These Highlights Do Not Include All The Information Needed To Use Aptiom Safely And Effectively. See Full Prescribing Information For Aptiom.
3d0c9554-eaeb-4694-8089-00133fcadce3
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
APTIOM is indicated for the treatment of partial-onset seizures in patients 4 years of age and older.
Indications and Usage
APTIOM is indicated for the treatment of partial-onset seizures in patients 4 years of age and older.
Dosage and Administration
Adult Patients: The recommended initial dosage of APTIOM is 400 mg once daily. For some patients, treatment may be initiated at 800 mg once daily if the need for seizure reduction outweighs an increased risk of adverse reactions. Increase the dose in weekly increments of 400 mg to 600 mg once daily, based on clinical response and tolerability, to a recommended maintenance dosage of 800 mg to 1600 mg once daily. ( 2.2 ) Pediatric Patients: The recommended dosage of APTIOM is based on body weight and is administered orally once daily. Increase the dose in weekly intervals based on clinical response and tolerability, to the recommended maintenance dosage. ( 2.2 ) Patients with Moderate or Severe Renal Impairment: Reduce dosage by 50%. ( 2.4 )
Warnings and Precautions
Suicidal Behavior and Ideation: Monitor for suicidal thoughts or behavior. ( 5.1 ) Serious Dermatologic Reactions, Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), Anaphylactic Reactions and Angioedema: Monitor and discontinue if another cause cannot be established. ( 5.2 , 5.3 , 5.4 ) Hyponatremia: Monitor sodium levels in patients at risk or patients experiencing hyponatremia symptoms. ( 5.5 ) Neurological Adverse Reactions: Monitor for dizziness, disturbance in gait and coordination, somnolence, fatigue, cognitive dysfunction, and visual changes. Use caution when driving or operating machinery. ( 5.6 ) Withdrawal of APTIOM: Withdraw APTIOM gradually to minimize the risk of increased seizure frequency and status epilepticus. ( 2.6 , 5.7 , 8.1 ) Drug Induced Liver Injury: Discontinue APTIOM in patients with jaundice or evidence of significant liver injury. ( 5.8 ) Hematologic Adverse Reactions: Consider discontinuing. ( 5.10 )
Contraindications
APTIOM is contraindicated in patients with a hypersensitivity to eslicarbazepine acetate or oxcarbazepine [see Warnings and Precautions ( 5.2 , 5.3 , and 5.4 )].
Adverse Reactions
Rare cases of pancytopenia, agranulocytosis, and leukopenia have been reported during postmarketing use in patients treated with APTIOM. Discontinuation of APTIOM should be considered in patients who develop pancytopenia, agranulocytosis, or leukopenia.
Drug Interactions
Carbamazepine: May need dose adjustment for APTIOM or carbamazepine. ( 2.3 , 5.6 , 7.1 ) Phenytoin: Higher dosage of APTIOM may be necessary and dose adjustment may be needed for phenytoin. ( 2.3 , 7.1 , 7.2 ) Phenobarbital or Primidone: Higher dosage of APTIOM may be necessary. ( 2.3 , 7.1 ) Hormonal Contraceptives: APTIOM may decrease the effectiveness of hormonal contraceptives. ( 7.4 , 8.3 )
Storage and Handling
Store APTIOM tablets at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
How Supplied
APTIOM tablets are white, oblong and with functional scoring on one side (200 mg, 600 mg, and 800 mg) or white, circular bi-convex and plain on one side (400 mg) and identified with strength-specific one-sided engraving on the other side, “ESL 200” (200 mg), “ESL 400” (400 mg), “ESL 600” (600 mg), or “ESL 800” (800 mg). Tablets are supplied in the following strengths and package configurations ( Table 6 ): Table 6: Package Configuration for APTIOM Tablets Tablet Strength Package Configuration NDC Code 200 mg Bottles of 30 63402-202-30 400 mg Bottles of 30 63402-204-30 600 mg Bottles of 60 63402-206-60 800 mg Bottles of 30 63402-208-30
Medication Information
Warnings and Precautions
Suicidal Behavior and Ideation: Monitor for suicidal thoughts or behavior. ( 5.1 ) Serious Dermatologic Reactions, Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), Anaphylactic Reactions and Angioedema: Monitor and discontinue if another cause cannot be established. ( 5.2 , 5.3 , 5.4 ) Hyponatremia: Monitor sodium levels in patients at risk or patients experiencing hyponatremia symptoms. ( 5.5 ) Neurological Adverse Reactions: Monitor for dizziness, disturbance in gait and coordination, somnolence, fatigue, cognitive dysfunction, and visual changes. Use caution when driving or operating machinery. ( 5.6 ) Withdrawal of APTIOM: Withdraw APTIOM gradually to minimize the risk of increased seizure frequency and status epilepticus. ( 2.6 , 5.7 , 8.1 ) Drug Induced Liver Injury: Discontinue APTIOM in patients with jaundice or evidence of significant liver injury. ( 5.8 ) Hematologic Adverse Reactions: Consider discontinuing. ( 5.10 )
Indications and Usage
APTIOM is indicated for the treatment of partial-onset seizures in patients 4 years of age and older.
Dosage and Administration
Adult Patients: The recommended initial dosage of APTIOM is 400 mg once daily. For some patients, treatment may be initiated at 800 mg once daily if the need for seizure reduction outweighs an increased risk of adverse reactions. Increase the dose in weekly increments of 400 mg to 600 mg once daily, based on clinical response and tolerability, to a recommended maintenance dosage of 800 mg to 1600 mg once daily. ( 2.2 ) Pediatric Patients: The recommended dosage of APTIOM is based on body weight and is administered orally once daily. Increase the dose in weekly intervals based on clinical response and tolerability, to the recommended maintenance dosage. ( 2.2 ) Patients with Moderate or Severe Renal Impairment: Reduce dosage by 50%. ( 2.4 )
Contraindications
APTIOM is contraindicated in patients with a hypersensitivity to eslicarbazepine acetate or oxcarbazepine [see Warnings and Precautions ( 5.2 , 5.3 , and 5.4 )].
Adverse Reactions
Rare cases of pancytopenia, agranulocytosis, and leukopenia have been reported during postmarketing use in patients treated with APTIOM. Discontinuation of APTIOM should be considered in patients who develop pancytopenia, agranulocytosis, or leukopenia.
Drug Interactions
Carbamazepine: May need dose adjustment for APTIOM or carbamazepine. ( 2.3 , 5.6 , 7.1 ) Phenytoin: Higher dosage of APTIOM may be necessary and dose adjustment may be needed for phenytoin. ( 2.3 , 7.1 , 7.2 ) Phenobarbital or Primidone: Higher dosage of APTIOM may be necessary. ( 2.3 , 7.1 ) Hormonal Contraceptives: APTIOM may decrease the effectiveness of hormonal contraceptives. ( 7.4 , 8.3 )
Storage and Handling
Store APTIOM tablets at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
How Supplied
APTIOM tablets are white, oblong and with functional scoring on one side (200 mg, 600 mg, and 800 mg) or white, circular bi-convex and plain on one side (400 mg) and identified with strength-specific one-sided engraving on the other side, “ESL 200” (200 mg), “ESL 400” (400 mg), “ESL 600” (600 mg), or “ESL 800” (800 mg). Tablets are supplied in the following strengths and package configurations ( Table 6 ): Table 6: Package Configuration for APTIOM Tablets Tablet Strength Package Configuration NDC Code 200 mg Bottles of 30 63402-202-30 400 mg Bottles of 30 63402-204-30 600 mg Bottles of 60 63402-206-60 800 mg Bottles of 30 63402-208-30
Description
APTIOM is indicated for the treatment of partial-onset seizures in patients 4 years of age and older.
Section 42229-5
Monotherapy and Adjunctive Therapy
Section 42231-1
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Revised 9/2017 |
|||
|
MEDICATION GUIDE
APTIOM (ap tee' om) (eslicarbazepine acetate) tablets |
||||
|
What is the most important information I should know about APTIOM?
|
||||
|
1. Like other antiepileptic drugs, APTIOM may cause suicidal thoughts or actions in a very small number of people, about 1 in 500.
Call a healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you: |
||||
|
|
|||
How can I watch for early symptoms of suicidal thoughts and actions?
|
||||
|
2. APTIOM may cause allergic reactions or serious problems which may affect organs and other parts of your body like the liver or blood cells. You may or may not have a rash with these types of reactions.
Call your healthcare provider right away if you have any of the following: |
||||
|
|
|||
|
||||
|
|
|||
| Some medicines can also cause low sodium in your blood. Be sure to tell your healthcare provider about all the other medicines that you are taking. | ||||
|
What is APTIOM?
