These Highlights Do Not Include All The Information Needed To Use Tepezza®
3e6c54a1-cefd-4a5b-a855-ab9f268b6cce
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration, Administration ( 2.3 ) 9/2025 Warnings, and Precautions, Inflammatory Bowel Disease ( 5.2 ) 8/2025
Indications and Usage
TEPEZZA is indicated for the treatment of Thyroid Eye Disease regardless of Thyroid Eye Disease activity or duration.
Dosage and Administration
Initiate dosing with 10 mg/kg for first infusion, followed by 20 mg/kg every 3 weeks for 7 additional infusions ( 2.1 ) Administer TEPEZZA by intravenous infusion over 60 to 90 minutes ( 2.3 )
Warnings and Precautions
Infusion Reactions : If an infusion reaction occurs, interrupt or slow the rate of infusion and use appropriate medical management ( 5.1 ) Inflammatory Bowel Disease (IBD) : Monitor patients; for signs and symptoms of disease, including patients without a history of IBD; discontinue TEPEZZA if IBD exacerbation is suspected ( 5.2 ) Hyperglycemia : Assess patients for elevated blood glucose and symptoms of hyperglycemia prior to infusion and continue to monitor while on treatment with TEPEZZA. Ensure patients with hyperglycemia or preexisting diabetes are under appropriate glycemic control before and while receiving TEPEZZA ( 5.3 ) Hearing Impairment Including Hearing Loss: TEPEZZA may cause severe hearing impairment including hearing loss, which in some cases may be permanent. Assess patients' hearing before, during, and after treatment with TEPEZZA and consider the benefit-risk of treatment with patients ( 5.4 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Infusion Reactions [see Warnings and Precautions (5.1) ] Inflammatory Bowel Disease [see Warnings and Precautions (5.2) ] Hyperglycemia [see Warnings and Precautions (5.3) ] Hearing Impairment Including Hearing Loss [see Warnings and Precautions (5.4) ]
Storage and Handling
TEPEZZA (teprotumumab-trbw) for injection is a sterile, preservative-free, white to off-white lyophilized powder available as follows: Carton containing one 500 mg single-dose vial NDC 75987-130-15
How Supplied
TEPEZZA (teprotumumab-trbw) for injection is a sterile, preservative-free, white to off-white lyophilized powder available as follows: Carton containing one 500 mg single-dose vial NDC 75987-130-15
Medication Information
Warnings and Precautions
Infusion Reactions : If an infusion reaction occurs, interrupt or slow the rate of infusion and use appropriate medical management ( 5.1 ) Inflammatory Bowel Disease (IBD) : Monitor patients; for signs and symptoms of disease, including patients without a history of IBD; discontinue TEPEZZA if IBD exacerbation is suspected ( 5.2 ) Hyperglycemia : Assess patients for elevated blood glucose and symptoms of hyperglycemia prior to infusion and continue to monitor while on treatment with TEPEZZA. Ensure patients with hyperglycemia or preexisting diabetes are under appropriate glycemic control before and while receiving TEPEZZA ( 5.3 ) Hearing Impairment Including Hearing Loss: TEPEZZA may cause severe hearing impairment including hearing loss, which in some cases may be permanent. Assess patients' hearing before, during, and after treatment with TEPEZZA and consider the benefit-risk of treatment with patients ( 5.4 )
Indications and Usage
TEPEZZA is indicated for the treatment of Thyroid Eye Disease regardless of Thyroid Eye Disease activity or duration.
Dosage and Administration
Initiate dosing with 10 mg/kg for first infusion, followed by 20 mg/kg every 3 weeks for 7 additional infusions ( 2.1 ) Administer TEPEZZA by intravenous infusion over 60 to 90 minutes ( 2.3 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Infusion Reactions [see Warnings and Precautions (5.1) ] Inflammatory Bowel Disease [see Warnings and Precautions (5.2) ] Hyperglycemia [see Warnings and Precautions (5.3) ] Hearing Impairment Including Hearing Loss [see Warnings and Precautions (5.4) ]
Storage and Handling
TEPEZZA (teprotumumab-trbw) for injection is a sterile, preservative-free, white to off-white lyophilized powder available as follows: Carton containing one 500 mg single-dose vial NDC 75987-130-15
How Supplied
TEPEZZA (teprotumumab-trbw) for injection is a sterile, preservative-free, white to off-white lyophilized powder available as follows: Carton containing one 500 mg single-dose vial NDC 75987-130-15
Description
Dosage and Administration, Administration ( 2.3 ) 9/2025 Warnings, and Precautions, Inflammatory Bowel Disease ( 5.2 ) 8/2025
Section 42229-5
Risk Summary
Based on findings in animals and its mechanism of action inhibiting insulin-like growth factor-1 receptor (IGF-1R) signaling, TEPEZZA may cause fetal harm when administered to a pregnant woman. Adequate and well-controlled studies with TEPEZZA have not been conducted in pregnant women. There are insufficient data with TEPEZZA use in pregnant women to inform any drug associated risks for adverse developmental outcomes. Cynomolgus monkeys dosed once weekly with teprotumumab during pregnancy resulted in placental changes, decreased fetal growth during pregnancy, decreased fetal size and weight, and multiple external and skeletal abnormalities in offspring. Teprotumumab exposure may lead to an increase in fetal loss [see Data]. Therefore, TEPEZZA should not be used in pregnancy, and appropriate forms of contraception should be implemented prior to initiation, during treatment and for 6 months following the last dose of TEPEZZA. Women of childbearing age should have a pregnancy test performed by their doctor before starting treatment with TEPEZZA. If the patient becomes pregnant during treatment, TEPEZZA should be discontinued and the patient advised of the potential risk to the fetus.
