These Highlights Do Not Include All The Information Needed To Use Zembrace®
3eb49807-fa9d-4a0d-9a3c-76de5b23b01b
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
ZEMBRACE SymTouch is indicated for the acute treatment of migraine with or without aura in adults.
Indications and Usage
ZEMBRACE SymTouch is indicated for the acute treatment of migraine with or without aura in adults.
Dosage and Administration
For subcutaneous use only. ( 2.1 ) Acute treatment of migraine: 3 mg Single dose. ( 2.1 ) Maximum dose in a 24-hour period: 12 mg. Separate doses by at least 1 hour. ( 2.1 )
Warnings and Precautions
Myocardial ischemia/infarction and Prinzmetal's angina: Perform cardiac evaluation in patients with multiple cardiovascular risk factors. ( 5.1 ) Arrhythmias: Discontinue ZEMBRACE SymTouch if occurs. ( 5.2 ) Chest/throat/neck/jaw pain, tightness, pressure, or heaviness: Generally, not associated with myocardial ischemia; evaluate for coronary artery disease in patients at high risk. ( 5.3 ) Cerebral hemorrhage, subarachnoid hemorrhage, and stroke: Discontinue ZEMBRACE SymTouch if occurs. ( 5.4 ) Gastrointestinal ischemia and reactions, peripheral vasospastic reactions: Discontinue ZEMBRACE SymTouch if occurs. ( 5.5 ) Medication overuse headache: Detoxification may be necessary. ( 5.6 ) Serotonin syndrome: Discontinue ZEMBRACE SymTouch if occurs. ( 5.7 ) Seizures: Use with caution in patients with epilepsy or a lowered seizure threshold. ( 5.10 )
Contraindications
ZEMBRACE SymTouch injection is contraindicated in patients with: Ischemic coronary artery disease (CAD) (angina pectoris, history of myocardial infarction, or documented silent ischemia) or coronary artery vasospasm, including Prinzmetal's angina [see Warnings and Precautions (5.1) ] . Wolff-Parkinson-White syndrome or arrhythmias associated with other cardiac accessory conduction pathway disorders [see Warnings and Precautions (5.2) ] . History of stroke or transient ischemic attack (TIA) or history of hemiplegic or basilar migraine because these patients are at a higher risk of stroke [see Warnings and Precautions (5.4) ] . Peripheral vascular disease [see Warnings and Precautions (5.5) ] . Ischemic bowel disease [see Warnings and Precautions (5.5) ] . Uncontrolled hypertension [see Warnings and Precautions (5.8) ] . Recent (i.e., within 24 hours) use of ergotamine-containing medication, ergot-type medication (such as dihydroergotamine or methysergide), or another 5-hydroxytryptamine1 (5-HT 1 ) agonist [see Drug Interactions (7.1 , 7.3) ] . Concurrent administration of an MAO-A inhibitor or recent (within 2 weeks) use of an MAO-A inhibitor [see Drug Interactions (7.2) and Clinical Pharmacology (12.3) ] . Known hypersensitivity to sumatriptan (angioedema and anaphylaxis seen) [see Warnings and Precautions (5.9) ] . Severe hepatic impairment [see Clinical Pharmacology (12.3) ] .
Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling: Myocardial ischemia, myocardial infarction, and Prinzmetal's angina [see Warnings and Precautions (5.1) ] Arrhythmias [see Warnings and Precautions (5.2) ] Chest, throat, neck, and/or jaw pain/tightness/pressure [see Warnings and Precautions (5.3) ] Cerebrovascular events [see Warnings and Precautions (5.4) ] Other vasospasm reactions [see Warnings and Precautions (5.5) ] Medication overuse headache [see Warnings and Precautions (5.6) ] Serotonin syndrome [see Warnings and Precautions (5.7) ] Increase in blood pressure [see Warnings and Precautions (5.8) ] Hypersensitivity reactions [see Contraindications (4) , Warnings and Precautions (5.9) ] Seizures [see Warnings and Precautions (5.10) ]
Storage and Handling
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F). Protect from light.
How Supplied
ZEMBRACE ® SymTouch ® 3 mg/0.5 mL Injection contains sumatriptan as the succinate salt and is supplied as a clear, colorless to pale yellow, sterile, nonpyrogenic solution in a prefilled, ready-to-use, single dose, disposable auto-injector unit (NDC # 70792-809-89). Each carton contains 4 units (NDC # 70792-809-38) and a Patient Information and Instructions for Use leaflet.
Medication Information
Warnings and Precautions
Myocardial ischemia/infarction and Prinzmetal's angina: Perform cardiac evaluation in patients with multiple cardiovascular risk factors. ( 5.1 ) Arrhythmias: Discontinue ZEMBRACE SymTouch if occurs. ( 5.2 ) Chest/throat/neck/jaw pain, tightness, pressure, or heaviness: Generally, not associated with myocardial ischemia; evaluate for coronary artery disease in patients at high risk. ( 5.3 ) Cerebral hemorrhage, subarachnoid hemorrhage, and stroke: Discontinue ZEMBRACE SymTouch if occurs. ( 5.4 ) Gastrointestinal ischemia and reactions, peripheral vasospastic reactions: Discontinue ZEMBRACE SymTouch if occurs. ( 5.5 ) Medication overuse headache: Detoxification may be necessary. ( 5.6 ) Serotonin syndrome: Discontinue ZEMBRACE SymTouch if occurs. ( 5.7 ) Seizures: Use with caution in patients with epilepsy or a lowered seizure threshold. ( 5.10 )
Indications and Usage
ZEMBRACE SymTouch is indicated for the acute treatment of migraine with or without aura in adults.
Dosage and Administration
For subcutaneous use only. ( 2.1 ) Acute treatment of migraine: 3 mg Single dose. ( 2.1 ) Maximum dose in a 24-hour period: 12 mg. Separate doses by at least 1 hour. ( 2.1 )
Contraindications
ZEMBRACE SymTouch injection is contraindicated in patients with: Ischemic coronary artery disease (CAD) (angina pectoris, history of myocardial infarction, or documented silent ischemia) or coronary artery vasospasm, including Prinzmetal's angina [see Warnings and Precautions (5.1) ] . Wolff-Parkinson-White syndrome or arrhythmias associated with other cardiac accessory conduction pathway disorders [see Warnings and Precautions (5.2) ] . History of stroke or transient ischemic attack (TIA) or history of hemiplegic or basilar migraine because these patients are at a higher risk of stroke [see Warnings and Precautions (5.4) ] . Peripheral vascular disease [see Warnings and Precautions (5.5) ] . Ischemic bowel disease [see Warnings and Precautions (5.5) ] . Uncontrolled hypertension [see Warnings and Precautions (5.8) ] . Recent (i.e., within 24 hours) use of ergotamine-containing medication, ergot-type medication (such as dihydroergotamine or methysergide), or another 5-hydroxytryptamine1 (5-HT 1 ) agonist [see Drug Interactions (7.1 , 7.3) ] . Concurrent administration of an MAO-A inhibitor or recent (within 2 weeks) use of an MAO-A inhibitor [see Drug Interactions (7.2) and Clinical Pharmacology (12.3) ] . Known hypersensitivity to sumatriptan (angioedema and anaphylaxis seen) [see Warnings and Precautions (5.9) ] . Severe hepatic impairment [see Clinical Pharmacology (12.3) ] .
Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling: Myocardial ischemia, myocardial infarction, and Prinzmetal's angina [see Warnings and Precautions (5.1) ] Arrhythmias [see Warnings and Precautions (5.2) ] Chest, throat, neck, and/or jaw pain/tightness/pressure [see Warnings and Precautions (5.3) ] Cerebrovascular events [see Warnings and Precautions (5.4) ] Other vasospasm reactions [see Warnings and Precautions (5.5) ] Medication overuse headache [see Warnings and Precautions (5.6) ] Serotonin syndrome [see Warnings and Precautions (5.7) ] Increase in blood pressure [see Warnings and Precautions (5.8) ] Hypersensitivity reactions [see Contraindications (4) , Warnings and Precautions (5.9) ] Seizures [see Warnings and Precautions (5.10) ]
Storage and Handling
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F). Protect from light.
How Supplied
ZEMBRACE ® SymTouch ® 3 mg/0.5 mL Injection contains sumatriptan as the succinate salt and is supplied as a clear, colorless to pale yellow, sterile, nonpyrogenic solution in a prefilled, ready-to-use, single dose, disposable auto-injector unit (NDC # 70792-809-89). Each carton contains 4 units (NDC # 70792-809-38) and a Patient Information and Instructions for Use leaflet.
Description
ZEMBRACE SymTouch is indicated for the acute treatment of migraine with or without aura in adults.
Section 42229-5
Limitations of Use:
- Use only if a clear diagnosis of migraine has been established. If a patient has no response to the first migraine attack treated with ZEMBRACE SymTouch, reconsider the diagnosis before ZEMBRACE SymTouch is administered to treat any subsequent attacks.
- ZEMBRACE SymTouch injection is not indicated for the prevention of migraine attacks.
Section 42230-3
| Patient Information ZEMBRACE® SymTouch® (Zem-brace Sim-Touch) (sumatriptan succinate) Injection |
|
|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 11/2023 |
|
What is the most important information I should know about ZEMBRACE SymTouch?
ZEMBRACE SymTouch can cause serious side effects, including: Heart attack and other heart problems. Heart problems may lead to death. Stop taking ZEMBRACE SymTouch and get emergency medical help right away if you have any of the following symptoms of a heart attack:
|
|
|
What is ZEMBRACE SymTouch?
ZEMBRACE SymTouch is a prescription medicine used to treat acute migraine headaches with or without aura in adults who have been diagnosed with migraine. ZEMBRACE SymTouch is not used to treat other types of headaches such as hemiplegic (that make you unable to move on one side of your body) or basilar (rare form of migraine with aura) migraines. ZEMBRACE SymTouch is not used to prevent or decrease the number of migraines you have. It is not known if ZEMBRACE SymTouch is safe and effective in children under 18 years of age. |
|
|
Who should not take ZEMBRACE SymTouch? Do not take ZEMBRACE SymTouch if you have:
|
|
|
What should I tell my healthcare provider before taking ZEMBRACE SymTouch?
Before taking ZEMBRACE SymTouch, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take anti-depressant medicines called:
Know the medicines you take. Keep a list of them to show your healthcare provider or pharmacist when you get a new medicine. |
|
How should I take ZEMBRACE SymTouch?
|
|
|
What should I avoid while taking ZEMBRACE SymTouch?
ZEMBRACE SymTouch can cause dizziness, weakness, or drowsiness. If you have these symptoms, do not drive a car, use machinery, or do anything where you need to be alert. |
|
|
What are the possible side effects of ZEMBRACE SymTouch?
See "What is the most important information I should know about ZEMBRACE SymTouch?" ZEMBRACE SymTouch may cause serious side effects, including:
These are not all the possible side effects of ZEMBRACE SymTouch. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store ZEMBRACE SymTouch?
|
|
|
General information about the safe and effective use of ZEMBRACE SymTouch.
Medicines are sometimes prescribed for purposes other than those listed in Patient Information leaflets. Do not use ZEMBRACE SymTouch for a condition for which it was not prescribed. Do not give ZEMBRACE SymTouch to other people, even if they have the same symptoms you have. It may harm them. This Patient Information leaflet summarizes the most important information about ZEMBRACE SymTouch. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about ZEMBRACE SymTouch that is written for healthcare professionals. For more information, go to www.Zembrace.com or call 1-888-869-7633 (1-888-TNXPMED). |
|
|
What are the ingredients in ZEMBRACE SymTouch Injection?
Active ingredient: sumatriptan succinate Inactive ingredients: sodium chloride, water for injection Manufactured for TONIX MEDICINES, INC. Chatham, NJ 07928 ZEMBRACE and SymTouch are registered trademarks of Tonix Medicines, Inc. This product may be covered by one or more U.S. patent(s). |
Section 59845-8
| Instructions for Use ZEMBRACE® SymTouch® (Zem-brace Sim-Touch) (sumatriptan succinate) Injection |
||
|---|---|---|
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. | Revised: 11/2023 | |
| Read this Instructions for Use before you start to use the ZEMBRACE® SymTouch®. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment. You and your healthcare provider should talk about ZEMBRACE SymTouch when you start taking it and at regular checkups. The ZEMBRACE SymTouch Auto-injector is a single-use, pre-filled injection device with the exact dose you need. The device should be used only 1 time and then discarded. |
Important Precautions Keep out of reach of children. Do not use after the expiration date has passed. Do not store or expose to high temperatures. Do not freeze. Do not remove the Red Cap until you are ready to inject. Do not use if dropped after removing the Red Cap. Do not put or press thumb, fingers or hand over the Yellow Needle Guard. |
|
|
Step 1 – Inspect Auto-injector and Gather Supplies 1A – Remove Auto-injector from the carton |
||
| 1B – Check the Expiration Date on the Auto-injector | Do not use if the expiration date has passed. | |
|
1C – Inspect the medicine through the Medicine Viewing Window
Inspect the appearance of ZEMBRACE SymTouch Auto-injector medicine through the medicine window. It must be a clear, colorless to pale yellow solution. |
||
| Do not inject the medicine if the solution looks discolored or cloudy or contains lumps, flakes, or particles. | ||
1D – Gather the following supplies
|
||
|
Step 2 – Choose and Prepare Your Injection Site 2A – Choose your injection site The recommended injection sites are the sides of the thighs and the arms. |
||
| Caution: Do not inject where your skin is scarred, bruised, tender, red or near any open wound. | ||
| 2B – Clean the injection site with an alcohol swab | Do not touch injection site area after cleaning. | |
|
2C – Remove the Red Cap
Hold the body of the Auto-injector in one hand and pull the Red Cap straight off. . |
||
| Warning: Do not put or press thumb, fingers or hand over the Yellow Needle Guard, doing so may result in a needle stick injury | ||
|
Step 3 – Inject the Medicine 3A – Place Auto-injector straight on your injection site Place the Auto-injector straight on your injection site (90 degrees) with the Yellow Needle Guard end gently pressed against your skin. |
||
|
3B – Perform Injection
Press and hold the Auto-injector down against your skin. You will hear the first "Click 1" as the injection starts. Continue to hold the device down until you hear the second "Click 2." Wait 5 seconds before you remove the Auto-injector! After "Click 2," continue to hold down and slowly count to 5 to make sure you receive your full dose. |
||
| Important: To receive your full dose, always hold the Auto-injector down on your injection site for additional time after you hear the second click. This allows all of the medicine to be delivered. | ||
|
3C – Remove Auto-injector from skin
Remove the Auto-injector by lifting it straight away from your skin. The Yellow Needle Guard will drop down and lock over the needle. |
||
|
Step 4 – Confirm Injection and Dispose the Auto-injector 4A – Confirm Red Color in Medicine Viewing Window Look to confirm that the Red Plunger Rod has filled the Medicine Viewing Window. This means you received the full dose. |
||
|
Caution: Call your healthcare provider if the Red Plunger Rod has not filled the Medicine Viewing Window. This may mean that only a partial dose was delivered. Do not try to reuse the Auto-injector. |
||
|
4B – Disposal of the Auto-injector
Put your used Auto-injector, caps, needles, and sharps in an FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) loose needles and syringes in your household trash. Do not recycle your used sharps disposal container. If you do not have a FDA-cleared sharps disposal container, you may use a household container that is: made of a heavy-duty plastic; can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out; upright and stable during use; leak-resistant; and properly labeled to warn of hazardous waste inside the container. When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal. Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container. If needed, make sure you get a refill of your Auto-injector. |
||
|
4C – Treat Injection Site
Treat the injection site with a cotton ball, gauze pad or bandage, as needed. Do not rub the injection site. Manufactured for TONIX MEDICINES, INC. Chatham, NJ 07928 ZEMBRACE and SymTouch are registered trademarks of Tonix Medicines, Inc. This product may be covered by one or more U.S. patent(s). |
10 Overdosage
Coronary vasospasm was observed after intravenous administration of sumatriptan injection [see Contraindications (4)]. Overdoses would be expected from animal data (dogs at 0.1 g/kg, rats at 2 g/kg) to possibly cause convulsions, tremor, inactivity, erythema of the extremities, reduced respiratory rate, cyanosis, ataxia, mydriasis, injection site reactions (desquamation, hair loss, and scab formation), and paralysis.
