These Highlights Do Not Include All The Information Needed To Use Metoprolol Tartrate Injection Safely And Effectively. See Full Prescribing Information For Metoprolol Tartrate Injection
3ef0affa-a2f7-92de-e063-6294a90ad4d0
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions, Hypoglycemia ( 5.5 ) 5/2024
Indications and Usage
Metoprolol tartrate Injection is indicated in the treatment of definite or suspected acute myocardial infarction in hemodynamically stable patients to reduce cardiovascular mortality when used in conjunction with oral metoprolol maintenance therapy.
Dosage and Administration
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Initiate treatment in a coronary care or similar unit immediately after the patient's hemodynamic condition has stabilized. Begin treatment in this early phase with the intravenous administration of three bolus injections of 5 mg of Metoprolol tartrate each; give the injections at approximately 2-minute intervals. During the intravenous administration of Metoprolol tartrate, monitor blood pressure, heart rate, and electrocardiogram.
Warnings and Precautions
Worsening cardiac failure may occur. ( 5.2 ) Bronchospastic Disease: Avoid beta-blockers. ( 5.3 ) Pheochromocytoma: First initiate therapy with an alpha blocker. ( 5.4 ) May aggravate symptoms of arterial insufficiency. ( 5.6 )
Contraindications
Hypersensitivity to Metoprolol tartrate and related derivatives, or to any of the excipients; hypersensitivity to other beta-blockers (cross sensitivity between beta-blockers can occur). Metoprolol tartrate is contraindicated in patients with a heart rate <45 beats/min; second- and third-degree heart block (unless a functioning pacemaker is present); significant first-degree heart block (P-R interval ≥0.24 sec); systolic blood pressure <100 mmHg; or decompensated cardiac failure.
Adverse Reactions
The following adverse reactions are described elsewhere in labeling: Worsening angina or myocardial infarction [see Warnings and Precautions (5) ] Worsening heart failure [see Warnings and Precautions (5) ] Worsening AV block [see Contraindications (4) ]
Drug Interactions
Catecholamine-depleting drugs may have an additive effect when given with beta-blocking agents. ( 7.1 ) Patients may be unresponsive to the usual doses of epinephrine used to treat allergic reaction. ( 7.2 ) CYP2D6 Inhibitors are likely to increase metoprolol concentration. ( 7.3 ) Concomitant use of glycosides, clonidine, and diltiazem and verapamil with beta-blockers can increase the risk of bradycardia. ( 7.4 ) Beta-blockers including metoprolol, may exacerbate the rebound hypertension that can follow the withdrawal of clonidine. ( 7.4 )
Storage and Handling
Metoprolol tartrate Injection, USP is available as: Unit of Sale Concentration NDC 84549-778-05 Single-dose glass Fliptop Vial 5 mg/5 mL (1 mg/mL)
How Supplied
Metoprolol tartrate Injection, USP is available as: Unit of Sale Concentration NDC 84549-778-05 Single-dose glass Fliptop Vial 5 mg/5 mL (1 mg/mL)
Medication Information
Warnings and Precautions
Worsening cardiac failure may occur. ( 5.2 ) Bronchospastic Disease: Avoid beta-blockers. ( 5.3 ) Pheochromocytoma: First initiate therapy with an alpha blocker. ( 5.4 ) May aggravate symptoms of arterial insufficiency. ( 5.6 )
Indications and Usage
Metoprolol tartrate Injection is indicated in the treatment of definite or suspected acute myocardial infarction in hemodynamically stable patients to reduce cardiovascular mortality when used in conjunction with oral metoprolol maintenance therapy.
Dosage and Administration
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Initiate treatment in a coronary care or similar unit immediately after the patient's hemodynamic condition has stabilized. Begin treatment in this early phase with the intravenous administration of three bolus injections of 5 mg of Metoprolol tartrate each; give the injections at approximately 2-minute intervals. During the intravenous administration of Metoprolol tartrate, monitor blood pressure, heart rate, and electrocardiogram.
Contraindications
Hypersensitivity to Metoprolol tartrate and related derivatives, or to any of the excipients; hypersensitivity to other beta-blockers (cross sensitivity between beta-blockers can occur). Metoprolol tartrate is contraindicated in patients with a heart rate <45 beats/min; second- and third-degree heart block (unless a functioning pacemaker is present); significant first-degree heart block (P-R interval ≥0.24 sec); systolic blood pressure <100 mmHg; or decompensated cardiac failure.