The background rate of major birth defects and miscarriage is unknown for the indicated population. In the U.S. general population, the estimated background risks of major birth defects and miscarriage in clinically recognized pregnancies are 2-4% and 15-20%, respectively.
Section 43683-2
Section 44425-7
Refrigerate at 2°C to 8°C (36°F to 46°F) in original carton until time of use to protect from light. Do not freeze.
10 Overdosage
No information is available for patients who have received an overdosage.
11 Description
Teprotumumab-trbw, an insulin-like growth factor-1 receptor inhibitor (IGF-1R), is a fully human IgG1 monoclonal antibody produced in Chinese hamster ovary (CHO-DG44) cells. It has a molecular weight of approximately 148 kilodaltons.
TEPEZZA (teprotumumab-trbw) for injection is supplied as a sterile, preservative-free, white to off-white, lyophilized powder for intravenous infusion. Each single-dose vial delivers 500 mg of teprotumumab-trbw, histidine (7.82 mg), L-histidine hydrochloride monohydrate (33.43 mg), polysorbate 20 (1.05 mg), and trehalose (898.43 mg). After reconstitution with 10 mL of Sterile Water for Injection, USP, the final concentration is 47.6 mg/mL with a pH of 5.5.
5.3 Hyperglycemia
Hyperglycemia or increased blood glucose may occur in patients treated with TEPEZZA. In the premarketing clinical trials, 10% of patients (two-thirds of whom had preexisting diabetes or impaired glucose tolerance) experienced hyperglycemia [see Adverse Reactions (6.1)]. Hyperglycemic events should be controlled with medications for glycemic control, if necessary.
Assess patients for elevated blood glucose and symptoms of hyperglycemia prior to infusion and continue to monitor while on treatment with TEPEZZA. Ensure patients with hyperglycemia or preexisting diabetes are under appropriate glycemic control before and while receiving TEPEZZA.
8.4 Pediatric Use
Safety and effectiveness have not been established in pediatric patients.
8.5 Geriatric Use
Of the 171 patients in the two randomized trials, 15% were 65 years of age or older; the number of patients 65 years or older was similar between treatment groups. No overall differences in efficacy or safety were observed between patients 65 years or older and younger patients (less than 65 years of age).
2.3 Administration
Administer the diluted solution intravenously over at least 90 minutes for the first two infusions. If well tolerated, the minimum time for subsequent infusions can be reduced to 60 minutes. If not well tolerated, the minimum time for subsequent infusions should remain at 90 minutes, and pre-medication is recommended for subsequent infusions.
Do not administer as an intravenous push or bolus. TEPEZZA should not be infused concomitantly with other agents.
12.6 Immunogenicity
The observed incidence of anti-drug antibody is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibody in the studies described below with the incidence of anti-drug antibodies in other studies, including those of TEPEZZA.
In a randomized placebo-controlled study, HZNP-TEP-301, where active TED patients were administered placebo or TEPEZZA over a 24-week period, 2.4% (1 of 42) of placebo and 0% (0 of 41) of TEPEZZA treated patients tested positive for anti-teprotumumab antibodies at post-baseline visits.
Because of the low occurrence of anti-teprotumumab antibodies, the effect of these antibodies on the pharmacokinetics, pharmacodynamics, safety, and/or effectiveness of teprotumumab products is unknown.
14 Clinical Studies
TEPEZZA was evaluated in 2 randomized, double-masked, placebo-controlled studies in 171 patients with Thyroid Eye Disease: Study 1 (NCT01868997) and Study 2 (NCT03298867). Patients were randomized to receive TEPEZZA or placebo in a 1:1 ratio. Patients were given intravenous infusions (10 mg/kg for first infusion and 20 mg/kg for the remaining 7 infusions) every 3 weeks for a total of 8 infusions. Patients had a clinical diagnosis of Thyroid Eye Disease with symptoms and were euthyroid or had thyroxine and free triiodothyronine levels less than 50% above or below normal limits. Prior surgical treatment for Thyroid Eye Disease was not permitted. Proptosis ranged from 16 to 33 mm and 125 patients (73%) had diplopia at baseline.
A total of 84 patients were randomized to TEPEZZA and 87 patients were randomized to placebo. The median age was 52 years (range 20 to 79 years), 86% were White, 9% were Black or African-American, 4% were Asian and 1% identified as Other. The majority (73%) were female. At baseline, 27% of patients were smokers.
