These Highlights Do Not Include All The Information Needed To Use Cibinqo Safely And Effectively. See Full Prescribing Information For Cibinqo.
3f6d6fcd-e9f9-42c8-bfa2-bb2cfc9ed258
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1 ) 02/2023 Dosage and Administration ( 2.2 , 2.3 , 2.4 , 2.5 ) 12/2023
Indications and Usage
CIBINQO is indicated for the treatment of adults and pediatric patients 12 years of age and older with refractory, moderate-to-severe atopic dermatitis whose disease is not adequately controlled with other systemic drug products, including biologics, or when use of those therapies is inadvisable.
Dosage and Administration
• For recommended testing, evaluations, and procedures prior to CIBINQO initiation, see Full Prescribing Information. ( 2.1 ) • Recommended dosage is 100 mg orally once daily. ( 2.2 ) • 200 mg orally once daily is recommended for those patients who are not responding to 100 mg once daily. ( 2.2 ) • Moderate renal impairment: 50 mg once daily or 100 mg once daily for those patients who are not responding to 50 mg once daily. ( 2.3 ) • CYP2C19 poor metabolizer: 50 mg once daily or 100 mg once daily for those patients who are not responding to 50 mg once daily. ( 2.4 ) • For dosage modifications for certain adverse reactions, see Full Prescribing Information. ( 2.6 )
Warnings and Precautions
Contraindications
CIBINQO is contraindicated in patients taking antiplatelet therapies, except for low-dose aspirin (≤81 mg daily), during the first 3 months of treatment [see Warnings and Precautions (5.6) , Drug Interactions (7.2) , and Clinical Pharmacology (12.2) ].
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: • Serious Infections [see Warnings and Precautions (5.1) ] • Mortality [see Warnings and Precautions (5.2) ] • Malignancy and Lymphoproliferative Disorders [see Warnings and Precautions (5.3) ] • Major Adverse Cardiovascular Events [see Warnings and Precautions (5.4) ] • Thrombosis [see Warnings and Precautions (5.5) ] • Laboratory Abnormalities [see Warnings and Precautions (5.6) ]
Drug Interactions
• Strong inhibitors of CYP2C19: The recommended dosage is 50 mg once daily or 100 mg once daily for those patients who are not responding to 50 mg once daily. ( 2.5 , 7.1 ) • Moderate to strong inhibitors of both CYP2C19 and CYP2C9, or strong CYP2C19 or CYP2C9 inducers: Avoid concomitant use. ( 7.1 ) • P-gp substrate where small concentration changes may lead to serious or life-threatening toxicities: Monitor or titrate dosage of P-gp substrate. ( 7.2 )
Storage and Handling
CIBINQO is supplied as: Dosage Form Strength Description Bottle Size (number of tablets) NDC Number Tablets 50 mg Pink, oval tablet debossed with "PFE" on one side and "ABR 50" on the other. 30 count bottle 0069-0235-30 Tablets 100 mg Pink, round tablet debossed with "PFE" on one side and "ABR 100" on the other. 30 count bottle 0069-0335-30 Tablets 200 mg Pink, oval tablet debossed with "PFE" on one side and "ABR 200" on the other. 30 count bottle 0069-0435-30
How Supplied
CIBINQO is supplied as: Dosage Form Strength Description Bottle Size (number of tablets) NDC Number Tablets 50 mg Pink, oval tablet debossed with "PFE" on one side and "ABR 50" on the other. 30 count bottle 0069-0235-30 Tablets 100 mg Pink, round tablet debossed with "PFE" on one side and "ABR 100" on the other. 30 count bottle 0069-0335-30 Tablets 200 mg Pink, oval tablet debossed with "PFE" on one side and "ABR 200" on the other. 30 count bottle 0069-0435-30
Medication Information
Warnings and Precautions
Indications and Usage
CIBINQO is indicated for the treatment of adults and pediatric patients 12 years of age and older with refractory, moderate-to-severe atopic dermatitis whose disease is not adequately controlled with other systemic drug products, including biologics, or when use of those therapies is inadvisable.
Dosage and Administration
• For recommended testing, evaluations, and procedures prior to CIBINQO initiation, see Full Prescribing Information. ( 2.1 ) • Recommended dosage is 100 mg orally once daily. ( 2.2 ) • 200 mg orally once daily is recommended for those patients who are not responding to 100 mg once daily. ( 2.2 ) • Moderate renal impairment: 50 mg once daily or 100 mg once daily for those patients who are not responding to 50 mg once daily. ( 2.3 ) • CYP2C19 poor metabolizer: 50 mg once daily or 100 mg once daily for those patients who are not responding to 50 mg once daily. ( 2.4 ) • For dosage modifications for certain adverse reactions, see Full Prescribing Information. ( 2.6 )
Contraindications
CIBINQO is contraindicated in patients taking antiplatelet therapies, except for low-dose aspirin (≤81 mg daily), during the first 3 months of treatment [see Warnings and Precautions (5.6) , Drug Interactions (7.2) , and Clinical Pharmacology (12.2) ].
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: • Serious Infections [see Warnings and Precautions (5.1) ] • Mortality [see Warnings and Precautions (5.2) ] • Malignancy and Lymphoproliferative Disorders [see Warnings and Precautions (5.3) ] • Major Adverse Cardiovascular Events [see Warnings and Precautions (5.4) ] • Thrombosis [see Warnings and Precautions (5.5) ] • Laboratory Abnormalities [see Warnings and Precautions (5.6) ]
Drug Interactions
• Strong inhibitors of CYP2C19: The recommended dosage is 50 mg once daily or 100 mg once daily for those patients who are not responding to 50 mg once daily. ( 2.5 , 7.1 ) • Moderate to strong inhibitors of both CYP2C19 and CYP2C9, or strong CYP2C19 or CYP2C9 inducers: Avoid concomitant use. ( 7.1 ) • P-gp substrate where small concentration changes may lead to serious or life-threatening toxicities: Monitor or titrate dosage of P-gp substrate. ( 7.2 )
Storage and Handling
CIBINQO is supplied as: Dosage Form Strength Description Bottle Size (number of tablets) NDC Number Tablets 50 mg Pink, oval tablet debossed with "PFE" on one side and "ABR 50" on the other. 30 count bottle 0069-0235-30 Tablets 100 mg Pink, round tablet debossed with "PFE" on one side and "ABR 100" on the other. 30 count bottle 0069-0335-30 Tablets 200 mg Pink, oval tablet debossed with "PFE" on one side and "ABR 200" on the other. 30 count bottle 0069-0435-30
How Supplied
CIBINQO is supplied as: Dosage Form Strength Description Bottle Size (number of tablets) NDC Number Tablets 50 mg Pink, oval tablet debossed with "PFE" on one side and "ABR 50" on the other. 30 count bottle 0069-0235-30 Tablets 100 mg Pink, round tablet debossed with "PFE" on one side and "ABR 100" on the other. 30 count bottle 0069-0335-30 Tablets 200 mg Pink, oval tablet debossed with "PFE" on one side and "ABR 200" on the other. 30 count bottle 0069-0435-30
Description
Indications and Usage ( 1 ) 02/2023 Dosage and Administration ( 2.2 , 2.3 , 2.4 , 2.5 ) 12/2023
Section 42229-5
Serious Infections
Patients treated with CIBINQO may be at increased risk for developing serious infections that may lead to hospitalization or death. The most frequent serious infections reported with CIBINQO were herpes simplex, herpes zoster, and pneumonia [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)].
If a serious or opportunistic infection develops, discontinue CIBINQO and control the infection.
Reported infections from Janus kinase (JAK) inhibitors used to treat inflammatory conditions:
-
•Active tuberculosis, which may present with pulmonary or extrapulmonary disease. Test for latent TB before and during therapy; treat latent TB prior to use. Monitor all patients for active TB during treatment, even patients with initial negative latent TB test.
-
•Invasive fungal infections, including cryptococcosis and pneumocystosis. Patients with invasive fungal infections may present with disseminated, rather than localized, disease.
-
•Bacterial, viral, including herpes zoster, and other infections due to opportunistic pathogens.
Avoid use of CIBINQO in patients with an active, serious infection including localized infections. The risks and benefits of treatment with CIBINQO should be carefully considered prior to initiating therapy in patients with chronic or recurrent infections.
Patients should be closely monitored for the development of signs and symptoms of infection during and after treatment with CIBINQO, including the possible development of tuberculosis in patients who tested negative for latent tuberculosis infection prior to initiating therapy [see Warnings and Precautions (5.1)].
Section 42231-1
|
Medication Guide
|
|||||
|
What is the most important information I should know about CIBINQO?
You should not start taking CIBINQO if you have any kind of infection unless your healthcare provider tells you it is okay.
|
|||||
|
|
|
|||
|
After starting CIBINQO, call your healthcare provider right away if you have any symptoms of an infection. CIBINQO can make you more likely to get infections or make any infections that you have worse. If you get a serious infection, your healthcare provider may stop treatment with CIBINQO until your infection is controlled.
3. Cancer and immune system problems
Tell your healthcare provider if you have ever had any type of cancer.
5. Blood clots
6. Changes in certain laboratory test results
You should not take CIBINQO if your lymphocyte counts, neutrophil counts, red blood cell counts, or platelet counts are too low. Your healthcare provider may stop your CIBINQO treatment for a period of time if needed because of changes in these blood test results.
Increased cholesterol levels. You may also have increases in the amount of fat found in your blood. Your healthcare provider should check your cholesterol about 4 weeks after you start CIBINQO, and then as needed. See "What are the possible side effects of CIBINQO?" for more information about side effects. |
|||||
|
What is CIBINQO?