The elimination half-life of sumatriptan is about 2 hours [see Clinical Pharmacology (12.3)], and therefore monitoring of patients after overdose with ZEMBRACE SymTouch injection should continue for at least 10 hours or while symptoms or signs persist.
It is unknown what effect hemodialysis or peritoneal dialysis has on the serum concentrations of sumatriptan.
5.10 Seizures
Seizures have been reported following administration of sumatriptan. Some have occurred in patients with either a history of seizures or concurrent conditions predisposing to seizures. There are also reports in patients where no such predisposing factors are apparent. ZEMBRACE SymTouch injection should be used with caution in patients with a history of epilepsy or conditions associated with a lowered seizure threshold.
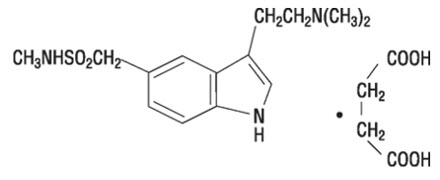
11 Description
ZEMBRACE SymTouch injection contains sumatriptan succinate, a selective 5-HT1B/1D receptor agonist. Sumatriptan succinate is chemically designated as 3-[2-(dimethylamino) ethyl]-N-methyl-indole-5-methanesulfonamide succinate (1:1), and it has the following structure:
The empirical formula is C14H21N3O2S∙C4H6O4, representing a molecular weight of 413.5. Sumatriptan succinate is a white to off-white powder that is readily soluble in water and in saline.
ZEMBRACE SymTouch is a clear, colorless to pale yellow, sterile, nonpyrogenic solution for subcutaneous injection. Each 0.5 mL of ZEMBRACE SymTouch contains 4.2 mg of sumatriptan succinate equivalent to 3 mg of sumatriptan (base) and 4.15 mg of sodium chloride, USP in Water for Injection, USP.
The pH range of solution is approximately 4.2 to 5.3 and the osmolality of injection is approximately 291 mOsmol (275 to 315 mOsmol).
5.2 Arrhythmias
Life-threatening disturbances of cardiac rhythm, including ventricular tachycardia and ventricular fibrillation leading to death, have been reported within a few hours following the administration of 5-HT1 agonists. Discontinue ZEMBRACE SymTouch injection if these disturbances occur. ZEMBRACE SymTouch injection is contraindicated in patients with Wolff-Parkinson-White syndrome or arrhythmias associated with other cardiac accessory conduction pathway disorders.
7.3 Other 5 Ht1
Because their vasospastic effects may be additive, coadministration of ZEMBRACE SymTouch injection and other 5-HT1 agonists (e.g., triptans) within 24 hours of each other is contraindicated.
16.1 How Supplied
- ZEMBRACE® SymTouch® 3 mg/0.5 mL Injection contains sumatriptan as the succinate salt and is supplied as a clear, colorless to pale yellow, sterile, nonpyrogenic solution in a prefilled, ready-to-use, single dose, disposable auto-injector unit (NDC # 70792-809-89).
- Each carton contains 4 units (NDC # 70792-809-38) and a Patient Information and Instructions for Use leaflet.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established. ZEMBRACE SymTouch injection is not recommended for use in patients younger than 18 years of age.
Two controlled clinical trials evaluated sumatriptan nasal spray (5 to 20 mg) in 1,248 pediatric migraineurs 12 to 17 years of age who treated a single attack. The trials did not establish the efficacy of sumatriptan nasal spray compared with placebo in the treatment of migraine in pediatric patients. Adverse reactions observed in these clinical trials were similar in nature to those reported in clinical trials in adults.
Five controlled clinical trials (2 single-attack trials, 3 multiple-attack trials) evaluating oral sumatriptan (25 to 100 mg) in pediatric subjects 12 to 17 years of age enrolled a total of 701 pediatric migraineurs. These trials did not establish the efficacy of oral sumatriptan compared with placebo in the treatment of migraine in pediatric patients. Adverse reactions observed in these clinical trials were similar in nature to those reported in clinical trials in adults. The frequency of all adverse reactions in these patients appeared to be both dose- and age-dependent, with younger patients reporting reactions more commonly than older pediatric patients.
Post-marketing experience documents that serious adverse reactions have occurred in the pediatric population after use of subcutaneous, oral, and/or intranasal sumatriptan. These reports include reactions similar in nature to those reported rarely in adults, including stroke, visual loss, and death. A myocardial infarction has been reported in a 14-year-old male following the use of oral sumatriptan; clinical signs occurred within 1 day of drug administration. Clinical data to determine the frequency of serious adverse reactions in pediatric patients who might receive subcutaneous, oral, or intranasal sumatriptan are not presently available.
8.5 Geriatric Use
Clinical trials of sumatriptan injection did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger patients. Other reported clinical experience has not identified differences in responses between the elderly and younger subjects. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
A cardiovascular evaluation is recommended for geriatric patients who have other cardiovascular risk factors (e.g., diabetes, hypertension, smoking, obesity, strong family history of CAD) prior to receiving ZEMBRACE SymTouch injection [see Warnings and Precautions (5.1)].
4 Contraindications
ZEMBRACE SymTouch injection is contraindicated in patients with:
- Ischemic coronary artery disease (CAD) (angina pectoris, history of myocardial infarction, or documented silent ischemia) or coronary artery vasospasm, including Prinzmetal's angina [see Warnings and Precautions (5.1)].
- Wolff-Parkinson-White syndrome or arrhythmias associated with other cardiac accessory conduction pathway disorders [see Warnings and Precautions (5.2)].