The proptosis responder rate at week 24 was defined as the percentage of patients with ≥ 2 mm reduction in proptosis in the study eye from baseline, without deterioration in the non-study eye (≥ 2 mm increase) in proptosis. Additional evaluations included signs and symptoms of Thyroid Eye Disease including pain, gaze evoked orbital pain, swelling, eyelid erythema, redness, chemosis, inflammation, clinical activity score and assessments of functional vision and patient appearance. Results for proptosis are found in Table 2.
| Study 1 | Study 2 | |||||
|---|---|---|---|---|---|---|
| Teprotumumab (N = 42) |
Placebo (N = 45) |
Difference (95% CI) |
Teprotumumab (N = 41) |
Placebo (N = 42) |
Difference (95% CI) |
|
| Proptosis responder rate at week 24, % (n) Difference and its corresponding 95% Confidence Interval (CI) is based on a weighted average of the difference within each randomization stratum (tobacco user, tobacco non-use) using CMH weights.
|
71% (30) | 20% (9) | 51% (33, 69) |
83% (34) | 10% (4) | 73% (59, 88) |
| Proptosis (mm) average change from baseline through week 24, LS Mean (SE) Results were obtained from an MMRM with an unstructured covariance matrix and including treatment, smoking status, baseline value, visit, treatment by visit, and visit by baseline value interaction as fixed effects. A change from baseline of 0 was imputed at the first post-baseline visit for any subject without a post baseline value.
|
-2.5 (0.2) | -0.2 (0.2) | -2.3 (-2.8, -1.8) |
-2.8 (0.2) | -0.5 (0.2) | -2.3 (-2.8, -1.8) |
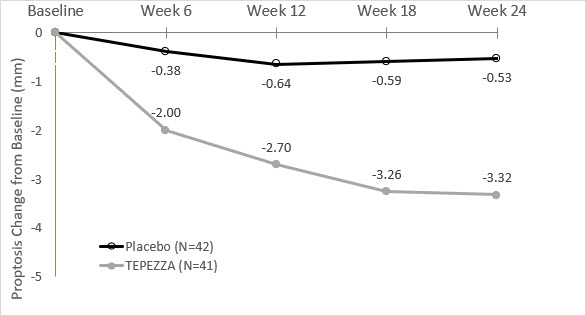
In Study 2, improvement of proptosis as measured by mean change from Baseline was observed as early as 6 weeks and continued to improve through week 24 as shown in Figure 1. Similar results were seen in Study 1.
Figure 1. Change from Baseline in Proptosis over 24 Weeks in Study 2
P < 0.01 at each timepoint
TEPEZZA also led to improvement in the less severely impacted "fellow" eye.
Diplopia (double vision) was evaluated in a subgroup of patients that had diplopia at baseline in Study 1 and 2. Results are shown in Table 3.
| Parameter | TEPEZZA (n = 66) |
Placebo (n = 59) |
|---|---|---|
| P < 0.01 | ||
| Diplopia | ||
| Responder rate Diplopia was evaluated on a 4-point scale where scores ranged from 0 for no diplopia to 3 for constant diplopia. A diplopia responder was defined as a patient with baseline diplopia > 0 and a score of 0 at week 24. at week 24, % (n) |
53% (35) | 25% (15) |
Following discontinuation of treatment in Study 1, 53% of patients (16 of 30 patients) who were proptosis responders at week 24 maintained proptosis response 51 weeks after the last TEPEZZA infusion. 67% of patients (12 of 18) who were diplopia responders at week 24 maintained diplopia response 51 weeks after the last TEPEZZA infusion.
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Infusion Reactions [see Warnings and Precautions (5.1)]
- Inflammatory Bowel Disease [see Warnings and Precautions (5.2)]
- Hyperglycemia [see Warnings and Precautions (5.3)]
- Hearing Impairment Including Hearing Loss [see Warnings and Precautions (5.4)]
12.2 Pharmacodynamics
No formal pharmacodynamic studies have been conducted with teprotumumab-trbw.
12.3 Pharmacokinetics
The pharmacokinetics of teprotumumab-trbw was described by a two compartment population PK model based on data from 40 patients with Thyroid Eye Disease receiving an initial intravenous infusion of 10 mg/kg, followed by infusions of 20 mg/kg TEPEZZA every 3 weeks in one clinical trial. Following this regimen, the mean (± standard deviation) estimates for steady-state area under the concentration curve (AUC), peak (Cmax), and trough (Ctrough) concentrations of teprotumumab-trbw were 138 (± 34) mg∙hr/mL, 632 (± 139) mcg/mL, and 176 (± 56) mcg/mL, respectively.
2.1 Recommended Dosing
The recommended dose of TEPEZZA is an intravenous infusion of 10 mg/kg for the initial dose followed by an intravenous infusion of 20 mg/kg every three weeks for 7 additional infusions.
5.1 Infusion Reactions
TEPEZZA may cause infusion reactions. Infusion reactions have been reported in approximately 4% of patients treated with TEPEZZA. Signs and symptoms of infusion-related reactions include transient increases in blood pressure, feeling hot, tachycardia, dyspnea, headache and muscular pain. Infusion reactions may occur during any of the infusions or within 1.5 hours after an infusion. Reported infusion reactions are usually mild or moderate in severity and can usually be successfully managed with corticosteroids and antihistamines. In patients who experience an infusion reaction, consideration should be given to pre-medicating with an antihistamine, antipyretic, corticosteroid and/or administering all subsequent infusions at a slower infusion rate.
1 Indications and Usage
TEPEZZA is indicated for the treatment of Thyroid Eye Disease regardless of Thyroid Eye Disease activity or duration.
12.1 Mechanism of Action
Teprotumumab-trbw's mechanism of action in patients with Thyroid Eye Disease has not been fully characterized. Teprotumumab-trbw binds to IGF-1R and blocks its activation and signaling.