It is not known if CIBINQO is safe and effective in children under 12 years of age. |
|||||
|
Who should not take CIBINQO? Do not take CIBINQO if you take medicines that prevent blood clots (antiplatelet medicines), except for low-dose aspirin up to a dose of 81 mg each day during the first 3 months of CIBINQO treatment. |
|||||
|
Before taking CIBINQO, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. CIBINQO and other medicines may affect each other causing side effects. |
|||||
|
How should I take CIBINQO?
|
|||||
|
What are the possible side effects of CIBINQO?
See "What is the most important information I should know about CIBINQO?" The most common side effects of CIBINQO include: |
|||||
|
|
||||
|
Separation or tear to the lining of the back part of the eye (retinal detachment) has happened in people with atopic dermatitis treated with CIBINQO. Call your healthcare provider right away if you have any sudden changes in your vision during treatment with CIBINQO.
|
|||||
|
How should I store CIBINQO?
Keep CIBINQO and all medicines out of the reach of children. |
|||||
|
General information about the safe and effective use of CIBINQO.
You can ask your pharmacist or healthcare provider for information about CIBINQO that is written for health professionals. |
|||||
|
What are the ingredients in CIBINQO?
LAB-1424-3.0 |
This Medication Guide has been approved by the U.S. Food and Drug Administration. Approved: 12/2023
Section 43683-2
Section 44425-7
Store CIBINQO at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C to 30°C (59°F to 86°F). Keep in original package. The container closure system is child resistant.
Limitations of Use
CIBINQO is not recommended for use in combination with other JAK inhibitors, biologic immunomodulators, or other immunosuppressants.
10 Overdosage
There is no experience regarding human overdosage with CIBINQO. There is no specific antidote for overdose with CIBINQO. In case of an overdose, call Poison Control Center at 1-800-222-1222 for latest recommendations.
5.2 Mortality
In a large, randomized, postmarketing safety trial of another JAK inhibitor in rheumatoid arthritis (RA) patients 50 years of age and older with at least one cardiovascular risk factor, a higher rate of all-cause mortality, including sudden cardiovascular death, was observed in subjects treated with the JAK inhibitor compared with TNF blockers. CIBINQO is not approved for use in RA.
Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with CIBINQO.
8.1 Pregnancy
11 Description
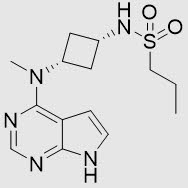
CIBINQO (abrocitinib) tablets contain the free base of abrocitinib, a Janus kinase (JAK) inhibitor, for oral administration.
Abrocitinib is a white to pale colored powder with the following chemical name: N-((1s,3s)-3-(methyl(7H-pyrrolo[2,3-d]pyrimidin-4-yl)amino)cyclobutyl)propane-1-sulfonamide
The solubility of abrocitinib in water is 0.04 mg/mL at 25ºC.
Abrocitinib has a molecular weight of 323.42 g/mol and a molecular formula of C14H21N5O2S. The structural formula of abrocitinib is:
Each film-coated tablet contains 50 mg or 100 mg or 200 mg of abrocitinib and the following inactive ingredients: dibasic calcium phosphate anhydrous, hypromellose, iron oxide red, lactose monohydrate, Macrogol, magnesium stearate, microcrystalline cellulose, sodium starch glycolate, titanium dioxide, and triacetin.
5.5 Thrombosis
Deep venous thrombosis (DVT) and pulmonary embolism (PE) were observed in subjects receiving CIBINQO in the clinical trials for atopic dermatitis [see Adverse Reactions (6.1)].
Thrombosis, including DVT, PE, and arterial thrombosis have been reported in patients receiving JAK inhibitors used to treat inflammatory conditions. Many of these adverse reactions were serious and some resulted in death.
In a large, randomized, postmarketing safety trial of another JAK inhibitor in RA subjects 50 years of age and older with at least one cardiovascular risk factor, higher rates of overall thrombosis, DVT, and PE were observed compared to those treated with TNF blockers. CIBINQO is not approved for use in RA.
Avoid CIBINQO in patients that may be at increased risk of thrombosis. If symptoms of thrombosis occur, discontinue CIBINQO and evaluate and treat patients appropriately.
5.7 Immunizations
Prior to initiating CIBINQO, complete all age-appropriate vaccinations as recommended by current immunization guidelines, including prophylactic herpes zoster vaccinations. Avoid vaccination with live vaccines immediately prior to, during and immediately after CIBINQO therapy.
8.4 Pediatric Use
The safety and effectiveness of CIBINQO in pediatric patients 12 years of age and older with atopic dermatitis have been established.
In trials Trial-AD-1 and Trial-AD-2, 124 pediatric subjects 12 to less than 18 years old weighing 25 kg or more with moderate-to-severe atopic dermatitis were enrolled and randomized to receive either CIBINQO 100 mg (N=51), 200 mg (N=48), or matching placebo (N=25) in monotherapy. Additional 284 pediatric subjects 12 to less than 18 years of age weighing 25 kg or more with moderate-to-severe atopic dermatitis, were enrolled and randomized to receive either CIBINQO 100 mg (N=95) or 200 mg (N=94) or matching placebo (N=95) in combination with topical corticosteroids in Trial-AD-4. Efficacy and adverse reaction profile were comparable between the pediatric patients and adults [see Clinical Studies (14) and Adverse Reactions (6.1)].
The safety and effectiveness of CIBINQO have not been established in pediatric patients below 12 years of age.
8.5 Geriatric Use
A total of 145 (4.6%) subjects 65 years of age and older, while 25 (0.8%) were 75 years of age and older, were enrolled in CIBINQO clinical trials. Clinical trials of CIBINQO did not include sufficient numbers of subjects 65 years of age and older to determine whether they respond differently from younger adult subjects.
A higher proportion of subjects 65 years of age and older discontinued from clinical trials compared to younger subjects. Among all subjects exposed to CIBINQO, including the long-term extension trial, confirmed ALC <500/mm3 occurred only in subjects 65 years of age and older. A higher proportion of subjects 65 years of age and older had platelet counts <75,000/mm3. The incidence rate of herpes zoster in subjects 65 years of age and older treated with CIBINQO (7.40 per 100 patient-years) was higher than that of subjects 18 to less than 65 years of age (3.44 per 100 patient-years).
14 Clinical Studies
The efficacy of CIBINQO as monotherapy and in combination with background topical corticosteroids was evaluated in 4 randomized, double-blind, placebo-controlled trials [Trial-AD-1 (NCT03349060), Trial-AD-2 (NCT03575871), Trial-AD-3 (NCT03720470), and Trial-AD-4 (NCT03796676)] in 1900 subjects (see Table 8). Trial-AD-1 and Trial-AD-2 enrolled adult and pediatric subjects 12 years of age and older. Trial-AD-3 enrolled only adults (≥18 years of age) and Trial-AD-4 enrolled only pediatric subjects 12 to less than 18 years of age. The trials enrolled subjects with moderate-to-severe atopic dermatitis as defined by Investigator’s Global Assessment (IGA) score ≥3, Eczema Area and Severity Index (EASI) score ≥16, body surface area (BSA) involvement ≥10%, and Peak Pruritus Numerical Rating Scale (PP-NRS) ≥4 at the baseline visit prior to randomization.
Baseline Characteristics
In Trial-AD-1, Trial-AD-2, and Trial-AD-3, 53% of subjects were male, 69% of subjects were white, 64% of subjects had a baseline IGA score of 3 (moderate AD), and 36% of subjects had a baseline IGA score of 4 (severe AD). The baseline mean EASI score was 30. The baseline mean age was 36 years old with 8% of subjects 12 to less than 18 years old and 92% of subjects 18 years of age or older. Subjects in these trials were those who had inadequate response to previous topical therapy or were subjects for whom topical treatments were medically inadvisable or who had received systemic therapies including dupilumab. In each of the trials, over 40% of subjects had prior exposure to systemic therapy. In Trial-AD-1 and Trial-AD-2, 6% of the subjects had received dupilumab, whereas prior use of dupilumab was not allowed in Trial-AD-3.
In Trial-AD-4, 49% of subjects were female, 56% of subjects were White, 33% of subjects were Asian and 6% of subjects were Black. The median age was 15 years and the proportion of subjects with severe atopic dermatitis (IGA of 4) was 38%.
Trial Designs and Endpoints
Trial-AD-1, Trial-AD-2, Trial-AD-3, and Trial-AD-4 assessed the co-primary endpoints of IGA and EASI-75 responses at Week 12. The designs of the trials are summarized in Table 8.
| Abbreviations: EASI=Eczema Area and Severity Index; IGA=Investigator’s Global Assessment; QD=once daily; Q2W=once every 2 weeks. | |||
|
Study Name
|
Population
|
Treatment Arms |
Co-Primary Endpoints |
|
Trial-AD-1 12 weeks |
Subjects 12 years of age or older (387) |
Oral administration of:
|
|
|
Trial-AD-2 12 weeks |
Subjects 12 years of age or older (391) |
Oral administration of:
|
|
|
Trial-AD-3 16 weeks |
Subjects 18 years of age or older (837) |
Oral administration of:
Subcutaneous administration of:
All subjects received background topical corticosteroids |
|
|
Trial-AD-4 (combination therapy) 12 weeks |
Subjects 12 to less than 18 years of age (285) |
Oral administration of:
All subjects received background topical corticosteroids |
4 Contraindications
CIBINQO is contraindicated in patients taking antiplatelet therapies, except for low-dose aspirin (≤81 mg daily), during the first 3 months of treatment [see Warnings and Precautions (5.6), Drug Interactions (7.2), and Clinical Pharmacology (12.2)].