- History of stroke or transient ischemic attack (TIA) or history of hemiplegic or basilar migraine because these patients are at a higher risk of stroke [see Warnings and Precautions (5.4)].
- Peripheral vascular disease [see Warnings and Precautions (5.5)].
- Ischemic bowel disease [see Warnings and Precautions (5.5)].
- Uncontrolled hypertension [see Warnings and Precautions (5.8)].
- Recent (i.e., within 24 hours) use of ergotamine-containing medication, ergot-type medication (such as dihydroergotamine or methysergide), or another 5-hydroxytryptamine1 (5-HT1) agonist [see Drug Interactions (7.1, 7.3)].
- Concurrent administration of an MAO-A inhibitor or recent (within 2 weeks) use of an MAO-A inhibitor [see Drug Interactions (7.2) and Clinical Pharmacology (12.3)].
- Known hypersensitivity to sumatriptan (angioedema and anaphylaxis seen) [see Warnings and Precautions (5.9)].
- Severe hepatic impairment [see Clinical Pharmacology (12.3)].
6 Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling:
- Myocardial ischemia, myocardial infarction, and Prinzmetal's angina [see Warnings and Precautions (5.1)]
- Arrhythmias [see Warnings and Precautions (5.2)]
- Chest, throat, neck, and/or jaw pain/tightness/pressure [see Warnings and Precautions (5.3)]
- Cerebrovascular events [see Warnings and Precautions (5.4)]
- Other vasospasm reactions [see Warnings and Precautions (5.5)]
- Medication overuse headache [see Warnings and Precautions (5.6)]
- Serotonin syndrome [see Warnings and Precautions (5.7)]
- Increase in blood pressure [see Warnings and Precautions (5.8)]
- Hypersensitivity reactions [see Contraindications (4), Warnings and Precautions (5.9)]
- Seizures [see Warnings and Precautions (5.10)]
12.3 Pharmacokinetics
After a single 3 mg dose, ZEMBRACE SymTouch was bioequivalent to IMITREX subcutaneous injection.
2.1 Dosing Information
The recommended dose of ZEMBRACE SymTouch is 3 mg injected subcutaneously.
The maximum cumulative injected dose that may be given in 24 hours is 12 mg, with doses of ZEMBRACE SymTouch separated by at least 1 hour. ZEMBRACE SymTouch may also be given at least 1 hour following a dose of another sumatriptan product.
5.7 Serotonin Syndrome
Serotonin syndrome may occur with ZEMBRACE SymTouch injection, particularly during co-administration with selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), and MAO inhibitors [see Drug Interactions (7.4)]. Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, coma), autonomic instability (e.g., tachycardia, labile blood pressure, hyperthermia), neuromuscular aberrations (e.g., hyperreflexia, incoordination), and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). The onset of symptoms usually occurs within minutes to hours of receiving a new or a greater dose of a serotonergic medication. Discontinue ZEMBRACE SymTouch injection if serotonin syndrome is suspected.
1 Indications and Usage
ZEMBRACE SymTouch is indicated for the acute treatment of migraine with or without aura in adults.
12.1 Mechanism of Action
Sumatriptan binds with high affinity to human cloned 5-HT1B/1D receptors. Sumatriptan presumably exerts its therapeutic effects in the treatment of migraine headache through agonist effects at the 5-HT1B/1D receptors on intracranial blood vessels and sensory nerves of the trigeminal system, which result in cranial vessel constriction and inhibition of pro-inflammatory neuropeptide release.
16.2 Storage and Handling
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F). Protect from light.
5 Warnings and Precautions
- Myocardial ischemia/infarction and Prinzmetal's angina: Perform cardiac evaluation in patients with multiple cardiovascular risk factors. (5.1)
- Arrhythmias: Discontinue ZEMBRACE SymTouch if occurs. (5.2)
- Chest/throat/neck/jaw pain, tightness, pressure, or heaviness: Generally, not associated with myocardial ischemia; evaluate for coronary artery disease in patients at high risk. (5.3)
- Cerebral hemorrhage, subarachnoid hemorrhage, and stroke: Discontinue ZEMBRACE SymTouch if occurs. (5.4)
- Gastrointestinal ischemia and reactions, peripheral vasospastic reactions: Discontinue ZEMBRACE SymTouch if occurs. (5.5)
- Medication overuse headache: Detoxification may be necessary. (5.6)
- Serotonin syndrome: Discontinue ZEMBRACE SymTouch if occurs. (5.7)
- Seizures: Use with caution in patients with epilepsy or a lowered seizure threshold. (5.10)
5.4 Cerebrovascular Events
Cerebral hemorrhage, subarachnoid hemorrhage, and stroke have occurred in patients treated with 5-HT1 agonists, and some have resulted in fatalities. In a number of cases, it appears possible that the cerebrovascular events were primary, the 5-HT1 agonist having been administered in the incorrect belief that the symptoms experienced were a consequence of migraine when they were not. Also, patients with migraine may be at increased risk of certain cerebrovascular events (e.g., stroke, hemorrhage, TIA). Discontinue ZEMBRACE SymTouch injection if a cerebrovascular event occurs.
Before treating headaches in patients not previously diagnosed with migraine or in patients who present with atypical symptoms, exclude other potentially serious neurological conditions. ZEMBRACE SymTouch injection is contraindicated in patients with a history of stroke or TIA.
7.1 Ergot Containing Drugs
Ergot-containing drugs have been reported to cause prolonged vasospastic reactions. Because these effects may be additive, use of ergotamine-containing or ergot-type medications (like dihydroergotamine or methysergide) and ZEMBRACE SymTouch within 24 hours of each other is contraindicated.
2 Dosage and Administration
3 Dosage Forms and Strengths
Injection: 3 mg sumatriptan in 0.5 mL prefilled, ready-to-use, single dose, disposable auto-injector.
5.5 Other Vasospasm Reactions
ZEMBRACE SymTouch injection may cause non-coronary vasospastic reactions, such as peripheral vascular ischemia, gastrointestinal vascular ischemia and infarction (presenting with abdominal pain and bloody diarrhea), splenic infarction, and Raynaud's syndrome. In patients who experience symptoms or signs suggestive of non-coronary vasospasm reaction following the use of any 5-HT1 agonist, rule out a vasospastic reaction before receiving additional ZEMBRACE SymTouch injections.
Reports of transient and permanent blindness and significant partial vision loss have been reported with the use of 5-HT1 agonists. Since visual disorders may be part of a migraine attack, a causal relationship between these events and the use of 5-HT1 agonists have not been clearly established.
6.2 Post Marketing Experience
The following adverse reactions have been identified during post-approval use of sumatriptan tablets, sumatriptan nasal spray, and sumatriptan injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiovascular
Hypotension, palpitations
Neurological
Dystonia, tremor
8 Use in Specific Populations
- Pregnancy: Based on animal data, may cause fetal harm. (8.1)
5.8 Increase in Blood Pressure
Significant elevation in blood pressure, including hypertensive crisis with acute impairment of organ systems, has been reported on rare occasions in patients treated with 5-HT1 agonists, including patients without a history of hypertension. Monitor blood pressure in patients treated with ZEMBRACE SymTouch. ZEMBRACE SymTouch injection is contraindicated in patients with uncontrolled hypertension.