5 Warnings and Precautions
- Infusion Reactions: If an infusion reaction occurs, interrupt or slow the rate of infusion and use appropriate medical management (5.1)
- Inflammatory Bowel Disease (IBD): Monitor patients; for signs and symptoms of disease, including patients without a history of IBD; discontinue TEPEZZA if IBD exacerbation is suspected (5.2)
- Hyperglycemia: Assess patients for elevated blood glucose and symptoms of hyperglycemia prior to infusion and continue to monitor while on treatment with TEPEZZA. Ensure patients with hyperglycemia or preexisting diabetes are under appropriate glycemic control before and while receiving TEPEZZA (5.3)
- Hearing Impairment Including Hearing Loss: TEPEZZA may cause severe hearing impairment including hearing loss, which in some cases may be permanent. Assess patients' hearing before, during, and after treatment with TEPEZZA and consider the benefit-risk of treatment with patients (5.4)
2 Dosage and Administration
3 Dosage Forms and Strengths
For injection (intravenous infusion): 500 mg of teprotumumab as a white to off-white lyophilized powder in a single-dose vial for reconstitution and dilution.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of TEPEZZA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: bowel perforation, exacerbation of inflammatory bowel disease (IBD), including patients without a prior diagnosis of IBD
Metabolism and Nutrition Disorders: diabetic ketoacidosis, hyperosmolar hyperglycemic state (HHS)
Otologic: severe hearing impairment including hearing loss, which in some cases may be permanent
8 Use in Specific Populations
Females of Reproductive Potential: Appropriate forms of contraception should be implemented prior to initiation, during treatment and for 6 months following the last dose of TEPEZZA (8.3)
5.2 Inflammatory Bowel Disease
TEPEZZA may cause an exacerbation of inflammatory bowel disease (IBD). IBD has been reported in some patients without a prior diagnosis of IBD [see Adverse Reactions (6.2)]. Monitor patients for signs and symptoms of IBD. If IBD exacerbation is suspected, discontinue use of TEPEZZA.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of TEPEZZA was evaluated in two premarketing randomized, double-masked, placebo-controlled clinical trials (Study 1 [NCT01868997] and Study 2 [NCT03298867]) consisting of 170 patients with Thyroid Eye Disease (84 received TEPEZZA and 86 received placebo). Patients were treated with TEPEZZA (10 mg/kg for first infusion and 20 mg/kg for the remaining 7 infusions) or placebo given as an intravenous infusion every 3 weeks for a total of 8 infusions. The majority of patients completed 8 infusions (89% of TEPEZZA patients and 93% of placebo patients).
The most common adverse reactions (≥ 5%) that occurred at greater incidence in the TEPEZZA group than in the control group during the treatment period of Studies 1 and 2 are summarized in Table 1.
In addition, menstrual disorders (amenorrhea, metrorrhagia, dysmenorrhea) were reported in approximately 23% (5 of 22 patients) of menstruating women treated with TEPEZZA compared to 4% (1 of 25 patients) treated with placebo in the premarketing clinical trials.
| Adverse Reactions | TEPEZZA N = 84 N (%) |
Placebo N = 86 N (%) |
|---|---|---|
| Muscle spasms | 21 (25%) | 6 (7%) |
| Nausea | 14 (17%) | 8 (9%) |
| Alopecia | 11 (13%) | 7 (8%) |
| Diarrhea | 10 (12%) | 7 (8%) |
| Fatigue Fatigue includes asthenia
|
10 (12%) | 6 (7%) |
| Hyperglycemia Hyperglycemia includes blood glucose increase
|
8 (10%) | 1 (1%) |
| Hearing impairment Hearing impairment including hearing loss (deafness, including sensorineural deafness, eustachian tube dysfunction, hyperacusis, hypoacusis, autophony and tinnitus)
|
8 (10%) | 0 |
| Dysgeusia | 7 (8%) | 0 |
| Headache | 7 (8%) | 6 (7%) |
| Dry skin | 7 (8%) | 0 |
| Weight decreased | 5 (6%) | 0 |
| Nail disorder Nail disorder (includes nail discoloration, nail disorder and onychoclasis)
|
4 (5%) | 0 |
In clinical trials, gastrointestinal complaints including exacerbation of inflammatory bowel disease were reported.
In two postapproval trials, the following were the adverse reactions occurring ≥ 5% of participants treated with TEPEZZA during the double-masked treatment period: muscle spasms (29%), hearing impairment (19%), hyperglycemia (18%) diarrhea (16%), fatigue (13%), headache (13%), alopecia (10%), dry skin (10%), dysgeusia (9%), ear discomfort (9%), and nail disorder (6%).
2.2 Reconstitution and Preparation
Step 1: Calculate the dose (mg) and determine the number of vials needed for the 10 or 20 mg/kg dosage based on patient weight. Each TEPEZZA vial contains 500 mg of the teprotumumab antibody.
Step 2: Using appropriate aseptic technique, reconstitute each TEPEZZA vial with 10 mL of Sterile Water for Injection, USP. Ensure that the stream of diluent is not directed onto the lyophilized powder, which has a cake-like appearance. Do not shake, but gently swirl the solution by rotating the vial until the lyophilized powder is dissolved. The reconstituted solution has a total volume of 10.5 mL. Withdraw 10.5 mL of reconstituted solution to obtain 500 mg. After reconstitution, the final concentration is 47.6 mg/mL.