Adverse Reactions
The following adverse reactions are described elsewhere in labeling: Worsening angina or myocardial infarction [see Warnings and Precautions (5) ] Worsening heart failure [see Warnings and Precautions (5) ] Worsening AV block [see Contraindications (4) ]
Drug Interactions
Catecholamine-depleting drugs may have an additive effect when given with beta-blocking agents. ( 7.1 ) Patients may be unresponsive to the usual doses of epinephrine used to treat allergic reaction. ( 7.2 ) CYP2D6 Inhibitors are likely to increase metoprolol concentration. ( 7.3 ) Concomitant use of glycosides, clonidine, and diltiazem and verapamil with beta-blockers can increase the risk of bradycardia. ( 7.4 ) Beta-blockers including metoprolol, may exacerbate the rebound hypertension that can follow the withdrawal of clonidine. ( 7.4 )
Storage and Handling
Metoprolol tartrate Injection, USP is available as: Unit of Sale Concentration NDC 84549-778-05 Single-dose glass Fliptop Vial 5 mg/5 mL (1 mg/mL)
How Supplied
Metoprolol tartrate Injection, USP is available as: Unit of Sale Concentration NDC 84549-778-05 Single-dose glass Fliptop Vial 5 mg/5 mL (1 mg/mL)
Description
Warnings and Precautions, Hypoglycemia ( 5.5 ) 5/2024
Section 42229-5
Transition to Oral Metoprolol:
Following administration of Metoprolol tartrate Injection, transition patients to an oral formulation of metoprolol. See prescribing information for oral metoprolol for dose selection.
Section 43683-2
|
Warnings and Precautions, Hypoglycemia ( 5.5) |
5/2024 |
Section 44425-7
Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room Temperature.]. Do not freeze.
PROTECT FROM LIGHT.Retain in carton until time of use.
Discard unused portion.
11 Description
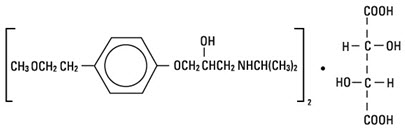
Metoprolol tartrate Injection, USP, is a selective beta 1-adrenoreceptor blocking agent, available in 5 mL vials for intravenous administration. Each vial contains a sterile solution of metoprolol tartrate USP, 5 mg, and sodium chloride USP, 45 mg. Metoprolol tartrate USP is (±)-1-(Isopropylamino)-3-[p-(2-methoxyethyl) phenoxy]-2-propanol (2:1) dextro-tartrate salt, and its structural formula is:
Metoprolol tartrate USP is a white, practically odorless, crystalline powder with a molecular weight of 684.83. It is very soluble in water; freely soluble in methylene chloride, in chloroform, and in alcohol; slightly soluble in acetone; and insoluble in ether.
5.1 Bradycardia
Bradycardia, including sinus pause, heart block, and cardiac arrest have occurred with the use of Metoprolol tartrate. Patients with first-degree atrioventricular block, sinus node dysfunction, or conduction disorders may be at increased risk. Monitor heart rate and rhythm in patients receiving Metoprolol tartrate. If severe bradycardia develops, reduce or stop Metoprolol tartrate.
7.2 Epinephrine
While taking beta-blockers, patients with a history of severe anaphylactic reactions to a variety of allergens may be more reactive to repeated challenge and may be unresponsive to the usual doses of epinephrine used to treat an allergic reaction.
5.5 Hypoglycemia
Beta-blockers may prevent early warning signs of hypoglycemia, such as tachycardia, and increase the risk for severe or prolonged hypoglycemia at any time during treatment, especially in patients with diabetes mellitus or children and patients who are fasting (i.e., surgery, not eating regularly, or are vomiting). If severe hypoglycemia occurs, patients should be instructed to seek emergency treatment.
5.2 Heart Failure
Worsening cardiac failure may occur during metoprolol use. If such symptoms occur, increase diuretics and restore clinical stability before administering the next dose of metoprolol [see Dosage and Administration (2)] . Such episodes do not preclude subsequent successful titration of oral metoprolol.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
In worldwide clinical trials of Metoprolol tartrate in myocardial infarction, where approximately 478 patients were over 65 years of age (0 over 75 years of age), no age-related differences in safety and effectiveness were found. Other reported clinical experience in myocardial infarction has not identified differences in response between the elderly and younger patients. However, greater sensitivity of some elderly individuals taking Metoprolol tartrate cannot be categorically ruled out. Therefore, in general, it is recommended that dosing proceed with caution in this population.
14 Clinical Studies
In a large (1,395 patients randomized), double-blind, placebo-controlled clinical study, Metoprolol tartrate was shown to reduce 3-month mortality by 36% in patients with suspected or definite myocardial infarction.
Patients were randomized and treated as soon as possible after their arrival in the hospital, once their clinical condition had stabilized and their hemodynamic status had been carefully evaluated. Subjects were ineligible if they had hypotension, bradycardia, peripheral signs of shock, and/or more than minimal basal rales as signs of congestive heart failure. Initial treatment consisted of intravenous followed by oral administration of Metoprolol tartrate or placebo, given in a coronary care or comparable unit. Oral maintenance therapy with Metoprolol tartrate or placebo was then continued for 3 months. After this double-blind period, all patients were given Metoprolol tartrate and followed up to 1 year.