APTIOM is a prescription medicine used to treat partial-onset seizures. It is not known if APTIOM is safe and effective in children under 4 years of age. |
||||
|
Who should not take APTIOM?
Do not take APTIOM if you are allergic to eslicarbazepine acetate, any of the other ingredients in APTIOM, or oxcarbazepine. See the end of this Medication Guide for a complete list of ingredients in APTIOM. |
||||
|
What should I tell my healthcare provider before taking APTIOM?
Before taking APTIOM, tell your healthcare provider about all your medical conditions, including if you:
|
||||
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking APTIOM with certain other medicines may cause side effects or affect how well they work. Do not start or stop other medicines without talking to your healthcare provider. Especially tell your healthcare provider if you take: |
||||
|
|
|
|
|
| Ask your healthcare provider or pharmacist for a list of these medicines, if you are not sure. Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine. |
||||
How should I take APTIOM?
|
||||
What should I avoid while taking APTIOM?
|
||||
|
What are the possible side effects of APTIOM?
See “What is the most important information I should know about APTIOM?” APTIOM may cause other serious side effects including:
|
||||
|
|
|
||
|
||||
|
|
|
||
|
Get medical help right away if you have any of the symptoms listed above or listed in “What is the most important information I should know about APTIOM?”
The most common side effects of APTIOM include: |
||||
|
|
|
|
|
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of APTIOM. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store APTIOM?
|
||||
| Keep APTIOM and all medicines out of reach of children. | ||||
|
What are the ingredients in APTIOM?
Active ingredient: eslicarbazepine acetate Inactive ingredients: croscarmellose sodium, magnesium stearate, and povidone |
||||
|
General information about the safe and effective use of APTIOM.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use APTIOM for a condition for which it was not prescribed. Do not give APTIOM to other people, even if they have the same symptoms that you have. It may harm them. This Medication Guide summarizes the most important information about APTIOM. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about APTIOM that is written for health professionals. For more information, go to www.aptiom.com or call 1-888-394-7377. |
||||
|
Manufactured for: Sunovion Pharmaceuticals Inc., Marlborough, MA 01752 USA Under license from © 2023 Sunovion Pharmaceuticals Inc. All rights reserved. |
Section 51945-4
PRINCIPAL DISPLAY PANEL – 200 MG 30 COUNT OBLONG BOTTLE
NDC 63402-202-30
30 Tablets
ONCE DAILY
Aptiom®
(eslicarbazepine acetate) Tablets
200 mg
Rx Only
ATTENTION DISPENSER: Each time Aptiom is
dispensed give the patient the accompanying
Medication Guide.
Sumitomo Pharma
Under license from Bial
9.2 Abuse
Prescription drug abuse is the intentional non-therapeutic use of a drug, even once, for its rewarding psychological or physiological effects. Drug addiction, which develops after repeated drug abuse, is characterized by a strong desire to take a drug despite harmful consequences, difficulty in controlling its use, giving a higher priority to drug use than to obligations, increased tolerance, and sometimes physical withdrawal. Drug abuse and drug addiction are separate and distinct from physical dependence (for example, abuse may not be accompanied by physical dependence) [see Drug Abuse and Dependence (9.3)].
In a human abuse study in recreational sedative abusers APTIOM showed no evidence of abuse. In Phase 1, 1.5% of the healthy volunteers taking APTIOM reported euphoria compared to 0.4% taking placebo.
15 References
French JA, Wang S, Warnock B, Temkin N. Historical control monotherapy design in the treatment of epilepsy. Epilepsia 2010;51(10):1936-43.
8.2 Lactation
Eslicarbazepine is present in human milk. The effects of APTIOM on the breastfed infant or on milk production are unknown. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for APTIOM and any potential adverse effects on the breastfed infant from APTIOM or from the underlying maternal condition.
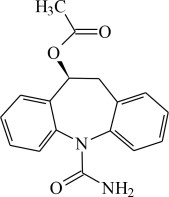
11 Description
The chemical name of APTIOM (eslicarbazepine acetate) is (S)-10-Acetoxy-10,11-dihydro-5H-dibenz[b,f]azepine-5-carboxamide. APTIOM is a dibenz[b,f]azepine-5-carboxamide derivative. Its molecular formula is C17H16N2O3 and its molecular weight is 296.32. The chemical structure is:
APTIOM is a white to off-white, odorless crystalline solid. It is insoluble in hexane, very slightly soluble in aqueous solvents and soluble in organic solvents such as acetone, acetonitrile, and methanol.
Each APTIOM tablet contains 200 mg, 400 mg, 600 mg or 800 mg of eslicarbazepine acetate and the following inactive ingredients: croscarmellose sodium, magnesium stearate, and povidone.
9.3 Dependence
Physical dependence is characterized by withdrawal symptoms after abrupt discontinuation or a significant dose reduction of a drug.
There was some evidence of physical dependence or a withdrawal syndrome with APTIOM in a physical dependence study conducted in healthy volunteers who were maintained at a daily dose of 800 mg APTIOM for 4 weeks prior to discontinuation. The primary endpoint was the maximum change from steady-state baseline in the total score of the Physician's Withdrawal Checklist (PWC-34) during the 21-day discontinuation period. APTIOM and placebo were shown to be equivalent on the primary endpoint. Two out of 8 secondary endpoints (visual analog scales for anxiety and nausea) showed some increase in these symptoms for subjects who were maintained on APTIOM and discontinued, versus subjects who were maintained on placebo. In general, AEDs should not be abruptly discontinued in patients with epilepsy because of the risk of increased seizure frequency and status epilepticus.
5.5 Hyponatremia
Clinically significant hyponatremia (sodium <125 mEq/L) can develop in patients taking APTIOM.
Measurement of serum sodium and chloride levels should be considered during maintenance treatment with APTIOM, particularly if the patient is receiving other medications known to decrease serum sodium levels, and should be performed if symptoms of hyponatremia develop (e.g., nausea/vomiting, malaise, headache, lethargy, confusion, irritability, muscle weakness/spasms, obtundation, or increase in seizure frequency or severity). Cases of symptomatic hyponatremia and syndrome of inappropriate antidiuretic hormone secretion (SIADH) have been reported during postmarketing use. In clinical trials, patients whose treatment with APTIOM was discontinued because of hyponatremia generally experienced normalization of serum sodium within a few days without additional treatment.
In the controlled adult adjunctive epilepsy trials, 4/415 patients (1.0%) treated with 800 mg and 6/410 (1.5%) patients treated with 1200 mg of APTIOM had at least one serum sodium value less than 125 mEq/L, compared to none of the patients assigned to placebo. A higher percentage of APTIOM-treated patients (5.1%) than placebo-treated patients (0.7%) experienced decreases in sodium values of more than 10 mEq/L. These effects were dose-related and generally appeared within the first 8 weeks of treatment (as early as after 3 days). Serious, life-threatening complications were reported with APTIOM-associated hyponatremia (as low as 112 mEq/L) including seizures, severe nausea/vomiting leading to dehydration, severe gait instability, and injury. Some patients required hospitalization and discontinuation of APTIOM. Concurrent hypochloremia was also present in patients with hyponatremia. Hyponatremia was also observed in adult monotherapy trials and in pediatric trials. Depending on the severity of hyponatremia, the dose of APTIOM may need to be reduced or discontinued.
16.1 How Supplied
APTIOM tablets are white, oblong and with functional scoring on one side (200 mg, 600 mg, and 800 mg) or white, circular bi-convex and plain on one side (400 mg) and identified with strength-specific one-sided engraving on the other side, “ESL 200” (200 mg), “ESL 400” (400 mg), “ESL 600” (600 mg), or “ESL 800” (800 mg). Tablets are supplied in the following strengths and package configurations (Table 6):
| Tablet Strength | Package Configuration | NDC Code |
| 200 mg | Bottles of 30 | 63402-202-30 |
| 400 mg | Bottles of 30 | 63402-204-30 |
| 600 mg | Bottles of 60 | 63402-206-60 |
| 800 mg | Bottles of 30 | 63402-208-30 |
8.4 Pediatric Use
Safety and effectiveness of APTIOM have been established in the age groups 4 to 17 years. Use of APTIOM in these age groups is supported by evidence from adequate and well-controlled studies of APTIOM in adults with partial-onset seizures, pharmacokinetic data from adult and pediatric patients, and safety data from clinical studies in 393 pediatric patients 4 to 17 years of age [see Adverse Reactions (6.1) and Clinical Pharmacology (12.3)].
Safety and effectiveness in pediatric patients below the age of 4 years have not been established.