Step 3: The reconstituted TEPEZZA solution must be further diluted in 0.9% Sodium Chloride Injection, USP prior to infusion. To prepare the diluted solution, use 100 mL infusion bags for a dose less than 1800 mg, and 250 mL infusion bags for a dose equal to or greater than 1800 mg. To maintain a constant volume in the infusion bag, a sterile syringe and needle should be used to remove the volume equivalent to the amount of the reconstituted TEPEZZA solution to be placed into the infusion bag. Discard the volume of 0.9% Sodium Chloride Injection, USP withdrawn.
Step 4: Withdraw the required volume from the reconstituted TEPEZZA vial(s) based on the patient's weight (in kg) and transfer into the intravenous bag containing 0.9% Sodium Chloride Injection, USP. Mix diluted solution by gentle inversion. Do not shake. If refrigerated prior to administration, allow the diluted solution to reach room temperature prior to infusion.
The product does not contain any preservative. The combined storage time of reconstituted TEPEZZA solution in the vial and the diluted solution in the infusion bag containing 0.9% Sodium Chloride Injection, USP is a total of 4 hours at room temperature 20°C to 25°C (68°F to 77°F) or up to 48 hours under refrigerated conditions 2°C to 8°C (36°F to 46°F) protected from light.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Upon reconstitution, TEPEZZA is a nearly colorless or slightly brown, clear to opalescent solution which is free of foreign particulate matter. Discard the solution if any particulate matter or discoloration are observed.
Do not freeze the reconstituted or diluted solution.
Discard vial(s) and all unused contents.
No incompatibilities between TEPEZZA and polyethylene (PE), polyvinyl chloride (PVC), polyurethane (PUR) or polyolefin (PO) bags and intravenous administration sets have been observed.
16 How Supplied/storage and Handling
TEPEZZA (teprotumumab-trbw) for injection is a sterile, preservative-free, white to off-white lyophilized powder available as follows:
| Carton containing one 500 mg single-dose vial | NDC 75987-130-15 |
Principal Display Panel 500 Mg Vial Carton
NDC-75987-130-15
TEPEZZA®
(teprotumumab-trbw)
for Injection
500 mg/vial
For Intravenous Infusion Only
Reconstitute and Further Dilute Prior to Use
Single-dose vial. Discard unused portion.
AMGEN
5.4 Hearing Impairment Including Hearing Loss
TEPEZZA may cause severe hearing impairment including hearing loss, which in some cases may be permanent. Assess patients' hearing before, during, and after treatment with TEPEZZA and consider the benefit-risk of treatment with patients.
Structured Label Content
Section 42229-5 (42229-5)
Risk Summary
Based on findings in animals and its mechanism of action inhibiting insulin-like growth factor-1 receptor (IGF-1R) signaling, TEPEZZA may cause fetal harm when administered to a pregnant woman. Adequate and well-controlled studies with TEPEZZA have not been conducted in pregnant women. There are insufficient data with TEPEZZA use in pregnant women to inform any drug associated risks for adverse developmental outcomes. Cynomolgus monkeys dosed once weekly with teprotumumab during pregnancy resulted in placental changes, decreased fetal growth during pregnancy, decreased fetal size and weight, and multiple external and skeletal abnormalities in offspring. Teprotumumab exposure may lead to an increase in fetal loss [see Data]. Therefore, TEPEZZA should not be used in pregnancy, and appropriate forms of contraception should be implemented prior to initiation, during treatment and for 6 months following the last dose of TEPEZZA. Women of childbearing age should have a pregnancy test performed by their doctor before starting treatment with TEPEZZA. If the patient becomes pregnant during treatment, TEPEZZA should be discontinued and the patient advised of the potential risk to the fetus.
The background rate of major birth defects and miscarriage is unknown for the indicated population. In the U.S. general population, the estimated background risks of major birth defects and miscarriage in clinically recognized pregnancies are 2-4% and 15-20%, respectively.
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Refrigerate at 2°C to 8°C (36°F to 46°F) in original carton until time of use to protect from light. Do not freeze.
10 Overdosage (10 OVERDOSAGE)
No information is available for patients who have received an overdosage.
11 Description (11 DESCRIPTION)
Teprotumumab-trbw, an insulin-like growth factor-1 receptor inhibitor (IGF-1R), is a fully human IgG1 monoclonal antibody produced in Chinese hamster ovary (CHO-DG44) cells. It has a molecular weight of approximately 148 kilodaltons.
TEPEZZA (teprotumumab-trbw) for injection is supplied as a sterile, preservative-free, white to off-white, lyophilized powder for intravenous infusion. Each single-dose vial delivers 500 mg of teprotumumab-trbw, histidine (7.82 mg), L-histidine hydrochloride monohydrate (33.43 mg), polysorbate 20 (1.05 mg), and trehalose (898.43 mg). After reconstitution with 10 mL of Sterile Water for Injection, USP, the final concentration is 47.6 mg/mL with a pH of 5.5.
5.3 Hyperglycemia
Hyperglycemia or increased blood glucose may occur in patients treated with TEPEZZA. In the premarketing clinical trials, 10% of patients (two-thirds of whom had preexisting diabetes or impaired glucose tolerance) experienced hyperglycemia [see Adverse Reactions (6.1)]. Hyperglycemic events should be controlled with medications for glycemic control, if necessary.
