These Highlights Do Not Include All The Information Needed To Use Avaclyr Safely And Effectively. See Full Prescribing Information For Avaclyr.
3ce26231-bc40-48b0-85e5-2e7622f334d7
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
AVACLYR is a sterile topical antiviral indicated for the treatment of acute herpetic keratitis (dendritic ulcers) in patients with herpes simplex (HSV-1 and HSV-2) virus.
Indications and Usage
AVACLYR is a sterile topical antiviral indicated for the treatment of acute herpetic keratitis (dendritic ulcers) in patients with herpes simplex (HSV-1 and HSV-2) virus.
Dosage and Administration
The recommended dosing regimen is to apply a 1 cm ribbon of ointment in the lower cul-de-sac of the affected eye 5 times per day (approximately every 3 hours while awake) until the corneal ulcer heals and then a 1 cm ribbon 3 times per day for 7 days.
Warnings and Precautions
AVACLYR is indicated for topical ophthalmic use. ( 5.1 ) Patients should not wear contact lenses if they have signs or symptoms of herpetic keratitis or during the course of therapy with AVACLYR. ( 5.2 )
Contraindications
AVACLYR is contraindicated for patients who develop sensitivity to acyclovir or valacyclovir.
Adverse Reactions
The most common adverse reactions (2-10%) reported in patients were eye pain (stinging), punctate keratitis and follicular conjunctivitis. Rare reports of blepharitis and very rare reports of immediate hypersensitivity reactions including angioedema and urticaria have been observed post-marketing in patients taking AVACLYR.
Drug Interactions
No clinically significant interactions have been identified resulting from topical administration of other drugs concomitantly with AVACLYR.
Storage and Handling
AVACLYR is available in a 3.5 g aluminum tube with a white high density polyethylene cap as a clear, colorless, sterile ophthalmic ointment for topical use containing 3% acyclovir active drug. Each tube is packaged in an individual carton. 3.5 g tube (NDC 48102-028-35) Store at 15°C to 25°C (59°F to 77°F).
How Supplied
AVACLYR is available in a 3.5 g aluminum tube with a white high density polyethylene cap as a clear, colorless, sterile ophthalmic ointment for topical use containing 3% acyclovir active drug. Each tube is packaged in an individual carton. 3.5 g tube (NDC 48102-028-35) Store at 15°C to 25°C (59°F to 77°F).
Medication Information
Warnings and Precautions
AVACLYR is indicated for topical ophthalmic use. ( 5.1 ) Patients should not wear contact lenses if they have signs or symptoms of herpetic keratitis or during the course of therapy with AVACLYR. ( 5.2 )
Indications and Usage
AVACLYR is a sterile topical antiviral indicated for the treatment of acute herpetic keratitis (dendritic ulcers) in patients with herpes simplex (HSV-1 and HSV-2) virus.
Dosage and Administration
The recommended dosing regimen is to apply a 1 cm ribbon of ointment in the lower cul-de-sac of the affected eye 5 times per day (approximately every 3 hours while awake) until the corneal ulcer heals and then a 1 cm ribbon 3 times per day for 7 days.
Contraindications
AVACLYR is contraindicated for patients who develop sensitivity to acyclovir or valacyclovir.
Adverse Reactions
The most common adverse reactions (2-10%) reported in patients were eye pain (stinging), punctate keratitis and follicular conjunctivitis. Rare reports of blepharitis and very rare reports of immediate hypersensitivity reactions including angioedema and urticaria have been observed post-marketing in patients taking AVACLYR.
Drug Interactions
No clinically significant interactions have been identified resulting from topical administration of other drugs concomitantly with AVACLYR.
Storage and Handling
AVACLYR is available in a 3.5 g aluminum tube with a white high density polyethylene cap as a clear, colorless, sterile ophthalmic ointment for topical use containing 3% acyclovir active drug. Each tube is packaged in an individual carton. 3.5 g tube (NDC 48102-028-35) Store at 15°C to 25°C (59°F to 77°F).
How Supplied
AVACLYR is available in a 3.5 g aluminum tube with a white high density polyethylene cap as a clear, colorless, sterile ophthalmic ointment for topical use containing 3% acyclovir active drug. Each tube is packaged in an individual carton. 3.5 g tube (NDC 48102-028-35) Store at 15°C to 25°C (59°F to 77°F).
Description
AVACLYR is a sterile topical antiviral indicated for the treatment of acute herpetic keratitis (dendritic ulcers) in patients with herpes simplex (HSV-1 and HSV-2) virus.
