These Highlights Do Not Include All The Information Needed To Use Briviact ®
3cf2f439-0e97-443e-8e33-25ecef616f6c
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
BRIVIACT is indicated for the treatment of partial-onset seizures in patients 1 month of age and older.
Indications and Usage
BRIVIACT is indicated for the treatment of partial-onset seizures in patients 1 month of age and older.
Dosage and Administration
Adults (16 Years and Older): The recommended starting dosage for monotherapy or adjunctive therapy is 50 mg twice daily (100 mg per day). Based on individual patient tolerability and therapeutic response, the dosage may be adjusted down to 25 mg twice daily (50 mg per day) or up to 100 mg twice daily (200 mg per day). ( 2.1 ) Pediatric Patients (1 Month to less than 16 Years): The recommended dosage is based on body weight and is administered orally twice daily ( 2.1 ) Injection: for intravenous use only when oral administration is temporarily not feasible; dosing is the same as oral regimen. ( 2.1 , 2.3 ) Hepatic Impairment: Dose adjustment is recommended for all stages of hepatic impairment. ( 2.5 )
Warnings and Precautions
Suicidal Behavior and Ideation : Monitor patients for suicidal behavior and ideation. ( 5.1 ) Neurological Adverse Reactions : Monitor for somnolence and fatigue, and advise patients not to drive or operate machinery until they have gained sufficient experience on BRIVIACT. ( 5.2 ) Psychiatric Adverse Reactions : Behavioral reactions including psychotic symptoms, irritability, depression, aggressive behavior, and anxiety; monitor patients for symptoms. ( 5.3 ) Hypersensitivity: Bronchospasm and Angioedema : Advise patients to seek immediate medical care. Discontinue and do not restart BRIVIACT if hypersensitivity occurs. ( 5.4 ) Withdrawal of Antiepileptic Drugs : BRIVIACT should be gradually withdrawn. ( 5.5 )
Contraindications
Hypersensitivity to brivaracetam or any of the inactive ingredients in BRIVIACT (bronchospasm and angioedema have occurred) [see Warnings and Precautions (5.4) ] .
Adverse Reactions
BRIVIACT causes psychiatric adverse reactions. In the Phase 3 controlled adjunctive epilepsy trials, psychiatric adverse reactions were reported in approximately 13% of patients who received BRIVIACT (at least 50 mg/day) compared to 8% of patients who received placebo. Psychiatric events included both non-psychotic symptoms (irritability, anxiety, nervousness, aggression, belligerence, anger, agitation, restlessness, depression, depressed mood, tearfulness, apathy, altered mood, mood swings, affect lability, psychomotor hyperactivity, abnormal behavior, and adjustment disorder) and psychotic symptoms (psychotic disorder along with hallucination, paranoia, acute psychosis, and psychotic behavior). A total of 1.7% of adult patients treated with BRIVIACT discontinued treatment because of psychiatric reactions compared to 1.3% of patients who received placebo. Psychiatric adverse reactions were also observed in open-label pediatric trials and were generally similar to those observed in adults [see Adverse Reactions (6.1) and Use in Specific Populations (8.4) ].
Drug Interactions
Rifampin: Because of decreased concentrations, increasing BRIVIACT dosage in patients on concomitant rifampin is recommended. ( 2.6 , 7.1 ) Carbamazepine: Because of increased exposure to carbamazepine metabolite, if tolerability issues arise, consider reducing carbamazepine dosage in patients on concomitant BRIVIACT. ( 7.2 ) Phenytoin: Because phenytoin concentrations can increase, phenytoin levels should be monitored in patients on concomitant BRIVIACT. ( 7.3 ) Levetiracetam: BRIVIACT had no added therapeutic benefit when co-administered with levetiracetam. ( 7.4 )
Storage and Handling
Store at 25°C (77°F); excursions permitted between 15°C to 30°C (59°F to 86°F). See USP Controlled Room Temperature. Do not freeze BRIVIACT injection or oral solution. Discard any unused BRIVIACT oral solution remaining after 5 months of first opening the bottle. BRIVIACT injection vials are single-dose only [see Dosage and Administration (2.3) ] .
Medication Information
Warnings and Precautions
Suicidal Behavior and Ideation : Monitor patients for suicidal behavior and ideation. ( 5.1 ) Neurological Adverse Reactions : Monitor for somnolence and fatigue, and advise patients not to drive or operate machinery until they have gained sufficient experience on BRIVIACT. ( 5.2 ) Psychiatric Adverse Reactions : Behavioral reactions including psychotic symptoms, irritability, depression, aggressive behavior, and anxiety; monitor patients for symptoms. ( 5.3 ) Hypersensitivity: Bronchospasm and Angioedema : Advise patients to seek immediate medical care. Discontinue and do not restart BRIVIACT if hypersensitivity occurs. ( 5.4 ) Withdrawal of Antiepileptic Drugs : BRIVIACT should be gradually withdrawn. ( 5.5 )
Indications and Usage
BRIVIACT is indicated for the treatment of partial-onset seizures in patients 1 month of age and older.
Dosage and Administration
Adults (16 Years and Older): The recommended starting dosage for monotherapy or adjunctive therapy is 50 mg twice daily (100 mg per day). Based on individual patient tolerability and therapeutic response, the dosage may be adjusted down to 25 mg twice daily (50 mg per day) or up to 100 mg twice daily (200 mg per day). ( 2.1 ) Pediatric Patients (1 Month to less than 16 Years): The recommended dosage is based on body weight and is administered orally twice daily ( 2.1 ) Injection: for intravenous use only when oral administration is temporarily not feasible; dosing is the same as oral regimen. ( 2.1 , 2.3 ) Hepatic Impairment: Dose adjustment is recommended for all stages of hepatic impairment. ( 2.5 )
Contraindications
Hypersensitivity to brivaracetam or any of the inactive ingredients in BRIVIACT (bronchospasm and angioedema have occurred) [see Warnings and Precautions (5.4) ] .
Adverse Reactions
BRIVIACT causes psychiatric adverse reactions. In the Phase 3 controlled adjunctive epilepsy trials, psychiatric adverse reactions were reported in approximately 13% of patients who received BRIVIACT (at least 50 mg/day) compared to 8% of patients who received placebo. Psychiatric events included both non-psychotic symptoms (irritability, anxiety, nervousness, aggression, belligerence, anger, agitation, restlessness, depression, depressed mood, tearfulness, apathy, altered mood, mood swings, affect lability, psychomotor hyperactivity, abnormal behavior, and adjustment disorder) and psychotic symptoms (psychotic disorder along with hallucination, paranoia, acute psychosis, and psychotic behavior). A total of 1.7% of adult patients treated with BRIVIACT discontinued treatment because of psychiatric reactions compared to 1.3% of patients who received placebo. Psychiatric adverse reactions were also observed in open-label pediatric trials and were generally similar to those observed in adults [see Adverse Reactions (6.1) and Use in Specific Populations (8.4) ].
Drug Interactions
Rifampin: Because of decreased concentrations, increasing BRIVIACT dosage in patients on concomitant rifampin is recommended. ( 2.6 , 7.1 ) Carbamazepine: Because of increased exposure to carbamazepine metabolite, if tolerability issues arise, consider reducing carbamazepine dosage in patients on concomitant BRIVIACT. ( 7.2 ) Phenytoin: Because phenytoin concentrations can increase, phenytoin levels should be monitored in patients on concomitant BRIVIACT. ( 7.3 ) Levetiracetam: BRIVIACT had no added therapeutic benefit when co-administered with levetiracetam. ( 7.4 )
Storage and Handling
Store at 25°C (77°F); excursions permitted between 15°C to 30°C (59°F to 86°F). See USP Controlled Room Temperature. Do not freeze BRIVIACT injection or oral solution. Discard any unused BRIVIACT oral solution remaining after 5 months of first opening the bottle. BRIVIACT injection vials are single-dose only [see Dosage and Administration (2.3) ] .
Description
BRIVIACT is indicated for the treatment of partial-onset seizures in patients 1 month of age and older.