8.5 Geriatric Use
There were insufficient numbers of patients ≥65 years old enrolled in the controlled adjunctive epilepsy trials (N=15) to determine the efficacy of APTIOM in this patient population. The pharmacokinetics of APTIOM were evaluated in elderly healthy subjects (N=12) (Figure 1). Although the pharmacokinetics of eslicarbazepine are not affected by age independently, dose selection should take in consideration the greater frequency of renal impairment and other concomitant medical conditions and drug therapies in the elderly patient. Dose adjustment is necessary if CrCl is <50 mL/min [see Clinical Pharmacology (12.3)].
4 Contraindications
6 Adverse Reactions
The following adverse reactions are described in more detail in the Warnings and Precautions section of the label:
- Suicidal Behavior and Ideation [see Warnings and Precautions (5.1)]
- Serious Dermatologic Reactions [see Warnings and Precautions (5.2)]
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity [see Warnings and Precautions (5.3)]
- Anaphylactic Reactions and Angioedema [see Warnings and Precautions (5.4)]
- Hyponatremia [see Warnings and Precautions (5.5)]
- Neurological Adverse Reactions [see Warnings and Precautions (5.6)]
- Drug Induced Liver Injury [see Warnings and Precautions (5.8)]
- Abnormal Thyroid Function Tests [see Warnings and Precautions (5.9)]
- Pancytopenia, Agranulocytosis, and Leukopenia [see Warnings and Precautions (5.10)]
7 Drug Interactions
- Carbamazepine: May need dose adjustment for APTIOM or carbamazepine. (2.3, 5.6, 7.1)
- Phenytoin: Higher dosage of APTIOM may be necessary and dose adjustment may be needed for phenytoin. (2.3, 7.1, 7.2)
- Phenobarbital or Primidone: Higher dosage of APTIOM may be necessary. (2.3, 7.1)
- Hormonal Contraceptives: APTIOM may decrease the effectiveness of hormonal contraceptives. (7.4, 8.3)
12.2 Pharmacodynamics
The effect of APTIOM on cardiac repolarization was evaluated in a randomized, double-blind, placebo- and active-controlled 4-period crossover trial in healthy adult men and women. Subjects received APTIOM 1200 mg once daily × 5 days, APTIOM 2400 mg once daily × 5 days, an active-control, moxifloxacin 400 mg × 1 dose on Day 5, and placebo once daily × 5 days. At both doses of APTIOM, no significant effect on the QTc interval was detected.
12.3 Pharmacokinetics
The pharmacokinetics of eslicarbazepine is linear and dose-proportional in the dose range of 400 mg to 1600 mg once daily, both in healthy adult subjects and patients. The apparent half-life of eslicarbazepine in plasma was 13-20 hours in adult epilepsy patients. Steady-state plasma concentrations are attained after 4 to 5 days of once daily dosing.
7.3 Cyp3a4 Substrates
In vivo studies suggest that APTIOM can induce CYP3A4, decreasing plasma concentrations of drugs that are metabolized by this isoenzyme (e.g., simvastatin, lovastatin) [see Clinical Pharmacology (12.3)]. Dose adjustment of simvastatin and lovastatin may be needed if a clinically significant change in lipids is noted.
5.7 Withdrawal of Aeds
As with all antiepileptic drugs, APTIOM should be withdrawn gradually because of the risk of increased seizure frequency and status epilepticus, but if withdrawal is needed because of a serious adverse event, rapid discontinuation can be considered.
7.2 Cyp2c19 Substrates
APTIOM can inhibit CYP2C19, which can cause increased plasma concentrations of drugs that are metabolized by this isoenzyme (e.g., phenytoin, clobazam, and omeprazole) [see Clinical Pharmacology (12.3)]. Dose adjustment may be needed.
1 Indications and Usage
APTIOM is indicated for the treatment of partial-onset seizures in patients 4 years of age and older.
7.4 Oral Contraceptives
Because concomitant use of APTIOM and ethinylestradiol and levonorgestrel is associated with lower plasma levels of these hormones, females of reproductive potential should use additional or alternative non-hormonal birth control.
12.1 Mechanism of Action
APTIOM is extensively converted to eslicarbazepine, which is considered to be responsible for therapeutic effects in humans. The precise mechanism(s) by which eslicarbazepine exerts anticonvulsant activity is unknown but is thought to involve inhibition of voltage-gated sodium channels.
9.1 Controlled Substance
APTIOM is not a controlled substance.
16.2 Storage and Handling
Store APTIOM tablets at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
5 Warnings and Precautions
- Suicidal Behavior and Ideation: Monitor for suicidal thoughts or behavior. (5.1)
- Serious Dermatologic Reactions, Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), Anaphylactic Reactions and Angioedema: Monitor and discontinue if another cause cannot be established. (5.2, 5.3, 5.4)
- Hyponatremia: Monitor sodium levels in patients at risk or patients experiencing hyponatremia symptoms. (5.5)
- Neurological Adverse Reactions: Monitor for dizziness, disturbance in gait and coordination, somnolence, fatigue, cognitive dysfunction, and visual changes. Use caution when driving or operating machinery. (5.6)
- Withdrawal of APTIOM: Withdraw APTIOM gradually to minimize the risk of increased seizure frequency and status epilepticus. (2.6, 5.7, 8.1)
- Drug Induced Liver Injury: Discontinue APTIOM in patients with jaundice or evidence of significant liver injury. (5.8)
- Hematologic Adverse Reactions: Consider discontinuing. (5.10)
2 Dosage and Administration
- Adult Patients: The recommended initial dosage of APTIOM is 400 mg once daily. For some patients, treatment may be initiated at 800 mg once daily if the need for seizure reduction outweighs an increased risk of adverse reactions. Increase the dose in weekly increments of 400 mg to 600 mg once daily, based on clinical response and tolerability, to a recommended maintenance dosage of 800 mg to 1600 mg once daily. (2.2)
- Pediatric Patients: The recommended dosage of APTIOM is based on body weight and is administered orally once daily. Increase the dose in weekly intervals based on clinical response and tolerability, to the recommended maintenance dosage. (2.2)
- Patients with Moderate or Severe Renal Impairment: Reduce dosage by 50%. (2.4)
3 Dosage Forms and Strengths
APTIOM tablets are available in the following shapes and color (Table 2) with respective one-sided engraving:
| Tablet Strength | Tablet Color/Shape | Tablet Markings | Functional Score |
| 200 mg | White oblong | ESL 200 | Yes |
| 400 mg | White circular bi-convex | ESL 400 | No |
| 600 mg | White oblong | ESL 600 | Yes |
| 800 mg | White oblong | ESL 800 | Yes |
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of APTIOM. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure:
Hematologic and Lymphatic Systems: leukopenia, agranulocytosis, thrombocytopenia, megaloblastic anemia, and pancytopenia [see Warnings and Precautions (5.10)]
Metabolism and Nutrition Disorders: syndrome of inappropriate antidiuretic hormone secretion (SIADH) [see Warnings and Precautions (5.5)]
2.6 Discontinuation of Aptiom
When discontinuing APTIOM, reduce the dosage gradually and avoid abrupt discontinuation in order to minimize the risk of increased seizure frequency and status epilepticus [see Warnings and Precautions (5.7)].
5.8 Drug Induced Liver Injury
Hepatic effects, ranging from mild to moderate elevations in transaminases (>3 times the upper limit of normal) to rare cases with concomitant elevations of total bilirubin (>2 times the upper limit of normal) have been reported with APTIOM use. Baseline evaluations of liver laboratory tests are recommended. The combination of transaminase elevations and elevated bilirubin without evidence of obstruction is generally recognized as an important predictor of severe liver injury. APTIOM should be discontinued in patients with jaundice or other evidence of significant liver injury (e.g., laboratory evidence).
7.1 Other Antiepileptic Drugs
8 Use in Specific Populations
Pregnancy: Based on animal data, may cause fetal harm. (8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
See FDA-approved patient labeling (Medication Guide).
Inform patients and caregivers of the availability of a Medication Guide, and instruct them to read the Medication Guide prior to taking APTIOM. Instruct patients and caregivers that APTIOM should only be taken as prescribed.
5.1 Suicidal Behavior and Ideation
Antiepileptic drugs (AEDs), including APTIOM, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted Relative Risk 1.8, 95% confidence interval [CI]: 1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number of events is too small to allow any conclusion about drug effect on suicide.
The increased risk of suicidal thoughts or behavior with AEDs was observed as early as one week after starting treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5-100 years) in the clinical trials analyzed.