Section 42229-5
Risk Summary
A prospective epidemiologic registry of acyclovir use from 1984 to 1999 indicated that the occurrence rate of birth defects in women exposed to systemically administered acyclovir during the first trimester of pregnancy (period of organogenesis) approximated that found in the general population. Likewise, oral and subcutaneous administration of acyclovir to pregnant mice, rats, and rabbits during organogenesis did not produce teratogenicity at clinically relevant doses (see Animal Data).
10 Overdosage
Overdosage of topical application of acyclovir 3% is unlikely because of minimal absorption.
11 Description
Acyclovir is a synthetic herpes simplex virus nucleoside analog DNA polymerase inhibitor. The drug substance is a white crystalline powder with the molecular formula of C8H11N5O3 and a molecular weight of 225.2. The maximum solubility in water at 25°C is 1.41 mg/mL. The pka's of acyclovir are 2.52 and 9.35.
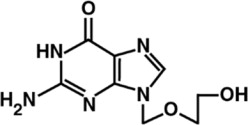
The chemical name of acyclovir is 2-amino-1,9-dihydro-9-[(2-hydroxyethoxy)methyl]6H-purin-6-one, it has the following chemical structure:
AVACLYR is a sterile ointment for topical administration in eyes. Each gram of ointment contains 30 mg of acyclovir in a white petrolatum base.
12.4 Microbiology
Acyclovir is a synthetic purine nucleoside analogue that is phosphorylated intracellularly by the viral encoded thymidine kinase (TK) of HSV into acyclovir monophosphate, a nucleotide analogue. The monophosphate is further converted into diphosphate by cellular guanylate kinase and into triphosphate by a number of cellular enzymes. In a biochemical reaction, acyclovir triphosphate inhibits replication of herpes viral DNA by competing with nucleotides for binding to the viral DNA polymerase and by incorporation into and termination of the growing viral DNA chain. The cellular thymidine kinase of normal, uninfected cells does not use acyclovir effectively as a substrate, hence toxicity to mammalian host cells is low.
8.4 Pediatric Use
Safety and efficacy of Acyclovir ophthalmic ointment in pediatric patients below the age of two years has not been established.
8.5 Geriatric Use
No overall differences in safety or effectiveness have been observed between elderly and other adult patients.
14 Clinical Studies
In five randomized, double masked studies which enrolled a total of 238 subjects with dendritic herpetic keratitis, acyclovir ophthalmic ointment, 3% was either superior or as effective as idoxuridine ophthalmic ointment 0.5% or 1% in subjects with dendritic ulcers. Clinical resolution (healed ulcers) at Day 7 averaged 83% for acyclovir and 50% for idoxuridine.
4 Contraindications
AVACLYR is contraindicated for patients who develop sensitivity to acyclovir or valacyclovir.
6 Adverse Reactions
The most common adverse reactions (2-10%) reported in patients were eye pain (stinging), punctate keratitis and follicular conjunctivitis. Rare reports of blepharitis and very rare reports of immediate hypersensitivity reactions including angioedema and urticaria have been observed post-marketing in patients taking AVACLYR.
7 Drug Interactions
No clinically significant interactions have been identified resulting from topical administration of other drugs concomitantly with AVACLYR.
12.3 Pharmacokinetics
It has not been possible to detect acyclovir concentrations in the blood by existing bioanalytical methods after topical application to the eye. Trace quantities are detectable in the urine but are not therapeutically relevant.
1 Indications and Usage
AVACLYR is a sterile topical antiviral indicated for the treatment of acute herpetic keratitis (dendritic ulcers) in patients with herpes simplex (HSV-1 and HSV-2) virus.
12.1 Mechanism of Action
Acyclovir is an antiviral drug [see Microbiology (12.4)].
5.3 Risk of Contamination
This product is sterile when packaged. Patients should be advised to not allow the tip of the container to touch any surface, as this may contaminate the ointment. If pain develops, or if redness, itching, or inflammation becomes aggravated, the patient should be advised to consult a physician.
5 Warnings and Precautions
5.1 Topical Ophthalmic Use
AVACLYR is indicated for topical ophthalmic use.
2 Dosage and Administration
The recommended dosing regimen is to apply a 1 cm ribbon of ointment in the lower cul-de-sac of the affected eye 5 times per day (approximately every 3 hours while awake) until the corneal ulcer heals and then a 1 cm ribbon 3 times per day for 7 days.
3 Dosage Forms and Strengths
AVACLYR is an ophthalmic ointment containing 3% acyclovir.
5.2 Avoidance of Contact Lenses
Patients should not wear contact lenses if they have signs or symptoms of herpetic keratitis or during the course of therapy with AVACLYR.