Section 34083-6
Carcinogenesis
In a carcinogenicity study in mice, oral administration of brivaracetam (0, 400, 550, or 700 mg/kg/day) for 104 weeks increased the incidence of liver tumors (hepatocellular adenoma and carcinoma) in male mice at the two highest doses tested. At the dose (400 mg/kg) not associated with an increase in liver tumors, plasma exposures (AUC) were approximately equal to those in humans at the maximum recommended dose (MRD) of 200 mg/day. Oral administration (0, 150, 230, 450, or 700 mg/kg/day) to rats for 104 weeks resulted in an increased incidence of thymus tumors (benign thymoma) in female rats at the highest dose tested. At the highest dose not associated with an increase in thymus tumors, plasma exposures were approximately 9 times those in humans at the MRD.
Section 42229-5
Monotherapy or Adjunctive Therapy
The recommended dosage for patients 1 month of age and older is included in Table 1. In pediatric patients weighing less than 50 kg, the recommended dosing regimen is dependent upon body weight. When initiating treatment, gradual dose escalation is not required. Dosage should be adjusted based on clinical response and tolerability.
| Age and Body Weight | Initial Dosage | Minimum and Maximum Maintenance Dosage |
|---|---|---|
| Adults (16 years and older) | 50 mg twice daily
(100 mg per day) |
25 mg to 100 mg twice daily
(50 mg to 200 mg per day) |
| Pediatric patients weighing 50 kg or more | 25 mg to 50 mg twice daily
(50 mg to 100 mg per day) |
25 mg to 100 mg twice daily
(50 mg to 200 mg per day) |
| Pediatric patients weighing 20 kg to less than 50 kg | 0.5 mg/kg to 1 mg/kg twice daily
(1 mg/kg to 2 mg/kg per day) |
0.5 mg/kg to 2 mg/kg twice daily
(1 mg/kg to 4 mg/kg per day) |
| Pediatric patients weighing 11 kg to less than 20 kg | 0.5 mg/kg to 1.25 mg/kg twice daily
(1 mg/kg to 2.5 mg/kg per day) |
0.5 mg/kg to 2.5 mg/kg twice daily
(1 mg/kg to 5 mg/kg per day) |
| Pediatric patients weighing less than 11 kg | 0.75 mg/kg to 1.5 mg/kg twice daily
(1.5 mg/kg to 3 mg/kg per day) |
0.75 mg/kg to 3 mg/kg twice daily
(1.5 mg/kg to 6 mg/kg per day) |
Section 42231-1
| MEDICATION GUIDE
BRIVIACT ®(briv ee akt) CV (brivaracetam) tablets, oral solution, and injection for intravenous use |
|||
|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Revised: 5/2023 | ||
|
What is the most important information I should know about BRIVIACT?
BRIVIACT is a federally controlled substance (CV) because it can be abused or lead to dependence. Keep BRIVIACT in a safe place to prevent misuse and abuse. Selling or giving away BRIVIACT may harm others and is against the law. Like other antiepileptic drugs, BRIVIACT may cause suicidal thoughts or actions in a very small number of people, about 1 in 500 people taking it. Call a healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you: |
|||
|
|
||
| Suicidal thoughts or actions can be caused by things other than medicines. If you have suicidal thoughts or actions, your healthcare provider may check for other causes. | |||
How can I watch for early symptoms of suicidal thoughts and actions?
|
|||
| Call your healthcare provider between visits as needed, especially if you are worried about symptoms. | |||
Do not stop BRIVIACT without first talking to a healthcare provider
.
|
|||
|
What is BRIVIACT?
BRIVIACT is a prescription medicine used to treat partial-onset seizures in people 1 month of age and older. It is not known if BRIVIACT is safe and effective in children younger than 1 month of age. |
|||
|
Who should not take BRIVIACT?
Do not take BRIVIACT if you are allergic to brivaracetam or any of the ingredients in BRIVIACT. See the end of this Medication Guide for a complete list of ingredients in BRIVIACT. |
|||
|
What should I tell my healthcare provider before starting BRIVIACT?
Before taking BRIVIACT, tell your healthcare provider about all of your medical conditions, including if you :
|
|||
| Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. BRIVIACT may affect the way other medicines work, and other medicines may affect how BRIVIACT works. Do not start a new medicine without first talking with your healthcare provider. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist each time you get a new medicine. | |||
How should I take BRIVIACT?
|
|||
|
What should I avoid while taking BRIVIACT?
Do not drive or operate machinery until you know how BRIVIACT affects you. BRIVIACT may cause drowsiness, tiredness, dizziness, and problems with your balance and coordination. |
|||
|
What are the possible side effects of BRIVIACT?
BRIVIACT may cause serious side effects, including:
|
|||
| The most common side effects of BRIVIACT in adults include: | |||
|
|
||
| Side effects of BRIVIACT in children 1 month to less than 16 years of age are similar to those seen in adults.
These are not all the possible side effects of BRIVIACT. For more information, ask your healthcare provider or pharmacist. Tell your healthcare provider about any side effect that bothers you or that does not go away. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
How should I store BRIVIACT?
|
|||
| Keep BRIVIACT and all medicines out of the reach of children. | |||
|
General information about the safe and effective use of BRIVIACT.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use BRIVIACT for a condition for which it was not prescribed. Do not give BRIVIACT to other people, even if they have the same symptoms that you have. It may harm them. This Medication Guide summarizes the most important information about BRIVIACT. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about BRIVIACT that is written for health professionals . |
|||
|
What are the ingredients in BRIVIACT?
Active ingredient: brivaracetam Tablet inactive ingredients:croscarmellose sodium, lactose monohydrate, betadex (β-cyclodextrin), anhydrous lactose, and magnesium stearate. The tablet film coating contains the inactive ingredients listed below: |
|||
|
|||
|
Oral solution inactive ingredients:sodium citrate, anhydrous citric acid, methylparaben, sodium carboxymethylcellulose, sucralose, sorbitol solution, glycerin, raspberry flavor, and purified water.
Injection inactive ingredients: sodium acetate, trihydrate (1.64 mg/mL), glacial acetic acid (for pH adjustment to 5.5), sodium chloride (9.00 mg/mL), and water for injection. |
|||
| Manufactured for UCB, Inc., Smyrna, GA 30080.
BRIVIACT ®is a registered trademark of the UCB Group of Companies. ©2023, UCB, Inc., Smyrna, GA 30080. All rights reserved. For more information, go to www.BRIVIACT.com or call 1-844-599-2273. |
9.2 Abuse
In a human abuse potential study, single doses of BRIVIACT at therapeutic and supratherapeutic doses were compared to alprazolam (C-IV) (1.5 mg and 3 mg). BRIVIACT at the recommended single dose (50 mg) caused fewer sedative and euphoric effects than alprazolam; however, BRIVIACT at supratherapeutic single doses (200 mg and 1000 mg) was similar to alprazolam on other measures of abuse.
7.1 Rifampin
Co-administration with rifampin decreases BRIVIACT plasma concentrations likely because of CYP2C19 induction [see Clinical Pharmacology (12.3)] . Prescribers should increase the BRIVIACT dose by up to 100% (i.e., double the dosage) in patients while receiving concomitant treatment with rifampin [see Dosage and Administration (2.6)] .
10 Overdosage
There is limited clinical experience with BRIVIACT overdose in humans. Somnolence and dizziness were reported in a patient taking a single dose of 1400 mg (14 times the highest recommended single dose) of BRIVIACT. The following adverse reactions were reported with BRIVIACT overdose: vertigo, balance disorder, fatigue, nausea, diplopia, anxiety, and bradycardia. In general, the adverse reactions associated with BRIVIACT overdose were consistent with the known adverse reactions.
There is no specific antidote for overdose with BRIVIACT. In the event of overdose, standard medical practice for the management of any overdose should be used. An adequate airway, oxygenation, and ventilation should be ensured; monitoring of cardiac rate and rhythm and vital signs is recommended. A certified poison control center should be contacted for updated information on the management of overdose with BRIVIACT. There are no data on the removal of brivaracetam using hemodialysis, but because less than 10% of brivaracetam is excreted in urine, hemodialysis is not expected to enhance BRIVIACT clearance.