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
-
•Serious Infections [see Warnings and Precautions (5.1)]
-
•Mortality [see Warnings and Precautions (5.2)]
-
•Malignancy and Lymphoproliferative Disorders [see Warnings and Precautions (5.3)]
-
•Major Adverse Cardiovascular Events [see Warnings and Precautions (5.4)]
-
•Thrombosis [see Warnings and Precautions (5.5)]
-
•Laboratory Abnormalities [see Warnings and Precautions (5.6)]
7 Drug Interactions
-
•Strong inhibitors of CYP2C19: The recommended dosage is 50 mg once daily or 100 mg once daily for those patients who are not responding to 50 mg once daily. (2.5, 7.1)
-
•Moderate to strong inhibitors of both CYP2C19 and CYP2C9, or strong CYP2C19 or CYP2C9 inducers: Avoid concomitant use. (7.1)
-
•P-gp substrate where small concentration changes may lead to serious or life-threatening toxicities: Monitor or titrate dosage of P-gp substrate. (7.2)
8.6 Renal Impairment
In patients with severe (eGFR <30 mL/min) and moderate (eGFR 30–59 mL/min) renal impairment, the combined exposure (AUCinf,u) of abrocitinib and its two active metabolites, M1 and M2, is increased compared to patients with normal renal function (eGFR ≥90 mL/min) [see Clinical Pharmacology (12.3)]. This may increase the risk of adverse reactions such as infections.
CIBINQO is not recommended for use in patients with severe renal impairment and ESRD including those on renal replacement therapy [see Dosage and Administration (2.3)].
A dosage reduction in patients with moderate renal impairment is recommended. No dosage adjustment is required in patients with mild renal impairment (eGFR 60–89 mL/min) [see Dosage and Administration (2.3)].
CIBINQO has not been studied in subjects on renal replacement therapy. In Phase 3 clinical trials, CIBINQO was not evaluated in subjects with atopic dermatitis with baseline creatinine clearance values less than 40 mL/min.
12.2 Pharmacodynamics
Treatment with CIBINQO was associated with dose-dependent reduction in serum markers of inflammation, including high sensitivity C-reactive protein (hsCRP), interleukin-31 (IL-31) and thymus and activation regulated chemokine (TARC). These changes returned to near baseline within 4 weeks of drug discontinuation.
12.3 Pharmacokinetics
Abrocitinib plasma Cmax and AUC increased dose proportionally up to 200 mg. Steady-state plasma concentrations of abrocitinib are achieved within 48 hours after once daily administration.
12.5 Pharmacogenomics
Patients who are CYP2C19 poor metabolizers have little to no CYP2C19 enzyme function compared to CYP2C19 normal metabolizers that have fully functional CYP2C19 enzymes.
After single doses of abrocitinib, CYP2C19 poor metabolizers demonstrated dose-normalized AUC of abrocitinib values that were 2.3-fold higher when compared to CYP2C19 normal metabolizers. Approximately 3–5% of Whites and Blacks and 15 to 20% of Asians are CYP2C19 poor metabolizers [see Dosage and Administration (2.4) and Use in Specific Populations (8.8)].
2.2 Recommended Dosage
The recommended dose is 100 mg once daily. If an adequate response is not achieved with CIBINQO 100 mg once daily, consider increasing the dosage to 200 mg once daily.
Discontinue CIBINQO if an adequate response is not achieved with 200 mg once daily.
Use the lowest efficacious dose to maintain response.
CIBINQO can be used with or without topical corticosteroids.
If a dose is missed, administer the dose as soon as possible unless it is less than 12 hours before the next dose, in which case skip the missed dose. Thereafter, resume dosing at the regular scheduled time.
5.1 Serious Infections
The most frequent serious infections reported in clinical studies with CIBINQO for atopic dermatitis were herpes simplex, herpes zoster, and pneumonia [see Adverse Reactions (6.1)]. Serious infections leading to hospitalization or death, including tuberculosis and bacterial, invasive fungal, viral, and other opportunistic infections, have occurred in patients receiving JAK inhibitors used to treat inflammatory conditions.
Avoid use of CIBINQO in patients with active, serious infection including localized infections.
Consider the risks and benefits of treatment prior to initiating CIBINQO in patients:
-
•with chronic or recurrent infection
-
•who have been exposed to tuberculosis
-
•with a history of a serious or an opportunistic infection
-
•who have resided or traveled in areas of endemic tuberculosis or endemic mycoses
-
•with underlying conditions that may predispose them to infection
Closely monitor patients for the development of signs and symptoms of infection during and after treatment with CIBINQO. If a patient develops a serious or opportunistic infection, discontinue CIBINQO. Initiate complete diagnostic testing and appropriate antimicrobial therapy. The risks and benefits of treatment with CIBINQO should be carefully considered prior to reinitiating therapy with CIBINQO.
8.7 Hepatic Impairment
Avoid use of CIBINQO in patients with severe (Child Pugh C) hepatic impairment. In clinical trials, CIBINQO was not evaluated in subjects with severe (Child Pugh C) hepatic impairment.
Dosage adjustment is not required in patients with mild (Child Pugh A) or moderate (Child Pugh B) hepatic impairment based on similar combined exposure (AUCinf,u) of abrocitinib and its two active metabolites, M1 and M2 compared to patients with normal hepatic function [see Clinical Pharmacology (12.3)].
1 Indications and Usage
CIBINQO is indicated for the treatment of adults and pediatric patients 12 years of age and older with refractory, moderate-to-severe atopic dermatitis whose disease is not adequately controlled with other systemic drug products, including biologics, or when use of those therapies is inadvisable.
12 Clinical Pharmacology
12.1 Mechanism of Action
CIBINQO is a Janus kinase (JAK) inhibitor.
Abrocitinib reversibly inhibits JAK1 by blocking the adenosine triphosphate (ATP) binding site. In a cell-free isolated enzyme assay, abrocitinib was selective for JAK1 over JAK2 (28-fold), JAK3 (>340-fold), and tyrosine kinase (TYK) 2 (43-fold), as well as the broader kinome. The relevance of inhibition of specific JAK enzymes to therapeutic effectiveness is not currently known. Both the parent compound and the active metabolites inhibit JAK1 activity in vitro with similar levels of selectivity.
13 Nonclinical Toxicology
5 Warnings and Precautions
2 Dosage and Administration
-
•For recommended testing, evaluations, and procedures prior to CIBINQO initiation, see Full Prescribing Information. (2.1)
-
•Recommended dosage is 100 mg orally once daily. (2.2)
-
•200 mg orally once daily is recommended for those patients who are not responding to 100 mg once daily. (2.2)
-
•Moderate renal impairment: 50 mg once daily or 100 mg once daily for those patients who are not responding to 50 mg once daily. (2.3)
-
•CYP2C19 poor metabolizer: 50 mg once daily or 100 mg once daily for those patients who are not responding to 50 mg once daily. (2.4)
-
•For dosage modifications for certain adverse reactions, see Full Prescribing Information. (2.6)
3 Dosage Forms and Strengths
-
•50 mg: Pink, oval, film-coated tablet debossed with "PFE" on one side and "ABR 50" on the other.
-
•100 mg: Pink, round, film-coated tablet debossed with "PFE" on one side and "ABR 100" on the other.
-
•200 mg: Pink, oval, film-coated tablet debossed with "PFE" on one side and "ABR 200" on the other.
8 Use in Specific Populations
8.8 Cyp2c19 Poor Metabolizers
In patients who are CYP2C19 poor metabolizers, the AUC of abrocitinib is increased compared to CYP2C19 normal metabolizers due to reduced metabolic clearance. Dosage reduction of CIBINQO is recommended in patients who are known or suspected to be CYP2C19 poor metabolizers based on genotype or previous history/experience with other CYP2C19 substrates [see Dosage and Administration (2.4) and Clinical Pharmacology (12.5)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of CIBINQO was evaluated in four randomized, placebo-controlled clinical trials (2 monotherapy, 1 combination therapy with topical corticosteroid, and 1 dose-ranging) and one long-term extension trial in subjects with moderate to severe atopic dermatitis (AD). A total of 1623 subjects with moderate to severe atopic dermatitis were treated with CIBINQO in these clinical trials representing 1428 patient-years of exposure. There were 634 subjects with at least 1 year of exposure to CIBINQO.
In the placebo-controlled clinical trials, a total of 1198 subjects were exposed to CIBINQO with 608 subjects receiving CIBINQO 100 mg once daily and 590 subjects receiving CIBINQO 200 mg once daily for up to 16 weeks. The median age of subjects was 33.0 years, 124 subjects (8.1%) were 12 to less than 18 years old and 94 subjects (6.1%) were 65 years of age or older. The majority of subjects were White (68.7%) and male (53.9%).