Table 3 shows absolute and relative risk by indication for all evaluated AEDs.
| Indication | Placebo Patients with Events Per 1000 Patients | Drug Patients with Events Per 1000 Patients | Relative Risk: Incidence of Events in Drug Patients/Incidence in Placebo Patients | Risk Differences: Additional Drug Patients with Events Per 1000 Patients |
| Epilepsy | 1.0 | 3.4 | 3.5 | 2.4 |
| Psychiatric | 5.7 | 8.5 | 1.5 | 2.9 |
| Other | 1.0 | 1.8 | 1.9 | 0.9 |
| Total | 2.4 | 4.3 | 1.8 | 1.9 |
The relative risk for suicidal thoughts or behavior was higher in clinical trials in patients with epilepsy than in clinical trials in patients with psychiatric or other conditions, but the absolute risk differences were similar for epilepsy and psychiatric indications.
Anyone considering prescribing APTIOM or any other AED must balance this risk with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
Patients, their caregivers, and families should be informed that AEDs increase the risk of suicidal thoughts and behavior and should be advised of the need to be alert for the emergence or worsening of the signs and symptoms of depression; any unusual changes in mood or behavior; or the emergence of suicidal thoughts, behavior, or thoughts about self-harm. Behaviors of concern should be reported immediately to healthcare providers.
5.10 Hematologic Adverse Reactions
Rare cases of pancytopenia, agranulocytosis, and leukopenia have been reported during postmarketing use in patients treated with APTIOM. Discontinuation of APTIOM should be considered in patients who develop pancytopenia, agranulocytosis, or leukopenia.
5.2 Serious Dermatologic Reactions
Serious dermatologic reactions including Stevens-Johnson Syndrome (SJS) and toxic epidermal necrolysis (TEN) have been reported in association with APTIOM use. Serious and sometimes fatal dermatologic reactions, including TEN and SJS, have also been reported in patients using oxcarbazepine or carbamazepine which are chemically related to APTIOM. The reporting rate of these reactions associated with oxcarbazepine use exceeds the background incidence rate estimates by a factor of 3- to 10-fold. The reporting rates for Aptiom have not been determined.
Risk factors for the development of serious and potentially fatal dermatologic reactions with APTIOM use have not been identified.
If a patient develops a dermatologic reaction while taking APTIOM, discontinue APTIOM use, unless the reaction is clearly not drug-related. Patients with a prior dermatologic reaction with oxcarbazepine, carbamazepine, or APTIOM should ordinarily not be treated with APTIOM [see Contraindications (4)].
8.6 Patients With Renal Impairment
5.9 Abnormal Thyroid Function Tests
Dose-dependent decreases in serum T3 and T4 (free and total) values have been observed in patients taking APTIOM. These changes were not associated with other abnormal thyroid function tests suggesting hypothyroidism. Abnormal thyroid function tests should be clinically evaluated.
2.5 Patients With Hepatic Impairment
8.7 Patients With Hepatic Impairment
10.2 Treatment Or Management of Overdose
There is no specific antidote for overdose with APTIOM. Symptomatic and supportive treatment should be administered as appropriate. Removal of the drug by gastric lavage and/or inactivation by administering activated charcoal should be considered.
Standard hemodialysis procedures result in partial clearance of APTIOM. Hemodialysis may be considered based on the patient's clinical state or in patients with significant renal impairment.
2.1 Important Administration Instructions
Instruct patients to administer APTIOM either as whole or as crushed tablets. Instruct patients to take APTIOM either with or without food. The APTIOM dosing regimen depends on age, weight, and renal function.
5.4 Anaphylactic Reactions and Angioedema
Rare cases of anaphylaxis and angioedema have been reported in patients taking APTIOM. Anaphylaxis and angioedema associated with laryngeal edema can be fatal. If a patient develops any of these reactions after treatment with APTIOM, the drug should be discontinued. Patients with a prior anaphylactic-type reaction with either oxcarbazepine or APTIOM should not be treated with APTIOM [see Contraindications (4)].
14.1 Monotherapy for Partial Onset Seizures
The effectiveness of APTIOM as monotherapy for partial-onset seizures was established in two identical, dose-blinded historical control trials in a total of 365 patients with epilepsy (Study 1 and Study 2). In these trials, patients were randomized in a 2:1 ratio to receive either APTIOM 1600 mg or 1200 mg once daily, and their responses were compared to those of a historical control group. The historical control methodology is described in a publication by French et al. [see References (15)]. The historical control consisted of a pooled analysis of the control groups from 8 trials of similar design, which utilized a subtherapeutic dose of an AED as a comparator. Statistical superiority to the historical control was considered to be demonstrated if the upper limit from a 2-sided 95% confidence interval for the percentage of patients meeting exit criteria in patients receiving APTIOM remained below the lower 95% prediction interval of 65% derived from the historical control data.
In Study 1 and Study 2, patients ≥16 years of age experienced at least 4 seizures during the baseline period with no 28-day seizure free period while receiving 1 or 2 AEDs (both could not be sodium-channel blocking drugs, and at least one AED was limited to 2/3 of a typical dose). APTIOM was titrated over a 1- to 2-week period followed by the gradual withdrawal of the background AED over a 6-week period, followed by a 10-week monotherapy period.
The exit criteria were one or more of the following: (1) an episode of status epilepticus, (2) emergence of a generalized tonic-clonic seizure in patients who had not had one in the past 6 months, (3) doubling of average monthly seizure count during any 28 consecutive days, (4) doubling of highest consecutive 2-day seizure frequency during the entire treatment phase, or (5) worsening of seizure severity considered by the investigator to require intervention. The primary endpoint was the cumulative 112-day exit rate in the efficacy population. Additionally, in Studies 1 and 2, if the discontinuation rate exceeded 10%, patients were randomly reassigned to be counted as exits.
The most commonly used baseline AEDs were carbamazepine, levetiracetam, valproic acid, and lamotrigine. Oxcarbazepine was used as a baseline AED in 6.6% of patients.
In Study 1, the Kaplan-Meier (K-M) estimate of the percentage of patients meeting at least 1 exit criterion was 29% (95% CI: 21%, 38%) in the 1600 mg group and 44% (95% CI 33%, 58%) in the 1200 mg group. In Study 2, the K-M estimate of the percentage of patients meeting at least 1 exit criterion was 13% (95% CI: 8%, 22%) in the 1600 mg group and 16% (95% CI: 8%, 29%) in the 1200 mg group. The upper limit of the 2-sided 95% CI of both doses in both trials were below the threshold of 65% derived from the historical control data, meeting the pre-specified criteria for efficacy (see Figure 4).
Figure 4: Kaplan-Meier Estimates of Cumulative 112-Day Exit Rates for Studies 1 and 2
14.2 Adjunctive Therapy for Partial Onset Seizures
The efficacy of APTIOM as adjunctive therapy in partial-onset seizures was established in three randomized, double-blind, placebo-controlled, multicenter trials in adult patients with epilepsy (Study 3, Study 4, and Study 5). Patients enrolled had partial-onset seizures with or without secondary generalization and were not adequately controlled with 1 to 3 concomitant AEDs. During an 8-week baseline period, patients were required to have an average of ≥4 partial-onset seizures per 28 days with no seizure-free period exceeding 21 days. In these three trials, patients had a median duration of epilepsy of 19 years and a median baseline seizure frequency of 8 seizures per 28 days. Two-thirds (69%) of subjects used 2 concomitant AEDs and 28% used 1 concomitant AED. The most commonly used AEDs were carbamazepine (50%), lamotrigine (24%), valproic acid (21%), and levetiracetam (18%). Oxcarbazepine was not allowed as a concomitant AED.
Studies 3 and 4 compared dosages of APTIOM 400, 800, and 1200 mg once daily with placebo. Study 5 compared dosages of APTIOM 800 and 1200 mg once daily with placebo. In all three trials, following an 8-week Baseline Phase, which established a baseline seizure frequency, subjects were randomized to a treatment arm. Patients entered a treatment period consisting of an initial titration phase (2 weeks), and a subsequent maintenance phase (12 weeks). The specific titration schedule differed amongst the three studies. Thus, patients were started on a daily dose of 400 mg or 800 mg and subsequently increased by 400 mg/day following one or two weeks, until the final daily target dose was achieved.