5.9 Hypersensitivity Reactions
Hypersensitivity reactions, including angioedema and anaphylaxis, have occurred in patients receiving sumatriptan. Such reactions can be life-threatening or fatal. In general, anaphylactic reactions to drugs are more likely to occur in individuals with a history of sensitivity to multiple allergens. ZEMBRACE SymTouch injection is contraindicated in patients with a history of hypersensitivity reaction to sumatriptan.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.6 Medication Overuse Headache
Overuse of acute migraine drugs (e.g., ergotamine, triptans, opioids, combination of these drugs for 10 or more days per month) may lead to exacerbation of headache (medication overuse headache). Medication overuse headache may present as migraine-like daily headaches, or as a marked increase in frequency of migraine attacks. Detoxification of patients, including withdrawal of the overused drugs, and treatment of withdrawal symptoms (which often includes a transient worsening of headache) may be necessary.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
7.2 Monoamine Oxidase A Inhibitors
MAO-A inhibitors increase systemic exposure by 2-fold. Therefore, the use of ZEMBRACE SymTouch injection in patients receiving MAO-A inhibitors is contraindicated [see Clinical Pharmacology (12.3)].
2.2 Administration Using Zembrace Symtouch
ZEMBRACE SymTouch is available as a prefilled, ready-to-use, single dose, disposable auto-injector containing 3 mg sumatriptan. With ZEMBRACE SymTouch, the needle penetrates approximately ¼ inch (6 mm). The injection is intended to be given subcutaneously. Do not administer by any other route. Instruct patients on the proper use of ZEMBRACE SymTouch and direct them to use injection sites with an adequate skin and subcutaneous thickness to accommodate the length of the needle.
Principal Display Panel 3 Mg Syringe Blister Pack Carton
NDC 70792-809-38
Rx only
TONIX
MEDICINES
Zembrace® SYMTOUCH®
(sumatriptan injection)
3 mg
For subcutaneous use only.
Each auto-injector contains a prefilled syringe with 0.5 mL of
sterile solution delivering 4.2 mg of sumatriptan succinate
equivalent to 3 mg of sumatriptan base, 4.15 mg sodium
chloride and water for injection.
See package insert for dosage information.
Store at 20° to 25°C (68° to 77°F).
Protect from light.
Manufactured for
TONIX MEDICINES, INC.
Chatham, NJ 07928
ZEMBRACE and SymTouch are registered trademarks of
Tonix Medicines, Inc.
5.3 Chest, Throat, Neck, And/or Jaw Pain/tightness/pressure
Sensations of tightness, pain, pressure, and heaviness in the precordium, throat, neck, and jaw commonly occur after treatment with sumatriptan injection and are usually non-cardiac in origin. However, perform a cardiac evaluation if these patients are at high cardiac risk. The use of ZEMBRACE SymTouch injection is contraindicated in patients with CAD and those with Prinzmetal's variant angina.
5.1 Myocardial Ischemia, Myocardial Infarction, and Prinzmetal's Angina
The use of ZEMBRACE SymTouch injection is contraindicated in patients with ischemic or vasospastic CAD. There have been rare reports of serious cardiac adverse reactions, including acute myocardial infarction, occurring within a few hours following administration of sumatriptan injection. Some of these reactions occurred in patients without known CAD. 5-HT1 agonists, including ZEMBRACE SymTouch injection, may cause coronary artery vasospasm (Prinzmetal's angina), even in patients without a history of CAD.
Perform a cardiovascular evaluation in triptan-naive patients who have multiple cardiovascular risk factors (e.g., increased age, diabetes, hypertension, smoking, obesity, strong family history of CAD) prior to receiving ZEMBRACE SymTouch injection. If there is evidence of CAD or coronary artery vasospasm, ZEMBRACE SymTouch injection is contraindicated. For patients with multiple cardiovascular risk factors who have a negative cardiovascular evaluation, consider administering the first dose of ZEMBRACE SymTouch injection in a medically supervised setting and performing an electrocardiogram (ECG) immediately following ZEMBRACE SymTouch injection. For such patients, consider periodic cardiovascular evaluation in intermittent long-term users of ZEMBRACE SymTouch injection.
7.4 Selective Serotonin Reuptake Inhibitors/serotonin Norepinephrine Reuptake Inhibitors and Serotonin Syndrome
Cases of serotonin syndrome have been reported during coadministration of triptans and SSRIs, SNRIs, TCAs, and MAO inhibitors [see Warnings and Precautions (5.7)].
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use:
- Use only if a clear diagnosis of migraine has been established. If a patient has no response to the first migraine attack treated with ZEMBRACE SymTouch, reconsider the diagnosis before ZEMBRACE SymTouch is administered to treat any subsequent attacks.
- ZEMBRACE SymTouch injection is not indicated for the prevention of migraine attacks.
Section 42230-3 (42230-3)
| Patient Information ZEMBRACE® SymTouch® (Zem-brace Sim-Touch) (sumatriptan succinate) Injection |
|
|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 11/2023 |
|
What is the most important information I should know about ZEMBRACE SymTouch?
ZEMBRACE SymTouch can cause serious side effects, including: Heart attack and other heart problems. Heart problems may lead to death. Stop taking ZEMBRACE SymTouch and get emergency medical help right away if you have any of the following symptoms of a heart attack:
|
|
|
What is ZEMBRACE SymTouch?
ZEMBRACE SymTouch is a prescription medicine used to treat acute migraine headaches with or without aura in adults who have been diagnosed with migraine. ZEMBRACE SymTouch is not used to treat other types of headaches such as hemiplegic (that make you unable to move on one side of your body) or basilar (rare form of migraine with aura) migraines. ZEMBRACE SymTouch is not used to prevent or decrease the number of migraines you have. It is not known if ZEMBRACE SymTouch is safe and effective in children under 18 years of age. |
|
|
Who should not take ZEMBRACE SymTouch? Do not take ZEMBRACE SymTouch if you have:
|
|
|
What should I tell my healthcare provider before taking ZEMBRACE SymTouch?
Before taking ZEMBRACE SymTouch, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take anti-depressant medicines called:
Know the medicines you take. Keep a list of them to show your healthcare provider or pharmacist when you get a new medicine. |
|
How should I take ZEMBRACE SymTouch?
|
|
|
What should I avoid while taking ZEMBRACE SymTouch?
ZEMBRACE SymTouch can cause dizziness, weakness, or drowsiness. If you have these symptoms, do not drive a car, use machinery, or do anything where you need to be alert. |
|
|
What are the possible side effects of ZEMBRACE SymTouch?
See "What is the most important information I should know about ZEMBRACE SymTouch?" ZEMBRACE SymTouch may cause serious side effects, including:
These are not all the possible side effects of ZEMBRACE SymTouch. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store ZEMBRACE SymTouch?
|
|
|
General information about the safe and effective use of ZEMBRACE SymTouch.
Medicines are sometimes prescribed for purposes other than those listed in Patient Information leaflets. Do not use ZEMBRACE SymTouch for a condition for which it was not prescribed. Do not give ZEMBRACE SymTouch to other people, even if they have the same symptoms you have. It may harm them. This Patient Information leaflet summarizes the most important information about ZEMBRACE SymTouch. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about ZEMBRACE SymTouch that is written for healthcare professionals. For more information, go to www.Zembrace.com or call 1-888-869-7633 (1-888-TNXPMED). |
|
|
What are the ingredients in ZEMBRACE SymTouch Injection?