Adverse reactions occurring at ≥1% in any of the treated groups and at a higher rate than in the placebo group are presented in Table 3. A total of 61 (5.1%) subjects treated with CIBINQO were discontinued from the trials due to adverse reactions. The safety profile of CIBINQO in the monotherapy and the combination trial(s) were similar.
|
Weeks 0–16 |
|||
|
CIBINQO
Study size adjusted percentages
)
|
CIBINQO
|
Placebo
|
|
|
Nasopharyngitis |
51 (8.7) |
75 (12.4) |
27 (7.9) |
|
Nausea |
86 (14.5) |
37 (6.0) |
7 (2.1) |
|
Headache |
46 (7.8) |
36 (6.0) |
12 (3.5) |
|
Herpes simplex Herpes simplex also includes oral herpes, ophthalmic herpes, herpes dermatitis, genital herpes.
|
25 (4.2) |
20 (3.3) |
6 (1.8) |
|
Increased blood creatine phosphokinase |
17 (2.9) |
14 (2.3) |
5 (1.5) |
|
Dizziness |
17 (2.9) |
11 (1.8) |
3 (0.9) |
|
Urinary tract infection |
13 (2.2) |
10 (1.7) |
4 (1.2) |
|
Fatigue |
8 (1.3) |
10 (1.6) |
2 (0.5) |
|
Acne |
28 (4.7) |
10 (1.6) |
0 (0.0) |
|
Vomiting |
19 (3.2) |
9 (1.5) |
3 (0.9) |
|
Impetigo |
3 (0.5) |
9 (1.5) |
1 (0.3) |
|
Oropharyngeal pain |
6 (1.0) |
8 (1.4) |
2 (0.6) |
|
Hypertension |
5 (0.8) |
7 (1.2) |
2 (0.7) |
|
Influenza |
6 (1.1) |
7 (1.2) |
0 (0.0) |
|
Gastroenteritis |
8 (1.3) |
7 (1.1) |
2 (0.6) |
|
Dermatitis contact |
3 (0.5) |
6 (1.1) |
1 (0.3) |
|
Abdominal pain upper |
11 (1.9) |
4 (0.6) |
0 (0.0) |
|
Abdominal discomfort |
7 (1.2) |
3 (0.5) |
1 (0.3) |
|
Herpes zoster |
7 (1.2) |
2 (0.3) |
0 (0.0) |
|
Thrombocytopenia |
9 (1.5) |
0 (0.0) |
0 (0.0) |
2.7 Administration Instructions
Administer CIBINQO with or without food at approximately the same time each day.
Swallow CIBINQO tablets whole with water. Do not crush, split, or chew CIBINQO tablets.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling
CIBINQO is supplied as:
|
Dosage Form |
Strength |
Description |
Bottle Size
|
NDC Number |
|
Tablets |
50 mg |
Pink, oval tablet debossed with "PFE" on one side and "ABR 50" on the other. |
30 count bottle |
0069-0235-30 |
|
Tablets |
100 mg |
Pink, round tablet debossed with "PFE" on one side and "ABR 100" on the other. |
30 count bottle |
0069-0335-30 |
|
Tablets |
200 mg |
Pink, oval tablet debossed with "PFE" on one side and "ABR 200" on the other. |
30 count bottle |
0069-0435-30 |
7.1 Effects of Other Drugs On Cibinqo
Table 4 includes drugs with clinically significant drug interactions affecting CIBINQO.
|
Strong CYP2C19 Inhibitors |
|
|
Clinical Impact |
Coadministration of CIBINQO with strong CYP2C19 inhibitors increases the combined exposure of abrocitinib and its two active metabolites, M1 and M2 which may increase the adverse reactions of CIBINQO [see Clinical Pharmacology (12.3)]. |
|
Intervention |
Dosage reduction of CIBINQO is recommended when coadministered with strong CYP2C19 inhibitors [see Dosage and Administration (2.5)]. |
|
Moderate to Strong Inhibitors of both CYP2C19 and CYP2C9 |
|
|
Clinical Impact |
Coadministration of CIBINQO with drugs that are moderate to strong inhibitors of both CYP2C19 and CYP2C9 increases the exposure of abrocitinib and its two active metabolites, M1 and M2 which may increase the adverse reactions of CIBINQO [Clinical Pharmacology (12.3)]. |
|
Intervention |
Avoid concomitant use of CIBINQO with drugs that are moderate to strong inhibitors of both CYP2C19 and CYP2C9. |
|
Strong CYP2C19 or CYP2C9 Inducers |
|
|
Clinical Impact |
Coadministration of CIBINQO with strong CYP2C19 or CYP2C9 inducers decreases the combined exposure of abrocitinib and its two active metabolites, M1 and M2, which may result in loss of or reduced clinical response [see Clinical Pharmacology (12.3)]. |
|
Intervention |
Avoid concomitant use of CIBINQO with strong CYP2C19 or CYP2C9 inducers. |
7.2 Effects of Cibinqo On Other Drugs
Table 5 includes clinically significant drug interactions affecting other drugs.
|
P-gp Substrate Where Small Concentration Changes May Lead to Serious or Life-threatening Toxicities |
|
|
Clinical Impact |
Coadministration of CIBINQO with P-gp substrate increases plasma concentrations of P-gp substrates and may result in potential adverse reactions of the P-gp substrate where small concentration changes may lead to serious or life-threatening toxicities (e.g., digoxin) [see Clinical Pharmacology (12.3)]. |
|
Intervention |
Monitor appropriately or dose titrate P-gp substrate where small concentration changes may lead to serious or life-threatening toxicities when coadministered with CIBINQO. |
|
Antiplatelet Therapy Drugs |
|
|
Clinical Impact |
Coadministration of CIBINQO with antiplatelet therapy drugs may increase the risk of bleeding with thrombocytopenia [see Warnings and Precautions (5.5) and Clinical Pharmacology (12.2)]. |
|
Intervention |
Antiplatelet drugs, except for low-dose aspirin (≤81 mg daily), during the first 3 months of treatment are contraindicated with CIBINQO [see Contraindications (4)]. |
5.4 Major Adverse Cardiovascular Events
Major adverse cardiovascular events were reported in clinical trials of CIBINQO for atopic dermatitis [see Adverse Reactions (6.1)].
In a large, randomized, postmarketing safety trial of another JAK inhibitor in RA subjects 50 years of age and older with at least one cardiovascular risk factor, a higher rate of major adverse cardiovascular events (MACE) defined as cardiovascular death, non-fatal myocardial infarction (MI), and non-fatal stroke was observed with the JAK inhibitor compared to those treated with TNF blockers. CIBINQO is not approved for use in RA. Patients who are current or past smokers are at additional increased risk.
Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with CIBINQO, particularly in patients who are current or past smokers and patients with other cardiovascular risk factors. Patients should be informed about the symptoms of serious cardiovascular events and the steps to take if they occur. Discontinue CIBINQO in patients that have experienced a myocardial infarction or stroke.
8.3 Females and Males of Reproductive Potential
5.3 Malignancy and Lymphoproliferative Disorders
Malignancies, including non-melanoma skin cancer (NMSC), were observed in clinical trials with CIBINQO for atopic dermatitis [see Adverse Reactions (6.1)].
Perform periodic skin examination for patients who are at increased risk for skin cancer. Exposure to sunlight and UV light should be limited by wearing protective clothing and using broad-spectrum sunscreen.
Malignancies, including lymphomas, have occurred in patients receiving JAK inhibitors used to treat inflammatory conditions. In a large, randomized, postmarketing safety trial of another JAK inhibitor in RA subjects, a higher rate of malignancies (excluding non-melanoma skin cancer (NMSC)) was observed in subjects treated with the JAK inhibitor compared to those treated with TNF blockers. CIBINQO is not approved for use in RA. A higher rate of lymphomas was observed in subjects treated with the JAK inhibitor compared to those treated with TNF blockers. A higher rate of lung cancers was observed in current or past smokers treated with the JAK inhibitor compared to those treated with TNF blockers. In this trial, current or past smokers had an additional increased risk of overall malignancies.
Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with CIBINQO, particularly in patients with a known malignancy (other than a successfully treated NMSC), patients who develop a malignancy when on treatment, and patients who are current or past smokers.
2.5 Dosage Modifications Due to Strong Inhibitors
In patients taking strong inhibitors of cytochrome P450 (CYP) 2C19, reduce the dosage to 50 mg once daily [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)]. If an adequate response is not achieved with CIBINQO 50 mg daily, consider increasing the dosage to 100 mg once daily. Discontinue therapy if inadequate response is seen after dosage increase to 100 mg once daily.
2.4 Recommended Dosage in Cyp2c19 Poor Metabolizers
In patients who are known or suspected to be CYP2C19 poor metabolizers, the recommended dosage of CIBINQO is 50 mg once daily [see Use in Specific Populations (8.8) and Clinical Pharmacology (12.5)]. If an adequate response is not achieved with CIBINQO 50 mg once daily, consider increasing the dosage to 100 mg once daily. Discontinue therapy if inadequate response is seen after dosage increase to 100 mg once daily.
Principal Display Panel 50 Mg Tablet Bottle Label
ALWAYS DISPENSE WITH
MEDICATION GUIDE
Pfizer
NDC 0069-0235-30
CIBINQO®
(abrocitinib) tablets
50 mg
Do not crush, split, or chew the tablets.
30 Tablets
Rx only
Principal Display Panel 100 Mg Tablet Bottle Label
ALWAYS DISPENSE WITH
MEDICATION GUIDE
Pfizer
NDC 0069-0335-30
CIBINQO®
(abrocitinib) tablets
100 mg
Do not crush, split, or chew the tablets.
30 Tablets
Rx only
Principal Display Panel 200 Mg Tablet Bottle Label
ALWAYS DISPENSE WITH
MEDICATION GUIDE
Pfizer
NDC 0069-0435-30
CIBINQO®
(abrocitinib) tablets
200 mg
Do not crush, split, or chew the tablets.
30 Tablets
Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2-year oral carcinogenicity study in rats, abrocitinib increased the incidence of benign thymomas in female rats at doses of 10 and 30 mg/kg/day (2.8 and 14 times the MRHD, respectively, based on AUC comparison). Abrocitinib was not carcinogenic in female rats at 3 mg/kg/day (0.6 times the MRHD based on AUC comparison) or male rats at doses up to 30 mg/kg/day (14 times the MRHD based on AUC comparison). Abrocitinib was not carcinogenic in Tg.rasH2 mice at oral doses up to 60 mg/kg/day in males and 75 mg/kg/day in females.