The median delay from the onset of symptoms to the initiation of therapy was 8 hours in both the Metoprolol tartrate- and placebo-treatment groups. Among patients treated with Metoprolol tartrate, there were comparable reductions in 3-month mortality for those treated early (≤8 hours) and those in whom treatment was started later. Significant reductions in the incidence of ventricular fibrillation and in chest pain following initial intravenous therapy were also observed with Metoprolol tartrate and were independent of the interval between onset of symptoms and initiation of therapy.
In this study, patients treated with metoprolol received the drug both very early (intravenously) and during a subsequent 3-month period, while placebo patients received no beta-blocker treatment for this period. The study thus was able to show a benefit from the overall metoprolol regimen but cannot separate the benefit of very early intravenous treatment from the benefit of later beta-blocker therapy. Nonetheless, because the overall regimen showed a clear beneficial effect on survival without evidence of an early adverse effect on survival, one acceptable dosage regimen is the precise regimen used in the trial. Because the specific benefit of very early treatment remains to be defined however, it is also reasonable to administer the drug orally to patients at a later time as is recommended for certain other beta-blockers.
4 Contraindications
Hypersensitivity to Metoprolol tartrate and related derivatives, or to any of the excipients; hypersensitivity to other beta-blockers (cross sensitivity between beta-blockers can occur).
Metoprolol tartrate is contraindicated in patients with a heart rate <45 beats/min; second- and third-degree heart block (unless a functioning pacemaker is present); significant first-degree heart block (P-R interval ≥0.24 sec); systolic blood pressure <100 mmHg; or decompensated cardiac failure.
6 Adverse Reactions
The following adverse reactions are described elsewhere in labeling:
- Worsening angina or myocardial infarction [see Warnings and Precautions (5)]
- Worsening heart failure [see Warnings and Precautions (5)]
- Worsening AV block [see Contraindications (4)]
7 Drug Interactions
- Catecholamine-depleting drugs may have an additive effect when given with beta-blocking agents. ( 7.1)
- Patients may be unresponsive to the usual doses of epinephrine used to treat allergic reaction. ( 7.2)
- CYP2D6 Inhibitors are likely to increase metoprolol concentration. ( 7.3)
- Concomitant use of glycosides, clonidine, and diltiazem and verapamil with beta-blockers can increase the risk of bradycardia. ( 7.4)
- Beta-blockers including metoprolol, may exacerbate the rebound hypertension that can follow the withdrawal of clonidine. ( 7.4)
5.4 Pheochromocytoma
If metoprolol tartrate is used in the setting of pheochromocytoma, it should be given in combination with an alpha blocker, and only after the alpha blocker has been initiated. Administration of beta-blockers alone in the setting of pheochromocytoma has been associated with a paradoxical increase in blood pressure due to the attenuation of beta-mediated vasodilatation in skeletal muscle.
12.2 Pharmacodynamics
When the drug was infused over a 10-minute period, in normal volunteers, maximum beta-blockade was achieved at approximately 20 minutes. Equivalent maximal beta-blocking effect is achieved with oral and intravenous doses in the ratio of approximately 2.5:1. There is a linear relationship between the log of plasma levels and reduction of exercise heart rate.
In several studies of patients with acute myocardial infarction, intravenous followed by oral administration of Metoprolol tartrate caused a reduction in heart rate, systolic blood pressure and cardiac output. Stroke volume, diastolic blood pressure and pulmonary artery end diastolic pressure remained unchanged.
12.5 Pharmacogenomics
CYP2D6 is absent in about 8% of Caucasians (poor metabolizers) and about 2% of most other populations. CYP2D6 can be inhibited by several drugs. Poor metabolizers of CYP2D6 will have increased (several-fold) metoprolol blood levels, decreasing metoprolol's cardioselectivity.
7.3 Cyp2d6 Inhibitors
Drugs that are strong inhibitors of CYP2D6, such as quinidine, fluoxetine, paroxetine, and propafenone, were shown to double metoprolol concentrations. While there is no information about moderate or weak inhibitors, these too are likely to increase metoprolol concentration. Increases in plasma concentration decrease the cardioselectivity of metoprolol [see Clinical Pharmacology (12.3)]. Monitor patients closely, when the combination cannot be avoided.
8.6 Hepatic Impairment
No studies have been performed with metoprolol in patients with hepatic impairment. Because metoprolol is metabolized by the liver, metoprolol blood levels are likely to increase substantially with poor hepatic function. Therefore, initiate therapy at doses lower than those recommended for a given indication.