7.3 Phenytoin
Because BRIVIACT can increase plasma concentrations of phenytoin, phenytoin levels should be monitored in patients when concomitant BRIVIACT is added to or discontinued from ongoing phenytoin therapy [see Clinical Pharmacology (12.3)] .
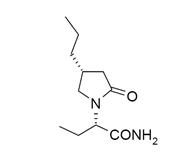
11 Description
The chemical name of BRIVIACT (brivaracetam) is (2S)-2-[(4R)-2-oxo-4-propyltetrahydro-1 H-pyrrol-1-yl] butanamide. Its molecular formula is C 11H 20N 2O 2and its molecular weight is 212.29. The chemical structure is:
Brivaracetam is a white to off-white crystalline powder. It is very soluble in water, buffer (pH 1.2, 4.5, and 7.4), ethanol, methanol, and glacial acetic acid. It is freely soluble in acetonitrile and acetone and soluble in toluene. It is very slightly soluble in n-hexane.
9.3 Dependence
There was no evidence of physical dependence potential or a withdrawal syndrome with BRIVIACT in a pooled review of placebo-controlled adjunctive therapy studies [see Warnings and Precautions (5.5)].
7.2 Carbamazepine
Co-administration with carbamazepine may increase exposure to carbamazepine-epoxide, the active metabolite of carbamazepine. Though available data did not reveal any safety concerns, if tolerability issues arise when co-administered, carbamazepine dose reduction should be considered [see Clinical Pharmacology (12.3)] .
7.4 Levetiracetam
BRIVIACT provided no added therapeutic benefit to levetiracetam when the two drugs were co-administered [see Clinical Studies (14)].
8.4 Pediatric Use
Safety and effectiveness of BRIVIACT have been established in pediatric patients 1 month to less than 16 years of age. Use of BRIVIACT in these age groups is supported by evidence from adequate and well-controlled studies of BRIVIACT in adults with partial-onset seizures, pharmacokinetic data from adult and pediatric patients, and safety data in pediatric patients 2 months to less than 16 years of age [see Dosage and Administration (2.1), Warnings and Precautions (5.3), Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)] .
Safety and effectiveness in pediatric patients below the age of 1 month have not been established.
8.5 Geriatric Use
There were insufficient numbers of patients 65 years of age and older in the double-blind, placebo-controlled epilepsy trials (n=38) to allow adequate assessment of the effectiveness of BRIVIACT in this population. In general, dose selection for an elderly patient should be judicious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy [see Clinical Pharmacology (12.3)] .
14 Clinical Studies
The effectiveness of BRIVIACT in partial-onset seizures with or without secondary generalization was established in 3 fixed-dose, randomized, double-blind, placebo-controlled, multicenter studies (Studies 1, 2, and 3), which included 1550 patients. Patients enrolled had partial-onset seizures that were not adequately controlled with 1 to 2 concomitant antiepileptic drugs (AEDs). In each of these studies, 72% to 86% of patients were taking 2 or more concomitant AEDs with or without vagal nerve stimulation. The median baseline seizure frequency across the 3 studies was 9 seizures per 28 days. Patients had a mean duration of epilepsy of approximately 23 years.
All trials had an 8-week baseline period, during which patients were required to have at least 8 partial-onset seizures. The baseline period was followed by a 12-week treatment period. There was no titration period in these studies. Study 1 compared doses of BRIVIACT 50 mg/day and 100 mg/day with placebo. Study 2 compared a dose of BRIVIACT 50 mg/day with placebo. Study 3 compared doses of BRIVIACT 100 mg/day and 200 mg/day with placebo. BRIVIACT was administered in equally divided twice daily doses. Upon termination of BRIVIACT treatment, patients were down-titrated over a 1-, 2-, and 4-week duration for patients receiving 25, 50, and 100 mg twice daily BRIVIACT, respectively.
The primary efficacy outcome in Study 1 and Study 2 was the percent reduction in 7-day partial-onset seizure frequency over placebo, while the primary outcome for Study 3 was the percent reduction in 28-day partial-onset seizure frequency over placebo. The criteria for statistical significance for all 3 studies was p<0.05. Table 6 presents the primary efficacy outcome of the percent change in seizure frequency over placebo, based upon each study's protocol-defined 7- and 28-day seizure frequency efficacy outcome.
| Percent Reduction Over Placebo
(%) |
|
|---|---|
|
STUDY 1
Based upon 7-day seizure frequency
|
|
| Placebo
(n=100) |
------- |
| 50 mg/day
(n=99) |
9.5 |
| 100 mg/day
(n=100) |
17.0 |
| STUDY 2 | |
| Placebo
(n=96) |
------- |
| 50 mg/day
(n=101) |
16.9
Statistically significant based on testing procedure with alpha = 0.05
|
|
STUDY 3
Based upon 28-day seizure frequency
|
|
| Placebo
(n=259) |
------ |
| 100 mg/day
(n=252) |
25.2 |
| 200 mg/day
(n= 249) |
25.7 |
Figure 1 presents the percentage of patients by category of reduction from baseline in partial-onset seizure frequency per 28 days for all pooled patients in the 3 pivotal studies. Patients in whom the seizure frequency increased are shown at left as "worse." Patients with an improvement in percent reduction from baseline partial-onset seizure frequency are shown in the 4 right-most categories.
Figure 1: Proportion of Patients by Category of Seizure Response for BRIVIACT and Placebo Across all Three Double-Blind Trials
4 Contraindications
Hypersensitivity to brivaracetam or any of the inactive ingredients in BRIVIACT (bronchospasm and angioedema have occurred) [see Warnings and Precautions (5.4)] .
6 Adverse Reactions
The following serious adverse reactions are described elsewhere in labeling:
- Suicidal Behavior and Ideation [see Warnings and Precautions (5.1)]
- Neurological Adverse Reactions [see Warnings and Precautions (5.2)]
- Psychiatric Adverse Reactions [see Warnings and Precautions (5.3)]
- Hypersensitivity: Bronchospasm and Angioedema [see Warnings and Precautions (5.4)]
- Withdrawal of Antiepileptic Drugs [see Warnings and Precautions (5.5)]
7 Drug Interactions
- Rifampin:Because of decreased concentrations, increasing BRIVIACT dosage in patients on concomitant rifampin is recommended. ( 2.6, 7.1)
- Carbamazepine:Because of increased exposure to carbamazepine metabolite, if tolerability issues arise, consider reducing carbamazepine dosage in patients on concomitant BRIVIACT. ( 7.2)
- Phenytoin:Because phenytoin concentrations can increase, phenytoin levels should be monitored in patients on concomitant BRIVIACT. ( 7.3)
- Levetiracetam:BRIVIACT had no added therapeutic benefit when co-administered with levetiracetam. ( 7.4)
8.6 Renal Impairment
Dose adjustments are not required for patients with impaired renal function. There are no data in patients with end-stage renal disease undergoing dialysis, and use of BRIVIACT is not recommended in this patient population [see Clinical Pharmacology (12.3)] .
12.3 Pharmacokinetics
BRIVIACT tablets, oral solution, and injection can be used interchangeably. Brivaracetam exhibits linear and time-independent pharmacokinetics at the approved doses.
The pharmacokinetics of brivaracetam are similar when used as monotherapy or as adjunctive therapy for the treatment of partial-onset seizures.
8.7 Hepatic Impairment
Because of increases in BRIVIACT exposure, dosage adjustment is recommended for all stages of hepatic impairment [see Dosage and Administration (2.5)and Clinical Pharmacology (12.3)] .
1 Indications and Usage
BRIVIACT is indicated for the treatment of partial-onset seizures in patients 1 month of age and older.
12.1 Mechanism of Action
The precise mechanism by which BRIVIACT exerts its anticonvulsant activity is not known. Brivaracetam displays a high and selective affinity for synaptic vesicle protein 2A (SV2A) in the brain, which may contribute to the anticonvulsant effect.
