guna®-diur
3f91cd92-eb6d-4bda-9cad-2e11e0b0fbe2
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
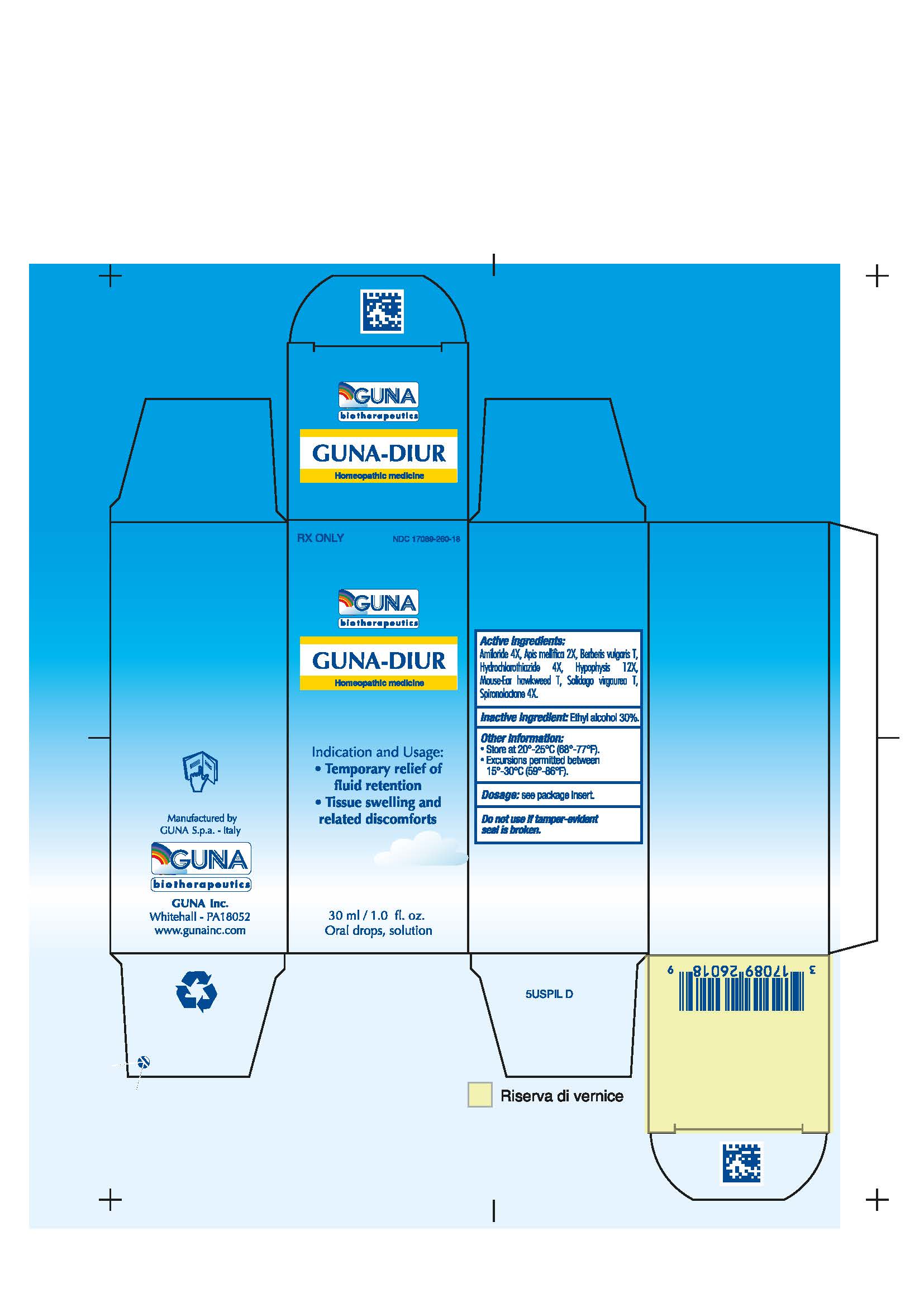
Composition & Product
Identifiers & Packaging
Indications and Usage
1.1 Temporary relief of fluid retention 1.2 Tissue swelling and related disconforts
Dosage and Administration
Adults: 20 drops in a little water, 2 times per day for an avarage of two months. Stop use and ask a doctor if symptoms persist more than 5 days. Administration may very according to individual needs. GUNA-DIUR may be used together with other homeopthic medicines.
Contraindications
4.1. There is no history of hypersensitivity to GUNA-DIUR. However, do not use if you are hypersensitive to any of the active ingredients of Guna-Diur.
Warnings and Precautions
5.1. GUNA-DIUR is contraindicated in patients with anuria and in patients with a history of hypersensitivity to Spironolactone, Amiloride, or Hydrocholorthiazide. 5.2. Use with caution in patients taking diuretic medications. 5.3 Keep out of reach of children.