1 Indications and Usage
Metoprolol tartrate Injection is indicated in the treatment of definite or suspected acute myocardial infarction in hemodynamically stable patients to reduce cardiovascular mortality when used in conjunction with oral metoprolol maintenance therapy.
12.1 Mechanism of Action
Metoprolol is a beta 1-selective (cardioselective) adrenergic receptor blocking agent. This preferential effect is not absolute, however, and at higher plasma concentrations, metoprolol also inhibits beta 2-adrenoreceptors, chiefly located in the bronchial and vascular musculature.
Metoprolol has no intrinsic sympathomimetic activity, and membrane-stabilizing activity is detectable only at plasma concentrations much greater than required for beta-blockade. Animal and human experiments indicate that metoprolol slows the sinus rate and decreases AV nodal conduction.
The relative beta 1-selectivity of metoprolol has been confirmed by the following: (1) In normal subjects, metoprolol is unable to reverse the beta 2-mediated vasodilating effects of epinephrine. This contrasts with the effect of nonselective beta-blockers, which completely reverse the vasodilating effects of epinephrine. (2) In asthmatic patients, metoprolol reduces FEV 1and FVC significantly less than a nonselective beta-blocker, propranolol, at equivalent beta 1-receptor blocking doses.
The precise mechanism of action of Metoprolol tartrate in patients with suspected or definite myocardial infarction is not known.
5 Warnings and Precautions
5.3 Bronchospastic Disease
PATIENTS WITH BRONCHOSPASTIC DISEASES, IN GENERAL, SHOULD NOT RECEIVE BETA-BLOCKERS because they can exacerbate bronchospasm. Because of its relative beta 1cardio-selectivity, however, metoprolol may be used in patients with bronchospastic disease for initial treatment of myocardial infarction. Bronchodilators, including beta 2-agonists, should be readily available or administered concomitantly [see Dosage and Administration (2)].
2 Dosage and Administration
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Initiate treatment in a coronary care or similar unit immediately after the patient's hemodynamic condition has stabilized.
Begin treatment in this early phase with the intravenous administration of three bolus injections of 5 mg of Metoprolol tartrate each; give the injections at approximately 2-minute intervals. During the intravenous administration of Metoprolol tartrate, monitor blood pressure, heart rate, and electrocardiogram.
3 Dosage Forms and Strengths
Injection: 5 mg metoprolol tartrate supplied in 5 mL Single-dose glass Fliptop Vial.
6.2 Post Marketing Experience
The following adverse reactions have been identified during post-approval use of metoprolol. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiovascular:Cold extremities, arterial insufficiency (usually of the Raynaud type), palpitations, peripheral edema, syncope, and hypotension.
Respiratory:Wheezing (bronchospasm), dyspnea.
Central Nervous System:Confusion, short-term memory loss, headache, nightmares, insomnia, nervousness, hallucinations.
Gastrointestinal:Nausea, dry mouth, constipation, flatulence, heartburn, hepatitis, vomiting.
Hypersensitive Reactions:Pruritus.
Miscellaneous:Musculoskeletal pain, arthritis, blurred vision, decreased libido, tinnitus, reversible alopecia, agranulocytosis, dry eyes, worsening of psoriasis, Peyronie's disease, photosensitivity.
8 Use in Specific Populations
- Hepatic Impairment: Consider initiating metoprolol tartrate therapy at low doses while monitoring closely for adverse events. ( 8.6)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The adverse reaction information from clinical trials does, however, provide a basis for identifying the adverse events that appear to be related to drug use and for approximating rates.
5.6 Peripheral Vascular Disease
Beta-blockers can precipitate or aggravate symptoms of arterial insufficiency in patients with peripheral vascular disease.
17 Patient Counseling Information
Advise patients (1) to avoid operating automobiles and machinery or engaging in other tasks requiring alertness until the patient's response to therapy with Metoprolol tartrate has been determined; (2) to contact the physician if any difficulty in breathing occurs; (3) to inform the physician or dentist before any type of surgery that he or she is taking Metoprolol tartrate.
Risk of Hypoglycemia
Inform patients or caregivers that there is a risk of hypoglycemia when Metoprolol tartrate Injection is given to patients who are fasting or who are vomiting. Monitor for signs of hypoglycemia [see Warnings and Precautions (5.5)] .
16 How Supplied/storage and Handling
Metoprolol tartrate Injection, USP is available as:
| Unit of Sale | Concentration |
|---|---|
|
NDC 84549-778-05 Single-dose glass Fliptop Vial |
5 mg/5 mL (1 mg/mL) |
7.5 Drugs That Decrease Blood Pressure
Concomitant administration of beta-blockers with other drugs known to decrease blood pressure may result in an enhanced hypotensive effect.
