautions (5.1)]
- Hypernatremia, Dehydration and Hypovolemia [see Warnings and Precautions (5.3)]
- Drug Interactions with Inhibitors of CYP3A [see Warnings and Precautions (5.4)]
7 Drug Interactions
12.2 Pharmacodynamics
In healthy subjects or patients with eGFRs as low as 10 mL/min/1.73m2 receiving a single dose of tolvaptan, the onset of the aquaretic effects occurs within 1 to 2 hours post-dose. In healthy subjects, single doses of 60 mg and 90 mg produce a peak effect of about a 9 mL/min increase in urine excretion rate is observed between 4 and 8 hours post-dose. Higher doses of tolvaptan do not increase the peak effect in urine excretion rate but sustain the effect for a longer period of time.
Urine excretion rate returns to baseline within 24 hours following the maximum recommended 90 mg dose of tolvaptan.
Changes in free water clearance mirror the changes in urine excretion rate. Increased free water clearance causes an increase in serum sodium concentration unless fluid intake is increased to match urine output.
Increases in urine excretion rate and free water clearance are positively correlated with baseline glomerular filtration rate with increases in both values observed in patients with creatinine clearance as low as 15 mL/min.
With the recommended split-dose regimens, tolvaptan inhibits vasopressin from binding to the V2-receptor in the kidney for the entire day, as indicated by increased urine output and decreased urine osmolality. Following a 90/30 mg split-dose regimen in patients with eGFR >60 mL/min/1.73 m2, the change in mean daily urine volume was about 4 L for a mean total daily volume of about 7 L. In patients with eGFR <30 mL/min/1.73 m2, the mean change in daily urine volume was about 2 L for a total daily urine volume of about 5 L.
Plasma concentrations of native AVP may increase (avg. 2 to 9 pg/mL) with tolvaptan treatment and return to baseline levels when treatment is stopped.
During tolvaptan treatment, small changes in renal function are expected and the changes are independent of baseline renal function. Glomerular filtration rate is decreased about 6% to 10% and uric acid clearance is decreased about 20% to 25%. Percent changes in renal plasma flow are highly correlated to percent changes in GFR. These changes are reversed upon discontinuation of tolvaptan.
12.3 Pharmacokinetics
In healthy subjects, the pharmacokinetics of tolvaptan after single doses of up to 480 mg and multiple doses up to 300 mg once daily have been studied. In ADPKD patients, single doses up to 120 mg and multiple split-doses up to 90/30 mg have been studied.
2.1 Recommended Dosage
The initial dosage for JYNARQUE is 60 mg orally per day as 45 mg taken on waking and 15 mg taken 8 hours later. Titrate to 60 mg plus 30 mg then to 90 mg plus 30 mg per day if tolerated with at least weekly intervals between titrations. Patients may down-titrate based on tolerability. Encourage patients to drink enough water to avoid thirst or dehydration.
1 Indications and Usage
JYNARQUE is indicated to slow kidney function decline in adults at risk of rapidly progressing autosomal dominant polycystic kidney disease (ADPKD).
12.1 Mechanism of Action
Tolvaptan is a selective vasopressin V2-receptor antagonist with an affinity for the V2-receptor that is 1.8 times that of native arginine vasopressin (AVP). Tolvaptan affinity for the V2-receptor is 29 times that for the V1a-receptor. Decreased binding of vasopressin to the V2-receptor in the kidney lowers adenylate cyclase activity resulting in a decrease in intracellular adenosine 3′, 5′-cyclic monophosphate (cAMP) concentrations. Decreased cAMP concentrations prevent aquaporin 2 containing vesicles from fusing with the plasma membrane, which in turn causes an increase in urine water excretion, an increase in free water clearance (aquaresis) and a decrease in urine osmolality. In human ADPKD cyst epithelial cells, tolvaptan inhibited AVP-stimulated in vitro cyst growth and chloride-dependent fluid secretion into cysts. In animal models, decreased cAMP concentrations were associated with decreases in the rate of growth of total kidney volume and the rate of formation and enlargement of kidney cysts. Tolvaptan metabolites have no or weak antagonist activity for human V2-receptors compared with tolvaptan.
5.1 Serious Liver Injury
JYNARQUE can cause serious and potentially fatal liver injury. Acute liver failure requiring liver transplantation has been reported in the post-marketing ADPKD experience. Discontinuation in response to laboratory abnormalities or signs or symptoms of liver injury (such as fatigue, anorexia, nausea, right upper abdominal discomfort, vomiting, fever, rash, pruritus, icterus, dark urine or jaundice) can reduce the risk of severe hepatotoxicity.
In a 3-year placebo-controlled trial and its open-label extension (in which patients' liver tests were monitored every 4 months), evidence of serious hepatocellular injury (elevations of hepatic transaminases of at least 3 times the upper limit of normal [ULN] combined with elevated bilirubin at least 2 times the ULN) occurred in 0.2% (3/1487) of tolvaptan-treated patients compared to none of the placebo-treated patients.
To reduce the risk of significant or irreversible liver injury, assess ALT, AST and bilirubin prior to initiation of JYNARQUE, at 2 weeks and 4 weeks after initiation, then monthly for 18 months and every 3 months thereafter.
At the onset of signs or symptoms consistent with hepatic injury or if ALT, AST, or bilirubin increase to >2 times ULN, immediately discontinue JYNARQUE, obtain repeat tests as soon as possible (within 48 to 72 hours), and continue testing as appropriate. If laboratory abnormalities stabilize or resolve, JYNARQUE may be reinitiated with increased frequency of monitoring as long as ALT and AST remain below 3 times the ULN.
Do not restart JYNARQUE in patients who experience signs or symptoms consistent with hepatic injury or whose ALT or AST ever exceeds 3 times the ULN during treatment with tolvaptan, unless there is another explanation for liver injury and the injury has resolved.