9.1 Controlled Substance
BRIVIACT contains brivaracetam and is listed as a Schedule V controlled substance.
16.2 Storage and Handling
Store at 25°C (77°F); excursions permitted between 15°C to 30°C (59°F to 86°F). See USP Controlled Room Temperature. Do not freeze BRIVIACT injection or oral solution.
Discard any unused BRIVIACT oral solution remaining after 5 months of first opening the bottle.
BRIVIACT injection vials are single-dose only [see Dosage and Administration (2.3)] .
5 Warnings and Precautions
- Suicidal Behavior and Ideation: Monitor patients for suicidal behavior and ideation. ( 5.1)
- Neurological Adverse Reactions: Monitor for somnolence and fatigue, and advise patients not to drive or operate machinery until they have gained sufficient experience on BRIVIACT. ( 5.2)
- Psychiatric Adverse Reactions: Behavioral reactions including psychotic symptoms, irritability, depression, aggressive behavior, and anxiety; monitor patients for symptoms. ( 5.3)
- Hypersensitivity: Bronchospasm and Angioedema: Advise patients to seek immediate medical care. Discontinue and do not restart BRIVIACT if hypersensitivity occurs. ( 5.4)
- Withdrawal of Antiepileptic Drugs: BRIVIACT should be gradually withdrawn. ( 5.5)
2 Dosage and Administration
- Adults (16 Years and Older):The recommended starting dosage for monotherapy or adjunctive therapy is 50 mg twice daily (100 mg per day). Based on individual patient tolerability and therapeutic response, the dosage may be adjusted down to 25 mg twice daily (50 mg per day) or up to 100 mg twice daily (200 mg per day). ( 2.1)
- Pediatric Patients (1 Month to less than 16 Years):The recommended dosage is based on body weight and is administered orally twice daily ( 2.1)
- Injection:for intravenous use only when oral administration is temporarily not feasible; dosing is the same as oral regimen. ( 2.1, 2.3)
- Hepatic Impairment:Dose adjustment is recommended for all stages of hepatic impairment. ( 2.5)
3 Dosage Forms and Strengths
8 Use in Specific Populations
Pregnancy: Based on animal data, may cause fetal harm. ( 8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In all controlled and uncontrolled trials performed in adult epilepsy patients, BRIVIACT was administered as adjunctive therapy to 2437 patients. Of these patients, 1929 were treated for at least 6 months, 1500 for at least 12 months, 1056 for at least 24 months, and 758 for at least 36 months. A total of 1558 patients (1099 patients treated with BRIVIACT and 459 patients treated with placebo) constituted the safety population in the pooled analysis of Phase 3 placebo-controlled studies in patients with partial-onset seizures (Studies 1, 2, and 3) [see Clinical Studies (14)] . The adverse reactions presented in Table 4 are based on this safety population; the median length of treatment in these studies was 12 weeks. Of the patients in those studies, approximately 51% were male, 74% were Caucasian, and the mean age was 38 years.
In the Phase 3 controlled epilepsy studies, adverse events occurred in 68% of patients treated with BRIVIACT and 62% treated with placebo. The most common adverse reactions occurring at a frequency of at least 5% in patients treated with BRIVIACT doses of at least 50 mg/day and greater than placebo were somnolence and sedation (16%), dizziness (12%), fatigue (9%), and nausea and vomiting symptoms (5%).
The discontinuation rates due to adverse events were 5%, 8%, and 7% for patients randomized to receive BRIVIACT at the recommended doses of 50 mg, 100 mg, and 200 mg/day, respectively, compared to 4% in patients randomized to receive placebo.
Table 4 lists adverse reactions for BRIVIACT that occurred at least 2% more frequently for BRIVIACT doses of at least 50 mg/day than placebo.
| Adverse Reactions | BRIVIACT
(N=803) % |
Placebo
(N=459) % |
|---|---|---|
| Gastrointestinal disorders | ||
| Nausea/vomiting symptoms | 5 | 3 |
| Constipation | 2 | 0 |
| Nervous system disorders | ||
| Somnolence and sedation | 16 | 8 |
| Dizziness | 12 | 7 |
| Fatigue | 9 | 4 |
| Cerebellar coordination and balance disturbances
Cerebellar coordination and balance disturbances includes ataxia, balance disorder, coordination abnormal, and nystagmus.
|
3 | 1 |
| Psychiatric disorders | ||
| Irritability | 3 | 1 |
There was no apparent dose-dependent increase in adverse reactions listed in Table 4 with the exception of somnolence and sedation.
2.4 Discontinuation of Briviact
Avoid abrupt withdrawal from BRIVIACT in order to minimize the risk of increased seizure frequency and status epilepticus [see Warnings and Precautions (5.5) and Clinical Studies (14)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide). The Medication Guide accompanies the product and can also be accessed on www.briviact.com or by calling 1-844-599-2273.
5.3 Psychiatric Adverse Reactions
BRIVIACT causes psychiatric adverse reactions. In the Phase 3 controlled adjunctive epilepsy trials, psychiatric adverse reactions were reported in approximately 13% of patients who received BRIVIACT (at least 50 mg/day) compared to 8% of patients who received placebo. Psychiatric events included both non-psychotic symptoms (irritability, anxiety, nervousness, aggression, belligerence, anger, agitation, restlessness, depression, depressed mood, tearfulness, apathy, altered mood, mood swings, affect lability, psychomotor hyperactivity, abnormal behavior, and adjustment disorder) and psychotic symptoms (psychotic disorder along with hallucination, paranoia, acute psychosis, and psychotic behavior). A total of 1.7% of adult patients treated with BRIVIACT discontinued treatment because of psychiatric reactions compared to 1.3% of patients who received placebo.
Psychiatric adverse reactions were also observed in open-label pediatric trials and were generally similar to those observed in adults [see Adverse Reactions (6.1)and Use in Specific Populations (8.4)].
5.1 Suicidal Behavior and Ideation
Antiepileptic drugs (AEDs), including BRIVIACT, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted Relative Risk 1.8, 95% CI:1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence rate of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number is too small to allow any conclusion about drug effect on suicide.
The increased risk of suicidal thoughts or behavior with AEDs was observed as early as one week after starting drug treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5-100 years) in the clinical trials analyzed. Table 3 shows absolute and relative risk by indication for all evaluated AEDs.
| Indication | Placebo Patients with Events Per 1000 Patients | Drug Patients with Events Per 1000 Patients | Relative Risk: Incidence of Events in Drug Patients/Incidence in Placebo Patients | Risk Difference: Additional Drug Patients with Events Per 1000 Patients |
|---|---|---|---|---|
| Epilepsy | 1.0 | 3.4 | 3.5 | 2.4 |
| Psychiatric | 5.7 | 8.5 | 1.5 | 2.9 |
| Other | 1.0 | 1.8 | 1.9 | 0.9 |
| Total | 2.4 | 4.3 | 1.8 | 1.9 |
The relative risk for suicidal thoughts or behavior was higher in clinical trials in patients with epilepsy than in clinical trials in patients with psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
Anyone considering prescribing BRIVIACT or any other AED must balance the risk of suicidal thoughts or behaviors with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
5.2 Neurological Adverse Reactions
BRIVIACT causes somnolence, fatigue, dizziness, and disturbance in coordination. Patients should be monitored for these signs and symptoms and advised not to drive or operate machinery until they have gained sufficient experience on BRIVIACT to gauge whether it adversely affects their ability to drive or operate machinery.
2.6 Co Administration With Rifampin
Increase the BRIVIACT dosage in patients on concomitant rifampin by up to 100% (i.e., double the dosage) [see Drug Interactions (7.1)and Clinical Pharmacology (12.3)] .