Principal Display Panel 5 Ml Vial Label
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have been conducted to evaluate the carcinogenic potential of metoprolol tartrate. In 2-year studies in rats at three oral dosage levels of up to 800 mg/kg/day (39 times, on a mg/m 2basis, the daily dose of 200 mg for a 60 kg patient), there was no increase in the development of spontaneously occurring benign or malignant neoplasms of any type. The only histologic changes that appeared to be drug related were an increased incidence of generally mild focal accumulation of foamy macrophages in pulmonary alveoli and a slight increase in biliary hyperplasia. In a 21-month study in Swiss albino mice at three oral dosage levels of up to 750 mg/kg/day (18 times, on a mg/m 2basis, the daily dose of 200 mg for a 60-kg patient), benign lung tumors (small adenomas) occurred more frequently in female mice receiving the highest dose than in untreated control animals. There was no increase in malignant or total (benign plus malignant) lung tumors, nor in the overall incidence of tumors or malignant tumors. This 21-month study was repeated in CD-1 mice, and no statistically or biologically significant differences were observed between treated and control mice of either sex for any type of tumor.
All genotoxicity tests performed on metoprolol tartrate (a dominant lethal study in mice, chromosome studies in somatic cells, a Salmonella/mammalian-microsome mutagenicity test, and a nucleus anomaly test in somatic interphase nuclei) and metoprolol succinate (a Salmonella/mammalian-microsome mutagenicity test) were negative.
No evidence of impaired fertility due to metoprolol tartrate was observed in a study performed in rats at doses up to 24 times, on a mg/m 2basis, the daily dose of 200 mg in a 60-kg patient.
7.1 Catecholamine Depleting Drugs and Monoamine Oxidate (mao) Inhibitors
Catecholamine depleting drugs (e.g., reserpine) and monoamine oxidase (MAO) inhibitors) may have an additive effect when given with beta-blocking agents. Observe patients treated with metoprolol plus a catecholamine depletor for evidence of hypotension or marked bradycardia, which may produce vertigo, syncope, or postural hypotension.
7.4 Digitalis, Clonidine, and Calcium Channel Blockers and Other Drugs That Decrease Heart Rate
Digitalis glycosides, clonidine, diltiazem and verapamil slow atrioventricular conduction and decrease heart rate. Concomitant administration of beta-blockers with these and other drugs known to decrease heart rate such as sphingosine-1-phosphate receptor modulators (e.g. fingolimod) may result in additive heart rate lowering effects.
If clonidine and metoprolol are coadministered, withdraw the metoprolol several days before the gradual withdrawal of clonidine because beta-blockers may exacerbate the rebound hypertension that can follow the withdrawal of clonidine. If replacing clonidine by beta-blocker therapy, delay the introduction of beta-blockers for several days after clonidine administration has stopped .
Structured Label Content
Section 42229-5 (42229-5)
Transition to Oral Metoprolol:
Following administration of Metoprolol tartrate Injection, transition patients to an oral formulation of metoprolol. See prescribing information for oral metoprolol for dose selection.
Section 43683-2 (43683-2)
|
Warnings and Precautions, Hypoglycemia ( 5.5) |
5/2024 |
Section 44425-7 (44425-7)
Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room Temperature.]. Do not freeze.
PROTECT FROM LIGHT.Retain in carton until time of use.
Discard unused portion.
11 Description (11 DESCRIPTION)
Metoprolol tartrate Injection, USP, is a selective beta 1-adrenoreceptor blocking agent, available in 5 mL vials for intravenous administration. Each vial contains a sterile solution of metoprolol tartrate USP, 5 mg, and sodium chloride USP, 45 mg. Metoprolol tartrate USP is (±)-1-(Isopropylamino)-3-[p-(2-methoxyethyl) phenoxy]-2-propanol (2:1) dextro-tartrate salt, and its structural formula is:
Metoprolol tartrate USP is a white, practically odorless, crystalline powder with a molecular weight of 684.83. It is very soluble in water; freely soluble in methylene chloride, in chloroform, and in alcohol; slightly soluble in acetone; and insoluble in ether.
5.1 Bradycardia
Bradycardia, including sinus pause, heart block, and cardiac arrest have occurred with the use of Metoprolol tartrate. Patients with first-degree atrioventricular block, sinus node dysfunction, or conduction disorders may be at increased risk. Monitor heart rate and rhythm in patients receiving Metoprolol tartrate. If severe bradycardia develops, reduce or stop Metoprolol tartrate.
7.2 Epinephrine
While taking beta-blockers, patients with a history of severe anaphylactic reactions to a variety of allergens may be more reactive to repeated challenge and may be unresponsive to the usual doses of epinephrine used to treat an allergic reaction.