In patients with a stable, low baseline AST or ALT, an increase above 2 times baseline, even if less than 2 times the ULN, may indicate early liver injury. Such elevations may warrant treatment suspension and prompt (48 to 72 hours) re-evaluation of liver test trends prior to reinitiating therapy with more frequent monitoring.
5 Warnings and Precautions
- Hypernatremia, dehydration and hypovolemia: May require intervention (5.3)
2 Dosage and Administration
- Recommended dosage (2.1)
Initial Dosage Titration Step Target Dosage 1st Dose 45 mg 1st Dose 60 mg 1st Dose 90 mg 2nd Dose
(8 hours later)15 mg 2nd Dose
(8 hours later)30 mg 2nd Dose
(8 hours later)30 mg Total Daily Dose 60 mg Total Daily Dose 90 mg Total Daily Dose 120 mg
3 Dosage Forms and Strengths
JYNARQUE (tolvaptan) is supplied as non-scored, blue, shallow-convex, immediate release tablets, debossed with "OTSUKA" and the tablet strength (mg) on one side as follows:
| Strength | Shape |
|---|---|
| 15 mg | triangular |
| 30 mg | round |
| 45 mg | square |
| 60 mg | rectangular |
| 90 mg | pentagonal |
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of tolvaptan. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hepatobiliary Disorders: Liver failure requiring transplant
Immune System Disorders: Anaphylaxis
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. JYNARQUE has been studied in over 3000 patients with ADPKD. Long-term, placebo-controlled safety information of JYNARQUE in ADPKD is principally derived from two trials where 1,413 subjects received tolvaptan and 1,098 received placebo for at least 12 months across both studies.
17 Patient Counseling Information
As part of patient counseling, healthcare providers must review the JYNARQUE Medication Guide with every patient [see Medication Guide].
Warning: Risk of Serious Liver Injury
JYNARQUE (tolvaptan) can cause serious and potentially fatal liver injury. Acute liver failure requiring liver transplantation has been reported [see Warnings and Precautions (5.1)].
Measure ALT, AST and bilirubin before initiating treatment, at 2 weeks and 4 weeks after initiation, then monthly for the first 18 months and every 3 months thereafter [see Warnings and Precautions (5.1)]. Prompt action in response to laboratory abnormalities, signs, or symptoms indicative of hepatic injury can mitigate, but not eliminate, the risk of serious hepatotoxicity.
Because of the risks of serious liver injury, JYNARQUE is available only through a restricted distribution program under a Risk Evaluation and Mitigation Strategy (REMS) called the Tolvaptan for ADPKD Shared System REMS [see Warnings and Precautions (5.2)].
8.7 Use in Patients With Renal Impairment
Efficacy studies included patients with normal and reduced renal function [see Clinical Studies (14)]. TEMPO 3:4 required patients to have an estimated creatinine clearance ≥60 mL/min, while REPRISE included patients with eGFRCKD-Epi 25 to 65 mL/min/1.73m2.
Principal Display Panel 15 15 Mg Sleeve
1
Press here
to open
14 Tablets
Weekly Pack contains
1 blister card with 14 tablets
(14 x 15 mg tablets).
NDC 59148-079-07
Rx only
JYNARQUE®
(tolvaptan) tablets
15 mg
per tablet
and
15 mg
per tablet
Instructions to the right
- Using thumbnail, press and hold down perforated button.
- While holding button down, pull out foil package.
- Push tablet through foil.
- Push weekly pack into carton.
DISPENSE THE ENCLOSED
MEDICATION GUIDE
TO EACH PATIENT
2
PULL HERE
Principal Display Panel 30 15 Mg Sleeve
1
Press here
to open
14 Tablets
Weekly Pack contains
1 blister card with 14 tablets
(7 x 30 mg tablets and 7 x 15 mg tablets).
NDC 59148-080-07
Rx only
JYNARQUE®
(tolvaptan) tablets
30 mg
per tablet
and
15 mg
per tablet
Instructions to the right
- Using thumbnail, press and hold down perforated button.
- While holding button down, pull out foil package.
- Push tablet through foil.
- Push weekly pack into carton.
DISPENSE THE ENCLOSED
MEDICATION GUIDE
TO EACH PATIENT
2
PULL HERE
Principal Display Panel 45 15 Mg Sleeve
1
Press here
to open
14 Tablets
Weekly Pack contains
1 blister card with 14 tablets
(7 x 45 mg tablets and 7 x 15 mg tablets).
NDC 59148-087-07
Rx only
JYNARQUE®
(tolvaptan) tablets
45 mg
per tablet
and
15 mg
per tablet
Instructions to the right
- Using thumbnail, press and hold down perforated button.
- While holding button down, pull out foil package.
- Push tablet through foil.
- Push weekly pack into carton.
DISPENSE THE ENCLOSED
MEDICATION GUIDE
TO EACH PATIENT
2
PULL HERE
Principal Display Panel 60 30 Mg Sleeve
1
Press here
to open
14 Tablets
Weekly Pack contains
1 blister card with 14 tablets
(7 x 60 mg tablets and 7 x 30 mg tablets).
NDC 59148-088-07
Rx only
JYNARQUE®
(tolvaptan) tablets
60 mg
per tablet
and
30 mg
per tablet
Instructions to the right
- Using thumbnail, press and hold down perforated button.
- While holding button down, pull out foil package.
- Push tablet through foil.
- Push weekly pack into carton.
DISPENSE THE ENCLOSED
MEDICATION GUIDE
TO EACH PATIENT
2
PULL HERE
Principal Display Panel 90 30 Mg Sleeve
1
Press here
to open
14 Tablets
Weekly Pack contains
1 blister card with 14 tablets
(7 x 90 mg tablets and 7 x 30 mg tablets).