2.5 Patients With Hepatic Impairment
The recommended dosage for patients with hepatic impairment is included in Table 2 [see Use in Specific Populations (8.7)and Clinical Pharmacology (12.3)] .
| Age and Body Weight | Initial Dosage | Maximum Maintenance Dosage |
|---|---|---|
| Adults (16 years and older) | 25 mg twice daily
(50 mg per day) |
75 mg twice daily
(150 mg per day) |
| Pediatric patients weighing
50 kg or more |
||
| Pediatric patients weighing 20 kg to less than 50 kg | 0.5 mg/kg twice daily
(1 mg/kg per day) |
1.5 mg/kg twice daily
(3 mg/kg per day) |
| Pediatric patients weighing 11 kg to less than 20 kg | 0.5 mg/kg twice daily
(1 mg/kg per day) |
2 mg/kg twice daily
(4 mg/kg per day) |
| Pediatric patients weighing less than 11 kg | 0.75 mg/kg twice daily
(1.5 mg/kg per day) |
2.25 mg/kg twice daily
(4.5 mg/kg per day) |
5.5 Withdrawal of Antiepileptic Drugs
As with most antiepileptic drugs, BRIVIACT should generally be withdrawn gradually because of the risk of increased seizure frequency and status epilepticus [see Dosage and Administration (2.4)and Clinical Studies (14)] . But if withdrawal is needed because of a serious adverse event, rapid discontinuation can be considered.
Principal Display Panel 10 Mg/ml Bottle Carton
NDC 50474-870-15
Rx only
BRIVIACT
®
(brivaracetam)
oral solution
CV
10 mg/mL
ATTENTION PHARMACIST:
Dispense accompanying
medication guide to
each patient
300 mL
Principal Display Panel 50 Mg/5 Ml Vial Carton
NDC 50474-970-75
Rx only
BRIVIACT
®
(brivaracetam) injection CV
50 mg/5 mL
(10 mg/mL)
For Intravenous Use Only
10 vials
5.4 Hypersensitivity: Bronchospasm and Angioedema
BRIVIACT can cause hypersensitivity reactions. Bronchospasm and angioedema have been reported in patients taking BRIVIACT. If a patient develops hypersensitivity reactions after treatment with BRIVIACT, the drug should be discontinued. BRIVIACT is contraindicated in patients with a prior hypersensitivity reaction to brivaracetam or any of the inactive ingredients [see Contraindications (4)] .
Principal Display Panel 10 Mg Tablet Bottle Carton
NDC 50474-370-66
Rx only
BRIVIACT
®
(brivaracetam)
tablets
CV
10 mg
ATTENTION PHARMACIST:
Dispense accompanying
medication guide to each patient.
60 tablets
Principal Display Panel 25 Mg Tablet Bottle Carton
NDC 50474-470-66
Rx only
BRIVIACT
®
(brivaracetam)
tablets
CV
25 mg
ATTENTION PHARMACIST:
Dispense accompanying
medication guide to each patient.
60 tablets
Principal Display Panel 50 Mg Tablet Bottle Carton
NDC 50474-570-66
Rx only
BRIVIACT
®
(brivaracetam)
tablets
CV
50 mg
ATTENTION PHARMACIST:
Dispense accompanying
medication guide to each patient.
60 tablets
Principal Display Panel 75 Mg Tablet Bottle Carton
NDC 50474-670-66
Rx only
BRIVIACT
®
(brivaracetam)
tablets
CV
75 mg
ATTENTION PHARMACIST:
Dispense accompanying
medication guide to each patient.
60 tablets
Principal Display Panel 100 Mg Tablet Bottle Carton
NDC 50474-770-66
Rx only
BRIVIACT
®
(brivaracetam)
tablets
CV
100 mg
ATTENTION PHARMACIST:
Dispense accompanying
medication guide to each patient.
60 tablets
2.3 Preparation and Administration Instructions for Briviact Injection
BRIVIACT injection is for intravenous use only.
2.2 Administration Instructions for Briviact Tablets and Briviact Oral Solution
BRIVIACT can be initiated with either intravenous or oral administration.
BRIVIACT tablets and oral solution may be taken with or without food.
Structured Label Content
Section 34083-6 (34083-6)
Carcinogenesis
In a carcinogenicity study in mice, oral administration of brivaracetam (0, 400, 550, or 700 mg/kg/day) for 104 weeks increased the incidence of liver tumors (hepatocellular adenoma and carcinoma) in male mice at the two highest doses tested. At the dose (400 mg/kg) not associated with an increase in liver tumors, plasma exposures (AUC) were approximately equal to those in humans at the maximum recommended dose (MRD) of 200 mg/day. Oral administration (0, 150, 230, 450, or 700 mg/kg/day) to rats for 104 weeks resulted in an increased incidence of thymus tumors (benign thymoma) in female rats at the highest dose tested. At the highest dose not associated with an increase in thymus tumors, plasma exposures were approximately 9 times those in humans at the MRD.
Section 42229-5 (42229-5)
Monotherapy or Adjunctive Therapy
The recommended dosage for patients 1 month of age and older is included in Table 1. In pediatric patients weighing less than 50 kg, the recommended dosing regimen is dependent upon body weight. When initiating treatment, gradual dose escalation is not required. Dosage should be adjusted based on clinical response and tolerability.
| Age and Body Weight | Initial Dosage | Minimum and Maximum Maintenance Dosage |
|---|---|---|
| Adults (16 years and older) | 50 mg twice daily
(100 mg per day) |
25 mg to 100 mg twice daily
(50 mg to 200 mg per day) |
| Pediatric patients weighing 50 kg or more | 25 mg to 50 mg twice daily
(50 mg to 100 mg per day) |
25 mg to 100 mg twice daily
(50 mg to 200 mg per day) |
| Pediatric patients weighing 20 kg to less than 50 kg | 0.5 mg/kg to 1 mg/kg twice daily
(1 mg/kg to 2 mg/kg per day) |
0.5 mg/kg to 2 mg/kg twice daily
(1 mg/kg to 4 mg/kg per day) |
| Pediatric patients weighing 11 kg to less than 20 kg | 0.5 mg/kg to 1.25 mg/kg twice daily
(1 mg/kg to 2.5 mg/kg per day) |
0.5 mg/kg to 2.5 mg/kg twice daily
(1 mg/kg to 5 mg/kg per day) |
| Pediatric patients weighing less than 11 kg | 0.75 mg/kg to 1.5 mg/kg twice daily
(1.5 mg/kg to 3 mg/kg per day) |
0.75 mg/kg to 3 mg/kg twice daily
(1.5 mg/kg to 6 mg/kg per day) |
Section 42231-1 (42231-1)
| MEDICATION GUIDE
BRIVIACT ®(briv ee akt) CV (brivaracetam) tablets, oral solution, and injection for intravenous use |
|||
|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Revised: 5/2023 | ||
|
What is the most important information I should know about BRIVIACT?
BRIVIACT is a federally controlled substance (CV) because it can be abused or lead to dependence. Keep BRIVIACT in a safe place to prevent misuse and abuse. Selling or giving away BRIVIACT may harm others and is against the law. Like other antiepileptic drugs, BRIVIACT may cause suicidal thoughts or actions in a very small number of people, about 1 in 500 people taking it. Call a healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you: |
|||
|
|
||
| Suicidal thoughts or actions can be caused by things other than medicines. If you have suicidal thoughts or actions, your healthcare provider may check for other causes. | |||
How can I watch for early symptoms of suicidal thoughts and actions?
|
|||
| Call your healthcare provider between visits as needed, especially if you are worried about symptoms. | |||
Do not stop BRIVIACT without first talking to a healthcare provider
.
|
|||
|
What is BRIVIACT?
BRIVIACT is a prescription medicine used to treat partial-onset seizures in people 1 month of age and older. It is not known if BRIVIACT is safe and effective in children younger than 1 month of age. |
|||
|
Who should not take BRIVIACT?
Do not take BRIVIACT if you are allergic to brivaracetam or any of the ingredients in BRIVIACT. See the end of this Medication Guide for a complete list of ingredients in BRIVIACT. |
|||
|
What should I tell my healthcare provider before starting BRIVIACT?