5.5 Hypoglycemia
Beta-blockers may prevent early warning signs of hypoglycemia, such as tachycardia, and increase the risk for severe or prolonged hypoglycemia at any time during treatment, especially in patients with diabetes mellitus or children and patients who are fasting (i.e., surgery, not eating regularly, or are vomiting). If severe hypoglycemia occurs, patients should be instructed to seek emergency treatment.
5.2 Heart Failure
Worsening cardiac failure may occur during metoprolol use. If such symptoms occur, increase diuretics and restore clinical stability before administering the next dose of metoprolol [see Dosage and Administration (2)] . Such episodes do not preclude subsequent successful titration of oral metoprolol.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
In worldwide clinical trials of Metoprolol tartrate in myocardial infarction, where approximately 478 patients were over 65 years of age (0 over 75 years of age), no age-related differences in safety and effectiveness were found. Other reported clinical experience in myocardial infarction has not identified differences in response between the elderly and younger patients. However, greater sensitivity of some elderly individuals taking Metoprolol tartrate cannot be categorically ruled out. Therefore, in general, it is recommended that dosing proceed with caution in this population.
14 Clinical Studies (14 CLINICAL STUDIES)
In a large (1,395 patients randomized), double-blind, placebo-controlled clinical study, Metoprolol tartrate was shown to reduce 3-month mortality by 36% in patients with suspected or definite myocardial infarction.
Patients were randomized and treated as soon as possible after their arrival in the hospital, once their clinical condition had stabilized and their hemodynamic status had been carefully evaluated. Subjects were ineligible if they had hypotension, bradycardia, peripheral signs of shock, and/or more than minimal basal rales as signs of congestive heart failure. Initial treatment consisted of intravenous followed by oral administration of Metoprolol tartrate or placebo, given in a coronary care or comparable unit. Oral maintenance therapy with Metoprolol tartrate or placebo was then continued for 3 months. After this double-blind period, all patients were given Metoprolol tartrate and followed up to 1 year.
The median delay from the onset of symptoms to the initiation of therapy was 8 hours in both the Metoprolol tartrate- and placebo-treatment groups. Among patients treated with Metoprolol tartrate, there were comparable reductions in 3-month mortality for those treated early (≤8 hours) and those in whom treatment was started later. Significant reductions in the incidence of ventricular fibrillation and in chest pain following initial intravenous therapy were also observed with Metoprolol tartrate and were independent of the interval between onset of symptoms and initiation of therapy.
In this study, patients treated with metoprolol received the drug both very early (intravenously) and during a subsequent 3-month period, while placebo patients received no beta-blocker treatment for this period. The study thus was able to show a benefit from the overall metoprolol regimen but cannot separate the benefit of very early intravenous treatment from the benefit of later beta-blocker therapy. Nonetheless, because the overall regimen showed a clear beneficial effect on survival without evidence of an early adverse effect on survival, one acceptable dosage regimen is the precise regimen used in the trial. Because the specific benefit of very early treatment remains to be defined however, it is also reasonable to administer the drug orally to patients at a later time as is recommended for certain other beta-blockers.
4 Contraindications (4 CONTRAINDICATIONS)
Hypersensitivity to Metoprolol tartrate and related derivatives, or to any of the excipients; hypersensitivity to other beta-blockers (cross sensitivity between beta-blockers can occur).
Metoprolol tartrate is contraindicated in patients with a heart rate <45 beats/min; second- and third-degree heart block (unless a functioning pacemaker is present); significant first-degree heart block (P-R interval ≥0.24 sec); systolic blood pressure <100 mmHg; or decompensated cardiac failure.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described elsewhere in labeling:
- Worsening angina or myocardial infarction [see Warnings and Precautions (5)]
- Worsening heart failure [see Warnings and Precautions (5)]
- Worsening AV block [see Contraindications (4)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Catecholamine-depleting drugs may have an additive effect when given with beta-blocking agents. ( 7.1)
- Patients may be unresponsive to the usual doses of epinephrine used to treat allergic reaction. ( 7.2)
- CYP2D6 Inhibitors are likely to increase metoprolol concentration. ( 7.3)
- Concomitant use of glycosides, clonidine, and diltiazem and verapamil with beta-blockers can increase the risk of bradycardia. ( 7.4)
- Beta-blockers including metoprolol, may exacerbate the rebound hypertension that can follow the withdrawal of clonidine. ( 7.4)
5.4 Pheochromocytoma
If metoprolol tartrate is used in the setting of pheochromocytoma, it should be given in combination with an alpha blocker, and only after the alpha blocker has been initiated. Administration of beta-blockers alone in the setting of pheochromocytoma has been associated with a paradoxical increase in blood pressure due to the attenuation of beta-mediated vasodilatation in skeletal muscle.