NDC 59148-089-07
Rx only
JYNARQUE®
(tolvaptan) tablets
90 mg
per tablet
and
30 mg
per tablet
Instructions to the right
- Using thumbnail, press and hold down perforated button.
- While holding button down, pull out foil package.
- Push tablet through foil.
- Push weekly pack into carton.
DISPENSE THE ENCLOSED
MEDICATION GUIDE
TO EACH PATIENT
2
PULL HERE
5.2 Tolvaptan for Adpkd Shared System Rems
JYNARQUE is available only through a restricted distribution program under a Risk Evaluation and Mitigation Strategy (REMS) called the Tolvaptan for ADPKD Shared System REMS, because of the risks of liver injury [see Warnings and Precautions (5.1)].
Notable requirements of the Tolvaptan for ADPKD Shared System REMS include the following:
- Prescribers must be certified by enrolling in the REMS program.
- Prescribers must inform patients receiving JYNARQUE about the risk of hepatotoxicity associated with its use and how to recognize the signs and symptoms of hepatotoxicity and the appropriate actions to take if it occurs.
- Patients must enroll in the REMS program and comply with ongoing monitoring requirements.
- Pharmacies must be certified by enrolling in the REMS program and must only dispense to patients who are authorized to receive JYNARQUE.
Further information, including a list of qualified pharmacies/distributors, is available at www.TolvaptanADPKDSharedREMS.com or by telephone at 1-866-244-9446.
8.6 Use in Patients With Hepatic Impairment
Because of the risk of serious liver injury, use is contraindicated in patients with a history, signs or symptoms of significant liver impairment or injury. This contraindication does not apply to uncomplicated polycystic liver disease which was present in 60% and 66% of patients in TEMPO 3:4 and REPRISE, respectively. No specific exclusion for hepatic impairment was implemented in TEMPO 3:4. However, REPRISE excluded patients with ADPKD who had hepatic impairment or liver function abnormalities other than that expected for ADPKD with typical cystic liver disease [see Contraindications (4)].
Principal Display Panel 15 Mg Tablet Label
NDC 59148-082-13
Rx only
JYNARQUE®
(tolvaptan) tablets
15 mg
DISPENSE THE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
30 tablets
Principal Display Panel 30 Mg Tablet Label
NDC 59148-083-13
Rx only
JYNARQUE®
(tolvaptan) tablets
30 mg
DISPENSE THE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
30 tablets
5.3 Hypernatremia, Dehydration and Hypovolemia
JYNARQUE increases free water clearance and, as a result, may cause dehydration, hypovolemia and hypernatremia. Therefore, ensure abnormalities in sodium concentrations are corrected prior to initiation of therapy.
Instruct patients to drink water when thirsty, and throughout the day and night if awake. Monitor for weight loss, tachycardia and hypotension because they may signal dehydration.
In the two double-blind, placebo-controlled trials of patients with ADPKD, hypernatremia (defined as any serum sodium concentration >150 mEq/L) was observed in 4.0% versus 0.6% and 1.4% versus 0% of tolvaptan-treated versus placebo-treated patients, respectively. The rate of dehydration and hypovolemia in the two studies was 2.1% versus 0.7% and 2.3% versus 0.4% for tolvaptan-treated versus placebo-treated patients, respectively.
During JYNARQUE therapy, if serum sodium increases above normal range or the patient becomes hypovolemic or dehydrated and fluid intake cannot be increased, then suspend JYNARQUE until serum sodium, hydration status and volume status is within the normal range.
5.4 Co Administration With Inhibitors of Cyp3a
Concomitant use of JYNARQUE with drugs that are moderate or strong CYP3A inhibitors (e.g., ketoconazole, itraconazole, lopinavir/ritonavir, indinavir/ritonavir, ritonavir, and conivaptan) increases tolvaptan exposure [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)]. Use with strong CYP3A inhibitors is contraindicated; dose reduction of JYNARQUE is recommended for patients while taking moderate CYP3A inhibitors [see Dosage and Administration (2.4) and Contraindications (4)].
Principal Display Panel 15 Mg Tablet Bottle Carton
NDC 59148-082-13
Rx only
JYNARQUE®
(tolvaptan) tablets
15 mg
DISPENSE THE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
30 tablets
Principal Display Panel 30 Mg Tablet Bottle Carton
NDC 59148-083-13
Rx only
JYNARQUE®
(tolvaptan) tablets
30 mg
DISPENSE THE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
30 tablets
Principal Display Panel Kit Carton 15 Mg and 15 Mg
56 Tablets
Monthly Carton contains
4 child resistant Weekly Packs.
Each Weekly Pack contains 1 blister card with
14 tablets (14 x 15 mg tablets).
NDC 59148-079-28
Rx only
JYNARQUE®
(tolvaptan) tablets
15 mg
per tablet
and
15 mg
per tablet
DISPENSE THE ENCLOSED MEDICATION GUIDE TO EACH PATIENT
Principal Display Panel Kit Carton 30 Mg and 15 Mg
56 Tablets
Monthly Carton contains
4 child resistant Weekly Packs.
Each Weekly Pack contains 1 blister card with
14 tablets (7 x 30 mg tablets and 7 x 15 mg tablets).
NDC 59148-080-28
Rx only
JYNARQUE®
(tolvaptan) tablets
30 mg
per tablet
and
15 mg
per tablet
DISPENSE THE ENCLOSED MEDICATION GUIDE TO EACH PATIENT
Principal Display Panel Kit Carton 45 Mg and 15 Mg
56 Tablets
Monthly Carton contains
4 child resistant Weekly Packs.
Each Weekly Pack contains 1 blister card with
14 tablets (7 x 45 mg tablets and 7 x 15 mg tablets).