The standardized seizure frequency during the Maintenance Phase over 28 days was the primary efficacy endpoint in all three trials. Table 5 presents the results for the primary endpoint, as well as the secondary endpoint of percent reduction from baseline in seizure frequency. The APTIOM treatment at 400 mg/day was studied in Studies 3 and 4 and did not show significant treatment effect. A statistically significant effect was observed with APTIOM treatment at doses of 800 mg/day in Studies 3 and 4, but not in Study 5, and at doses of 1200 mg/day in all 3 studies.
| Placebo | APTIOM | ||
|---|---|---|---|
| 800 mg | 1200 mg | ||
|
*statistically significant compared to placebo |
|||
| Study 3 | |||
| N | 95 | 88 | 87 |
| Seizure Frequency (LS Mean seizures per 28 days) (p-value) |
6.6 | 5.0 (0.047*) |
4.3 (0.001*) |
| Median Percent Reduction from Baseline in Seizure Frequency (%) | -15 | -36 | -39 |
| Study 4 | |||
| N | 99 | 87 | 81 |
| Seizure Frequency (LS Mean seizures per 28 days) (p-value) |
8.6 | 6.2 (0.006*) |
6.6 (0.042*) |
| Median Percent Reduction from Baseline in Seizure Frequency (%) | -6 | -33 | -28 |
| Study 5 | |||
| N | 212 | 200 | 184 |
| Seizure Frequency (LS Mean seizures per 28 days) (p-value) |
7.9 | 6.5 (0.058) |
6.0 (0.004*) |
| Median Percent Reduction from Baseline in Seizure Frequency (%) | -22 | -30 | -36 |
Figure 5 shows changes from baseline in the 28-day total partial seizure frequency by category of reduction in seizure frequency from baseline for patients treated with APTIOM and placebo in an integrated analysis across the three clinical trials. Patients in whom the seizure frequency increased are shown to the left as “Worse.” Patients in whom the seizure frequency decreased are shown in four categories.
Figure 5: Proportion of Patients by Category of Seizure Reduction for APTIOM and Placebo Across All Three Double-blind Trials
2.3 Dosage Modifications With Other Antiepileptic Drugs
Some adverse reactions occur more frequently when patients take APTIOM adjunctively with carbamazepine [see Warnings and Precautions (5.6)]. However, carbamazepine reduces the plasma concentration of eslicarbazepine [see Drug Interactions (7.1)]. When APTIOM and carbamazepine are taken concomitantly, the dose of APTIOM or carbamazepine may need to be adjusted based on efficacy and tolerability. For patients taking other enzyme-inducing AEDs (i.e., phenobarbital, phenytoin, and primidone), higher doses of APTIOM may be needed [see Drug Interactions (7.1)].
APTIOM should not be taken as an adjunctive therapy with oxcarbazepine.
2.4 Dosage Modifications in Patients With Renal Impairment
In patients with moderate and severe renal impairment (i.e., creatinine clearance < 50 mL/min), the initial, titration, and maintenance dosages should generally be reduced by 50%. Titration and maintenance dosages may be adjusted according to clinical response [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
10.1 Signs, Symptoms, and Laboratory Findings of Acute Overdose in Humans
Symptoms of overdose are consistent with the known adverse reactions of APTIOM and include hyponatremia (sometimes severe), dizziness, nausea, vomiting, somnolence, euphoria, oral paraesthesia, ataxia, walking difficulties, and diplopia. The maximum dosage studied in open-label adult monotherapy treatment following withdrawal of concomitant AEDs was 2400 mg once daily.
5.3 Drug Reaction With Eosinophilia and Systemic Symptoms (dress)/multiorgan Hypersensitivity
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), also known as Multiorgan Hypersensitivity, has been reported in patients taking APTIOM. DRESS may be fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, and/or lymphadenopathy, in association with other organ system involvement, such as hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis sometimes resembling an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its expression, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, the patient should be evaluated immediately. APTIOM should be discontinued and not be resumed if an alternative etiology for the signs or symptoms cannot be established. Patients with a prior DRESS reaction with either oxcarbazepine or APTIOM should not be treated with APTIOM [see Contraindications (4)].
Structured Label Content
Section 42229-5 (42229-5)
Monotherapy and Adjunctive Therapy
Section 42231-1 (42231-1)
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Revised 9/2017 |
|||
|
MEDICATION GUIDE
APTIOM (ap tee' om) (eslicarbazepine acetate) tablets |
||||
|
What is the most important information I should know about APTIOM?
|
||||
|
1. Like other antiepileptic drugs, APTIOM may cause suicidal thoughts or actions in a very small number of people, about 1 in 500.
Call a healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you: |
||||
|
|
|||
How can I watch for early symptoms of suicidal thoughts and actions?
|
||||
|
2. APTIOM may cause allergic reactions or serious problems which may affect organs and other parts of your body like the liver or blood cells. You may or may not have a rash with these types of reactions.
Call your healthcare provider right away if you have any of the following: |
||||
|
|
|||
|
||||
|
|
|||
| Some medicines can also cause low sodium in your blood. Be sure to tell your healthcare provider about all the other medicines that you are taking. | ||||
|
What is APTIOM?
APTIOM is a prescription medicine used to treat partial-onset seizures. It is not known if APTIOM is safe and effective in children under 4 years of age. |
||||
|
Who should not take APTIOM?
Do not take APTIOM if you are allergic to eslicarbazepine acetate, any of the other ingredients in APTIOM, or oxcarbazepine. See the end of this Medication Guide for a complete list of ingredients in APTIOM. |
||||
|
What should I tell my healthcare provider before taking APTIOM?
Before taking APTIOM, tell your healthcare provider about all your medical conditions, including if you:
|
||||
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking APTIOM with certain other medicines may cause side effects or affect how well they work. Do not start or stop other medicines without talking to your healthcare provider. Especially tell your healthcare provider if you take: |
||||
|
|
|
|
|
| Ask your healthcare provider or pharmacist for a list of these medicines, if you are not sure. Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine. |
||||
How should I take APTIOM?
|
||||
What should I avoid while taking APTIOM?
|
||||
|
What are the possible side effects of APTIOM?
See “What is the most important information I should know about APTIOM?” APTIOM may cause other serious side effects including:
|
||||
|
|
|
||
|
||||
|
|
|
||
|
Get medical help right away if you have any of the symptoms listed above or listed in “What is the most important information I should know about APTIOM?”
The most common side effects of APTIOM include: |
||||
|
|
|
|
|
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of APTIOM. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store APTIOM?
|
||||
| Keep APTIOM and all medicines out of reach of children. | ||||
|
What are the ingredients in APTIOM?
Active ingredient: eslicarbazepine acetate Inactive ingredients: croscarmellose sodium, magnesium stearate, and povidone |
||||
|
General information about the safe and effective use of APTIOM.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use APTIOM for a condition for which it was not prescribed. Do not give APTIOM to other people, even if they have the same symptoms that you have. It may harm them. This Medication Guide summarizes the most important information about APTIOM. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about APTIOM that is written for health professionals. For more information, go to www.aptiom.com or call 1-888-394-7377. |
||||
|
Manufactured for: Sunovion Pharmaceuticals Inc., Marlborough, MA 01752 USA Under license from © 2023 Sunovion Pharmaceuticals Inc. All rights reserved. |
Section 51945-4 (51945-4)
PRINCIPAL DISPLAY PANEL – 200 MG 30 COUNT OBLONG BOTTLE
NDC 63402-202-30
30 Tablets
ONCE DAILY
Aptiom®
(eslicarbazepine acetate) Tablets
200 mg
Rx Only
ATTENTION DISPENSER: Each time Aptiom is
dispensed give the patient the accompanying
Medication Guide.
Sumitomo Pharma
Under license from Bial
9.2 Abuse
Prescription drug abuse is the intentional non-therapeutic use of a drug, even once, for its rewarding psychological or physiological effects. Drug addiction, which develops after repeated drug abuse, is characterized by a strong desire to take a drug despite harmful consequences, difficulty in controlling its use, giving a higher priority to drug use than to obligations, increased tolerance, and sometimes physical withdrawal. Drug abuse and drug addiction are separate and distinct from physical dependence (for example, abuse may not be accompanied by physical dependence) [see Drug Abuse and Dependence (9.3)].
In a human abuse study in recreational sedative abusers APTIOM showed no evidence of abuse. In Phase 1, 1.5% of the healthy volunteers taking APTIOM reported euphoria compared to 0.4% taking placebo.
15 References (15 REFERENCES)
French JA, Wang S, Warnock B, Temkin N. Historical control monotherapy design in the treatment of epilepsy. Epilepsia 2010;51(10):1936-43.
8.2 Lactation
Eslicarbazepine is present in human milk. The effects of APTIOM on the breastfed infant or on milk production are unknown. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for APTIOM and any potential adverse effects on the breastfed infant from APTIOM or from the underlying maternal condition.