12.2 Pharmacodynamics
When the drug was infused over a 10-minute period, in normal volunteers, maximum beta-blockade was achieved at approximately 20 minutes. Equivalent maximal beta-blocking effect is achieved with oral and intravenous doses in the ratio of approximately 2.5:1. There is a linear relationship between the log of plasma levels and reduction of exercise heart rate.
In several studies of patients with acute myocardial infarction, intravenous followed by oral administration of Metoprolol tartrate caused a reduction in heart rate, systolic blood pressure and cardiac output. Stroke volume, diastolic blood pressure and pulmonary artery end diastolic pressure remained unchanged.
12.5 Pharmacogenomics
CYP2D6 is absent in about 8% of Caucasians (poor metabolizers) and about 2% of most other populations. CYP2D6 can be inhibited by several drugs. Poor metabolizers of CYP2D6 will have increased (several-fold) metoprolol blood levels, decreasing metoprolol's cardioselectivity.
7.3 Cyp2d6 Inhibitors (7.3 CYP2D6 Inhibitors)
Drugs that are strong inhibitors of CYP2D6, such as quinidine, fluoxetine, paroxetine, and propafenone, were shown to double metoprolol concentrations. While there is no information about moderate or weak inhibitors, these too are likely to increase metoprolol concentration. Increases in plasma concentration decrease the cardioselectivity of metoprolol [see Clinical Pharmacology (12.3)]. Monitor patients closely, when the combination cannot be avoided.
8.6 Hepatic Impairment
No studies have been performed with metoprolol in patients with hepatic impairment. Because metoprolol is metabolized by the liver, metoprolol blood levels are likely to increase substantially with poor hepatic function. Therefore, initiate therapy at doses lower than those recommended for a given indication.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Metoprolol tartrate Injection is indicated in the treatment of definite or suspected acute myocardial infarction in hemodynamically stable patients to reduce cardiovascular mortality when used in conjunction with oral metoprolol maintenance therapy.
12.1 Mechanism of Action
Metoprolol is a beta 1-selective (cardioselective) adrenergic receptor blocking agent. This preferential effect is not absolute, however, and at higher plasma concentrations, metoprolol also inhibits beta 2-adrenoreceptors, chiefly located in the bronchial and vascular musculature.
Metoprolol has no intrinsic sympathomimetic activity, and membrane-stabilizing activity is detectable only at plasma concentrations much greater than required for beta-blockade. Animal and human experiments indicate that metoprolol slows the sinus rate and decreases AV nodal conduction.
The relative beta 1-selectivity of metoprolol has been confirmed by the following: (1) In normal subjects, metoprolol is unable to reverse the beta 2-mediated vasodilating effects of epinephrine. This contrasts with the effect of nonselective beta-blockers, which completely reverse the vasodilating effects of epinephrine. (2) In asthmatic patients, metoprolol reduces FEV 1and FVC significantly less than a nonselective beta-blocker, propranolol, at equivalent beta 1-receptor blocking doses.
The precise mechanism of action of Metoprolol tartrate in patients with suspected or definite myocardial infarction is not known.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
5.3 Bronchospastic Disease
PATIENTS WITH BRONCHOSPASTIC DISEASES, IN GENERAL, SHOULD NOT RECEIVE BETA-BLOCKERS because they can exacerbate bronchospasm. Because of its relative beta 1cardio-selectivity, however, metoprolol may be used in patients with bronchospastic disease for initial treatment of myocardial infarction. Bronchodilators, including beta 2-agonists, should be readily available or administered concomitantly [see Dosage and Administration (2)].
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Initiate treatment in a coronary care or similar unit immediately after the patient's hemodynamic condition has stabilized.
Begin treatment in this early phase with the intravenous administration of three bolus injections of 5 mg of Metoprolol tartrate each; give the injections at approximately 2-minute intervals. During the intravenous administration of Metoprolol tartrate, monitor blood pressure, heart rate, and electrocardiogram.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 5 mg metoprolol tartrate supplied in 5 mL Single-dose glass Fliptop Vial.
6.2 Post Marketing Experience (6.2 Post-Marketing Experience)
The following adverse reactions have been identified during post-approval use of metoprolol. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiovascular:Cold extremities, arterial insufficiency (usually of the Raynaud type), palpitations, peripheral edema, syncope, and hypotension.
Respiratory:Wheezing (bronchospasm), dyspnea.
Central Nervous System:Confusion, short-term memory loss, headache, nightmares, insomnia, nervousness, hallucinations.
Gastrointestinal:Nausea, dry mouth, constipation, flatulence, heartburn, hepatitis, vomiting.
Hypersensitive Reactions:Pruritus.