NDC 59148-087-28
Rx only
JYNARQUE®
(tolvaptan) tablets
45 mg
per tablet
and
15 mg
per tablet
DISPENSE THE ENCLOSED MEDICATION GUIDE TO EACH PATIENT
Principal Display Panel Kit Carton 60 Mg and 30 Mg
56 Tablets
Monthly Carton contains
4 child resistant Weekly Packs.
Each Weekly Pack contains 1 blister card with
14 tablets (7 x 60 mg tablets and 7 x 30 mg tablets).
NDC 59148-088-28
Rx only
JYNARQUE®
(tolvaptan) tablets
60 mg
per tablet
and
30 mg
per tablet
DISPENSE THE ENCLOSED MEDICATION GUIDE TO EACH PATIENT
Principal Display Panel Kit Carton 90 Mg and 30 Mg
56 Tablets
Monthly Carton contains
4 child resistant Weekly Packs.
Each Weekly Pack contains 1 blister card with
14 tablets (7 x 90 mg tablets and 7 x 30 mg tablets).
NDC 59148-089-28
Rx only
JYNARQUE®
(tolvaptan) tablets
90 mg
per tablet
and
30 mg
per tablet
DISPENSE THE ENCLOSED MEDICATION GUIDE TO EACH PATIENT
Structured Label Content
7.2 V2
As a V2-receptor antagonist, tolvaptan will interfere with the V2-agonist activity of desmopressin (dDAVP). Avoid concomitant use of JYNARQUE with a V2-agonist.
Section 42229-5 (42229-5)
CYP3A Inhibitors
Concomitant use of strong CYP3A inhibitors is contraindicated [see Contraindications (4) and Warnings and Precautions (5.4)].
In patients taking concomitant moderate CYP3A inhibitors, reduce the dose of JYNARQUE per Table 1. Consider further reductions if patients cannot tolerate the reduced dose [see Warnings and Precautions (5.4) and Drug Interactions (7.1)]. Interrupt JYNARQUE temporarily for short term therapy with moderate CYP3A inhibitors if the recommended reduced doses are not available.
| Standard Morning and Afternoon Dose (mg) | Dose (mg) with Moderate CYP3A Inhibitors |
|---|---|
| 90 mg and 30 mg | 45 mg and 15 mg |
| 60 mg and 30 mg | 30 mg and 15 mg |
| 45 mg and 15 mg | 15 mg and 15 mg |
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 3/2025 | ||
|
MEDICATION GUIDE JYNARQUE® (jin-AR-kew) (tolvaptan) Tablets |
|||
|
What is the most important information I should know about JYNARQUE? JYNARQUE can cause serious side effects, including:
|
|||
|
|
||
| To help reduce your risk of liver problems, your healthcare provider will do a blood test to check your liver: | |||
|
|||
| It is important to stay under the care of your healthcare provider during treatment with JYNARQUE. Because of the risk of serious liver problems JYNARQUE is only available through a restricted distribution program called the Tolvaptan for ADPKD Shared System REMS.
|
|||
|
What is JYNARQUE?
JYNARQUE is a prescription medicine used to slow kidney function decline in adults who are at risk of rapidly progressing autosomal dominant polycystic kidney disease (ADPKD). It is not known if JYNARQUE is safe and effective in children. |
|||
Do not take JYNARQUE if you:
|
|||
Before taking JYNARQUE, tell your healthcare provider about all your medical conditions, including if you:
|
|||
How should I take JYNARQUE?
|
|||
|
What are the possible side effects of JYNARQUE? JYNARQUE may cause serious side effects, including: See "What is the most important information I should know about JYNARQUE?"
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
How should I store JYNARQUE?
JYNARQUE comes in a child-resistant package. Store JYNARQUE between 68°F to 77°F (20°C to 25°C). Keep JYNARQUE and all medicines out of the reach of children. |
|||
|
General information about the safe and effective use of JYNARQUE.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use JYNARQUE for a condition for which it was not prescribed. Do not give JYNARQUE to other people, even if they have the same symptoms you have. It may harm them. |
|||
|
What are the ingredients in JYNARQUE? Active ingredient: tolvaptan Inactive ingredients: corn starch, hydroxypropyl cellulose, lactose monohydrate, low-substituted hydroxypropyl cellulose, magnesium stearate and microcrystalline cellulose, and FD&C Blue no. 2 Aluminum Lake as colorant. |
|||
| Manufactured by Otsuka Pharmaceutical Co., Ltd., Tokyo, 101-8535 Japan Distributed and marketed by Otsuka America Pharmaceutical, Inc., Rockville, MD 20850 USA JYNARQUE is a registered trademark of Otsuka Pharmaceutical Co., Ltd., Tokyo, 101-8535 Japan ©2025, Otsuka Pharmaceutical Co., Ltd., Tokyo, 101-8535 Japan For more information about JYNARQUE, go to www.TolvaptanADPKDSharedREMS.com or call 1-866-244-9446. |
Section 43683-2 (43683-2)
|
Warnings and Precautions
Tolvaptan for ADPKD Shared System REMS (5.2) |
3/2025 |
Section 44425-7 (44425-7)
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (59°F to 86°F) [see USP controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
Single oral doses up to 480 mg (4 times the maximum recommended daily dose) and multiple doses up to 300 mg once daily for 5 days have been well tolerated in trials in healthy subjects. There is no specific antidote for tolvaptan intoxication. The signs and symptoms of an acute overdose can be anticipated to be those of excessive pharmacologic effect: a rise in serum sodium concentration, polyuria, thirst, and dehydration/hypovolemia.
No mortality was observed in rats or dogs following single oral doses of 2000 mg/kg (maximum feasible dose). A single oral dose of 2000 mg/kg was lethal in mice, and symptoms of toxicity in affected mice included decreased locomotor activity, staggering gait, tremor and hypothermia.
