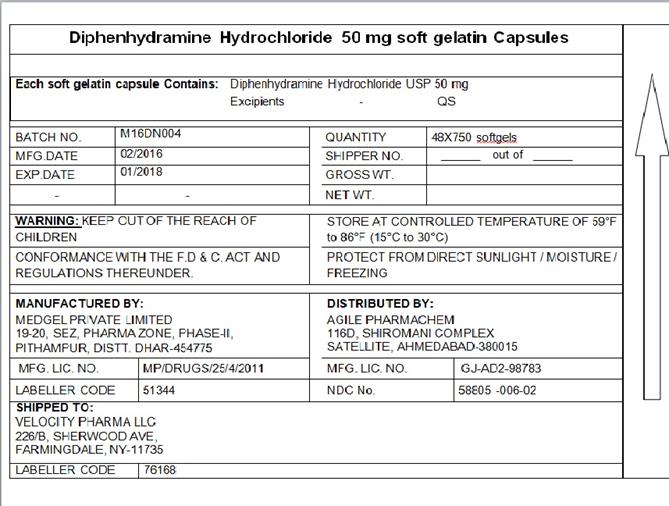
Diphenhydramine Hcl 50mg, Usp
3f12bd24-8265-44a0-9112-92c026c8e2fa
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
(in each softgel) Diphenhydramine HCl 50 mg
Purpose
Nighttime sleep aid
Medication Information
Purpose
Nighttime sleep aid
Description
(in each softgel) Diphenhydramine HCl 50 mg
Uses
- For relief of occasional sleeplessness.
Warnings
Do not use
- in children under 12 years of age.
- with any other product containing diphenhydramine, even one used on skin
Ask a doctor before use if you have
- glaucoma
- trouble urinating due to an enlarged prostate gland
- a breathing problem such as emphysema or chronic bronchitis
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers
When using this product
- avoid alcoholic drinks
If pregnant or breast-feeding, ask a health professional before use.
Directions
- adults and children 12 years and over : one softgel (50 mg) at bed time if needed or as directed by a doctor.
- children under 12 years: do not use
Active Ingredient
(in each softgel)
Diphenhydramine HCl 50 mg
Other Information
- save carton for full directions and warnings
Inactive Ingredients
Blue 1, gelatin, glycerin, polyethylene glycol, polyvinyl acetate phthalate, propylene glycol, sorbitol, titanium dioxide and water.
Questions Or Comments
Call 1-855-314-1850
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Package Label.principal Display Panel
DIPHENHYDRAMINE HYDROCHLORIDE SOFTGEL CAPSULE, USP 50 MG
NDC:58805-006-02
Structured Label Content
Uses
- For relief of occasional sleeplessness.
Purpose
Nighttime sleep aid
Warnings
Do not use
- in children under 12 years of age.
- with any other product containing diphenhydramine, even one used on skin
Ask a doctor before use if you have
- glaucoma
- trouble urinating due to an enlarged prostate gland
- a breathing problem such as emphysema or chronic bronchitis
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers
When using this product
- avoid alcoholic drinks
If pregnant or breast-feeding, ask a health professional before use.
Directions
- adults and children 12 years and over : one softgel (50 mg) at bed time if needed or as directed by a doctor.
- children under 12 years: do not use
Active Ingredient
(in each softgel)
Diphenhydramine HCl 50 mg
Other Information
- save carton for full directions and warnings
Inactive Ingredients
Blue 1, gelatin, glycerin, polyethylene glycol, polyvinyl acetate phthalate, propylene glycol, sorbitol, titanium dioxide and water.
Questions Or Comments (Questions or Comments)
Call 1-855-314-1850
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
DIPHENHYDRAMINE HYDROCHLORIDE SOFTGEL CAPSULE, USP 50 MG
NDC:58805-006-02
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:34.250841 · Updated: 2026-03-14T22:55:07.187033