Abrocitinib was not mutagenic in the bacterial mutagenicity assay (Ames assay). Although abrocitinib was aneugenic in the in vitro TK6 micronucleus assay, abrocitinib was not aneugenic or clastogenic in an in vivo rat bone marrow micronucleus assay.
Abrocitinib did not impair male fertility at doses up to 70 mg/kg/day (26 times the MRHD based on AUC comparison) or female fertility at 10 mg/kg/day (2 times the MRHD based on AUC comparison). Abrocitinib impaired female fertility (reducing fertility index, corpora lutea, and implantation sites) at 70 mg/kg/day (29 times the MRHD based on AUC comparison). Impaired fertility in female rats reversed 1 month after cessation of abrocitinib administration.
2.1 Recommended Testing, Evaluations, and Procedures Prior to Treatment Initiation
Perform the following tests and evaluations prior to CIBINQO initiation:
-
•Tuberculosis (TB) infection evaluation – CIBINQO initiation is not recommended in patients with active TB. For patients with latent TB or those with a negative latent TB test who are at high risk for TB, start preventive therapy for latent TB prior to initiation of CIBINQO [see Warnings and Precautions (5.1)].
-
•Viral hepatitis screening in accordance with clinical guidelines – CIBINQO initiation is not recommended in patients with active hepatitis B or hepatitis C [see Warnings and Precautions (5.1)].
-
•A complete blood count (CBC) – CIBINQO initiation is not recommended in patients with a platelet count <150,000/mm3, an absolute lymphocyte count <500/mm3, an absolute neutrophil count <1,000/mm3, or a hemoglobin value <8 g/dL [see Warnings and Precautions (5.6)].
Complete any necessary immunizations, including herpes zoster vaccinations, in agreement with current immunization guidelines prior to CIBINQO initiation [see Warnings and Precautions (5.7)].
Warning: Serious Infections, Mortality, Malignancy, Major Adverse Cardiovascular Events, and Thrombosis
WARNING: SERIOUS INFECTIONS, MORTALITY, MALIGNANCY, MAJOR ADVERSE CARDIOVASCULAR EVENTS (MACE), and THROMBOSIS
See full prescribing information for complete boxed warning.
-
•Increased risk of serious bacterial, fungal, viral and opportunistic infections leading to hospitalization or death, including tuberculosis (TB). Discontinue treatment with CIBINQO if serious or opportunistic infection occurs. Test for latent TB before and during therapy; treat latent TB prior to use. Monitor all patients for active TB during treatment, even patients with initial negative latent TB test. (5.1)
-
•Higher rate of all-cause mortality, including sudden cardiovascular death, with another JAK inhibitor vs. TNF blockers in rheumatoid arthritis (RA) patients. CIBINQO is not approved for use in RA patients. (5.2)
-
•Malignancies have occurred with CIBINQO. Higher rate of lymphomas and lung cancers with another JAK inhibitor vs. TNF blockers in RA patients. (5.3)
-
•MACE has occurred with CIBINQO. Higher rate of MACE (defined as cardiovascular death, myocardial infarction, and stroke) with another JAK inhibitor vs. TNF blockers in RA patients. (5.4)
-
•Thrombosis has occurred with CIBINQO. Increased incidence of pulmonary embolism, venous and arterial thrombosis with another JAK inhibitor vs. TNF blockers. (5.5)
Structured Label Content
Section 42229-5 (42229-5)
Serious Infections
Patients treated with CIBINQO may be at increased risk for developing serious infections that may lead to hospitalization or death. The most frequent serious infections reported with CIBINQO were herpes simplex, herpes zoster, and pneumonia [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)].
If a serious or opportunistic infection develops, discontinue CIBINQO and control the infection.
Reported infections from Janus kinase (JAK) inhibitors used to treat inflammatory conditions:
-
•Active tuberculosis, which may present with pulmonary or extrapulmonary disease. Test for latent TB before and during therapy; treat latent TB prior to use. Monitor all patients for active TB during treatment, even patients with initial negative latent TB test.
-
•Invasive fungal infections, including cryptococcosis and pneumocystosis. Patients with invasive fungal infections may present with disseminated, rather than localized, disease.
-
•Bacterial, viral, including herpes zoster, and other infections due to opportunistic pathogens.
Avoid use of CIBINQO in patients with an active, serious infection including localized infections. The risks and benefits of treatment with CIBINQO should be carefully considered prior to initiating therapy in patients with chronic or recurrent infections.
Patients should be closely monitored for the development of signs and symptoms of infection during and after treatment with CIBINQO, including the possible development of tuberculosis in patients who tested negative for latent tuberculosis infection prior to initiating therapy [see Warnings and Precautions (5.1)].
Section 42231-1 (42231-1)
|
Medication Guide
|
|||||
|
What is the most important information I should know about CIBINQO?
You should not start taking CIBINQO if you have any kind of infection unless your healthcare provider tells you it is okay.
|
|||||
|
|
|
|||
|
After starting CIBINQO, call your healthcare provider right away if you have any symptoms of an infection. CIBINQO can make you more likely to get infections or make any infections that you have worse. If you get a serious infection, your healthcare provider may stop treatment with CIBINQO until your infection is controlled.
3. Cancer and immune system problems
Tell your healthcare provider if you have ever had any type of cancer.
5. Blood clots
6. Changes in certain laboratory test results
You should not take CIBINQO if your lymphocyte counts, neutrophil counts, red blood cell counts, or platelet counts are too low. Your healthcare provider may stop your CIBINQO treatment for a period of time if needed because of changes in these blood test results.
Increased cholesterol levels. You may also have increases in the amount of fat found in your blood. Your healthcare provider should check your cholesterol about 4 weeks after you start CIBINQO, and then as needed. See "What are the possible side effects of CIBINQO?" for more information about side effects. |
|||||
|
What is CIBINQO?
It is not known if CIBINQO is safe and effective in children under 12 years of age. |
|||||
|
Who should not take CIBINQO? Do not take CIBINQO if you take medicines that prevent blood clots (antiplatelet medicines), except for low-dose aspirin up to a dose of 81 mg each day during the first 3 months of CIBINQO treatment. |
|||||
|
Before taking CIBINQO, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. CIBINQO and other medicines may affect each other causing side effects. |
|||||
|
How should I take CIBINQO?
|
|||||
|
What are the possible side effects of CIBINQO?
See "What is the most important information I should know about CIBINQO?" The most common side effects of CIBINQO include: |
|||||
|
|
||||
|
Separation or tear to the lining of the back part of the eye (retinal detachment) has happened in people with atopic dermatitis treated with CIBINQO. Call your healthcare provider right away if you have any sudden changes in your vision during treatment with CIBINQO.
|
|||||
|
How should I store CIBINQO?
Keep CIBINQO and all medicines out of the reach of children. |
|||||
|
General information about the safe and effective use of CIBINQO.
You can ask your pharmacist or healthcare provider for information about CIBINQO that is written for health professionals. |
|||||
|
What are the ingredients in CIBINQO?
LAB-1424-3.0 |
This Medication Guide has been approved by the U.S. Food and Drug Administration. Approved: 12/2023
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store CIBINQO at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C to 30°C (59°F to 86°F). Keep in original package. The container closure system is child resistant.
Limitations of Use
CIBINQO is not recommended for use in combination with other JAK inhibitors, biologic immunomodulators, or other immunosuppressants.
10 Overdosage (10 OVERDOSAGE)
There is no experience regarding human overdosage with CIBINQO. There is no specific antidote for overdose with CIBINQO. In case of an overdose, call Poison Control Center at 1-800-222-1222 for latest recommendations.
5.2 Mortality
In a large, randomized, postmarketing safety trial of another JAK inhibitor in rheumatoid arthritis (RA) patients 50 years of age and older with at least one cardiovascular risk factor, a higher rate of all-cause mortality, including sudden cardiovascular death, was observed in subjects treated with the JAK inhibitor compared with TNF blockers. CIBINQO is not approved for use in RA.
Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with CIBINQO.
8.1 Pregnancy
11 Description (11 DESCRIPTION)
CIBINQO (abrocitinib) tablets contain the free base of abrocitinib, a Janus kinase (JAK) inhibitor, for oral administration.
Abrocitinib is a white to pale colored powder with the following chemical name: N-((1s,3s)-3-(methyl(7H-pyrrolo[2,3-d]pyrimidin-4-yl)amino)cyclobutyl)propane-1-sulfonamide
The solubility of abrocitinib in water is 0.04 mg/mL at 25ºC.
Abrocitinib has a molecular weight of 323.42 g/mol and a molecular formula of C14H21N5O2S. The structural formula of abrocitinib is:
Each film-coated tablet contains 50 mg or 100 mg or 200 mg of abrocitinib and the following inactive ingredients: dibasic calcium phosphate anhydrous, hypromellose, iron oxide red, lactose monohydrate, Macrogol, magnesium stearate, microcrystalline cellulose, sodium starch glycolate, titanium dioxide, and triacetin.
5.5 Thrombosis
Deep venous thrombosis (DVT) and pulmonary embolism (PE) were observed in subjects receiving CIBINQO in the clinical trials for atopic dermatitis [see Adverse Reactions (6.1)].
Thrombosis, including DVT, PE, and arterial thrombosis have been reported in patients receiving JAK inhibitors used to treat inflammatory conditions. Many of these adverse reactions were serious and some resulted in death.
In a large, randomized, postmarketing safety trial of another JAK inhibitor in RA subjects 50 years of age and older with at least one cardiovascular risk factor, higher rates of overall thrombosis, DVT, and PE were observed compared to those treated with TNF blockers. CIBINQO is not approved for use in RA.