In patients with suspected JYNARQUE overdosage, assessment of vital signs, electrolyte concentrations, ECG and fluid status is recommended. Continue replacement of water and electrolytes until aquaresis abates. Dialysis may not be effective in removing JYNARQUE because of its high binding affinity for human plasma protein (>98%).
11 Description (11 DESCRIPTION)
JYNARQUE contains tolvaptan, a selective vasopressin V2-receptor antagonist in immediate release tablets for oral administration available in 15 mg, 30 mg, 45 mg, 60 mg and 90 mg strengths. Tolvaptan is (±)-4'-[(7-chloro-2,3,4,5-tetrahydro-5-hydroxy-1H-1-benzazepin-1-yl) carbonyl]-o-tolu-m-toluidide. The empirical formula is C26H25ClN2O3. Molecular weight is 448.94. The chemical structure is:
Inactive ingredients include corn starch, hydroxypropyl cellulose, lactose monohydrate, low-substituted hydroxypropyl cellulose, magnesium stearate and microcrystalline cellulose and FD&C Blue No. 2 Aluminum Lake as colorant.
2.2 Monitoring
To mitigate the risk of significant or irreversible liver injury, perform blood testing for ALT, AST and bilirubin prior to initiation of JYNARQUE, at 2 and 4 weeks after initiation, monthly for 18 months and every 3 months thereafter. Monitor for concurrent symptoms that may indicate liver injury [see Warnings and Precautions (5.1)].
2.3 Missed Doses
If a dose of JYNARQUE is not taken at the scheduled time, take the next dose at its scheduled time.
8.4 Pediatric Use
Safety and effectiveness of JYNARQUE in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of tolvaptan did not include sufficient numbers of subjects aged 65 years old and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
14 Clinical Studies (14 CLINICAL STUDIES)
JYNARQUE was shown to slow the rate of decline in renal function in adult patients at risk of rapidly progressing ADPKD in two trials: TEMPO 3:4 in patients at earlier stages of disease and REPRISE in patients at later stages. The findings from these trials, when taken together, suggest that JYNARQUE slows the loss of renal function progressively through the course of the disease.
4 Contraindications (4 CONTRAINDICATIONS)
JYNARQUE is contraindicated in patients:
- With a history, signs or symptoms of significant liver impairment or injury. This contraindication does not apply to uncomplicated polycystic liver disease [see Warnings and Precautions (5.1)]
- Taking strong CYP3A inhibitors
- With uncorrected abnormal blood sodium concentrations [see Warnings and Precautions (5.3)]
- Unable to sense or respond to thirst [see Warnings and Precautions (5.3)]
- Hypovolemia [see Warnings and Precautions (5.3)]
- Hypersensitivity (e.g., anaphylaxis, rash) to tolvaptan or any component of the product [see Adverse Reactions (6)]
- Uncorrected urinary outflow obstruction
- Anuria
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Serious Liver Injury [see Boxed Warning and Warnings and Precautions (5.1)]
- Hypernatremia, Dehydration and Hypovolemia [see Warnings and Precautions (5.3)]
- Drug Interactions with Inhibitors of CYP3A [see Warnings and Precautions (5.4)]
7 Drug Interactions (7 DRUG INTERACTIONS)
12.2 Pharmacodynamics
In healthy subjects or patients with eGFRs as low as 10 mL/min/1.73m2 receiving a single dose of tolvaptan, the onset of the aquaretic effects occurs within 1 to 2 hours post-dose. In healthy subjects, single doses of 60 mg and 90 mg produce a peak effect of about a 9 mL/min increase in urine excretion rate is observed between 4 and 8 hours post-dose. Higher doses of tolvaptan do not increase the peak effect in urine excretion rate but sustain the effect for a longer period of time.
Urine excretion rate returns to baseline within 24 hours following the maximum recommended 90 mg dose of tolvaptan.
Changes in free water clearance mirror the changes in urine excretion rate. Increased free water clearance causes an increase in serum sodium concentration unless fluid intake is increased to match urine output.
Increases in urine excretion rate and free water clearance are positively correlated with baseline glomerular filtration rate with increases in both values observed in patients with creatinine clearance as low as 15 mL/min.
With the recommended split-dose regimens, tolvaptan inhibits vasopressin from binding to the V2-receptor in the kidney for the entire day, as indicated by increased urine output and decreased urine osmolality. Following a 90/30 mg split-dose regimen in patients with eGFR >60 mL/min/1.73 m2, the change in mean daily urine volume was about 4 L for a mean total daily volume of about 7 L. In patients with eGFR <30 mL/min/1.73 m2, the mean change in daily urine volume was about 2 L for a total daily urine volume of about 5 L.
Plasma concentrations of native AVP may increase (avg. 2 to 9 pg/mL) with tolvaptan treatment and return to baseline levels when treatment is stopped.
During tolvaptan treatment, small changes in renal function are expected and the changes are independent of baseline renal function. Glomerular filtration rate is decreased about 6% to 10% and uric acid clearance is decreased about 20% to 25%. Percent changes in renal plasma flow are highly correlated to percent changes in GFR. These changes are reversed upon discontinuation of tolvaptan.
12.3 Pharmacokinetics
In healthy subjects, the pharmacokinetics of tolvaptan after single doses of up to 480 mg and multiple doses up to 300 mg once daily have been studied. In ADPKD patients, single doses up to 120 mg and multiple split-doses up to 90/30 mg have been studied.
2.1 Recommended Dosage
The initial dosage for JYNARQUE is 60 mg orally per day as 45 mg taken on waking and 15 mg taken 8 hours later. Titrate to 60 mg plus 30 mg then to 90 mg plus 30 mg per day if tolerated with at least weekly intervals between titrations. Patients may down-titrate based on tolerability. Encourage patients to drink enough water to avoid thirst or dehydration.