11 Description (11 DESCRIPTION)
The chemical name of APTIOM (eslicarbazepine acetate) is (S)-10-Acetoxy-10,11-dihydro-5H-dibenz[b,f]azepine-5-carboxamide. APTIOM is a dibenz[b,f]azepine-5-carboxamide derivative. Its molecular formula is C17H16N2O3 and its molecular weight is 296.32. The chemical structure is:
APTIOM is a white to off-white, odorless crystalline solid. It is insoluble in hexane, very slightly soluble in aqueous solvents and soluble in organic solvents such as acetone, acetonitrile, and methanol.
Each APTIOM tablet contains 200 mg, 400 mg, 600 mg or 800 mg of eslicarbazepine acetate and the following inactive ingredients: croscarmellose sodium, magnesium stearate, and povidone.
9.3 Dependence
Physical dependence is characterized by withdrawal symptoms after abrupt discontinuation or a significant dose reduction of a drug.
There was some evidence of physical dependence or a withdrawal syndrome with APTIOM in a physical dependence study conducted in healthy volunteers who were maintained at a daily dose of 800 mg APTIOM for 4 weeks prior to discontinuation. The primary endpoint was the maximum change from steady-state baseline in the total score of the Physician's Withdrawal Checklist (PWC-34) during the 21-day discontinuation period. APTIOM and placebo were shown to be equivalent on the primary endpoint. Two out of 8 secondary endpoints (visual analog scales for anxiety and nausea) showed some increase in these symptoms for subjects who were maintained on APTIOM and discontinued, versus subjects who were maintained on placebo. In general, AEDs should not be abruptly discontinued in patients with epilepsy because of the risk of increased seizure frequency and status epilepticus.
5.5 Hyponatremia
Clinically significant hyponatremia (sodium <125 mEq/L) can develop in patients taking APTIOM.
Measurement of serum sodium and chloride levels should be considered during maintenance treatment with APTIOM, particularly if the patient is receiving other medications known to decrease serum sodium levels, and should be performed if symptoms of hyponatremia develop (e.g., nausea/vomiting, malaise, headache, lethargy, confusion, irritability, muscle weakness/spasms, obtundation, or increase in seizure frequency or severity). Cases of symptomatic hyponatremia and syndrome of inappropriate antidiuretic hormone secretion (SIADH) have been reported during postmarketing use. In clinical trials, patients whose treatment with APTIOM was discontinued because of hyponatremia generally experienced normalization of serum sodium within a few days without additional treatment.
In the controlled adult adjunctive epilepsy trials, 4/415 patients (1.0%) treated with 800 mg and 6/410 (1.5%) patients treated with 1200 mg of APTIOM had at least one serum sodium value less than 125 mEq/L, compared to none of the patients assigned to placebo. A higher percentage of APTIOM-treated patients (5.1%) than placebo-treated patients (0.7%) experienced decreases in sodium values of more than 10 mEq/L. These effects were dose-related and generally appeared within the first 8 weeks of treatment (as early as after 3 days). Serious, life-threatening complications were reported with APTIOM-associated hyponatremia (as low as 112 mEq/L) including seizures, severe nausea/vomiting leading to dehydration, severe gait instability, and injury. Some patients required hospitalization and discontinuation of APTIOM. Concurrent hypochloremia was also present in patients with hyponatremia. Hyponatremia was also observed in adult monotherapy trials and in pediatric trials. Depending on the severity of hyponatremia, the dose of APTIOM may need to be reduced or discontinued.
16.1 How Supplied
APTIOM tablets are white, oblong and with functional scoring on one side (200 mg, 600 mg, and 800 mg) or white, circular bi-convex and plain on one side (400 mg) and identified with strength-specific one-sided engraving on the other side, “ESL 200” (200 mg), “ESL 400” (400 mg), “ESL 600” (600 mg), or “ESL 800” (800 mg). Tablets are supplied in the following strengths and package configurations (Table 6):
| Tablet Strength | Package Configuration | NDC Code |
| 200 mg | Bottles of 30 | 63402-202-30 |
| 400 mg | Bottles of 30 | 63402-204-30 |
| 600 mg | Bottles of 60 | 63402-206-60 |
| 800 mg | Bottles of 30 | 63402-208-30 |
8.4 Pediatric Use
Safety and effectiveness of APTIOM have been established in the age groups 4 to 17 years. Use of APTIOM in these age groups is supported by evidence from adequate and well-controlled studies of APTIOM in adults with partial-onset seizures, pharmacokinetic data from adult and pediatric patients, and safety data from clinical studies in 393 pediatric patients 4 to 17 years of age [see Adverse Reactions (6.1) and Clinical Pharmacology (12.3)].
Safety and effectiveness in pediatric patients below the age of 4 years have not been established.
8.5 Geriatric Use
There were insufficient numbers of patients ≥65 years old enrolled in the controlled adjunctive epilepsy trials (N=15) to determine the efficacy of APTIOM in this patient population. The pharmacokinetics of APTIOM were evaluated in elderly healthy subjects (N=12) (Figure 1). Although the pharmacokinetics of eslicarbazepine are not affected by age independently, dose selection should take in consideration the greater frequency of renal impairment and other concomitant medical conditions and drug therapies in the elderly patient. Dose adjustment is necessary if CrCl is <50 mL/min [see Clinical Pharmacology (12.3)].
4 Contraindications (4 CONTRAINDICATIONS)
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described in more detail in the Warnings and Precautions section of the label:
- Suicidal Behavior and Ideation [see Warnings and Precautions (5.1)]
- Serious Dermatologic Reactions [see Warnings and Precautions (5.2)]
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity [see Warnings and Precautions (5.3)]
- Anaphylactic Reactions and Angioedema [see Warnings and Precautions (5.4)]
- Hyponatremia [see Warnings and Precautions (5.5)]
- Neurological Adverse Reactions [see Warnings and Precautions (5.6)]
- Drug Induced Liver Injury [see Warnings and Precautions (5.8)]
- Abnormal Thyroid Function Tests [see Warnings and Precautions (5.9)]
- Pancytopenia, Agranulocytosis, and Leukopenia [see Warnings and Precautions (5.10)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Carbamazepine: May need dose adjustment for APTIOM or carbamazepine. (2.3, 5.6, 7.1)
- Phenytoin: Higher dosage of APTIOM may be necessary and dose adjustment may be needed for phenytoin. (2.3, 7.1, 7.2)
- Phenobarbital or Primidone: Higher dosage of APTIOM may be necessary. (2.3, 7.1)
- Hormonal Contraceptives: APTIOM may decrease the effectiveness of hormonal contraceptives. (7.4, 8.3)
12.2 Pharmacodynamics
The effect of APTIOM on cardiac repolarization was evaluated in a randomized, double-blind, placebo- and active-controlled 4-period crossover trial in healthy adult men and women. Subjects received APTIOM 1200 mg once daily × 5 days, APTIOM 2400 mg once daily × 5 days, an active-control, moxifloxacin 400 mg × 1 dose on Day 5, and placebo once daily × 5 days. At both doses of APTIOM, no significant effect on the QTc interval was detected.
12.3 Pharmacokinetics
The pharmacokinetics of eslicarbazepine is linear and dose-proportional in the dose range of 400 mg to 1600 mg once daily, both in healthy adult subjects and patients. The apparent half-life of eslicarbazepine in plasma was 13-20 hours in adult epilepsy patients. Steady-state plasma concentrations are attained after 4 to 5 days of once daily dosing.
7.3 Cyp3a4 Substrates (7.3 CYP3A4 Substrates)
In vivo studies suggest that APTIOM can induce CYP3A4, decreasing plasma concentrations of drugs that are metabolized by this isoenzyme (e.g., simvastatin, lovastatin) [see Clinical Pharmacology (12.3)]. Dose adjustment of simvastatin and lovastatin may be needed if a clinically significant change in lipids is noted.
5.7 Withdrawal of Aeds (5.7 Withdrawal of AEDs)
As with all antiepileptic drugs, APTIOM should be withdrawn gradually because of the risk of increased seizure frequency and status epilepticus, but if withdrawal is needed because of a serious adverse event, rapid discontinuation can be considered.
7.2 Cyp2c19 Substrates (7.2 CYP2C19 Substrates)
APTIOM can inhibit CYP2C19, which can cause increased plasma concentrations of drugs that are metabolized by this isoenzyme (e.g., phenytoin, clobazam, and omeprazole) [see Clinical Pharmacology (12.3)]. Dose adjustment may be needed.
1 Indications and Usage (1 INDICATIONS AND USAGE)
APTIOM is indicated for the treatment of partial-onset seizures in patients 4 years of age and older.
7.4 Oral Contraceptives
Because concomitant use of APTIOM and ethinylestradiol and levonorgestrel is associated with lower plasma levels of these hormones, females of reproductive potential should use additional or alternative non-hormonal birth control.