Active ingredient: sumatriptan succinate Inactive ingredients: sodium chloride, water for injection Manufactured for TONIX MEDICINES, INC. Chatham, NJ 07928 ZEMBRACE and SymTouch are registered trademarks of Tonix Medicines, Inc. This product may be covered by one or more U.S. patent(s). |
Section 59845-8 (59845-8)
| Instructions for Use ZEMBRACE® SymTouch® (Zem-brace Sim-Touch) (sumatriptan succinate) Injection |
||
|---|---|---|
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. | Revised: 11/2023 | |
| Read this Instructions for Use before you start to use the ZEMBRACE® SymTouch®. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment. You and your healthcare provider should talk about ZEMBRACE SymTouch when you start taking it and at regular checkups. The ZEMBRACE SymTouch Auto-injector is a single-use, pre-filled injection device with the exact dose you need. The device should be used only 1 time and then discarded. |
Important Precautions Keep out of reach of children. Do not use after the expiration date has passed. Do not store or expose to high temperatures. Do not freeze. Do not remove the Red Cap until you are ready to inject. Do not use if dropped after removing the Red Cap. Do not put or press thumb, fingers or hand over the Yellow Needle Guard. |
|
|
Step 1 – Inspect Auto-injector and Gather Supplies 1A – Remove Auto-injector from the carton |
||
| 1B – Check the Expiration Date on the Auto-injector | Do not use if the expiration date has passed. | |
|
1C – Inspect the medicine through the Medicine Viewing Window
Inspect the appearance of ZEMBRACE SymTouch Auto-injector medicine through the medicine window. It must be a clear, colorless to pale yellow solution. |
||
| Do not inject the medicine if the solution looks discolored or cloudy or contains lumps, flakes, or particles. | ||
1D – Gather the following supplies
|
||
|
Step 2 – Choose and Prepare Your Injection Site 2A – Choose your injection site The recommended injection sites are the sides of the thighs and the arms. |
||
| Caution: Do not inject where your skin is scarred, bruised, tender, red or near any open wound. | ||
| 2B – Clean the injection site with an alcohol swab | Do not touch injection site area after cleaning. | |
|
2C – Remove the Red Cap
Hold the body of the Auto-injector in one hand and pull the Red Cap straight off. . |
||
| Warning: Do not put or press thumb, fingers or hand over the Yellow Needle Guard, doing so may result in a needle stick injury | ||
|
Step 3 – Inject the Medicine 3A – Place Auto-injector straight on your injection site Place the Auto-injector straight on your injection site (90 degrees) with the Yellow Needle Guard end gently pressed against your skin. |
||
|
3B – Perform Injection
Press and hold the Auto-injector down against your skin. You will hear the first "Click 1" as the injection starts. Continue to hold the device down until you hear the second "Click 2." Wait 5 seconds before you remove the Auto-injector! After "Click 2," continue to hold down and slowly count to 5 to make sure you receive your full dose. |
||
| Important: To receive your full dose, always hold the Auto-injector down on your injection site for additional time after you hear the second click. This allows all of the medicine to be delivered. | ||
|
3C – Remove Auto-injector from skin
Remove the Auto-injector by lifting it straight away from your skin. The Yellow Needle Guard will drop down and lock over the needle. |
||
|
Step 4 – Confirm Injection and Dispose the Auto-injector 4A – Confirm Red Color in Medicine Viewing Window Look to confirm that the Red Plunger Rod has filled the Medicine Viewing Window. This means you received the full dose. |
||
|
Caution: Call your healthcare provider if the Red Plunger Rod has not filled the Medicine Viewing Window. This may mean that only a partial dose was delivered. Do not try to reuse the Auto-injector. |
||
|
4B – Disposal of the Auto-injector
Put your used Auto-injector, caps, needles, and sharps in an FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) loose needles and syringes in your household trash. Do not recycle your used sharps disposal container. If you do not have a FDA-cleared sharps disposal container, you may use a household container that is: made of a heavy-duty plastic; can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out; upright and stable during use; leak-resistant; and properly labeled to warn of hazardous waste inside the container. When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal. Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container. If needed, make sure you get a refill of your Auto-injector. |
||
|
4C – Treat Injection Site
Treat the injection site with a cotton ball, gauze pad or bandage, as needed. Do not rub the injection site. Manufactured for TONIX MEDICINES, INC. Chatham, NJ 07928 ZEMBRACE and SymTouch are registered trademarks of Tonix Medicines, Inc. This product may be covered by one or more U.S. patent(s). |
10 Overdosage (10 OVERDOSAGE)
Coronary vasospasm was observed after intravenous administration of sumatriptan injection [see Contraindications (4)]. Overdoses would be expected from animal data (dogs at 0.1 g/kg, rats at 2 g/kg) to possibly cause convulsions, tremor, inactivity, erythema of the extremities, reduced respiratory rate, cyanosis, ataxia, mydriasis, injection site reactions (desquamation, hair loss, and scab formation), and paralysis.
The elimination half-life of sumatriptan is about 2 hours [see Clinical Pharmacology (12.3)], and therefore monitoring of patients after overdose with ZEMBRACE SymTouch injection should continue for at least 10 hours or while symptoms or signs persist.
It is unknown what effect hemodialysis or peritoneal dialysis has on the serum concentrations of sumatriptan.
5.10 Seizures
Seizures have been reported following administration of sumatriptan. Some have occurred in patients with either a history of seizures or concurrent conditions predisposing to seizures. There are also reports in patients where no such predisposing factors are apparent. ZEMBRACE SymTouch injection should be used with caution in patients with a history of epilepsy or conditions associated with a lowered seizure threshold.
11 Description (11 DESCRIPTION)
ZEMBRACE SymTouch injection contains sumatriptan succinate, a selective 5-HT1B/1D receptor agonist. Sumatriptan succinate is chemically designated as 3-[2-(dimethylamino) ethyl]-N-methyl-indole-5-methanesulfonamide succinate (1:1), and it has the following structure:
The empirical formula is C14H21N3O2S∙C4H6O4, representing a molecular weight of 413.5. Sumatriptan succinate is a white to off-white powder that is readily soluble in water and in saline.
ZEMBRACE SymTouch is a clear, colorless to pale yellow, sterile, nonpyrogenic solution for subcutaneous injection. Each 0.5 mL of ZEMBRACE SymTouch contains 4.2 mg of sumatriptan succinate equivalent to 3 mg of sumatriptan (base) and 4.15 mg of sodium chloride, USP in Water for Injection, USP.
The pH range of solution is approximately 4.2 to 5.3 and the osmolality of injection is approximately 291 mOsmol (275 to 315 mOsmol).
5.2 Arrhythmias
Life-threatening disturbances of cardiac rhythm, including ventricular tachycardia and ventricular fibrillation leading to death, have been reported within a few hours following the administration of 5-HT1 agonists. Discontinue ZEMBRACE SymTouch injection if these disturbances occur. ZEMBRACE SymTouch injection is contraindicated in patients with Wolff-Parkinson-White syndrome or arrhythmias associated with other cardiac accessory conduction pathway disorders.
7.3 Other 5 Ht1 (7.3 Other 5-HT1)
Because their vasospastic effects may be additive, coadministration of ZEMBRACE SymTouch injection and other 5-HT1 agonists (e.g., triptans) within 24 hours of each other is contraindicated.
16.1 How Supplied
- ZEMBRACE® SymTouch® 3 mg/0.5 mL Injection contains sumatriptan as the succinate salt and is supplied as a clear, colorless to pale yellow, sterile, nonpyrogenic solution in a prefilled, ready-to-use, single dose, disposable auto-injector unit (NDC # 70792-809-89).