Adverse Reactions
6.1. None known (see CONTRAINDICATIONS for hypersensitivity information).
Drug Interactions
7.1. None Known
How Supplied
16.1. NDC 17089-260-18 Oral Solution/Drops 30 mL 16.2. Store at 20-25°C (68-77° F). Excursions permitted between 15°-30°C (59°-86°F).
Storage and Handling
16.1. NDC 17089-260-18 Oral Solution/Drops 30 mL 16.2. Store at 20-25°C (68-77° F). Excursions permitted between 15°-30°C (59°-86°F).
Description
1.1 Temporary relief of fluid retention 1.2 Tissue swelling and related disconforts
Medication Information
Warnings and Precautions
5.1. GUNA-DIUR is contraindicated in patients with anuria and in patients with a history of hypersensitivity to Spironolactone, Amiloride, or Hydrocholorthiazide. 5.2. Use with caution in patients taking diuretic medications. 5.3 Keep out of reach of children.
Indications and Usage
1.1 Temporary relief of fluid retention 1.2 Tissue swelling and related disconforts
Dosage and Administration
Adults: 20 drops in a little water, 2 times per day for an avarage of two months. Stop use and ask a doctor if symptoms persist more than 5 days. Administration may very according to individual needs. GUNA-DIUR may be used together with other homeopthic medicines.
Contraindications
4.1. There is no history of hypersensitivity to GUNA-DIUR. However, do not use if you are hypersensitive to any of the active ingredients of Guna-Diur.
Adverse Reactions
6.1. None known (see CONTRAINDICATIONS for hypersensitivity information).
Drug Interactions
7.1. None Known
Storage and Handling
16.1. NDC 17089-260-18 Oral Solution/Drops 30 mL 16.2. Store at 20-25°C (68-77° F). Excursions permitted between 15°-30°C (59°-86°F).
How Supplied
16.1. NDC 17089-260-18 Oral Solution/Drops 30 mL 16.2. Store at 20-25°C (68-77° F). Excursions permitted between 15°-30°C (59°-86°F).
Description
1.1 Temporary relief of fluid retention 1.2 Tissue swelling and related disconforts
Package Label
10. Overdosage
10.1. No Known.
15. References
15.1. H.H. Reckeweg. Homeopathic Materia Medica. Aurelia Verlag.
15.2. Boericke, William, Materia Medica with Reperatory, 1927, ninth edition
11. Description
11. 1 GUNA-DIUR is a homeopathic medicine indicated for the temporary relief of fluid retention, tissue swelling and related disconforts.
14. Clinical Studies
14.1. GUNA-DIUR efficacy is not supported by clinical studies. It is based on homeopathic Materia Medica and scientific literature.
4. Contraindications
4.1. There is no history of hypersensitivity to GUNA-DIUR. However, do not use if you are hypersensitive to any of the active ingredients of Guna-Diur.
6. Adverse Reactions
6.1. None known (see CONTRAINDICATIONS for hypersensitivity information).
7. Drug Interactions
7.1. None Known
1. Indications and Usage
1.1 Temporary relief of fluid retention
1.2 Tissue swelling and related disconforts
12. Clinical Pharmacology
12.1. GUNA-DIUR exerts a diuretic effect. This is based on homeopthica Materia Medica and homeopathic principles.
12.2. Pharmacodynamics
Not applicable to homeopthic medicinal products.
12.3. Pharmacokinetics
Not applicable to homeopthic medicinal products.
13. Nonclinical Toxicology
13.1. Not available.
5. Warnings and Precautions
5.1. GUNA-DIUR is contraindicated in patients with anuria and in patients with a history of hypersensitivity to Spironolactone, Amiloride, or Hydrocholorthiazide.
5.2. Use with caution in patients taking diuretic medications.
5.3 Keep out of reach of children.
2. Dosage and Administration
Adults: 20 drops in a little water, 2 times per day for an avarage of two months.
Stop use and ask a doctor if symptoms persist more than 5 days.
Administration may very according to individual needs.
GUNA-DIUR may be used together with other homeopthic medicines.
9. Drug Abuse and Dependence
9.1. No Known.
3. Dosage Forms and Strengths
3.1. 30 ml Bottle dropper container contains:
Active ingredients: Amiloride 4X 0.006 ml, Apis Mellifica 2X 0.626 ml, Berberis Vulgaris T 0.314 ml, Hydrochlorothiazide 4X 0.006 ml, Hypophysis 12X 6.314 ml, Mouse-Ear Hawkweed T 6.314 ml, Solidago Virgaurea T 0.314 ml, Spironolactone 4X 0.006 ml.
Inactive Ingredient: Ethylic Alcohol 30%
8. Use in Specific Populations
8.1.
Pregnancy
: Pregnancy category C. Animal reproduction studies have not been conducted with GUNA-DIUR. GUNA®- DIUR should not be given to a pregnant woman.
8.2.
Lactation
: It is not known whether any of the ingredients in GUNA- DIUR are secreted in human milk. However, since many drugs are secreted in human milk, caution should be exercised when GUNA- DIUR is administered to a nursing woman.