Before taking BRIVIACT, tell your healthcare provider about all of your medical conditions, including if you :
|
|||
| Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. BRIVIACT may affect the way other medicines work, and other medicines may affect how BRIVIACT works. Do not start a new medicine without first talking with your healthcare provider. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist each time you get a new medicine. | |||
How should I take BRIVIACT?
|
|||
|
What should I avoid while taking BRIVIACT?
Do not drive or operate machinery until you know how BRIVIACT affects you. BRIVIACT may cause drowsiness, tiredness, dizziness, and problems with your balance and coordination. |
|||
|
What are the possible side effects of BRIVIACT?
BRIVIACT may cause serious side effects, including:
|
|||
| The most common side effects of BRIVIACT in adults include: | |||
|
|
||
| Side effects of BRIVIACT in children 1 month to less than 16 years of age are similar to those seen in adults.
These are not all the possible side effects of BRIVIACT. For more information, ask your healthcare provider or pharmacist. Tell your healthcare provider about any side effect that bothers you or that does not go away. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
How should I store BRIVIACT?
|
|||
| Keep BRIVIACT and all medicines out of the reach of children. | |||
|
General information about the safe and effective use of BRIVIACT.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use BRIVIACT for a condition for which it was not prescribed. Do not give BRIVIACT to other people, even if they have the same symptoms that you have. It may harm them. This Medication Guide summarizes the most important information about BRIVIACT. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about BRIVIACT that is written for health professionals . |
|||
|
What are the ingredients in BRIVIACT?
Active ingredient: brivaracetam Tablet inactive ingredients:croscarmellose sodium, lactose monohydrate, betadex (β-cyclodextrin), anhydrous lactose, and magnesium stearate. The tablet film coating contains the inactive ingredients listed below: |
|||
|
|||
|
Oral solution inactive ingredients:sodium citrate, anhydrous citric acid, methylparaben, sodium carboxymethylcellulose, sucralose, sorbitol solution, glycerin, raspberry flavor, and purified water.
Injection inactive ingredients: sodium acetate, trihydrate (1.64 mg/mL), glacial acetic acid (for pH adjustment to 5.5), sodium chloride (9.00 mg/mL), and water for injection. |
|||
| Manufactured for UCB, Inc., Smyrna, GA 30080.
BRIVIACT ®is a registered trademark of the UCB Group of Companies. ©2023, UCB, Inc., Smyrna, GA 30080. All rights reserved. For more information, go to www.BRIVIACT.com or call 1-844-599-2273. |
9.2 Abuse
In a human abuse potential study, single doses of BRIVIACT at therapeutic and supratherapeutic doses were compared to alprazolam (C-IV) (1.5 mg and 3 mg). BRIVIACT at the recommended single dose (50 mg) caused fewer sedative and euphoric effects than alprazolam; however, BRIVIACT at supratherapeutic single doses (200 mg and 1000 mg) was similar to alprazolam on other measures of abuse.
7.1 Rifampin
Co-administration with rifampin decreases BRIVIACT plasma concentrations likely because of CYP2C19 induction [see Clinical Pharmacology (12.3)] . Prescribers should increase the BRIVIACT dose by up to 100% (i.e., double the dosage) in patients while receiving concomitant treatment with rifampin [see Dosage and Administration (2.6)] .
10 Overdosage (10 OVERDOSAGE)
There is limited clinical experience with BRIVIACT overdose in humans. Somnolence and dizziness were reported in a patient taking a single dose of 1400 mg (14 times the highest recommended single dose) of BRIVIACT. The following adverse reactions were reported with BRIVIACT overdose: vertigo, balance disorder, fatigue, nausea, diplopia, anxiety, and bradycardia. In general, the adverse reactions associated with BRIVIACT overdose were consistent with the known adverse reactions.
There is no specific antidote for overdose with BRIVIACT. In the event of overdose, standard medical practice for the management of any overdose should be used. An adequate airway, oxygenation, and ventilation should be ensured; monitoring of cardiac rate and rhythm and vital signs is recommended. A certified poison control center should be contacted for updated information on the management of overdose with BRIVIACT. There are no data on the removal of brivaracetam using hemodialysis, but because less than 10% of brivaracetam is excreted in urine, hemodialysis is not expected to enhance BRIVIACT clearance.
7.3 Phenytoin
Because BRIVIACT can increase plasma concentrations of phenytoin, phenytoin levels should be monitored in patients when concomitant BRIVIACT is added to or discontinued from ongoing phenytoin therapy [see Clinical Pharmacology (12.3)] .
11 Description (11 DESCRIPTION)
The chemical name of BRIVIACT (brivaracetam) is (2S)-2-[(4R)-2-oxo-4-propyltetrahydro-1 H-pyrrol-1-yl] butanamide. Its molecular formula is C 11H 20N 2O 2and its molecular weight is 212.29. The chemical structure is:
Brivaracetam is a white to off-white crystalline powder. It is very soluble in water, buffer (pH 1.2, 4.5, and 7.4), ethanol, methanol, and glacial acetic acid. It is freely soluble in acetonitrile and acetone and soluble in toluene. It is very slightly soluble in n-hexane.
9.3 Dependence
There was no evidence of physical dependence potential or a withdrawal syndrome with BRIVIACT in a pooled review of placebo-controlled adjunctive therapy studies [see Warnings and Precautions (5.5)].
7.2 Carbamazepine
Co-administration with carbamazepine may increase exposure to carbamazepine-epoxide, the active metabolite of carbamazepine. Though available data did not reveal any safety concerns, if tolerability issues arise when co-administered, carbamazepine dose reduction should be considered [see Clinical Pharmacology (12.3)] .
7.4 Levetiracetam
BRIVIACT provided no added therapeutic benefit to levetiracetam when the two drugs were co-administered [see Clinical Studies (14)].
8.4 Pediatric Use
Safety and effectiveness of BRIVIACT have been established in pediatric patients 1 month to less than 16 years of age. Use of BRIVIACT in these age groups is supported by evidence from adequate and well-controlled studies of BRIVIACT in adults with partial-onset seizures, pharmacokinetic data from adult and pediatric patients, and safety data in pediatric patients 2 months to less than 16 years of age [see Dosage and Administration (2.1), Warnings and Precautions (5.3), Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)] .
Safety and effectiveness in pediatric patients below the age of 1 month have not been established.
8.5 Geriatric Use
There were insufficient numbers of patients 65 years of age and older in the double-blind, placebo-controlled epilepsy trials (n=38) to allow adequate assessment of the effectiveness of BRIVIACT in this population. In general, dose selection for an elderly patient should be judicious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy [see Clinical Pharmacology (12.3)] .
14 Clinical Studies (14 CLINICAL STUDIES)
The effectiveness of BRIVIACT in partial-onset seizures with or without secondary generalization was established in 3 fixed-dose, randomized, double-blind, placebo-controlled, multicenter studies (Studies 1, 2, and 3), which included 1550 patients. Patients enrolled had partial-onset seizures that were not adequately controlled with 1 to 2 concomitant antiepileptic drugs (AEDs). In each of these studies, 72% to 86% of patients were taking 2 or more concomitant AEDs with or without vagal nerve stimulation. The median baseline seizure frequency across the 3 studies was 9 seizures per 28 days. Patients had a mean duration of epilepsy of approximately 23 years.
All trials had an 8-week baseline period, during which patients were required to have at least 8 partial-onset seizures. The baseline period was followed by a 12-week treatment period. There was no titration period in these studies. Study 1 compared doses of BRIVIACT 50 mg/day and 100 mg/day with placebo. Study 2 compared a dose of BRIVIACT 50 mg/day with placebo. Study 3 compared doses of BRIVIACT 100 mg/day and 200 mg/day with placebo. BRIVIACT was administered in equally divided twice daily doses. Upon termination of BRIVIACT treatment, patients were down-titrated over a 1-, 2-, and 4-week duration for patients receiving 25, 50, and 100 mg twice daily BRIVIACT, respectively.