- Each carton contains 4 units (NDC # 70792-809-38) and a Patient Information and Instructions for Use leaflet.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established. ZEMBRACE SymTouch injection is not recommended for use in patients younger than 18 years of age.
Two controlled clinical trials evaluated sumatriptan nasal spray (5 to 20 mg) in 1,248 pediatric migraineurs 12 to 17 years of age who treated a single attack. The trials did not establish the efficacy of sumatriptan nasal spray compared with placebo in the treatment of migraine in pediatric patients. Adverse reactions observed in these clinical trials were similar in nature to those reported in clinical trials in adults.
Five controlled clinical trials (2 single-attack trials, 3 multiple-attack trials) evaluating oral sumatriptan (25 to 100 mg) in pediatric subjects 12 to 17 years of age enrolled a total of 701 pediatric migraineurs. These trials did not establish the efficacy of oral sumatriptan compared with placebo in the treatment of migraine in pediatric patients. Adverse reactions observed in these clinical trials were similar in nature to those reported in clinical trials in adults. The frequency of all adverse reactions in these patients appeared to be both dose- and age-dependent, with younger patients reporting reactions more commonly than older pediatric patients.
Post-marketing experience documents that serious adverse reactions have occurred in the pediatric population after use of subcutaneous, oral, and/or intranasal sumatriptan. These reports include reactions similar in nature to those reported rarely in adults, including stroke, visual loss, and death. A myocardial infarction has been reported in a 14-year-old male following the use of oral sumatriptan; clinical signs occurred within 1 day of drug administration. Clinical data to determine the frequency of serious adverse reactions in pediatric patients who might receive subcutaneous, oral, or intranasal sumatriptan are not presently available.
8.5 Geriatric Use
Clinical trials of sumatriptan injection did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger patients. Other reported clinical experience has not identified differences in responses between the elderly and younger subjects. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
A cardiovascular evaluation is recommended for geriatric patients who have other cardiovascular risk factors (e.g., diabetes, hypertension, smoking, obesity, strong family history of CAD) prior to receiving ZEMBRACE SymTouch injection [see Warnings and Precautions (5.1)].
4 Contraindications (4 CONTRAINDICATIONS)
ZEMBRACE SymTouch injection is contraindicated in patients with:
- Ischemic coronary artery disease (CAD) (angina pectoris, history of myocardial infarction, or documented silent ischemia) or coronary artery vasospasm, including Prinzmetal's angina [see Warnings and Precautions (5.1)].
- Wolff-Parkinson-White syndrome or arrhythmias associated with other cardiac accessory conduction pathway disorders [see Warnings and Precautions (5.2)].
- History of stroke or transient ischemic attack (TIA) or history of hemiplegic or basilar migraine because these patients are at a higher risk of stroke [see Warnings and Precautions (5.4)].
- Peripheral vascular disease [see Warnings and Precautions (5.5)].
- Ischemic bowel disease [see Warnings and Precautions (5.5)].
- Uncontrolled hypertension [see Warnings and Precautions (5.8)].
- Recent (i.e., within 24 hours) use of ergotamine-containing medication, ergot-type medication (such as dihydroergotamine or methysergide), or another 5-hydroxytryptamine1 (5-HT1) agonist [see Drug Interactions (7.1, 7.3)].
- Concurrent administration of an MAO-A inhibitor or recent (within 2 weeks) use of an MAO-A inhibitor [see Drug Interactions (7.2) and Clinical Pharmacology (12.3)].
- Known hypersensitivity to sumatriptan (angioedema and anaphylaxis seen) [see Warnings and Precautions (5.9)].
- Severe hepatic impairment [see Clinical Pharmacology (12.3)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described below and elsewhere in the labeling:
- Myocardial ischemia, myocardial infarction, and Prinzmetal's angina [see Warnings and Precautions (5.1)]
- Arrhythmias [see Warnings and Precautions (5.2)]
- Chest, throat, neck, and/or jaw pain/tightness/pressure [see Warnings and Precautions (5.3)]
- Cerebrovascular events [see Warnings and Precautions (5.4)]
- Other vasospasm reactions [see Warnings and Precautions (5.5)]
- Medication overuse headache [see Warnings and Precautions (5.6)]
- Serotonin syndrome [see Warnings and Precautions (5.7)]
- Increase in blood pressure [see Warnings and Precautions (5.8)]
- Hypersensitivity reactions [see Contraindications (4), Warnings and Precautions (5.9)]
- Seizures [see Warnings and Precautions (5.10)]
12.3 Pharmacokinetics
After a single 3 mg dose, ZEMBRACE SymTouch was bioequivalent to IMITREX subcutaneous injection.
2.1 Dosing Information
The recommended dose of ZEMBRACE SymTouch is 3 mg injected subcutaneously.
The maximum cumulative injected dose that may be given in 24 hours is 12 mg, with doses of ZEMBRACE SymTouch separated by at least 1 hour. ZEMBRACE SymTouch may also be given at least 1 hour following a dose of another sumatriptan product.
5.7 Serotonin Syndrome
Serotonin syndrome may occur with ZEMBRACE SymTouch injection, particularly during co-administration with selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), and MAO inhibitors [see Drug Interactions (7.4)]. Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, coma), autonomic instability (e.g., tachycardia, labile blood pressure, hyperthermia), neuromuscular aberrations (e.g., hyperreflexia, incoordination), and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). The onset of symptoms usually occurs within minutes to hours of receiving a new or a greater dose of a serotonergic medication. Discontinue ZEMBRACE SymTouch injection if serotonin syndrome is suspected.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ZEMBRACE SymTouch is indicated for the acute treatment of migraine with or without aura in adults.
12.1 Mechanism of Action
Sumatriptan binds with high affinity to human cloned 5-HT1B/1D receptors. Sumatriptan presumably exerts its therapeutic effects in the treatment of migraine headache through agonist effects at the 5-HT1B/1D receptors on intracranial blood vessels and sensory nerves of the trigeminal system, which result in cranial vessel constriction and inhibition of pro-inflammatory neuropeptide release.
16.2 Storage and Handling
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F). Protect from light.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Myocardial ischemia/infarction and Prinzmetal's angina: Perform cardiac evaluation in patients with multiple cardiovascular risk factors. (5.1)
- Arrhythmias: Discontinue ZEMBRACE SymTouch if occurs. (5.2)
- Chest/throat/neck/jaw pain, tightness, pressure, or heaviness: Generally, not associated with myocardial ischemia; evaluate for coronary artery disease in patients at high risk. (5.3)
- Cerebral hemorrhage, subarachnoid hemorrhage, and stroke: Discontinue ZEMBRACE SymTouch if occurs. (5.4)
- Gastrointestinal ischemia and reactions, peripheral vasospastic reactions: Discontinue ZEMBRACE SymTouch if occurs. (5.5)
- Medication overuse headache: Detoxification may be necessary. (5.6)
- Serotonin syndrome: Discontinue ZEMBRACE SymTouch if occurs. (5.7)
- Seizures: Use with caution in patients with epilepsy or a lowered seizure threshold. (5.10)
5.4 Cerebrovascular Events
Cerebral hemorrhage, subarachnoid hemorrhage, and stroke have occurred in patients treated with 5-HT1 agonists, and some have resulted in fatalities. In a number of cases, it appears possible that the cerebrovascular events were primary, the 5-HT1 agonist having been administered in the incorrect belief that the symptoms experienced were a consequence of migraine when they were not. Also, patients with migraine may be at increased risk of certain cerebrovascular events (e.g., stroke, hemorrhage, TIA). Discontinue ZEMBRACE SymTouch injection if a cerebrovascular event occurs.