8.3.
Pediatric use:
Safety and effectiveness in pediatric patients have not been established.
8.4.
Geriatric use
: No restrictions.
17. Patient Counseling Information
17.1. Patients should be informed about Homeopathy and the main differences with conventional clinical approaches.
16. How Supplied/storage and Handling
16.1. NDC 17089-260-18 Oral Solution/Drops 30 mL
16.2. Store at 20-25°C (68-77° F). Excursions permitted between 15°-30°C (59°-86°F).
Structured Label Content
Package Label (PACKAGE LABEL)
10. Overdosage (10. OVERDOSAGE)
10.1. No Known.
15. References (15. REFERENCES)
15.1. H.H. Reckeweg. Homeopathic Materia Medica. Aurelia Verlag.
15.2. Boericke, William, Materia Medica with Reperatory, 1927, ninth edition
11. Description (11. DESCRIPTION)
11. 1 GUNA-DIUR is a homeopathic medicine indicated for the temporary relief of fluid retention, tissue swelling and related disconforts.
14. Clinical Studies (14. CLINICAL STUDIES)
14.1. GUNA-DIUR efficacy is not supported by clinical studies. It is based on homeopathic Materia Medica and scientific literature.
4. Contraindications (4. CONTRAINDICATIONS)
4.1. There is no history of hypersensitivity to GUNA-DIUR. However, do not use if you are hypersensitive to any of the active ingredients of Guna-Diur.
6. Adverse Reactions (6. ADVERSE REACTIONS)
6.1. None known (see CONTRAINDICATIONS for hypersensitivity information).
7. Drug Interactions (7. DRUG INTERACTIONS)
7.1. None Known
1. Indications and Usage (1. INDICATIONS AND USAGE)
1.1 Temporary relief of fluid retention
1.2 Tissue swelling and related disconforts
12. Clinical Pharmacology (12. CLINICAL PHARMACOLOGY)
12.1. GUNA-DIUR exerts a diuretic effect. This is based on homeopthica Materia Medica and homeopathic principles.
12.2. Pharmacodynamics
Not applicable to homeopthic medicinal products.
12.3. Pharmacokinetics
Not applicable to homeopthic medicinal products.
13. Nonclinical Toxicology (13. NONCLINICAL TOXICOLOGY)
13.1. Not available.
5. Warnings and Precautions (5. WARNINGS AND PRECAUTIONS)
5.1. GUNA-DIUR is contraindicated in patients with anuria and in patients with a history of hypersensitivity to Spironolactone, Amiloride, or Hydrocholorthiazide.
5.2. Use with caution in patients taking diuretic medications.
5.3 Keep out of reach of children.
2. Dosage and Administration (2. DOSAGE AND ADMINISTRATION)
Adults: 20 drops in a little water, 2 times per day for an avarage of two months.
Stop use and ask a doctor if symptoms persist more than 5 days.
Administration may very according to individual needs.
GUNA-DIUR may be used together with other homeopthic medicines.
9. Drug Abuse and Dependence (9. DRUG ABUSE AND DEPENDENCE)
9.1. No Known.
3. Dosage Forms and Strengths (3. DOSAGE FORMS AND STRENGTHS)
3.1. 30 ml Bottle dropper container contains:
Active ingredients: Amiloride 4X 0.006 ml, Apis Mellifica 2X 0.626 ml, Berberis Vulgaris T 0.314 ml, Hydrochlorothiazide 4X 0.006 ml, Hypophysis 12X 6.314 ml, Mouse-Ear Hawkweed T 6.314 ml, Solidago Virgaurea T 0.314 ml, Spironolactone 4X 0.006 ml.
Inactive Ingredient: Ethylic Alcohol 30%
8. Use in Specific Populations (8. USE IN SPECIFIC POPULATIONS)
8.1.
Pregnancy
: Pregnancy category C. Animal reproduction studies have not been conducted with GUNA-DIUR. GUNA®- DIUR should not be given to a pregnant woman.
8.2.
Lactation
: It is not known whether any of the ingredients in GUNA- DIUR are secreted in human milk. However, since many drugs are secreted in human milk, caution should be exercised when GUNA- DIUR is administered to a nursing woman.
8.3.
Pediatric use:
Safety and effectiveness in pediatric patients have not been established.
8.4.
Geriatric use
: No restrictions.
17. Patient Counseling Information (17. PATIENT COUNSELING INFORMATION)
17.1. Patients should be informed about Homeopathy and the main differences with conventional clinical approaches.
16. How Supplied/storage and Handling (16. HOW SUPPLIED/STORAGE AND HANDLING)
16.1. NDC 17089-260-18 Oral Solution/Drops 30 mL
16.2. Store at 20-25°C (68-77° F). Excursions permitted between 15°-30°C (59°-86°F).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:09:50.368594 · Updated: 2026-03-14T23:14:49.979928