Miscellaneous:Musculoskeletal pain, arthritis, blurred vision, decreased libido, tinnitus, reversible alopecia, agranulocytosis, dry eyes, worsening of psoriasis, Peyronie's disease, photosensitivity.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Hepatic Impairment: Consider initiating metoprolol tartrate therapy at low doses while monitoring closely for adverse events. ( 8.6)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The adverse reaction information from clinical trials does, however, provide a basis for identifying the adverse events that appear to be related to drug use and for approximating rates.
5.6 Peripheral Vascular Disease
Beta-blockers can precipitate or aggravate symptoms of arterial insufficiency in patients with peripheral vascular disease.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients (1) to avoid operating automobiles and machinery or engaging in other tasks requiring alertness until the patient's response to therapy with Metoprolol tartrate has been determined; (2) to contact the physician if any difficulty in breathing occurs; (3) to inform the physician or dentist before any type of surgery that he or she is taking Metoprolol tartrate.
Risk of Hypoglycemia
Inform patients or caregivers that there is a risk of hypoglycemia when Metoprolol tartrate Injection is given to patients who are fasting or who are vomiting. Monitor for signs of hypoglycemia [see Warnings and Precautions (5.5)] .
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Metoprolol tartrate Injection, USP is available as:
| Unit of Sale | Concentration |
|---|---|
|
NDC 84549-778-05 Single-dose glass Fliptop Vial |
5 mg/5 mL (1 mg/mL) |
7.5 Drugs That Decrease Blood Pressure (7.5 Drugs that Decrease Blood Pressure)
Concomitant administration of beta-blockers with other drugs known to decrease blood pressure may result in an enhanced hypotensive effect.
Principal Display Panel 5 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 5 mL Vial Label)
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have been conducted to evaluate the carcinogenic potential of metoprolol tartrate. In 2-year studies in rats at three oral dosage levels of up to 800 mg/kg/day (39 times, on a mg/m 2basis, the daily dose of 200 mg for a 60 kg patient), there was no increase in the development of spontaneously occurring benign or malignant neoplasms of any type. The only histologic changes that appeared to be drug related were an increased incidence of generally mild focal accumulation of foamy macrophages in pulmonary alveoli and a slight increase in biliary hyperplasia. In a 21-month study in Swiss albino mice at three oral dosage levels of up to 750 mg/kg/day (18 times, on a mg/m 2basis, the daily dose of 200 mg for a 60-kg patient), benign lung tumors (small adenomas) occurred more frequently in female mice receiving the highest dose than in untreated control animals. There was no increase in malignant or total (benign plus malignant) lung tumors, nor in the overall incidence of tumors or malignant tumors. This 21-month study was repeated in CD-1 mice, and no statistically or biologically significant differences were observed between treated and control mice of either sex for any type of tumor.
All genotoxicity tests performed on metoprolol tartrate (a dominant lethal study in mice, chromosome studies in somatic cells, a Salmonella/mammalian-microsome mutagenicity test, and a nucleus anomaly test in somatic interphase nuclei) and metoprolol succinate (a Salmonella/mammalian-microsome mutagenicity test) were negative.
No evidence of impaired fertility due to metoprolol tartrate was observed in a study performed in rats at doses up to 24 times, on a mg/m 2basis, the daily dose of 200 mg in a 60-kg patient.
7.1 Catecholamine Depleting Drugs and Monoamine Oxidate (mao) Inhibitors (7.1 Catecholamine Depleting Drugs and Monoamine Oxidate (MAO) Inhibitors)
Catecholamine depleting drugs (e.g., reserpine) and monoamine oxidase (MAO) inhibitors) may have an additive effect when given with beta-blocking agents. Observe patients treated with metoprolol plus a catecholamine depletor for evidence of hypotension or marked bradycardia, which may produce vertigo, syncope, or postural hypotension.
7.4 Digitalis, Clonidine, and Calcium Channel Blockers and Other Drugs That Decrease Heart Rate (7.4 Digitalis, Clonidine, and Calcium Channel Blockers and Other Drugs that Decrease Heart Rate)
Digitalis glycosides, clonidine, diltiazem and verapamil slow atrioventricular conduction and decrease heart rate. Concomitant administration of beta-blockers with these and other drugs known to decrease heart rate such as sphingosine-1-phosphate receptor modulators (e.g. fingolimod) may result in additive heart rate lowering effects.
If clonidine and metoprolol are coadministered, withdraw the metoprolol several days before the gradual withdrawal of clonidine because beta-blockers may exacerbate the rebound hypertension that can follow the withdrawal of clonidine. If replacing clonidine by beta-blocker therapy, delay the introduction of beta-blockers for several days after clonidine administration has stopped .
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:46.002865 · Updated: 2026-03-14T22:34:21.968394