3dbe6e5b-1ca7-6941-e063-6294a90ab042
34390-5
HUMAN OTC DRUG LABEL
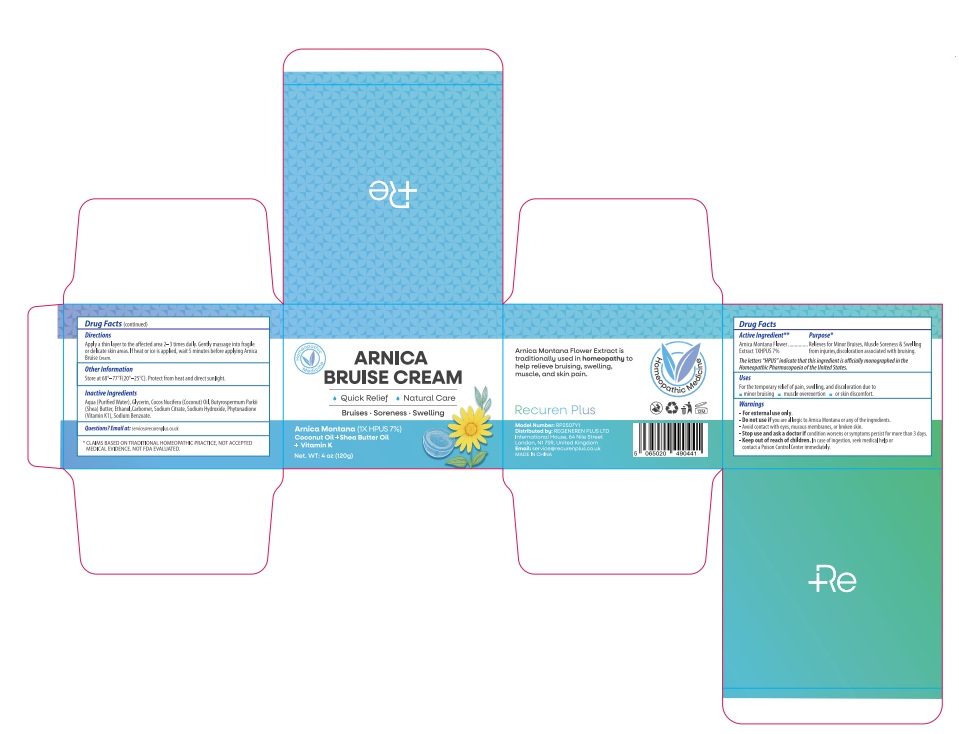
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Arnica montana 1X HPUS 7% The letters "HPUS" indicate that the components in this product are officially monographed in the Homeopathic Pharmacopoeia of the United States.
Medication Information
Warnings and Precautions
For external use only.
Indications and Usage
For the temporary relief of pain, swelling, and discoloration due to minor bruising, muscle overexertion, or skin discomfort.
Dosage and Administration
Apply a thin layer to the affected area 2–3 times daily. Gently massage into fragile or delicate skin areas. If heat or ice is applied, wait 5 minutes before applying Arnica Bruise Cream.
Description
Arnica montana 1X HPUS 7% The letters "HPUS" indicate that the components in this product are officially monographed in the Homeopathic Pharmacopoeia of the United States.
Section 44425-7
Store at 68°–77°F(20°–25°C). Protect from heat and direct sunlight.
Section 50565-1
Keep out of reach of children. In case of ingestion, seek medical help or contact a Poison Control Center immediately.
Section 50566-9
Stop use and ask a doctor if condition worsens or symptoms persist for more than 3 days.
Section 50570-1
if you are allergic to Arnica Montana or any of the ingredients.
Section 51727-6
Aqua (Purified Water), Glycerin, Cocos Nucifera (Coconut) Oil, Butyrospermum Parkii (Shea) Butter, Ethanol,Carbomer, Sodium Citrate, Sodium Hydroxide, Phytonadione (Vitamin K1), Sodium Benzoate.
Section 51945-4
Section 53413-1
Email at: [email protected]
Section 55105-1
Relieves for Minor Bruises, Muscle Soreness & Swelling from injuries,discoloration associated with bruising.
Section 55106-9
Arnica montana 1X HPUS 7%
The letters "HPUS" indicate that the components in this product are officially monographed in the Homeopathic Pharmacopoeia of the United States.
Section 60561-8
Avoid contact with eyes, mucous membranes, or broken skin.
Structured Label Content
Indications and Usage (34067-9)
For the temporary relief of pain, swelling, and discoloration due to minor bruising, muscle overexertion, or skin discomfort.
Dosage and Administration (34068-7)
Apply a thin layer to the affected area 2–3 times daily. Gently massage into fragile or delicate skin areas. If heat or ice is applied, wait 5 minutes before applying Arnica Bruise Cream.
Warnings and Precautions (34071-1)
For external use only.
Section 44425-7 (44425-7)
Store at 68°–77°F(20°–25°C). Protect from heat and direct sunlight.
Section 50565-1 (50565-1)
Keep out of reach of children. In case of ingestion, seek medical help or contact a Poison Control Center immediately.
Section 50566-9 (50566-9)
Stop use and ask a doctor if condition worsens or symptoms persist for more than 3 days.
Section 50570-1 (50570-1)
if you are allergic to Arnica Montana or any of the ingredients.
Section 51727-6 (51727-6)
Aqua (Purified Water), Glycerin, Cocos Nucifera (Coconut) Oil, Butyrospermum Parkii (Shea) Butter, Ethanol,Carbomer, Sodium Citrate, Sodium Hydroxide, Phytonadione (Vitamin K1), Sodium Benzoate.
Section 51945-4 (51945-4)
Section 53413-1 (53413-1)
Email at: [email protected]
Section 55105-1 (55105-1)
Relieves for Minor Bruises, Muscle Soreness & Swelling from injuries,discoloration associated with bruising.
Section 55106-9 (55106-9)
Arnica montana 1X HPUS 7%
The letters "HPUS" indicate that the components in this product are officially monographed in the Homeopathic Pharmacopoeia of the United States.
Section 60561-8 (60561-8)
Avoid contact with eyes, mucous membranes, or broken skin.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:12:06.878337 · Updated: 2026-03-14T23:17:41.257754