Avoid CIBINQO in patients that may be at increased risk of thrombosis. If symptoms of thrombosis occur, discontinue CIBINQO and evaluate and treat patients appropriately.
5.7 Immunizations
Prior to initiating CIBINQO, complete all age-appropriate vaccinations as recommended by current immunization guidelines, including prophylactic herpes zoster vaccinations. Avoid vaccination with live vaccines immediately prior to, during and immediately after CIBINQO therapy.
8.4 Pediatric Use
The safety and effectiveness of CIBINQO in pediatric patients 12 years of age and older with atopic dermatitis have been established.
In trials Trial-AD-1 and Trial-AD-2, 124 pediatric subjects 12 to less than 18 years old weighing 25 kg or more with moderate-to-severe atopic dermatitis were enrolled and randomized to receive either CIBINQO 100 mg (N=51), 200 mg (N=48), or matching placebo (N=25) in monotherapy. Additional 284 pediatric subjects 12 to less than 18 years of age weighing 25 kg or more with moderate-to-severe atopic dermatitis, were enrolled and randomized to receive either CIBINQO 100 mg (N=95) or 200 mg (N=94) or matching placebo (N=95) in combination with topical corticosteroids in Trial-AD-4. Efficacy and adverse reaction profile were comparable between the pediatric patients and adults [see Clinical Studies (14) and Adverse Reactions (6.1)].
The safety and effectiveness of CIBINQO have not been established in pediatric patients below 12 years of age.
8.5 Geriatric Use
A total of 145 (4.6%) subjects 65 years of age and older, while 25 (0.8%) were 75 years of age and older, were enrolled in CIBINQO clinical trials. Clinical trials of CIBINQO did not include sufficient numbers of subjects 65 years of age and older to determine whether they respond differently from younger adult subjects.
A higher proportion of subjects 65 years of age and older discontinued from clinical trials compared to younger subjects. Among all subjects exposed to CIBINQO, including the long-term extension trial, confirmed ALC <500/mm3 occurred only in subjects 65 years of age and older. A higher proportion of subjects 65 years of age and older had platelet counts <75,000/mm3. The incidence rate of herpes zoster in subjects 65 years of age and older treated with CIBINQO (7.40 per 100 patient-years) was higher than that of subjects 18 to less than 65 years of age (3.44 per 100 patient-years).
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of CIBINQO as monotherapy and in combination with background topical corticosteroids was evaluated in 4 randomized, double-blind, placebo-controlled trials [Trial-AD-1 (NCT03349060), Trial-AD-2 (NCT03575871), Trial-AD-3 (NCT03720470), and Trial-AD-4 (NCT03796676)] in 1900 subjects (see Table 8). Trial-AD-1 and Trial-AD-2 enrolled adult and pediatric subjects 12 years of age and older. Trial-AD-3 enrolled only adults (≥18 years of age) and Trial-AD-4 enrolled only pediatric subjects 12 to less than 18 years of age. The trials enrolled subjects with moderate-to-severe atopic dermatitis as defined by Investigator’s Global Assessment (IGA) score ≥3, Eczema Area and Severity Index (EASI) score ≥16, body surface area (BSA) involvement ≥10%, and Peak Pruritus Numerical Rating Scale (PP-NRS) ≥4 at the baseline visit prior to randomization.
Baseline Characteristics
In Trial-AD-1, Trial-AD-2, and Trial-AD-3, 53% of subjects were male, 69% of subjects were white, 64% of subjects had a baseline IGA score of 3 (moderate AD), and 36% of subjects had a baseline IGA score of 4 (severe AD). The baseline mean EASI score was 30. The baseline mean age was 36 years old with 8% of subjects 12 to less than 18 years old and 92% of subjects 18 years of age or older. Subjects in these trials were those who had inadequate response to previous topical therapy or were subjects for whom topical treatments were medically inadvisable or who had received systemic therapies including dupilumab. In each of the trials, over 40% of subjects had prior exposure to systemic therapy. In Trial-AD-1 and Trial-AD-2, 6% of the subjects had received dupilumab, whereas prior use of dupilumab was not allowed in Trial-AD-3.
In Trial-AD-4, 49% of subjects were female, 56% of subjects were White, 33% of subjects were Asian and 6% of subjects were Black. The median age was 15 years and the proportion of subjects with severe atopic dermatitis (IGA of 4) was 38%.
Trial Designs and Endpoints
Trial-AD-1, Trial-AD-2, Trial-AD-3, and Trial-AD-4 assessed the co-primary endpoints of IGA and EASI-75 responses at Week 12. The designs of the trials are summarized in Table 8.
| Abbreviations: EASI=Eczema Area and Severity Index; IGA=Investigator’s Global Assessment; QD=once daily; Q2W=once every 2 weeks. | |||
|
Study Name
|
Population
|
Treatment Arms |
Co-Primary Endpoints |
|
Trial-AD-1 12 weeks |
Subjects 12 years of age or older (387) |
Oral administration of:
|
|
|
Trial-AD-2 12 weeks |
Subjects 12 years of age or older (391) |
Oral administration of:
|
|
|
Trial-AD-3 16 weeks |
Subjects 18 years of age or older (837) |
Oral administration of:
Subcutaneous administration of:
All subjects received background topical corticosteroids |
|
|
Trial-AD-4 (combination therapy) 12 weeks |
Subjects 12 to less than 18 years of age (285) |
Oral administration of:
All subjects received background topical corticosteroids |
4 Contraindications (4 CONTRAINDICATIONS)
CIBINQO is contraindicated in patients taking antiplatelet therapies, except for low-dose aspirin (≤81 mg daily), during the first 3 months of treatment [see Warnings and Precautions (5.6), Drug Interactions (7.2), and Clinical Pharmacology (12.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
-
•Serious Infections [see Warnings and Precautions (5.1)]
-
•Mortality [see Warnings and Precautions (5.2)]
-
•Malignancy and Lymphoproliferative Disorders [see Warnings and Precautions (5.3)]
-
•Major Adverse Cardiovascular Events [see Warnings and Precautions (5.4)]
-
•Thrombosis [see Warnings and Precautions (5.5)]
-
•Laboratory Abnormalities [see Warnings and Precautions (5.6)]
7 Drug Interactions (7 DRUG INTERACTIONS)
-
•Strong inhibitors of CYP2C19: The recommended dosage is 50 mg once daily or 100 mg once daily for those patients who are not responding to 50 mg once daily. (2.5, 7.1)
-
•Moderate to strong inhibitors of both CYP2C19 and CYP2C9, or strong CYP2C19 or CYP2C9 inducers: Avoid concomitant use. (7.1)
-
•P-gp substrate where small concentration changes may lead to serious or life-threatening toxicities: Monitor or titrate dosage of P-gp substrate. (7.2)
8.6 Renal Impairment
In patients with severe (eGFR <30 mL/min) and moderate (eGFR 30–59 mL/min) renal impairment, the combined exposure (AUCinf,u) of abrocitinib and its two active metabolites, M1 and M2, is increased compared to patients with normal renal function (eGFR ≥90 mL/min) [see Clinical Pharmacology (12.3)]. This may increase the risk of adverse reactions such as infections.
CIBINQO is not recommended for use in patients with severe renal impairment and ESRD including those on renal replacement therapy [see Dosage and Administration (2.3)].
A dosage reduction in patients with moderate renal impairment is recommended. No dosage adjustment is required in patients with mild renal impairment (eGFR 60–89 mL/min) [see Dosage and Administration (2.3)].
CIBINQO has not been studied in subjects on renal replacement therapy. In Phase 3 clinical trials, CIBINQO was not evaluated in subjects with atopic dermatitis with baseline creatinine clearance values less than 40 mL/min.
12.2 Pharmacodynamics
Treatment with CIBINQO was associated with dose-dependent reduction in serum markers of inflammation, including high sensitivity C-reactive protein (hsCRP), interleukin-31 (IL-31) and thymus and activation regulated chemokine (TARC). These changes returned to near baseline within 4 weeks of drug discontinuation.
12.3 Pharmacokinetics
Abrocitinib plasma Cmax and AUC increased dose proportionally up to 200 mg. Steady-state plasma concentrations of abrocitinib are achieved within 48 hours after once daily administration.
12.5 Pharmacogenomics
Patients who are CYP2C19 poor metabolizers have little to no CYP2C19 enzyme function compared to CYP2C19 normal metabolizers that have fully functional CYP2C19 enzymes.
After single doses of abrocitinib, CYP2C19 poor metabolizers demonstrated dose-normalized AUC of abrocitinib values that were 2.3-fold higher when compared to CYP2C19 normal metabolizers. Approximately 3–5% of Whites and Blacks and 15 to 20% of Asians are CYP2C19 poor metabolizers [see Dosage and Administration (2.4) and Use in Specific Populations (8.8)].
2.2 Recommended Dosage
The recommended dose is 100 mg once daily. If an adequate response is not achieved with CIBINQO 100 mg once daily, consider increasing the dosage to 200 mg once daily.
Discontinue CIBINQO if an adequate response is not achieved with 200 mg once daily.
Use the lowest efficacious dose to maintain response.
CIBINQO can be used with or without topical corticosteroids.
If a dose is missed, administer the dose as soon as possible unless it is less than 12 hours before the next dose, in which case skip the missed dose. Thereafter, resume dosing at the regular scheduled time.
5.1 Serious Infections
The most frequent serious infections reported in clinical studies with CIBINQO for atopic dermatitis were herpes simplex, herpes zoster, and pneumonia [see Adverse Reactions (6.1)]. Serious infections leading to hospitalization or death, including tuberculosis and bacterial, invasive fungal, viral, and other opportunistic infections, have occurred in patients receiving JAK inhibitors used to treat inflammatory conditions.