The primary efficacy outcome in Study 1 and Study 2 was the percent reduction in 7-day partial-onset seizure frequency over placebo, while the primary outcome for Study 3 was the percent reduction in 28-day partial-onset seizure frequency over placebo. The criteria for statistical significance for all 3 studies was p<0.05. Table 6 presents the primary efficacy outcome of the percent change in seizure frequency over placebo, based upon each study's protocol-defined 7- and 28-day seizure frequency efficacy outcome.
| Percent Reduction Over Placebo
(%) |
|
|---|---|
|
STUDY 1
Based upon 7-day seizure frequency
|
|
| Placebo
(n=100) |
------- |
| 50 mg/day
(n=99) |
9.5 |
| 100 mg/day
(n=100) |
17.0 |
| STUDY 2 | |
| Placebo
(n=96) |
------- |
| 50 mg/day
(n=101) |
16.9
Statistically significant based on testing procedure with alpha = 0.05
|
|
STUDY 3
Based upon 28-day seizure frequency
|
|
| Placebo
(n=259) |
------ |
| 100 mg/day
(n=252) |
25.2 |
| 200 mg/day
(n= 249) |
25.7 |
Figure 1 presents the percentage of patients by category of reduction from baseline in partial-onset seizure frequency per 28 days for all pooled patients in the 3 pivotal studies. Patients in whom the seizure frequency increased are shown at left as "worse." Patients with an improvement in percent reduction from baseline partial-onset seizure frequency are shown in the 4 right-most categories.
Figure 1: Proportion of Patients by Category of Seizure Response for BRIVIACT and Placebo Across all Three Double-Blind Trials
4 Contraindications (4 CONTRAINDICATIONS)
Hypersensitivity to brivaracetam or any of the inactive ingredients in BRIVIACT (bronchospasm and angioedema have occurred) [see Warnings and Precautions (5.4)] .
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described elsewhere in labeling:
- Suicidal Behavior and Ideation [see Warnings and Precautions (5.1)]
- Neurological Adverse Reactions [see Warnings and Precautions (5.2)]
- Psychiatric Adverse Reactions [see Warnings and Precautions (5.3)]
- Hypersensitivity: Bronchospasm and Angioedema [see Warnings and Precautions (5.4)]
- Withdrawal of Antiepileptic Drugs [see Warnings and Precautions (5.5)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Rifampin:Because of decreased concentrations, increasing BRIVIACT dosage in patients on concomitant rifampin is recommended. ( 2.6, 7.1)
- Carbamazepine:Because of increased exposure to carbamazepine metabolite, if tolerability issues arise, consider reducing carbamazepine dosage in patients on concomitant BRIVIACT. ( 7.2)
- Phenytoin:Because phenytoin concentrations can increase, phenytoin levels should be monitored in patients on concomitant BRIVIACT. ( 7.3)
- Levetiracetam:BRIVIACT had no added therapeutic benefit when co-administered with levetiracetam. ( 7.4)
8.6 Renal Impairment
Dose adjustments are not required for patients with impaired renal function. There are no data in patients with end-stage renal disease undergoing dialysis, and use of BRIVIACT is not recommended in this patient population [see Clinical Pharmacology (12.3)] .
12.3 Pharmacokinetics
BRIVIACT tablets, oral solution, and injection can be used interchangeably. Brivaracetam exhibits linear and time-independent pharmacokinetics at the approved doses.
The pharmacokinetics of brivaracetam are similar when used as monotherapy or as adjunctive therapy for the treatment of partial-onset seizures.
8.7 Hepatic Impairment
Because of increases in BRIVIACT exposure, dosage adjustment is recommended for all stages of hepatic impairment [see Dosage and Administration (2.5)and Clinical Pharmacology (12.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
BRIVIACT is indicated for the treatment of partial-onset seizures in patients 1 month of age and older.
12.1 Mechanism of Action
The precise mechanism by which BRIVIACT exerts its anticonvulsant activity is not known. Brivaracetam displays a high and selective affinity for synaptic vesicle protein 2A (SV2A) in the brain, which may contribute to the anticonvulsant effect.
9.1 Controlled Substance
BRIVIACT contains brivaracetam and is listed as a Schedule V controlled substance.
16.2 Storage and Handling
Store at 25°C (77°F); excursions permitted between 15°C to 30°C (59°F to 86°F). See USP Controlled Room Temperature. Do not freeze BRIVIACT injection or oral solution.
Discard any unused BRIVIACT oral solution remaining after 5 months of first opening the bottle.
BRIVIACT injection vials are single-dose only [see Dosage and Administration (2.3)] .
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Suicidal Behavior and Ideation: Monitor patients for suicidal behavior and ideation. ( 5.1)
- Neurological Adverse Reactions: Monitor for somnolence and fatigue, and advise patients not to drive or operate machinery until they have gained sufficient experience on BRIVIACT. ( 5.2)
- Psychiatric Adverse Reactions: Behavioral reactions including psychotic symptoms, irritability, depression, aggressive behavior, and anxiety; monitor patients for symptoms. ( 5.3)
- Hypersensitivity: Bronchospasm and Angioedema: Advise patients to seek immediate medical care. Discontinue and do not restart BRIVIACT if hypersensitivity occurs. ( 5.4)
- Withdrawal of Antiepileptic Drugs: BRIVIACT should be gradually withdrawn. ( 5.5)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Adults (16 Years and Older):The recommended starting dosage for monotherapy or adjunctive therapy is 50 mg twice daily (100 mg per day). Based on individual patient tolerability and therapeutic response, the dosage may be adjusted down to 25 mg twice daily (50 mg per day) or up to 100 mg twice daily (200 mg per day). ( 2.1)
- Pediatric Patients (1 Month to less than 16 Years):The recommended dosage is based on body weight and is administered orally twice daily ( 2.1)
- Injection:for intravenous use only when oral administration is temporarily not feasible; dosing is the same as oral regimen. ( 2.1, 2.3)
- Hepatic Impairment:Dose adjustment is recommended for all stages of hepatic impairment. ( 2.5)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy: Based on animal data, may cause fetal harm. ( 8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In all controlled and uncontrolled trials performed in adult epilepsy patients, BRIVIACT was administered as adjunctive therapy to 2437 patients. Of these patients, 1929 were treated for at least 6 months, 1500 for at least 12 months, 1056 for at least 24 months, and 758 for at least 36 months. A total of 1558 patients (1099 patients treated with BRIVIACT and 459 patients treated with placebo) constituted the safety population in the pooled analysis of Phase 3 placebo-controlled studies in patients with partial-onset seizures (Studies 1, 2, and 3) [see Clinical Studies (14)] . The adverse reactions presented in Table 4 are based on this safety population; the median length of treatment in these studies was 12 weeks. Of the patients in those studies, approximately 51% were male, 74% were Caucasian, and the mean age was 38 years.
In the Phase 3 controlled epilepsy studies, adverse events occurred in 68% of patients treated with BRIVIACT and 62% treated with placebo. The most common adverse reactions occurring at a frequency of at least 5% in patients treated with BRIVIACT doses of at least 50 mg/day and greater than placebo were somnolence and sedation (16%), dizziness (12%), fatigue (9%), and nausea and vomiting symptoms (5%).
The discontinuation rates due to adverse events were 5%, 8%, and 7% for patients randomized to receive BRIVIACT at the recommended doses of 50 mg, 100 mg, and 200 mg/day, respectively, compared to 4% in patients randomized to receive placebo.
Table 4 lists adverse reactions for BRIVIACT that occurred at least 2% more frequently for BRIVIACT doses of at least 50 mg/day than placebo.
| Adverse Reactions | BRIVIACT
(N=803) % |
Placebo
(N=459) % |
|---|---|---|
| Gastrointestinal disorders | ||
| Nausea/vomiting symptoms | 5 | 3 |
| Constipation | 2 | 0 |
| Nervous system disorders | ||
| Somnolence and sedation | 16 | 8 |
| Dizziness | 12 | 7 |
| Fatigue | 9 | 4 |
| Cerebellar coordination and balance disturbances
Cerebellar coordination and balance disturbances includes ataxia, balance disorder, coordination abnormal, and nystagmus.
|
3 | 1 |
| Psychiatric disorders | ||
| Irritability | 3 | 1 |
There was no apparent dose-dependent increase in adverse reactions listed in Table 4 with the exception of somnolence and sedation.
