83766-107 Drawzone Toenail Fungus Treatment Pen
3dcdb0c4-e0ec-dfee-e063-6294a90a0bda
34390-5
HUMAN OTC DRUG LABEL
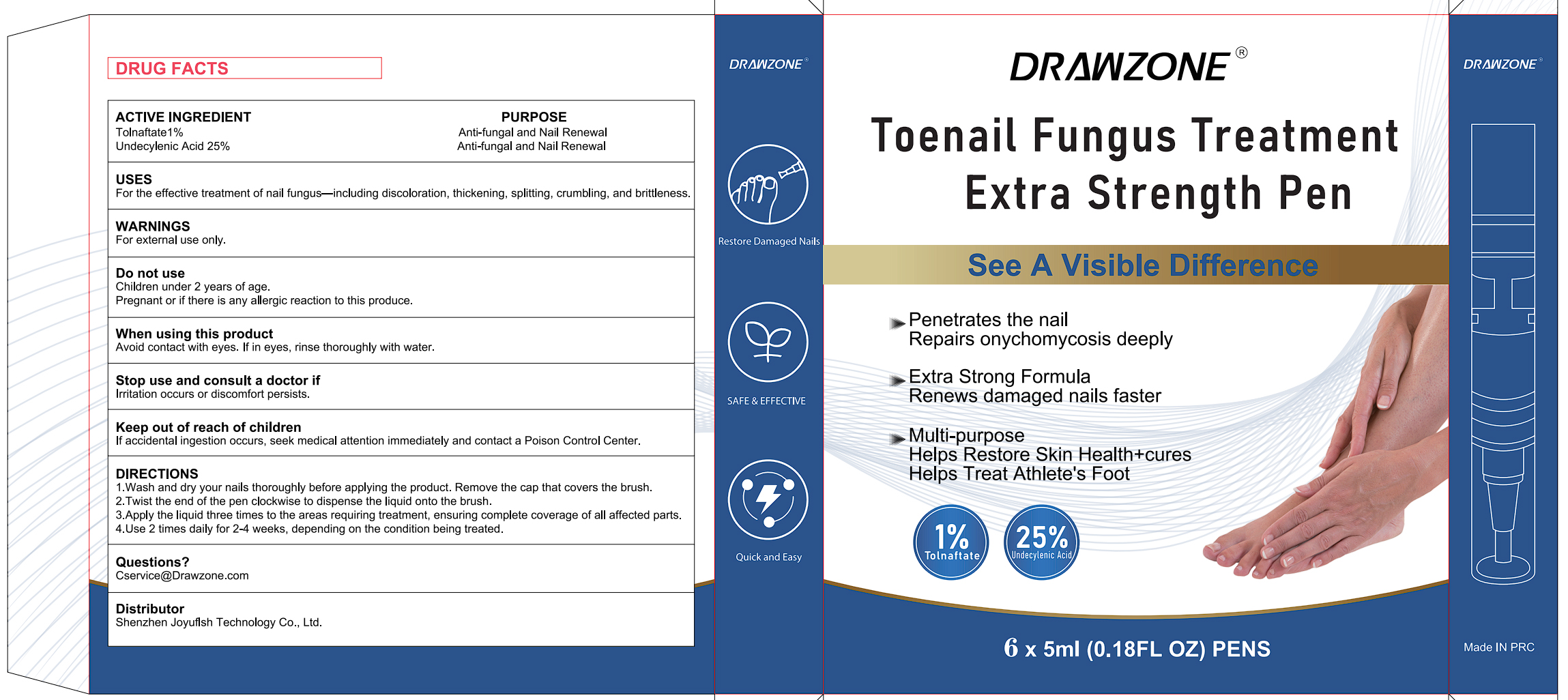
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Tolnaftate 1% UNDECYLENIC ACID 25%
Medication Information
Warnings and Precautions
For external use only.
Indications and Usage
For the effective treatment of nail fungus-including discoloration, thickening, spliting, crumbling, and britleness
Dosage and Administration
1.Wash and dry your nails thoroughly before applying the product. Remove the cap that covers the brush.
2.Twist the end of the pen clockwise to dispense the liquid onto the brush.
3.Apply the liquid three times to the areas requiring treatment, ensuring complete coverage of all affected parts.
4.Use 2 times daily for 2-4 weeks, depending on the condition being treated.
Description
Tolnaftate 1% UNDECYLENIC ACID 25%
Section 50565-1
lf accidental ingestion occurs, seek medical attention immediately and contact a Poison Control Center.
Section 50566-9
lrritation occurs or discomfort persists
Section 50567-7
Avoid contact with eyes. lf in eyes, rinse thoroughly with water.
Section 50570-1
Children under 2 years of age.
Pregnant or if there is any allergic reaction to this produce.
Section 51727-6
ALCOHOL
POLYETHYLENE GLYCOL
PROPYLENE GLYCOL
DIMETHICONE
C12-15 ALKYL LACTATE
ETHYLHEXYLGLYCERIN
GLYCERIN
LAUROCAPRAM
SOPHORA FLAVESCENS ROOT
TOCOPHEROL
PHENOXYETHANOL
WATER
Section 51945-4
Section 55105-1
Anti-fungal and Nail Renewal
Section 55106-9
Tolnaftate 1%
UNDECYLENIC ACID 25%
Structured Label Content
Indications and Usage (34067-9)
For the effective treatment of nail fungus-including discoloration, thickening, spliting, crumbling, and britleness
Dosage and Administration (34068-7)
1.Wash and dry your nails thoroughly before applying the product. Remove the cap that covers the brush.
2.Twist the end of the pen clockwise to dispense the liquid onto the brush.
3.Apply the liquid three times to the areas requiring treatment, ensuring complete coverage of all affected parts.
4.Use 2 times daily for 2-4 weeks, depending on the condition being treated.
Warnings and Precautions (34071-1)
For external use only.
Section 50565-1 (50565-1)
lf accidental ingestion occurs, seek medical attention immediately and contact a Poison Control Center.
Section 50566-9 (50566-9)
lrritation occurs or discomfort persists
Section 50567-7 (50567-7)
Avoid contact with eyes. lf in eyes, rinse thoroughly with water.
Section 50570-1 (50570-1)
Children under 2 years of age.
Pregnant or if there is any allergic reaction to this produce.
Section 51727-6 (51727-6)
ALCOHOL
POLYETHYLENE GLYCOL
PROPYLENE GLYCOL
DIMETHICONE
C12-15 ALKYL LACTATE
ETHYLHEXYLGLYCERIN
GLYCERIN
LAUROCAPRAM
SOPHORA FLAVESCENS ROOT
TOCOPHEROL
PHENOXYETHANOL
WATER
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Anti-fungal and Nail Renewal
Section 55106-9 (55106-9)
Tolnaftate 1%
UNDECYLENIC ACID 25%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:55.494556 · Updated: 2026-03-14T23:10:01.237919