1 Indications and Usage (1 INDICATIONS AND USAGE)
JYNARQUE is indicated to slow kidney function decline in adults at risk of rapidly progressing autosomal dominant polycystic kidney disease (ADPKD).
12.1 Mechanism of Action
Tolvaptan is a selective vasopressin V2-receptor antagonist with an affinity for the V2-receptor that is 1.8 times that of native arginine vasopressin (AVP). Tolvaptan affinity for the V2-receptor is 29 times that for the V1a-receptor. Decreased binding of vasopressin to the V2-receptor in the kidney lowers adenylate cyclase activity resulting in a decrease in intracellular adenosine 3′, 5′-cyclic monophosphate (cAMP) concentrations. Decreased cAMP concentrations prevent aquaporin 2 containing vesicles from fusing with the plasma membrane, which in turn causes an increase in urine water excretion, an increase in free water clearance (aquaresis) and a decrease in urine osmolality. In human ADPKD cyst epithelial cells, tolvaptan inhibited AVP-stimulated in vitro cyst growth and chloride-dependent fluid secretion into cysts. In animal models, decreased cAMP concentrations were associated with decreases in the rate of growth of total kidney volume and the rate of formation and enlargement of kidney cysts. Tolvaptan metabolites have no or weak antagonist activity for human V2-receptors compared with tolvaptan.
5.1 Serious Liver Injury
JYNARQUE can cause serious and potentially fatal liver injury. Acute liver failure requiring liver transplantation has been reported in the post-marketing ADPKD experience. Discontinuation in response to laboratory abnormalities or signs or symptoms of liver injury (such as fatigue, anorexia, nausea, right upper abdominal discomfort, vomiting, fever, rash, pruritus, icterus, dark urine or jaundice) can reduce the risk of severe hepatotoxicity.
In a 3-year placebo-controlled trial and its open-label extension (in which patients' liver tests were monitored every 4 months), evidence of serious hepatocellular injury (elevations of hepatic transaminases of at least 3 times the upper limit of normal [ULN] combined with elevated bilirubin at least 2 times the ULN) occurred in 0.2% (3/1487) of tolvaptan-treated patients compared to none of the placebo-treated patients.
To reduce the risk of significant or irreversible liver injury, assess ALT, AST and bilirubin prior to initiation of JYNARQUE, at 2 weeks and 4 weeks after initiation, then monthly for 18 months and every 3 months thereafter.
At the onset of signs or symptoms consistent with hepatic injury or if ALT, AST, or bilirubin increase to >2 times ULN, immediately discontinue JYNARQUE, obtain repeat tests as soon as possible (within 48 to 72 hours), and continue testing as appropriate. If laboratory abnormalities stabilize or resolve, JYNARQUE may be reinitiated with increased frequency of monitoring as long as ALT and AST remain below 3 times the ULN.
Do not restart JYNARQUE in patients who experience signs or symptoms consistent with hepatic injury or whose ALT or AST ever exceeds 3 times the ULN during treatment with tolvaptan, unless there is another explanation for liver injury and the injury has resolved.
In patients with a stable, low baseline AST or ALT, an increase above 2 times baseline, even if less than 2 times the ULN, may indicate early liver injury. Such elevations may warrant treatment suspension and prompt (48 to 72 hours) re-evaluation of liver test trends prior to reinitiating therapy with more frequent monitoring.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypernatremia, dehydration and hypovolemia: May require intervention (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Recommended dosage (2.1)
Initial Dosage Titration Step Target Dosage 1st Dose 45 mg 1st Dose 60 mg 1st Dose 90 mg 2nd Dose
(8 hours later)15 mg 2nd Dose
(8 hours later)30 mg 2nd Dose
(8 hours later)30 mg Total Daily Dose 60 mg Total Daily Dose 90 mg Total Daily Dose 120 mg
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
JYNARQUE (tolvaptan) is supplied as non-scored, blue, shallow-convex, immediate release tablets, debossed with "OTSUKA" and the tablet strength (mg) on one side as follows:
| Strength | Shape |
|---|---|
| 15 mg | triangular |
| 30 mg | round |
| 45 mg | square |
| 60 mg | rectangular |
| 90 mg | pentagonal |
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of tolvaptan. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hepatobiliary Disorders: Liver failure requiring transplant
Immune System Disorders: Anaphylaxis
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. JYNARQUE has been studied in over 3000 patients with ADPKD. Long-term, placebo-controlled safety information of JYNARQUE in ADPKD is principally derived from two trials where 1,413 subjects received tolvaptan and 1,098 received placebo for at least 12 months across both studies.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
As part of patient counseling, healthcare providers must review the JYNARQUE Medication Guide with every patient [see Medication Guide].
Warning: Risk of Serious Liver Injury (WARNING: RISK OF SERIOUS LIVER INJURY)
JYNARQUE (tolvaptan) can cause serious and potentially fatal liver injury. Acute liver failure requiring liver transplantation has been reported [see Warnings and Precautions (5.1)].
Measure ALT, AST and bilirubin before initiating treatment, at 2 weeks and 4 weeks after initiation, then monthly for the first 18 months and every 3 months thereafter [see Warnings and Precautions (5.1)]. Prompt action in response to laboratory abnormalities, signs, or symptoms indicative of hepatic injury can mitigate, but not eliminate, the risk of serious hepatotoxicity.
Because of the risks of serious liver injury, JYNARQUE is available only through a restricted distribution program under a Risk Evaluation and Mitigation Strategy (REMS) called the Tolvaptan for ADPKD Shared System REMS [see Warnings and Precautions (5.2)].