2.4 Discontinuation of Briviact (2.4 Discontinuation of BRIVIACT)
Avoid abrupt withdrawal from BRIVIACT in order to minimize the risk of increased seizure frequency and status epilepticus [see Warnings and Precautions (5.5) and Clinical Studies (14)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide). The Medication Guide accompanies the product and can also be accessed on www.briviact.com or by calling 1-844-599-2273.
5.3 Psychiatric Adverse Reactions
BRIVIACT causes psychiatric adverse reactions. In the Phase 3 controlled adjunctive epilepsy trials, psychiatric adverse reactions were reported in approximately 13% of patients who received BRIVIACT (at least 50 mg/day) compared to 8% of patients who received placebo. Psychiatric events included both non-psychotic symptoms (irritability, anxiety, nervousness, aggression, belligerence, anger, agitation, restlessness, depression, depressed mood, tearfulness, apathy, altered mood, mood swings, affect lability, psychomotor hyperactivity, abnormal behavior, and adjustment disorder) and psychotic symptoms (psychotic disorder along with hallucination, paranoia, acute psychosis, and psychotic behavior). A total of 1.7% of adult patients treated with BRIVIACT discontinued treatment because of psychiatric reactions compared to 1.3% of patients who received placebo.
Psychiatric adverse reactions were also observed in open-label pediatric trials and were generally similar to those observed in adults [see Adverse Reactions (6.1)and Use in Specific Populations (8.4)].
5.1 Suicidal Behavior and Ideation
Antiepileptic drugs (AEDs), including BRIVIACT, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted Relative Risk 1.8, 95% CI:1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence rate of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number is too small to allow any conclusion about drug effect on suicide.
The increased risk of suicidal thoughts or behavior with AEDs was observed as early as one week after starting drug treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5-100 years) in the clinical trials analyzed. Table 3 shows absolute and relative risk by indication for all evaluated AEDs.
| Indication | Placebo Patients with Events Per 1000 Patients | Drug Patients with Events Per 1000 Patients | Relative Risk: Incidence of Events in Drug Patients/Incidence in Placebo Patients | Risk Difference: Additional Drug Patients with Events Per 1000 Patients |
|---|---|---|---|---|
| Epilepsy | 1.0 | 3.4 | 3.5 | 2.4 |
| Psychiatric | 5.7 | 8.5 | 1.5 | 2.9 |
| Other | 1.0 | 1.8 | 1.9 | 0.9 |
| Total | 2.4 | 4.3 | 1.8 | 1.9 |
The relative risk for suicidal thoughts or behavior was higher in clinical trials in patients with epilepsy than in clinical trials in patients with psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
Anyone considering prescribing BRIVIACT or any other AED must balance the risk of suicidal thoughts or behaviors with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
5.2 Neurological Adverse Reactions
BRIVIACT causes somnolence, fatigue, dizziness, and disturbance in coordination. Patients should be monitored for these signs and symptoms and advised not to drive or operate machinery until they have gained sufficient experience on BRIVIACT to gauge whether it adversely affects their ability to drive or operate machinery.
2.6 Co Administration With Rifampin (2.6 Co-administration with Rifampin)
Increase the BRIVIACT dosage in patients on concomitant rifampin by up to 100% (i.e., double the dosage) [see Drug Interactions (7.1)and Clinical Pharmacology (12.3)] .
2.5 Patients With Hepatic Impairment (2.5 Patients with Hepatic Impairment)
The recommended dosage for patients with hepatic impairment is included in Table 2 [see Use in Specific Populations (8.7)and Clinical Pharmacology (12.3)] .
| Age and Body Weight | Initial Dosage | Maximum Maintenance Dosage |
|---|---|---|
| Adults (16 years and older) | 25 mg twice daily
(50 mg per day) |
75 mg twice daily
(150 mg per day) |
| Pediatric patients weighing
50 kg or more |
||
| Pediatric patients weighing 20 kg to less than 50 kg | 0.5 mg/kg twice daily
(1 mg/kg per day) |
1.5 mg/kg twice daily
(3 mg/kg per day) |
| Pediatric patients weighing 11 kg to less than 20 kg | 0.5 mg/kg twice daily
(1 mg/kg per day) |
2 mg/kg twice daily
(4 mg/kg per day) |
| Pediatric patients weighing less than 11 kg | 0.75 mg/kg twice daily
(1.5 mg/kg per day) |
2.25 mg/kg twice daily
(4.5 mg/kg per day) |
5.5 Withdrawal of Antiepileptic Drugs
As with most antiepileptic drugs, BRIVIACT should generally be withdrawn gradually because of the risk of increased seizure frequency and status epilepticus [see Dosage and Administration (2.4)and Clinical Studies (14)] . But if withdrawal is needed because of a serious adverse event, rapid discontinuation can be considered.
Principal Display Panel 10 Mg/ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 10 mg/mL Bottle Carton)
NDC 50474-870-15
Rx only
BRIVIACT
®
(brivaracetam)
oral solution
CV
10 mg/mL
ATTENTION PHARMACIST:
Dispense accompanying
medication guide to
each patient
300 mL
Principal Display Panel 50 Mg/5 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 50 mg/5 mL Vial Carton)
NDC 50474-970-75
Rx only
BRIVIACT
®
(brivaracetam) injection CV
50 mg/5 mL
(10 mg/mL)
For Intravenous Use Only
10 vials
5.4 Hypersensitivity: Bronchospasm and Angioedema
BRIVIACT can cause hypersensitivity reactions. Bronchospasm and angioedema have been reported in patients taking BRIVIACT. If a patient develops hypersensitivity reactions after treatment with BRIVIACT, the drug should be discontinued. BRIVIACT is contraindicated in patients with a prior hypersensitivity reaction to brivaracetam or any of the inactive ingredients [see Contraindications (4)] .
Principal Display Panel 10 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 10 mg Tablet Bottle Carton)
NDC 50474-370-66
Rx only
BRIVIACT
®
(brivaracetam)
tablets
CV
10 mg
ATTENTION PHARMACIST:
Dispense accompanying
medication guide to each patient.
60 tablets
Principal Display Panel 25 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 25 mg Tablet Bottle Carton)
NDC 50474-470-66
Rx only
BRIVIACT
®
(brivaracetam)
tablets
CV
25 mg
ATTENTION PHARMACIST:
Dispense accompanying
medication guide to each patient.
60 tablets
Principal Display Panel 50 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 50 mg Tablet Bottle Carton)
NDC 50474-570-66
Rx only
BRIVIACT
®
(brivaracetam)
tablets
CV
50 mg
ATTENTION PHARMACIST:
Dispense accompanying
medication guide to each patient.
60 tablets
Principal Display Panel 75 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 75 mg Tablet Bottle Carton)
NDC 50474-670-66
Rx only
BRIVIACT
®
(brivaracetam)
tablets
CV
75 mg
ATTENTION PHARMACIST:
Dispense accompanying
medication guide to each patient.
60 tablets
Principal Display Panel 100 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 100 mg Tablet Bottle Carton)
NDC 50474-770-66
Rx only
BRIVIACT
®
(brivaracetam)
tablets
CV
100 mg
ATTENTION PHARMACIST:
Dispense accompanying
medication guide to each patient.
60 tablets
2.3 Preparation and Administration Instructions for Briviact Injection (2.3 Preparation and Administration Instructions for BRIVIACT Injection)
BRIVIACT injection is for intravenous use only.
2.2 Administration Instructions for Briviact Tablets and Briviact Oral Solution (2.2 Administration Instructions for BRIVIACT Tablets and BRIVIACT Oral Solution)
BRIVIACT can be initiated with either intravenous or oral administration.
BRIVIACT tablets and oral solution may be taken with or without food.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:26.550386 · Updated: 2026-03-14T22:51:41.355943