Assess patients for elevated blood glucose and symptoms of hyperglycemia prior to infusion and continue to monitor while on treatment with TEPEZZA. Ensure patients with hyperglycemia or preexisting diabetes are under appropriate glycemic control before and while receiving TEPEZZA.
8.4 Pediatric Use
Safety and effectiveness have not been established in pediatric patients.
8.5 Geriatric Use
Of the 171 patients in the two randomized trials, 15% were 65 years of age or older; the number of patients 65 years or older was similar between treatment groups. No overall differences in efficacy or safety were observed between patients 65 years or older and younger patients (less than 65 years of age).
2.3 Administration
Administer the diluted solution intravenously over at least 90 minutes for the first two infusions. If well tolerated, the minimum time for subsequent infusions can be reduced to 60 minutes. If not well tolerated, the minimum time for subsequent infusions should remain at 90 minutes, and pre-medication is recommended for subsequent infusions.
Do not administer as an intravenous push or bolus. TEPEZZA should not be infused concomitantly with other agents.
12.6 Immunogenicity
The observed incidence of anti-drug antibody is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibody in the studies described below with the incidence of anti-drug antibodies in other studies, including those of TEPEZZA.
In a randomized placebo-controlled study, HZNP-TEP-301, where active TED patients were administered placebo or TEPEZZA over a 24-week period, 2.4% (1 of 42) of placebo and 0% (0 of 41) of TEPEZZA treated patients tested positive for anti-teprotumumab antibodies at post-baseline visits.
Because of the low occurrence of anti-teprotumumab antibodies, the effect of these antibodies on the pharmacokinetics, pharmacodynamics, safety, and/or effectiveness of teprotumumab products is unknown.
14 Clinical Studies (14 CLINICAL STUDIES)
TEPEZZA was evaluated in 2 randomized, double-masked, placebo-controlled studies in 171 patients with Thyroid Eye Disease: Study 1 (NCT01868997) and Study 2 (NCT03298867). Patients were randomized to receive TEPEZZA or placebo in a 1:1 ratio. Patients were given intravenous infusions (10 mg/kg for first infusion and 20 mg/kg for the remaining 7 infusions) every 3 weeks for a total of 8 infusions. Patients had a clinical diagnosis of Thyroid Eye Disease with symptoms and were euthyroid or had thyroxine and free triiodothyronine levels less than 50% above or below normal limits. Prior surgical treatment for Thyroid Eye Disease was not permitted. Proptosis ranged from 16 to 33 mm and 125 patients (73%) had diplopia at baseline.
A total of 84 patients were randomized to TEPEZZA and 87 patients were randomized to placebo. The median age was 52 years (range 20 to 79 years), 86% were White, 9% were Black or African-American, 4% were Asian and 1% identified as Other. The majority (73%) were female. At baseline, 27% of patients were smokers.
The proptosis responder rate at week 24 was defined as the percentage of patients with ≥ 2 mm reduction in proptosis in the study eye from baseline, without deterioration in the non-study eye (≥ 2 mm increase) in proptosis. Additional evaluations included signs and symptoms of Thyroid Eye Disease including pain, gaze evoked orbital pain, swelling, eyelid erythema, redness, chemosis, inflammation, clinical activity score and assessments of functional vision and patient appearance. Results for proptosis are found in Table 2.
| Study 1 | Study 2 | |||||
|---|---|---|---|---|---|---|
| Teprotumumab (N = 42) |
Placebo (N = 45) |
Difference (95% CI) |
Teprotumumab (N = 41) |
Placebo (N = 42) |
Difference (95% CI) |
|
| Proptosis responder rate at week 24, % (n) Difference and its corresponding 95% Confidence Interval (CI) is based on a weighted average of the difference within each randomization stratum (tobacco user, tobacco non-use) using CMH weights.
|
71% (30) | 20% (9) | 51% (33, 69) |
83% (34) | 10% (4) | 73% (59, 88) |
| Proptosis (mm) average change from baseline through week 24, LS Mean (SE) Results were obtained from an MMRM with an unstructured covariance matrix and including treatment, smoking status, baseline value, visit, treatment by visit, and visit by baseline value interaction as fixed effects. A change from baseline of 0 was imputed at the first post-baseline visit for any subject without a post baseline value.
|
-2.5 (0.2) | -0.2 (0.2) | -2.3 (-2.8, -1.8) |
-2.8 (0.2) | -0.5 (0.2) | -2.3 (-2.8, -1.8) |
In Study 2, improvement of proptosis as measured by mean change from Baseline was observed as early as 6 weeks and continued to improve through week 24 as shown in Figure 1. Similar results were seen in Study 1.
Figure 1. Change from Baseline in Proptosis over 24 Weeks in Study 2
P < 0.01 at each timepoint
TEPEZZA also led to improvement in the less severely impacted "fellow" eye.
