Signature Numb Cream 10g/0.35 Oz
3d2ecb86-6bb1-2528-e063-6394a90a8d89
34390-5
HUMAN OTC DRUG LABEL
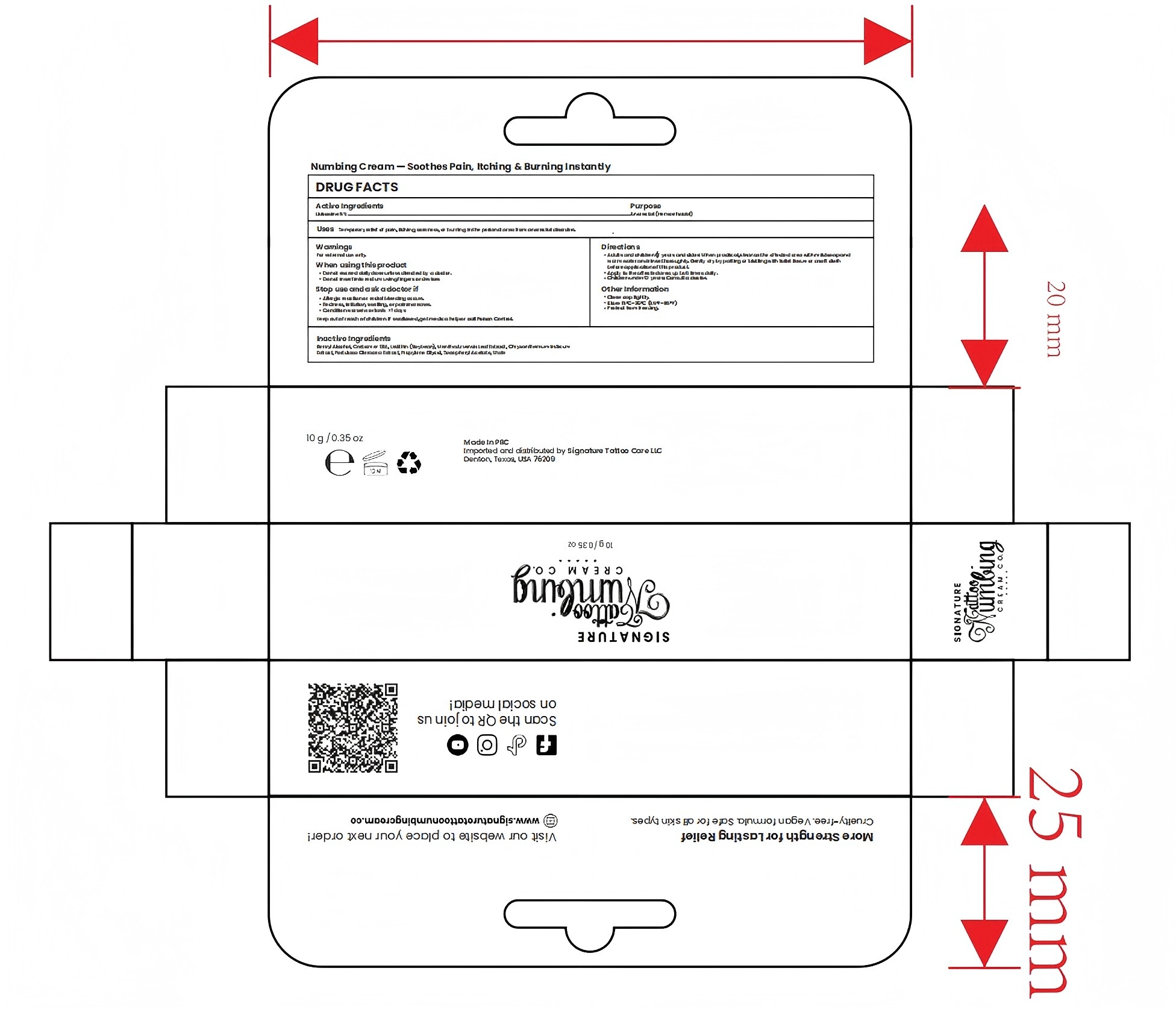
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Stop use and consulta doctor if an allergicreaction occurs. Keep out of reach of children.
Medication Information
Warnings and Precautions
Stop use and consulta doctor if an allergicreaction occurs.
Keep out of reach of children.
Indications and Usage
Adults and children 12 years and older: When practical, cleanse the affected area with mild soap andwarm water and rinse thoroughly. Gently dry by patting or blotting with toilet tissue or a soft clothbefore application of this product.Apply to the affected area up to 6 times daily.Children under 12 years: Consult a doctor.
Dosage and Administration
Apply 1-3 times a week
Description
Stop use and consulta doctor if an allergicreaction occurs. Keep out of reach of children.
Section 50565-1
KEEP OUT OF REACH OF CHILDREN SECTION
Section 50566-9
Allergic reaction or rectal bleeding occurs.Redness,irritation, swelling, or pain increases.Condition worsens or lasts >7 days
Section 50567-7
Wash the area with soap and water.
Section 51727-6
Benzyl Alcohol, Carbomer 934, Lecithin (Soybean), Mentha Arvensis Leaf Extract, Chrysanthemum Indicum
Extract, Portulaca Oleracea Extract, Propylene Glycol, Tocopheryl Acetate, Water
Section 51945-4
Section 55105-1
Used for numbing local area prior to tattooing
Section 55106-9
5% Lidocaine
Structured Label Content
Indications and Usage (34067-9)
Adults and children 12 years and older: When practical, cleanse the affected area with mild soap andwarm water and rinse thoroughly. Gently dry by patting or blotting with toilet tissue or a soft clothbefore application of this product.Apply to the affected area up to 6 times daily.Children under 12 years: Consult a doctor.
Dosage and Administration (34068-7)
Apply 1-3 times a week
Warnings and Precautions (34071-1)
Stop use and consulta doctor if an allergicreaction occurs.
Keep out of reach of children.
Section 50565-1 (50565-1)
KEEP OUT OF REACH OF CHILDREN SECTION
Section 50566-9 (50566-9)
Allergic reaction or rectal bleeding occurs.Redness,irritation, swelling, or pain increases.Condition worsens or lasts >7 days
Section 50567-7 (50567-7)
Wash the area with soap and water.
Section 51727-6 (51727-6)
Benzyl Alcohol, Carbomer 934, Lecithin (Soybean), Mentha Arvensis Leaf Extract, Chrysanthemum Indicum
Extract, Portulaca Oleracea Extract, Propylene Glycol, Tocopheryl Acetate, Water
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Used for numbing local area prior to tattooing
Section 55106-9 (55106-9)
5% Lidocaine
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:51.585033 · Updated: 2026-03-14T23:09:51.959406