Before treating headaches in patients not previously diagnosed with migraine or in patients who present with atypical symptoms, exclude other potentially serious neurological conditions. ZEMBRACE SymTouch injection is contraindicated in patients with a history of stroke or TIA.
7.1 Ergot Containing Drugs (7.1 Ergot-Containing Drugs)
Ergot-containing drugs have been reported to cause prolonged vasospastic reactions. Because these effects may be additive, use of ergotamine-containing or ergot-type medications (like dihydroergotamine or methysergide) and ZEMBRACE SymTouch within 24 hours of each other is contraindicated.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 3 mg sumatriptan in 0.5 mL prefilled, ready-to-use, single dose, disposable auto-injector.
5.5 Other Vasospasm Reactions
ZEMBRACE SymTouch injection may cause non-coronary vasospastic reactions, such as peripheral vascular ischemia, gastrointestinal vascular ischemia and infarction (presenting with abdominal pain and bloody diarrhea), splenic infarction, and Raynaud's syndrome. In patients who experience symptoms or signs suggestive of non-coronary vasospasm reaction following the use of any 5-HT1 agonist, rule out a vasospastic reaction before receiving additional ZEMBRACE SymTouch injections.
Reports of transient and permanent blindness and significant partial vision loss have been reported with the use of 5-HT1 agonists. Since visual disorders may be part of a migraine attack, a causal relationship between these events and the use of 5-HT1 agonists have not been clearly established.
6.2 Post Marketing Experience (6.2 Post-marketing Experience)
The following adverse reactions have been identified during post-approval use of sumatriptan tablets, sumatriptan nasal spray, and sumatriptan injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiovascular
Hypotension, palpitations
Neurological
Dystonia, tremor
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy: Based on animal data, may cause fetal harm. (8.1)
5.8 Increase in Blood Pressure
Significant elevation in blood pressure, including hypertensive crisis with acute impairment of organ systems, has been reported on rare occasions in patients treated with 5-HT1 agonists, including patients without a history of hypertension. Monitor blood pressure in patients treated with ZEMBRACE SymTouch. ZEMBRACE SymTouch injection is contraindicated in patients with uncontrolled hypertension.
5.9 Hypersensitivity Reactions
Hypersensitivity reactions, including angioedema and anaphylaxis, have occurred in patients receiving sumatriptan. Such reactions can be life-threatening or fatal. In general, anaphylactic reactions to drugs are more likely to occur in individuals with a history of sensitivity to multiple allergens. ZEMBRACE SymTouch injection is contraindicated in patients with a history of hypersensitivity reaction to sumatriptan.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.6 Medication Overuse Headache
Overuse of acute migraine drugs (e.g., ergotamine, triptans, opioids, combination of these drugs for 10 or more days per month) may lead to exacerbation of headache (medication overuse headache). Medication overuse headache may present as migraine-like daily headaches, or as a marked increase in frequency of migraine attacks. Detoxification of patients, including withdrawal of the overused drugs, and treatment of withdrawal symptoms (which often includes a transient worsening of headache) may be necessary.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
7.2 Monoamine Oxidase A Inhibitors (7.2 Monoamine Oxidase-A Inhibitors)
MAO-A inhibitors increase systemic exposure by 2-fold. Therefore, the use of ZEMBRACE SymTouch injection in patients receiving MAO-A inhibitors is contraindicated [see Clinical Pharmacology (12.3)].
2.2 Administration Using Zembrace Symtouch (2.2 Administration Using ZEMBRACE SymTouch)
ZEMBRACE SymTouch is available as a prefilled, ready-to-use, single dose, disposable auto-injector containing 3 mg sumatriptan. With ZEMBRACE SymTouch, the needle penetrates approximately ¼ inch (6 mm). The injection is intended to be given subcutaneously. Do not administer by any other route. Instruct patients on the proper use of ZEMBRACE SymTouch and direct them to use injection sites with an adequate skin and subcutaneous thickness to accommodate the length of the needle.
Principal Display Panel 3 Mg Syringe Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 3 mg Syringe Blister Pack Carton)
NDC 70792-809-38
Rx only
TONIX
MEDICINES
Zembrace® SYMTOUCH®
(sumatriptan injection)
3 mg
For subcutaneous use only.
Each auto-injector contains a prefilled syringe with 0.5 mL of
sterile solution delivering 4.2 mg of sumatriptan succinate
equivalent to 3 mg of sumatriptan base, 4.15 mg sodium
chloride and water for injection.
See package insert for dosage information.
Store at 20° to 25°C (68° to 77°F).
Protect from light.
Manufactured for
TONIX MEDICINES, INC.
Chatham, NJ 07928
ZEMBRACE and SymTouch are registered trademarks of
Tonix Medicines, Inc.
5.3 Chest, Throat, Neck, And/or Jaw Pain/tightness/pressure (5.3 Chest, Throat, Neck, and/or Jaw Pain/Tightness/Pressure)
Sensations of tightness, pain, pressure, and heaviness in the precordium, throat, neck, and jaw commonly occur after treatment with sumatriptan injection and are usually non-cardiac in origin. However, perform a cardiac evaluation if these patients are at high cardiac risk. The use of ZEMBRACE SymTouch injection is contraindicated in patients with CAD and those with Prinzmetal's variant angina.
5.1 Myocardial Ischemia, Myocardial Infarction, and Prinzmetal's Angina
The use of ZEMBRACE SymTouch injection is contraindicated in patients with ischemic or vasospastic CAD. There have been rare reports of serious cardiac adverse reactions, including acute myocardial infarction, occurring within a few hours following administration of sumatriptan injection. Some of these reactions occurred in patients without known CAD. 5-HT1 agonists, including ZEMBRACE SymTouch injection, may cause coronary artery vasospasm (Prinzmetal's angina), even in patients without a history of CAD.
Perform a cardiovascular evaluation in triptan-naive patients who have multiple cardiovascular risk factors (e.g., increased age, diabetes, hypertension, smoking, obesity, strong family history of CAD) prior to receiving ZEMBRACE SymTouch injection. If there is evidence of CAD or coronary artery vasospasm, ZEMBRACE SymTouch injection is contraindicated. For patients with multiple cardiovascular risk factors who have a negative cardiovascular evaluation, consider administering the first dose of ZEMBRACE SymTouch injection in a medically supervised setting and performing an electrocardiogram (ECG) immediately following ZEMBRACE SymTouch injection. For such patients, consider periodic cardiovascular evaluation in intermittent long-term users of ZEMBRACE SymTouch injection.
7.4 Selective Serotonin Reuptake Inhibitors/serotonin Norepinephrine Reuptake Inhibitors and Serotonin Syndrome (7.4 Selective Serotonin Reuptake Inhibitors/Serotonin Norepinephrine Reuptake Inhibitors and Serotonin Syndrome)
Cases of serotonin syndrome have been reported during coadministration of triptans and SSRIs, SNRIs, TCAs, and MAO inhibitors [see Warnings and Precautions (5.7)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:02.695286 · Updated: 2026-03-14T22:23:51.806226