Diplopia (double vision) was evaluated in a subgroup of patients that had diplopia at baseline in Study 1 and 2. Results are shown in Table 3.
| Parameter | TEPEZZA (n = 66) |
Placebo (n = 59) |
|---|---|---|
| P < 0.01 | ||
| Diplopia | ||
| Responder rate Diplopia was evaluated on a 4-point scale where scores ranged from 0 for no diplopia to 3 for constant diplopia. A diplopia responder was defined as a patient with baseline diplopia > 0 and a score of 0 at week 24. at week 24, % (n) |
53% (35) | 25% (15) |
Following discontinuation of treatment in Study 1, 53% of patients (16 of 30 patients) who were proptosis responders at week 24 maintained proptosis response 51 weeks after the last TEPEZZA infusion. 67% of patients (12 of 18) who were diplopia responders at week 24 maintained diplopia response 51 weeks after the last TEPEZZA infusion.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Infusion Reactions [see Warnings and Precautions (5.1)]
- Inflammatory Bowel Disease [see Warnings and Precautions (5.2)]
- Hyperglycemia [see Warnings and Precautions (5.3)]
- Hearing Impairment Including Hearing Loss [see Warnings and Precautions (5.4)]
12.2 Pharmacodynamics
No formal pharmacodynamic studies have been conducted with teprotumumab-trbw.
12.3 Pharmacokinetics
The pharmacokinetics of teprotumumab-trbw was described by a two compartment population PK model based on data from 40 patients with Thyroid Eye Disease receiving an initial intravenous infusion of 10 mg/kg, followed by infusions of 20 mg/kg TEPEZZA every 3 weeks in one clinical trial. Following this regimen, the mean (± standard deviation) estimates for steady-state area under the concentration curve (AUC), peak (Cmax), and trough (Ctrough) concentrations of teprotumumab-trbw were 138 (± 34) mg∙hr/mL, 632 (± 139) mcg/mL, and 176 (± 56) mcg/mL, respectively.
2.1 Recommended Dosing
The recommended dose of TEPEZZA is an intravenous infusion of 10 mg/kg for the initial dose followed by an intravenous infusion of 20 mg/kg every three weeks for 7 additional infusions.
5.1 Infusion Reactions
TEPEZZA may cause infusion reactions. Infusion reactions have been reported in approximately 4% of patients treated with TEPEZZA. Signs and symptoms of infusion-related reactions include transient increases in blood pressure, feeling hot, tachycardia, dyspnea, headache and muscular pain. Infusion reactions may occur during any of the infusions or within 1.5 hours after an infusion. Reported infusion reactions are usually mild or moderate in severity and can usually be successfully managed with corticosteroids and antihistamines. In patients who experience an infusion reaction, consideration should be given to pre-medicating with an antihistamine, antipyretic, corticosteroid and/or administering all subsequent infusions at a slower infusion rate.
1 Indications and Usage (1 INDICATIONS AND USAGE)
TEPEZZA is indicated for the treatment of Thyroid Eye Disease regardless of Thyroid Eye Disease activity or duration.
12.1 Mechanism of Action
Teprotumumab-trbw's mechanism of action in patients with Thyroid Eye Disease has not been fully characterized. Teprotumumab-trbw binds to IGF-1R and blocks its activation and signaling.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Infusion Reactions: If an infusion reaction occurs, interrupt or slow the rate of infusion and use appropriate medical management (5.1)
- Inflammatory Bowel Disease (IBD): Monitor patients; for signs and symptoms of disease, including patients without a history of IBD; discontinue TEPEZZA if IBD exacerbation is suspected (5.2)
- Hyperglycemia: Assess patients for elevated blood glucose and symptoms of hyperglycemia prior to infusion and continue to monitor while on treatment with TEPEZZA. Ensure patients with hyperglycemia or preexisting diabetes are under appropriate glycemic control before and while receiving TEPEZZA (5.3)
- Hearing Impairment Including Hearing Loss: TEPEZZA may cause severe hearing impairment including hearing loss, which in some cases may be permanent. Assess patients' hearing before, during, and after treatment with TEPEZZA and consider the benefit-risk of treatment with patients (5.4)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For injection (intravenous infusion): 500 mg of teprotumumab as a white to off-white lyophilized powder in a single-dose vial for reconstitution and dilution.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of TEPEZZA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: bowel perforation, exacerbation of inflammatory bowel disease (IBD), including patients without a prior diagnosis of IBD
Metabolism and Nutrition Disorders: diabetic ketoacidosis, hyperosmolar hyperglycemic state (HHS)
Otologic: severe hearing impairment including hearing loss, which in some cases may be permanent
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Females of Reproductive Potential: Appropriate forms of contraception should be implemented prior to initiation, during treatment and for 6 months following the last dose of TEPEZZA (8.3)
5.2 Inflammatory Bowel Disease
TEPEZZA may cause an exacerbation of inflammatory bowel disease (IBD). IBD has been reported in some patients without a prior diagnosis of IBD [see Adverse Reactions (6.2)]. Monitor patients for signs and symptoms of IBD. If IBD exacerbation is suspected, discontinue use of TEPEZZA.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of TEPEZZA was evaluated in two premarketing randomized, double-masked, placebo-controlled clinical trials (Study 1 [NCT01868997] and Study 2 [NCT03298867]) consisting of 170 patients with Thyroid Eye Disease (84 received TEPEZZA and 86 received placebo). Patients were treated with TEPEZZA (10 mg/kg for first infusion and 20 mg/kg for the remaining 7 infusions) or placebo given as an intravenous infusion every 3 weeks for a total of 8 infusions. The majority of patients completed 8 infusions (89% of TEPEZZA patients and 93% of placebo patients).
The most common adverse reactions (≥ 5%) that occurred at greater incidence in the TEPEZZA group than in the control group during the treatment period of Studies 1 and 2 are summarized in Table 1.