12.1 Mechanism of Action
APTIOM is extensively converted to eslicarbazepine, which is considered to be responsible for therapeutic effects in humans. The precise mechanism(s) by which eslicarbazepine exerts anticonvulsant activity is unknown but is thought to involve inhibition of voltage-gated sodium channels.
9.1 Controlled Substance
APTIOM is not a controlled substance.
16.2 Storage and Handling
Store APTIOM tablets at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Suicidal Behavior and Ideation: Monitor for suicidal thoughts or behavior. (5.1)
- Serious Dermatologic Reactions, Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), Anaphylactic Reactions and Angioedema: Monitor and discontinue if another cause cannot be established. (5.2, 5.3, 5.4)
- Hyponatremia: Monitor sodium levels in patients at risk or patients experiencing hyponatremia symptoms. (5.5)
- Neurological Adverse Reactions: Monitor for dizziness, disturbance in gait and coordination, somnolence, fatigue, cognitive dysfunction, and visual changes. Use caution when driving or operating machinery. (5.6)
- Withdrawal of APTIOM: Withdraw APTIOM gradually to minimize the risk of increased seizure frequency and status epilepticus. (2.6, 5.7, 8.1)
- Drug Induced Liver Injury: Discontinue APTIOM in patients with jaundice or evidence of significant liver injury. (5.8)
- Hematologic Adverse Reactions: Consider discontinuing. (5.10)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Adult Patients: The recommended initial dosage of APTIOM is 400 mg once daily. For some patients, treatment may be initiated at 800 mg once daily if the need for seizure reduction outweighs an increased risk of adverse reactions. Increase the dose in weekly increments of 400 mg to 600 mg once daily, based on clinical response and tolerability, to a recommended maintenance dosage of 800 mg to 1600 mg once daily. (2.2)
- Pediatric Patients: The recommended dosage of APTIOM is based on body weight and is administered orally once daily. Increase the dose in weekly intervals based on clinical response and tolerability, to the recommended maintenance dosage. (2.2)
- Patients with Moderate or Severe Renal Impairment: Reduce dosage by 50%. (2.4)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
APTIOM tablets are available in the following shapes and color (Table 2) with respective one-sided engraving:
| Tablet Strength | Tablet Color/Shape | Tablet Markings | Functional Score |
| 200 mg | White oblong | ESL 200 | Yes |
| 400 mg | White circular bi-convex | ESL 400 | No |
| 600 mg | White oblong | ESL 600 | Yes |
| 800 mg | White oblong | ESL 800 | Yes |
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of APTIOM. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure:
Hematologic and Lymphatic Systems: leukopenia, agranulocytosis, thrombocytopenia, megaloblastic anemia, and pancytopenia [see Warnings and Precautions (5.10)]
Metabolism and Nutrition Disorders: syndrome of inappropriate antidiuretic hormone secretion (SIADH) [see Warnings and Precautions (5.5)]
2.6 Discontinuation of Aptiom (2.6 Discontinuation of APTIOM)
When discontinuing APTIOM, reduce the dosage gradually and avoid abrupt discontinuation in order to minimize the risk of increased seizure frequency and status epilepticus [see Warnings and Precautions (5.7)].
5.8 Drug Induced Liver Injury
Hepatic effects, ranging from mild to moderate elevations in transaminases (>3 times the upper limit of normal) to rare cases with concomitant elevations of total bilirubin (>2 times the upper limit of normal) have been reported with APTIOM use. Baseline evaluations of liver laboratory tests are recommended. The combination of transaminase elevations and elevated bilirubin without evidence of obstruction is generally recognized as an important predictor of severe liver injury. APTIOM should be discontinued in patients with jaundice or other evidence of significant liver injury (e.g., laboratory evidence).
7.1 Other Antiepileptic Drugs
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy: Based on animal data, may cause fetal harm. (8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
See FDA-approved patient labeling (Medication Guide).
Inform patients and caregivers of the availability of a Medication Guide, and instruct them to read the Medication Guide prior to taking APTIOM. Instruct patients and caregivers that APTIOM should only be taken as prescribed.
5.1 Suicidal Behavior and Ideation
Antiepileptic drugs (AEDs), including APTIOM, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted Relative Risk 1.8, 95% confidence interval [CI]: 1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number of events is too small to allow any conclusion about drug effect on suicide.
The increased risk of suicidal thoughts or behavior with AEDs was observed as early as one week after starting treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5-100 years) in the clinical trials analyzed.
Table 3 shows absolute and relative risk by indication for all evaluated AEDs.
| Indication | Placebo Patients with Events Per 1000 Patients | Drug Patients with Events Per 1000 Patients | Relative Risk: Incidence of Events in Drug Patients/Incidence in Placebo Patients | Risk Differences: Additional Drug Patients with Events Per 1000 Patients |
| Epilepsy | 1.0 | 3.4 | 3.5 | 2.4 |
| Psychiatric | 5.7 | 8.5 | 1.5 | 2.9 |
| Other | 1.0 | 1.8 | 1.9 | 0.9 |
| Total | 2.4 | 4.3 | 1.8 | 1.9 |
The relative risk for suicidal thoughts or behavior was higher in clinical trials in patients with epilepsy than in clinical trials in patients with psychiatric or other conditions, but the absolute risk differences were similar for epilepsy and psychiatric indications.
Anyone considering prescribing APTIOM or any other AED must balance this risk with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
Patients, their caregivers, and families should be informed that AEDs increase the risk of suicidal thoughts and behavior and should be advised of the need to be alert for the emergence or worsening of the signs and symptoms of depression; any unusual changes in mood or behavior; or the emergence of suicidal thoughts, behavior, or thoughts about self-harm. Behaviors of concern should be reported immediately to healthcare providers.
5.10 Hematologic Adverse Reactions
Rare cases of pancytopenia, agranulocytosis, and leukopenia have been reported during postmarketing use in patients treated with APTIOM. Discontinuation of APTIOM should be considered in patients who develop pancytopenia, agranulocytosis, or leukopenia.
5.2 Serious Dermatologic Reactions
Serious dermatologic reactions including Stevens-Johnson Syndrome (SJS) and toxic epidermal necrolysis (TEN) have been reported in association with APTIOM use. Serious and sometimes fatal dermatologic reactions, including TEN and SJS, have also been reported in patients using oxcarbazepine or carbamazepine which are chemically related to APTIOM. The reporting rate of these reactions associated with oxcarbazepine use exceeds the background incidence rate estimates by a factor of 3- to 10-fold. The reporting rates for Aptiom have not been determined.
Risk factors for the development of serious and potentially fatal dermatologic reactions with APTIOM use have not been identified.
If a patient develops a dermatologic reaction while taking APTIOM, discontinue APTIOM use, unless the reaction is clearly not drug-related. Patients with a prior dermatologic reaction with oxcarbazepine, carbamazepine, or APTIOM should ordinarily not be treated with APTIOM [see Contraindications (4)].
8.6 Patients With Renal Impairment (8.6 Patients with Renal Impairment)
5.9 Abnormal Thyroid Function Tests
Dose-dependent decreases in serum T3 and T4 (free and total) values have been observed in patients taking APTIOM. These changes were not associated with other abnormal thyroid function tests suggesting hypothyroidism. Abnormal thyroid function tests should be clinically evaluated.
2.5 Patients With Hepatic Impairment (2.5 Patients with Hepatic Impairment)
8.7 Patients With Hepatic Impairment (8.7 Patients with Hepatic Impairment)
10.2 Treatment Or Management of Overdose (10.2 Treatment or Management of Overdose)
There is no specific antidote for overdose with APTIOM. Symptomatic and supportive treatment should be administered as appropriate. Removal of the drug by gastric lavage and/or inactivation by administering activated charcoal should be considered.
Standard hemodialysis procedures result in partial clearance of APTIOM. Hemodialysis may be considered based on the patient's clinical state or in patients with significant renal impairment.
2.1 Important Administration Instructions
Instruct patients to administer APTIOM either as whole or as crushed tablets. Instruct patients to take APTIOM either with or without food. The APTIOM dosing regimen depends on age, weight, and renal function.
5.4 Anaphylactic Reactions and Angioedema
Rare cases of anaphylaxis and angioedema have been reported in patients taking APTIOM. Anaphylaxis and angioedema associated with laryngeal edema can be fatal. If a patient develops any of these reactions after treatment with APTIOM, the drug should be discontinued. Patients with a prior anaphylactic-type reaction with either oxcarbazepine or APTIOM should not be treated with APTIOM [see Contraindications (4)].