8.7 Use in Patients With Renal Impairment (8.7 Use in Patients with Renal Impairment)
Efficacy studies included patients with normal and reduced renal function [see Clinical Studies (14)]. TEMPO 3:4 required patients to have an estimated creatinine clearance ≥60 mL/min, while REPRISE included patients with eGFRCKD-Epi 25 to 65 mL/min/1.73m2.
Principal Display Panel 15 15 Mg Sleeve (PRINCIPAL DISPLAY PANEL - 15-15 mg Sleeve)
1
Press here
to open
14 Tablets
Weekly Pack contains
1 blister card with 14 tablets
(14 x 15 mg tablets).
NDC 59148-079-07
Rx only
JYNARQUE®
(tolvaptan) tablets
15 mg
per tablet
and
15 mg
per tablet
Instructions to the right
- Using thumbnail, press and hold down perforated button.
- While holding button down, pull out foil package.
- Push tablet through foil.
- Push weekly pack into carton.
DISPENSE THE ENCLOSED
MEDICATION GUIDE
TO EACH PATIENT
2
PULL HERE
Principal Display Panel 30 15 Mg Sleeve (PRINCIPAL DISPLAY PANEL - 30-15 mg Sleeve)
1
Press here
to open
14 Tablets
Weekly Pack contains
1 blister card with 14 tablets
(7 x 30 mg tablets and 7 x 15 mg tablets).
NDC 59148-080-07
Rx only
JYNARQUE®
(tolvaptan) tablets
30 mg
per tablet
and
15 mg
per tablet
Instructions to the right
- Using thumbnail, press and hold down perforated button.
- While holding button down, pull out foil package.
- Push tablet through foil.
- Push weekly pack into carton.
DISPENSE THE ENCLOSED
MEDICATION GUIDE
TO EACH PATIENT
2
PULL HERE
Principal Display Panel 45 15 Mg Sleeve (PRINCIPAL DISPLAY PANEL - 45-15 mg Sleeve)
1
Press here
to open
14 Tablets
Weekly Pack contains
1 blister card with 14 tablets
(7 x 45 mg tablets and 7 x 15 mg tablets).
NDC 59148-087-07
Rx only
JYNARQUE®
(tolvaptan) tablets
45 mg
per tablet
and
15 mg
per tablet
Instructions to the right
- Using thumbnail, press and hold down perforated button.
- While holding button down, pull out foil package.
- Push tablet through foil.
- Push weekly pack into carton.
DISPENSE THE ENCLOSED
MEDICATION GUIDE
TO EACH PATIENT
2
PULL HERE
Principal Display Panel 60 30 Mg Sleeve (PRINCIPAL DISPLAY PANEL - 60-30 mg Sleeve)
1
Press here
to open
14 Tablets
Weekly Pack contains
1 blister card with 14 tablets
(7 x 60 mg tablets and 7 x 30 mg tablets).
NDC 59148-088-07
Rx only
JYNARQUE®
(tolvaptan) tablets
60 mg
per tablet
and
30 mg
per tablet
Instructions to the right
- Using thumbnail, press and hold down perforated button.
- While holding button down, pull out foil package.
- Push tablet through foil.
- Push weekly pack into carton.
DISPENSE THE ENCLOSED
MEDICATION GUIDE
TO EACH PATIENT
2
PULL HERE
Principal Display Panel 90 30 Mg Sleeve (PRINCIPAL DISPLAY PANEL - 90-30 mg Sleeve)
1
Press here
to open
14 Tablets
Weekly Pack contains
1 blister card with 14 tablets
(7 x 90 mg tablets and 7 x 30 mg tablets).
NDC 59148-089-07
Rx only
JYNARQUE®
(tolvaptan) tablets
90 mg
per tablet
and
30 mg
per tablet
Instructions to the right
- Using thumbnail, press and hold down perforated button.
- While holding button down, pull out foil package.
- Push tablet through foil.
- Push weekly pack into carton.
DISPENSE THE ENCLOSED
MEDICATION GUIDE
TO EACH PATIENT
2
PULL HERE
5.2 Tolvaptan for Adpkd Shared System Rems (5.2 Tolvaptan for ADPKD Shared System REMS)
JYNARQUE is available only through a restricted distribution program under a Risk Evaluation and Mitigation Strategy (REMS) called the Tolvaptan for ADPKD Shared System REMS, because of the risks of liver injury [see Warnings and Precautions (5.1)].
Notable requirements of the Tolvaptan for ADPKD Shared System REMS include the following:
- Prescribers must be certified by enrolling in the REMS program.
- Prescribers must inform patients receiving JYNARQUE about the risk of hepatotoxicity associated with its use and how to recognize the signs and symptoms of hepatotoxicity and the appropriate actions to take if it occurs.
- Patients must enroll in the REMS program and comply with ongoing monitoring requirements.
- Pharmacies must be certified by enrolling in the REMS program and must only dispense to patients who are authorized to receive JYNARQUE.
Further information, including a list of qualified pharmacies/distributors, is available at www.TolvaptanADPKDSharedREMS.com or by telephone at 1-866-244-9446.
8.6 Use in Patients With Hepatic Impairment (8.6 Use in Patients with Hepatic Impairment)
Because of the risk of serious liver injury, use is contraindicated in patients with a history, signs or symptoms of significant liver impairment or injury. This contraindication does not apply to uncomplicated polycystic liver disease which was present in 60% and 66% of patients in TEMPO 3:4 and REPRISE, respectively. No specific exclusion for hepatic impairment was implemented in TEMPO 3:4. However, REPRISE excluded patients with ADPKD who had hepatic impairment or liver function abnormalities other than that expected for ADPKD with typical cystic liver disease [see Contraindications (4)].