In addition, menstrual disorders (amenorrhea, metrorrhagia, dysmenorrhea) were reported in approximately 23% (5 of 22 patients) of menstruating women treated with TEPEZZA compared to 4% (1 of 25 patients) treated with placebo in the premarketing clinical trials.
| Adverse Reactions | TEPEZZA N = 84 N (%) |
Placebo N = 86 N (%) |
|---|---|---|
| Muscle spasms | 21 (25%) | 6 (7%) |
| Nausea | 14 (17%) | 8 (9%) |
| Alopecia | 11 (13%) | 7 (8%) |
| Diarrhea | 10 (12%) | 7 (8%) |
| Fatigue Fatigue includes asthenia
|
10 (12%) | 6 (7%) |
| Hyperglycemia Hyperglycemia includes blood glucose increase
|
8 (10%) | 1 (1%) |
| Hearing impairment Hearing impairment including hearing loss (deafness, including sensorineural deafness, eustachian tube dysfunction, hyperacusis, hypoacusis, autophony and tinnitus)
|
8 (10%) | 0 |
| Dysgeusia | 7 (8%) | 0 |
| Headache | 7 (8%) | 6 (7%) |
| Dry skin | 7 (8%) | 0 |
| Weight decreased | 5 (6%) | 0 |
| Nail disorder Nail disorder (includes nail discoloration, nail disorder and onychoclasis)
|
4 (5%) | 0 |
In clinical trials, gastrointestinal complaints including exacerbation of inflammatory bowel disease were reported.
In two postapproval trials, the following were the adverse reactions occurring ≥ 5% of participants treated with TEPEZZA during the double-masked treatment period: muscle spasms (29%), hearing impairment (19%), hyperglycemia (18%) diarrhea (16%), fatigue (13%), headache (13%), alopecia (10%), dry skin (10%), dysgeusia (9%), ear discomfort (9%), and nail disorder (6%).
2.2 Reconstitution and Preparation
Step 1: Calculate the dose (mg) and determine the number of vials needed for the 10 or 20 mg/kg dosage based on patient weight. Each TEPEZZA vial contains 500 mg of the teprotumumab antibody.
Step 2: Using appropriate aseptic technique, reconstitute each TEPEZZA vial with 10 mL of Sterile Water for Injection, USP. Ensure that the stream of diluent is not directed onto the lyophilized powder, which has a cake-like appearance. Do not shake, but gently swirl the solution by rotating the vial until the lyophilized powder is dissolved. The reconstituted solution has a total volume of 10.5 mL. Withdraw 10.5 mL of reconstituted solution to obtain 500 mg. After reconstitution, the final concentration is 47.6 mg/mL.
Step 3: The reconstituted TEPEZZA solution must be further diluted in 0.9% Sodium Chloride Injection, USP prior to infusion. To prepare the diluted solution, use 100 mL infusion bags for a dose less than 1800 mg, and 250 mL infusion bags for a dose equal to or greater than 1800 mg. To maintain a constant volume in the infusion bag, a sterile syringe and needle should be used to remove the volume equivalent to the amount of the reconstituted TEPEZZA solution to be placed into the infusion bag. Discard the volume of 0.9% Sodium Chloride Injection, USP withdrawn.
Step 4: Withdraw the required volume from the reconstituted TEPEZZA vial(s) based on the patient's weight (in kg) and transfer into the intravenous bag containing 0.9% Sodium Chloride Injection, USP. Mix diluted solution by gentle inversion. Do not shake. If refrigerated prior to administration, allow the diluted solution to reach room temperature prior to infusion.
The product does not contain any preservative. The combined storage time of reconstituted TEPEZZA solution in the vial and the diluted solution in the infusion bag containing 0.9% Sodium Chloride Injection, USP is a total of 4 hours at room temperature 20°C to 25°C (68°F to 77°F) or up to 48 hours under refrigerated conditions 2°C to 8°C (36°F to 46°F) protected from light.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Upon reconstitution, TEPEZZA is a nearly colorless or slightly brown, clear to opalescent solution which is free of foreign particulate matter. Discard the solution if any particulate matter or discoloration are observed.
Do not freeze the reconstituted or diluted solution.
Discard vial(s) and all unused contents.
No incompatibilities between TEPEZZA and polyethylene (PE), polyvinyl chloride (PVC), polyurethane (PUR) or polyolefin (PO) bags and intravenous administration sets have been observed.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
TEPEZZA (teprotumumab-trbw) for injection is a sterile, preservative-free, white to off-white lyophilized powder available as follows:
| Carton containing one 500 mg single-dose vial | NDC 75987-130-15 |
Principal Display Panel 500 Mg Vial Carton (PRINCIPAL DISPLAY PANEL - 500 mg Vial Carton)
NDC-75987-130-15
TEPEZZA®
(teprotumumab-trbw)
for Injection
500 mg/vial
For Intravenous Infusion Only
Reconstitute and Further Dilute Prior to Use
Single-dose vial. Discard unused portion.
AMGEN
5.4 Hearing Impairment Including Hearing Loss
TEPEZZA may cause severe hearing impairment including hearing loss, which in some cases may be permanent. Assess patients' hearing before, during, and after treatment with TEPEZZA and consider the benefit-risk of treatment with patients.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:11.571270 · Updated: 2026-03-14T22:49:26.517746