Avoid use of CIBINQO in patients with active, serious infection including localized infections.
Consider the risks and benefits of treatment prior to initiating CIBINQO in patients:
-
•with chronic or recurrent infection
-
•who have been exposed to tuberculosis
-
•with a history of a serious or an opportunistic infection
-
•who have resided or traveled in areas of endemic tuberculosis or endemic mycoses
-
•with underlying conditions that may predispose them to infection
Closely monitor patients for the development of signs and symptoms of infection during and after treatment with CIBINQO. If a patient develops a serious or opportunistic infection, discontinue CIBINQO. Initiate complete diagnostic testing and appropriate antimicrobial therapy. The risks and benefits of treatment with CIBINQO should be carefully considered prior to reinitiating therapy with CIBINQO.
8.7 Hepatic Impairment
Avoid use of CIBINQO in patients with severe (Child Pugh C) hepatic impairment. In clinical trials, CIBINQO was not evaluated in subjects with severe (Child Pugh C) hepatic impairment.
Dosage adjustment is not required in patients with mild (Child Pugh A) or moderate (Child Pugh B) hepatic impairment based on similar combined exposure (AUCinf,u) of abrocitinib and its two active metabolites, M1 and M2 compared to patients with normal hepatic function [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
CIBINQO is indicated for the treatment of adults and pediatric patients 12 years of age and older with refractory, moderate-to-severe atopic dermatitis whose disease is not adequately controlled with other systemic drug products, including biologics, or when use of those therapies is inadvisable.
12 Clinical Pharmacology (12 CLINICAL PHARMACOLOGY)
12.1 Mechanism of Action
CIBINQO is a Janus kinase (JAK) inhibitor.
Abrocitinib reversibly inhibits JAK1 by blocking the adenosine triphosphate (ATP) binding site. In a cell-free isolated enzyme assay, abrocitinib was selective for JAK1 over JAK2 (28-fold), JAK3 (>340-fold), and tyrosine kinase (TYK) 2 (43-fold), as well as the broader kinome. The relevance of inhibition of specific JAK enzymes to therapeutic effectiveness is not currently known. Both the parent compound and the active metabolites inhibit JAK1 activity in vitro with similar levels of selectivity.
13 Nonclinical Toxicology (13 NONCLINICAL TOXICOLOGY)
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•For recommended testing, evaluations, and procedures prior to CIBINQO initiation, see Full Prescribing Information. (2.1)
-
•Recommended dosage is 100 mg orally once daily. (2.2)
-
•200 mg orally once daily is recommended for those patients who are not responding to 100 mg once daily. (2.2)
-
•Moderate renal impairment: 50 mg once daily or 100 mg once daily for those patients who are not responding to 50 mg once daily. (2.3)
-
•CYP2C19 poor metabolizer: 50 mg once daily or 100 mg once daily for those patients who are not responding to 50 mg once daily. (2.4)
-
•For dosage modifications for certain adverse reactions, see Full Prescribing Information. (2.6)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
-
•50 mg: Pink, oval, film-coated tablet debossed with "PFE" on one side and "ABR 50" on the other.
-
•100 mg: Pink, round, film-coated tablet debossed with "PFE" on one side and "ABR 100" on the other.
-
•200 mg: Pink, oval, film-coated tablet debossed with "PFE" on one side and "ABR 200" on the other.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
8.8 Cyp2c19 Poor Metabolizers (8.8 CYP2C19 Poor Metabolizers)
In patients who are CYP2C19 poor metabolizers, the AUC of abrocitinib is increased compared to CYP2C19 normal metabolizers due to reduced metabolic clearance. Dosage reduction of CIBINQO is recommended in patients who are known or suspected to be CYP2C19 poor metabolizers based on genotype or previous history/experience with other CYP2C19 substrates [see Dosage and Administration (2.4) and Clinical Pharmacology (12.5)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of CIBINQO was evaluated in four randomized, placebo-controlled clinical trials (2 monotherapy, 1 combination therapy with topical corticosteroid, and 1 dose-ranging) and one long-term extension trial in subjects with moderate to severe atopic dermatitis (AD). A total of 1623 subjects with moderate to severe atopic dermatitis were treated with CIBINQO in these clinical trials representing 1428 patient-years of exposure. There were 634 subjects with at least 1 year of exposure to CIBINQO.
In the placebo-controlled clinical trials, a total of 1198 subjects were exposed to CIBINQO with 608 subjects receiving CIBINQO 100 mg once daily and 590 subjects receiving CIBINQO 200 mg once daily for up to 16 weeks. The median age of subjects was 33.0 years, 124 subjects (8.1%) were 12 to less than 18 years old and 94 subjects (6.1%) were 65 years of age or older. The majority of subjects were White (68.7%) and male (53.9%).
Adverse reactions occurring at ≥1% in any of the treated groups and at a higher rate than in the placebo group are presented in Table 3. A total of 61 (5.1%) subjects treated with CIBINQO were discontinued from the trials due to adverse reactions. The safety profile of CIBINQO in the monotherapy and the combination trial(s) were similar.
|
Weeks 0–16 |
|||
|
CIBINQO
Study size adjusted percentages
)
|
CIBINQO
|
Placebo
|
|
|
Nasopharyngitis |
51 (8.7) |
75 (12.4) |
27 (7.9) |
|
Nausea |
86 (14.5) |
37 (6.0) |
7 (2.1) |
|
Headache |
46 (7.8) |
36 (6.0) |
12 (3.5) |
|
Herpes simplex Herpes simplex also includes oral herpes, ophthalmic herpes, herpes dermatitis, genital herpes.
|
25 (4.2) |
20 (3.3) |
6 (1.8) |
|
Increased blood creatine phosphokinase |
17 (2.9) |
14 (2.3) |
5 (1.5) |
|
Dizziness |
17 (2.9) |
11 (1.8) |
3 (0.9) |
|
Urinary tract infection |
13 (2.2) |
10 (1.7) |
4 (1.2) |
|
Fatigue |
8 (1.3) |
10 (1.6) |
2 (0.5) |
|
Acne |
28 (4.7) |
10 (1.6) |
0 (0.0) |
|
Vomiting |
19 (3.2) |
9 (1.5) |
3 (0.9) |
|
Impetigo |
3 (0.5) |
9 (1.5) |
1 (0.3) |
|
Oropharyngeal pain |
6 (1.0) |
8 (1.4) |
2 (0.6) |
|
Hypertension |
5 (0.8) |
7 (1.2) |
2 (0.7) |
|
Influenza |
6 (1.1) |
7 (1.2) |
0 (0.0) |
|
Gastroenteritis |
8 (1.3) |
7 (1.1) |
2 (0.6) |
|
Dermatitis contact |
3 (0.5) |
6 (1.1) |
1 (0.3) |
|
Abdominal pain upper |
11 (1.9) |
4 (0.6) |
0 (0.0) |
|
Abdominal discomfort |
7 (1.2) |
3 (0.5) |
1 (0.3) |
|
Herpes zoster |
7 (1.2) |
2 (0.3) |
0 (0.0) |
|
Thrombocytopenia |
9 (1.5) |
0 (0.0) |
0 (0.0) |
2.7 Administration Instructions
Administer CIBINQO with or without food at approximately the same time each day.
Swallow CIBINQO tablets whole with water. Do not crush, split, or chew CIBINQO tablets.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
CIBINQO is supplied as:
|
Dosage Form |
Strength |
Description |
Bottle Size
|
NDC Number |
|
Tablets |
50 mg |
Pink, oval tablet debossed with "PFE" on one side and "ABR 50" on the other. |
30 count bottle |
0069-0235-30 |
|
Tablets |
100 mg |
Pink, round tablet debossed with "PFE" on one side and "ABR 100" on the other. |
30 count bottle |
0069-0335-30 |
|
Tablets |
200 mg |
Pink, oval tablet debossed with "PFE" on one side and "ABR 200" on the other. |
30 count bottle |
0069-0435-30 |
7.1 Effects of Other Drugs On Cibinqo (7.1 Effects of Other Drugs on CIBINQO)
Table 4 includes drugs with clinically significant drug interactions affecting CIBINQO.
|
Strong CYP2C19 Inhibitors |
|
|
Clinical Impact |
Coadministration of CIBINQO with strong CYP2C19 inhibitors increases the combined exposure of abrocitinib and its two active metabolites, M1 and M2 which may increase the adverse reactions of CIBINQO [see Clinical Pharmacology (12.3)]. |
|
Intervention |
Dosage reduction of CIBINQO is recommended when coadministered with strong CYP2C19 inhibitors [see Dosage and Administration (2.5)]. |
|
Moderate to Strong Inhibitors of both CYP2C19 and CYP2C9 |
|
|
Clinical Impact |
Coadministration of CIBINQO with drugs that are moderate to strong inhibitors of both CYP2C19 and CYP2C9 increases the exposure of abrocitinib and its two active metabolites, M1 and M2 which may increase the adverse reactions of CIBINQO [Clinical Pharmacology (12.3)]. |
|
Intervention |
Avoid concomitant use of CIBINQO with drugs that are moderate to strong inhibitors of both CYP2C19 and CYP2C9. |
|
Strong CYP2C19 or CYP2C9 Inducers |
|
|
Clinical Impact |
Coadministration of CIBINQO with strong CYP2C19 or CYP2C9 inducers decreases the combined exposure of abrocitinib and its two active metabolites, M1 and M2, which may result in loss of or reduced clinical response [see Clinical Pharmacology (12.3)]. |
|
Intervention |
Avoid concomitant use of CIBINQO with strong CYP2C19 or CYP2C9 inducers. |
7.2 Effects of Cibinqo On Other Drugs (7.2 Effects of CIBINQO on Other Drugs)
Table 5 includes clinically significant drug interactions affecting other drugs.