Principal Display Panel 15 Mg Tablet Label (PRINCIPAL DISPLAY PANEL - 15 mg Tablet Label)
NDC 59148-082-13
Rx only
JYNARQUE®
(tolvaptan) tablets
15 mg
DISPENSE THE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
30 tablets
Principal Display Panel 30 Mg Tablet Label (PRINCIPAL DISPLAY PANEL - 30 mg Tablet Label)
NDC 59148-083-13
Rx only
JYNARQUE®
(tolvaptan) tablets
30 mg
DISPENSE THE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
30 tablets
5.3 Hypernatremia, Dehydration and Hypovolemia
JYNARQUE increases free water clearance and, as a result, may cause dehydration, hypovolemia and hypernatremia. Therefore, ensure abnormalities in sodium concentrations are corrected prior to initiation of therapy.
Instruct patients to drink water when thirsty, and throughout the day and night if awake. Monitor for weight loss, tachycardia and hypotension because they may signal dehydration.
In the two double-blind, placebo-controlled trials of patients with ADPKD, hypernatremia (defined as any serum sodium concentration >150 mEq/L) was observed in 4.0% versus 0.6% and 1.4% versus 0% of tolvaptan-treated versus placebo-treated patients, respectively. The rate of dehydration and hypovolemia in the two studies was 2.1% versus 0.7% and 2.3% versus 0.4% for tolvaptan-treated versus placebo-treated patients, respectively.
During JYNARQUE therapy, if serum sodium increases above normal range or the patient becomes hypovolemic or dehydrated and fluid intake cannot be increased, then suspend JYNARQUE until serum sodium, hydration status and volume status is within the normal range.
5.4 Co Administration With Inhibitors of Cyp3a (5.4 Co-Administration with Inhibitors of CYP3A)
Concomitant use of JYNARQUE with drugs that are moderate or strong CYP3A inhibitors (e.g., ketoconazole, itraconazole, lopinavir/ritonavir, indinavir/ritonavir, ritonavir, and conivaptan) increases tolvaptan exposure [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)]. Use with strong CYP3A inhibitors is contraindicated; dose reduction of JYNARQUE is recommended for patients while taking moderate CYP3A inhibitors [see Dosage and Administration (2.4) and Contraindications (4)].
Principal Display Panel 15 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 15 mg Tablet Bottle Carton)
NDC 59148-082-13
Rx only
JYNARQUE®
(tolvaptan) tablets
15 mg
DISPENSE THE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
30 tablets
Principal Display Panel 30 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 30 mg Tablet Bottle Carton)
NDC 59148-083-13
Rx only
JYNARQUE®
(tolvaptan) tablets
30 mg
DISPENSE THE ENCLOSED MEDICATION
GUIDE TO EACH PATIENT
30 tablets
Principal Display Panel Kit Carton 15 Mg and 15 Mg (PRINCIPAL DISPLAY PANEL - Kit Carton - 15 mg and 15 mg)
56 Tablets
Monthly Carton contains
4 child resistant Weekly Packs.
Each Weekly Pack contains 1 blister card with
14 tablets (14 x 15 mg tablets).
NDC 59148-079-28
Rx only
JYNARQUE®
(tolvaptan) tablets
15 mg
per tablet
and
15 mg
per tablet
DISPENSE THE ENCLOSED MEDICATION GUIDE TO EACH PATIENT
Principal Display Panel Kit Carton 30 Mg and 15 Mg (PRINCIPAL DISPLAY PANEL - Kit Carton - 30 mg and 15 mg)
56 Tablets
Monthly Carton contains
4 child resistant Weekly Packs.
Each Weekly Pack contains 1 blister card with
14 tablets (7 x 30 mg tablets and 7 x 15 mg tablets).
NDC 59148-080-28
Rx only
JYNARQUE®
(tolvaptan) tablets
30 mg
per tablet
and
15 mg
per tablet
DISPENSE THE ENCLOSED MEDICATION GUIDE TO EACH PATIENT
Principal Display Panel Kit Carton 45 Mg and 15 Mg (PRINCIPAL DISPLAY PANEL - Kit Carton - 45 mg and 15 mg)
56 Tablets
Monthly Carton contains
4 child resistant Weekly Packs.
Each Weekly Pack contains 1 blister card with
14 tablets (7 x 45 mg tablets and 7 x 15 mg tablets).
NDC 59148-087-28
Rx only
JYNARQUE®
(tolvaptan) tablets
45 mg
per tablet
and
15 mg
per tablet
DISPENSE THE ENCLOSED MEDICATION GUIDE TO EACH PATIENT
Principal Display Panel Kit Carton 60 Mg and 30 Mg (PRINCIPAL DISPLAY PANEL - Kit Carton - 60 mg and 30 mg)
56 Tablets
Monthly Carton contains
4 child resistant Weekly Packs.
Each Weekly Pack contains 1 blister card with
14 tablets (7 x 60 mg tablets and 7 x 30 mg tablets).
NDC 59148-088-28
Rx only
JYNARQUE®
(tolvaptan) tablets
60 mg
per tablet
and
30 mg
per tablet
DISPENSE THE ENCLOSED MEDICATION GUIDE TO EACH PATIENT
Principal Display Panel Kit Carton 90 Mg and 30 Mg (PRINCIPAL DISPLAY PANEL - Kit Carton - 90 mg and 30 mg)
56 Tablets
Monthly Carton contains
4 child resistant Weekly Packs.
Each Weekly Pack contains 1 blister card with
14 tablets (7 x 90 mg tablets and 7 x 30 mg tablets).
NDC 59148-089-28
Rx only
JYNARQUE®
(tolvaptan) tablets
90 mg
per tablet
and
30 mg
per tablet
DISPENSE THE ENCLOSED MEDICATION GUIDE TO EACH PATIENT
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:02.232065 · Updated: 2026-03-14T22:49:18.583726