14.1 Monotherapy for Partial Onset Seizures (14.1 Monotherapy for Partial-Onset Seizures)
The effectiveness of APTIOM as monotherapy for partial-onset seizures was established in two identical, dose-blinded historical control trials in a total of 365 patients with epilepsy (Study 1 and Study 2). In these trials, patients were randomized in a 2:1 ratio to receive either APTIOM 1600 mg or 1200 mg once daily, and their responses were compared to those of a historical control group. The historical control methodology is described in a publication by French et al. [see References (15)]. The historical control consisted of a pooled analysis of the control groups from 8 trials of similar design, which utilized a subtherapeutic dose of an AED as a comparator. Statistical superiority to the historical control was considered to be demonstrated if the upper limit from a 2-sided 95% confidence interval for the percentage of patients meeting exit criteria in patients receiving APTIOM remained below the lower 95% prediction interval of 65% derived from the historical control data.
In Study 1 and Study 2, patients ≥16 years of age experienced at least 4 seizures during the baseline period with no 28-day seizure free period while receiving 1 or 2 AEDs (both could not be sodium-channel blocking drugs, and at least one AED was limited to 2/3 of a typical dose). APTIOM was titrated over a 1- to 2-week period followed by the gradual withdrawal of the background AED over a 6-week period, followed by a 10-week monotherapy period.
The exit criteria were one or more of the following: (1) an episode of status epilepticus, (2) emergence of a generalized tonic-clonic seizure in patients who had not had one in the past 6 months, (3) doubling of average monthly seizure count during any 28 consecutive days, (4) doubling of highest consecutive 2-day seizure frequency during the entire treatment phase, or (5) worsening of seizure severity considered by the investigator to require intervention. The primary endpoint was the cumulative 112-day exit rate in the efficacy population. Additionally, in Studies 1 and 2, if the discontinuation rate exceeded 10%, patients were randomly reassigned to be counted as exits.
The most commonly used baseline AEDs were carbamazepine, levetiracetam, valproic acid, and lamotrigine. Oxcarbazepine was used as a baseline AED in 6.6% of patients.
In Study 1, the Kaplan-Meier (K-M) estimate of the percentage of patients meeting at least 1 exit criterion was 29% (95% CI: 21%, 38%) in the 1600 mg group and 44% (95% CI 33%, 58%) in the 1200 mg group. In Study 2, the K-M estimate of the percentage of patients meeting at least 1 exit criterion was 13% (95% CI: 8%, 22%) in the 1600 mg group and 16% (95% CI: 8%, 29%) in the 1200 mg group. The upper limit of the 2-sided 95% CI of both doses in both trials were below the threshold of 65% derived from the historical control data, meeting the pre-specified criteria for efficacy (see Figure 4).
Figure 4: Kaplan-Meier Estimates of Cumulative 112-Day Exit Rates for Studies 1 and 2
14.2 Adjunctive Therapy for Partial Onset Seizures (14.2 Adjunctive Therapy for Partial-Onset Seizures)
The efficacy of APTIOM as adjunctive therapy in partial-onset seizures was established in three randomized, double-blind, placebo-controlled, multicenter trials in adult patients with epilepsy (Study 3, Study 4, and Study 5). Patients enrolled had partial-onset seizures with or without secondary generalization and were not adequately controlled with 1 to 3 concomitant AEDs. During an 8-week baseline period, patients were required to have an average of ≥4 partial-onset seizures per 28 days with no seizure-free period exceeding 21 days. In these three trials, patients had a median duration of epilepsy of 19 years and a median baseline seizure frequency of 8 seizures per 28 days. Two-thirds (69%) of subjects used 2 concomitant AEDs and 28% used 1 concomitant AED. The most commonly used AEDs were carbamazepine (50%), lamotrigine (24%), valproic acid (21%), and levetiracetam (18%). Oxcarbazepine was not allowed as a concomitant AED.
Studies 3 and 4 compared dosages of APTIOM 400, 800, and 1200 mg once daily with placebo. Study 5 compared dosages of APTIOM 800 and 1200 mg once daily with placebo. In all three trials, following an 8-week Baseline Phase, which established a baseline seizure frequency, subjects were randomized to a treatment arm. Patients entered a treatment period consisting of an initial titration phase (2 weeks), and a subsequent maintenance phase (12 weeks). The specific titration schedule differed amongst the three studies. Thus, patients were started on a daily dose of 400 mg or 800 mg and subsequently increased by 400 mg/day following one or two weeks, until the final daily target dose was achieved.
The standardized seizure frequency during the Maintenance Phase over 28 days was the primary efficacy endpoint in all three trials. Table 5 presents the results for the primary endpoint, as well as the secondary endpoint of percent reduction from baseline in seizure frequency. The APTIOM treatment at 400 mg/day was studied in Studies 3 and 4 and did not show significant treatment effect. A statistically significant effect was observed with APTIOM treatment at doses of 800 mg/day in Studies 3 and 4, but not in Study 5, and at doses of 1200 mg/day in all 3 studies.
| Placebo | APTIOM | ||
|---|---|---|---|
| 800 mg | 1200 mg | ||
|
*statistically significant compared to placebo |
|||
| Study 3 | |||
| N | 95 | 88 | 87 |
| Seizure Frequency (LS Mean seizures per 28 days) (p-value) |
6.6 | 5.0 (0.047*) |
4.3 (0.001*) |
| Median Percent Reduction from Baseline in Seizure Frequency (%) | -15 | -36 | -39 |
| Study 4 | |||
| N | 99 | 87 | 81 |
| Seizure Frequency (LS Mean seizures per 28 days) (p-value) |
8.6 | 6.2 (0.006*) |
6.6 (0.042*) |
| Median Percent Reduction from Baseline in Seizure Frequency (%) | -6 | -33 | -28 |
| Study 5 | |||
| N | 212 | 200 | 184 |
| Seizure Frequency (LS Mean seizures per 28 days) (p-value) |
7.9 | 6.5 (0.058) |
6.0 (0.004*) |
| Median Percent Reduction from Baseline in Seizure Frequency (%) | -22 | -30 | -36 |
Figure 5 shows changes from baseline in the 28-day total partial seizure frequency by category of reduction in seizure frequency from baseline for patients treated with APTIOM and placebo in an integrated analysis across the three clinical trials. Patients in whom the seizure frequency increased are shown to the left as “Worse.” Patients in whom the seizure frequency decreased are shown in four categories.
Figure 5: Proportion of Patients by Category of Seizure Reduction for APTIOM and Placebo Across All Three Double-blind Trials
2.3 Dosage Modifications With Other Antiepileptic Drugs (2.3 Dosage Modifications with Other Antiepileptic Drugs)
Some adverse reactions occur more frequently when patients take APTIOM adjunctively with carbamazepine [see Warnings and Precautions (5.6)]. However, carbamazepine reduces the plasma concentration of eslicarbazepine [see Drug Interactions (7.1)]. When APTIOM and carbamazepine are taken concomitantly, the dose of APTIOM or carbamazepine may need to be adjusted based on efficacy and tolerability. For patients taking other enzyme-inducing AEDs (i.e., phenobarbital, phenytoin, and primidone), higher doses of APTIOM may be needed [see Drug Interactions (7.1)].
APTIOM should not be taken as an adjunctive therapy with oxcarbazepine.
2.4 Dosage Modifications in Patients With Renal Impairment (2.4 Dosage Modifications in Patients with Renal Impairment)
In patients with moderate and severe renal impairment (i.e., creatinine clearance < 50 mL/min), the initial, titration, and maintenance dosages should generally be reduced by 50%. Titration and maintenance dosages may be adjusted according to clinical response [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
10.1 Signs, Symptoms, and Laboratory Findings of Acute Overdose in Humans
Symptoms of overdose are consistent with the known adverse reactions of APTIOM and include hyponatremia (sometimes severe), dizziness, nausea, vomiting, somnolence, euphoria, oral paraesthesia, ataxia, walking difficulties, and diplopia. The maximum dosage studied in open-label adult monotherapy treatment following withdrawal of concomitant AEDs was 2400 mg once daily.
5.3 Drug Reaction With Eosinophilia and Systemic Symptoms (dress)/multiorgan Hypersensitivity (5.3 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity)
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), also known as Multiorgan Hypersensitivity, has been reported in patients taking APTIOM. DRESS may be fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, and/or lymphadenopathy, in association with other organ system involvement, such as hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis sometimes resembling an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its expression, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, the patient should be evaluated immediately. APTIOM should be discontinued and not be resumed if an alternative etiology for the signs or symptoms cannot be established. Patients with a prior DRESS reaction with either oxcarbazepine or APTIOM should not be treated with APTIOM [see Contraindications (4)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:29.203311 · Updated: 2026-03-14T21:58:01.008833