|
P-gp Substrate Where Small Concentration Changes May Lead to Serious or Life-threatening Toxicities |
|
|
Clinical Impact |
Coadministration of CIBINQO with P-gp substrate increases plasma concentrations of P-gp substrates and may result in potential adverse reactions of the P-gp substrate where small concentration changes may lead to serious or life-threatening toxicities (e.g., digoxin) [see Clinical Pharmacology (12.3)]. |
|
Intervention |
Monitor appropriately or dose titrate P-gp substrate where small concentration changes may lead to serious or life-threatening toxicities when coadministered with CIBINQO. |
|
Antiplatelet Therapy Drugs |
|
|
Clinical Impact |
Coadministration of CIBINQO with antiplatelet therapy drugs may increase the risk of bleeding with thrombocytopenia [see Warnings and Precautions (5.5) and Clinical Pharmacology (12.2)]. |
|
Intervention |
Antiplatelet drugs, except for low-dose aspirin (≤81 mg daily), during the first 3 months of treatment are contraindicated with CIBINQO [see Contraindications (4)]. |
5.4 Major Adverse Cardiovascular Events
Major adverse cardiovascular events were reported in clinical trials of CIBINQO for atopic dermatitis [see Adverse Reactions (6.1)].
In a large, randomized, postmarketing safety trial of another JAK inhibitor in RA subjects 50 years of age and older with at least one cardiovascular risk factor, a higher rate of major adverse cardiovascular events (MACE) defined as cardiovascular death, non-fatal myocardial infarction (MI), and non-fatal stroke was observed with the JAK inhibitor compared to those treated with TNF blockers. CIBINQO is not approved for use in RA. Patients who are current or past smokers are at additional increased risk.
Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with CIBINQO, particularly in patients who are current or past smokers and patients with other cardiovascular risk factors. Patients should be informed about the symptoms of serious cardiovascular events and the steps to take if they occur. Discontinue CIBINQO in patients that have experienced a myocardial infarction or stroke.
8.3 Females and Males of Reproductive Potential
5.3 Malignancy and Lymphoproliferative Disorders
Malignancies, including non-melanoma skin cancer (NMSC), were observed in clinical trials with CIBINQO for atopic dermatitis [see Adverse Reactions (6.1)].
Perform periodic skin examination for patients who are at increased risk for skin cancer. Exposure to sunlight and UV light should be limited by wearing protective clothing and using broad-spectrum sunscreen.
Malignancies, including lymphomas, have occurred in patients receiving JAK inhibitors used to treat inflammatory conditions. In a large, randomized, postmarketing safety trial of another JAK inhibitor in RA subjects, a higher rate of malignancies (excluding non-melanoma skin cancer (NMSC)) was observed in subjects treated with the JAK inhibitor compared to those treated with TNF blockers. CIBINQO is not approved for use in RA. A higher rate of lymphomas was observed in subjects treated with the JAK inhibitor compared to those treated with TNF blockers. A higher rate of lung cancers was observed in current or past smokers treated with the JAK inhibitor compared to those treated with TNF blockers. In this trial, current or past smokers had an additional increased risk of overall malignancies.
Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with CIBINQO, particularly in patients with a known malignancy (other than a successfully treated NMSC), patients who develop a malignancy when on treatment, and patients who are current or past smokers.
2.5 Dosage Modifications Due to Strong Inhibitors (2.5 Dosage Modifications due to Strong Inhibitors)
In patients taking strong inhibitors of cytochrome P450 (CYP) 2C19, reduce the dosage to 50 mg once daily [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)]. If an adequate response is not achieved with CIBINQO 50 mg daily, consider increasing the dosage to 100 mg once daily. Discontinue therapy if inadequate response is seen after dosage increase to 100 mg once daily.
2.4 Recommended Dosage in Cyp2c19 Poor Metabolizers (2.4 Recommended Dosage in CYP2C19 Poor Metabolizers)
In patients who are known or suspected to be CYP2C19 poor metabolizers, the recommended dosage of CIBINQO is 50 mg once daily [see Use in Specific Populations (8.8) and Clinical Pharmacology (12.5)]. If an adequate response is not achieved with CIBINQO 50 mg once daily, consider increasing the dosage to 100 mg once daily. Discontinue therapy if inadequate response is seen after dosage increase to 100 mg once daily.
Principal Display Panel 50 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 50 mg Tablet Bottle Label)
ALWAYS DISPENSE WITH
MEDICATION GUIDE
Pfizer
NDC 0069-0235-30
CIBINQO®
(abrocitinib) tablets
50 mg
Do not crush, split, or chew the tablets.
30 Tablets
Rx only
Principal Display Panel 100 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 100 mg Tablet Bottle Label)
ALWAYS DISPENSE WITH
MEDICATION GUIDE
Pfizer
NDC 0069-0335-30
CIBINQO®
(abrocitinib) tablets
100 mg
Do not crush, split, or chew the tablets.
30 Tablets
Rx only
Principal Display Panel 200 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 200 mg Tablet Bottle Label)
ALWAYS DISPENSE WITH
MEDICATION GUIDE
Pfizer
NDC 0069-0435-30
CIBINQO®
(abrocitinib) tablets
200 mg
Do not crush, split, or chew the tablets.
30 Tablets
Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2-year oral carcinogenicity study in rats, abrocitinib increased the incidence of benign thymomas in female rats at doses of 10 and 30 mg/kg/day (2.8 and 14 times the MRHD, respectively, based on AUC comparison). Abrocitinib was not carcinogenic in female rats at 3 mg/kg/day (0.6 times the MRHD based on AUC comparison) or male rats at doses up to 30 mg/kg/day (14 times the MRHD based on AUC comparison). Abrocitinib was not carcinogenic in Tg.rasH2 mice at oral doses up to 60 mg/kg/day in males and 75 mg/kg/day in females.
Abrocitinib was not mutagenic in the bacterial mutagenicity assay (Ames assay). Although abrocitinib was aneugenic in the in vitro TK6 micronucleus assay, abrocitinib was not aneugenic or clastogenic in an in vivo rat bone marrow micronucleus assay.
Abrocitinib did not impair male fertility at doses up to 70 mg/kg/day (26 times the MRHD based on AUC comparison) or female fertility at 10 mg/kg/day (2 times the MRHD based on AUC comparison). Abrocitinib impaired female fertility (reducing fertility index, corpora lutea, and implantation sites) at 70 mg/kg/day (29 times the MRHD based on AUC comparison). Impaired fertility in female rats reversed 1 month after cessation of abrocitinib administration.
2.1 Recommended Testing, Evaluations, and Procedures Prior to Treatment Initiation
Perform the following tests and evaluations prior to CIBINQO initiation:
-
•Tuberculosis (TB) infection evaluation – CIBINQO initiation is not recommended in patients with active TB. For patients with latent TB or those with a negative latent TB test who are at high risk for TB, start preventive therapy for latent TB prior to initiation of CIBINQO [see Warnings and Precautions (5.1)].
-
•Viral hepatitis screening in accordance with clinical guidelines – CIBINQO initiation is not recommended in patients with active hepatitis B or hepatitis C [see Warnings and Precautions (5.1)].
-
•A complete blood count (CBC) – CIBINQO initiation is not recommended in patients with a platelet count <150,000/mm3, an absolute lymphocyte count <500/mm3, an absolute neutrophil count <1,000/mm3, or a hemoglobin value <8 g/dL [see Warnings and Precautions (5.6)].
Complete any necessary immunizations, including herpes zoster vaccinations, in agreement with current immunization guidelines prior to CIBINQO initiation [see Warnings and Precautions (5.7)].
Warning: Serious Infections, Mortality, Malignancy, Major Adverse Cardiovascular Events, and Thrombosis (WARNING: SERIOUS INFECTIONS, MORTALITY, MALIGNANCY, MAJOR ADVERSE CARDIOVASCULAR EVENTS, and THROMBOSIS)
WARNING: SERIOUS INFECTIONS, MORTALITY, MALIGNANCY, MAJOR ADVERSE CARDIOVASCULAR EVENTS (MACE), and THROMBOSIS
See full prescribing information for complete boxed warning.
-
•Increased risk of serious bacterial, fungal, viral and opportunistic infections leading to hospitalization or death, including tuberculosis (TB). Discontinue treatment with CIBINQO if serious or opportunistic infection occurs. Test for latent TB before and during therapy; treat latent TB prior to use. Monitor all patients for active TB during treatment, even patients with initial negative latent TB test. (5.1)
-
•Higher rate of all-cause mortality, including sudden cardiovascular death, with another JAK inhibitor vs. TNF blockers in rheumatoid arthritis (RA) patients. CIBINQO is not approved for use in RA patients. (5.2)
-
•Malignancies have occurred with CIBINQO. Higher rate of lymphomas and lung cancers with another JAK inhibitor vs. TNF blockers in RA patients. (5.3)
-
•MACE has occurred with CIBINQO. Higher rate of MACE (defined as cardiovascular death, myocardial infarction, and stroke) with another JAK inhibitor vs. TNF blockers in RA patients. (5.4)
-
•Thrombosis has occurred with CIBINQO. Increased incidence of pulmonary embolism, venous and arterial thrombosis with another JAK inhibitor vs. TNF blockers. (5.5)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:45:51.249265 · Updated: 2026-03-14T22:23:30.091915