16 How Supplied/storage and Handling
AVACLYR is available in a 3.5 g aluminum tube with a white high density polyethylene cap as a clear, colorless, sterile ophthalmic ointment for topical use containing 3% acyclovir active drug. Each tube is packaged in an individual carton.
3.5 g tube (NDC 48102-028-35)
Store at 15°C to 25°C (59°F to 77°F).
Principal Display Panel 30 Mg Tube Label
FERA
NDC 48102-028-35
Avaclyr®
(Acyclovir
Ophthalmic
Ointment) 3%
Each gram contains:
acyclovir USP 30 mg
in an ointment base
of white petrolatum.
For Topical
Ophthalmic Use.
Sterile
3.5g Net Wt. Rx only
Lot:
Principal Display Panel 30 Mg Carton Label
FERA
NDC 48102-028-35
Avaclyr®
(Acyclovir
Ophthalmic
Ointment) 3%
For
Topical
Ophthalmic
Use.
STERILE
3.5g Net Wt. Rx only
Structured Label Content
Section 42229-5 (42229-5)
Risk Summary
A prospective epidemiologic registry of acyclovir use from 1984 to 1999 indicated that the occurrence rate of birth defects in women exposed to systemically administered acyclovir during the first trimester of pregnancy (period of organogenesis) approximated that found in the general population. Likewise, oral and subcutaneous administration of acyclovir to pregnant mice, rats, and rabbits during organogenesis did not produce teratogenicity at clinically relevant doses (see Animal Data).
10 Overdosage (10 OVERDOSAGE)
Overdosage of topical application of acyclovir 3% is unlikely because of minimal absorption.
11 Description (11 DESCRIPTION)
Acyclovir is a synthetic herpes simplex virus nucleoside analog DNA polymerase inhibitor. The drug substance is a white crystalline powder with the molecular formula of C8H11N5O3 and a molecular weight of 225.2. The maximum solubility in water at 25°C is 1.41 mg/mL. The pka's of acyclovir are 2.52 and 9.35.
The chemical name of acyclovir is 2-amino-1,9-dihydro-9-[(2-hydroxyethoxy)methyl]6H-purin-6-one, it has the following chemical structure:
AVACLYR is a sterile ointment for topical administration in eyes. Each gram of ointment contains 30 mg of acyclovir in a white petrolatum base.
12.4 Microbiology
Acyclovir is a synthetic purine nucleoside analogue that is phosphorylated intracellularly by the viral encoded thymidine kinase (TK) of HSV into acyclovir monophosphate, a nucleotide analogue. The monophosphate is further converted into diphosphate by cellular guanylate kinase and into triphosphate by a number of cellular enzymes. In a biochemical reaction, acyclovir triphosphate inhibits replication of herpes viral DNA by competing with nucleotides for binding to the viral DNA polymerase and by incorporation into and termination of the growing viral DNA chain. The cellular thymidine kinase of normal, uninfected cells does not use acyclovir effectively as a substrate, hence toxicity to mammalian host cells is low.
8.4 Pediatric Use
Safety and efficacy of Acyclovir ophthalmic ointment in pediatric patients below the age of two years has not been established.
8.5 Geriatric Use
No overall differences in safety or effectiveness have been observed between elderly and other adult patients.
14 Clinical Studies (14 CLINICAL STUDIES)
In five randomized, double masked studies which enrolled a total of 238 subjects with dendritic herpetic keratitis, acyclovir ophthalmic ointment, 3% was either superior or as effective as idoxuridine ophthalmic ointment 0.5% or 1% in subjects with dendritic ulcers. Clinical resolution (healed ulcers) at Day 7 averaged 83% for acyclovir and 50% for idoxuridine.
4 Contraindications (4 CONTRAINDICATIONS)
AVACLYR is contraindicated for patients who develop sensitivity to acyclovir or valacyclovir.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reactions (2-10%) reported in patients were eye pain (stinging), punctate keratitis and follicular conjunctivitis. Rare reports of blepharitis and very rare reports of immediate hypersensitivity reactions including angioedema and urticaria have been observed post-marketing in patients taking AVACLYR.
7 Drug Interactions (7 DRUG INTERACTIONS)
No clinically significant interactions have been identified resulting from topical administration of other drugs concomitantly with AVACLYR.
12.3 Pharmacokinetics
It has not been possible to detect acyclovir concentrations in the blood by existing bioanalytical methods after topical application to the eye. Trace quantities are detectable in the urine but are not therapeutically relevant.
1 Indications and Usage (1 INDICATIONS AND USAGE)
AVACLYR is a sterile topical antiviral indicated for the treatment of acute herpetic keratitis (dendritic ulcers) in patients with herpes simplex (HSV-1 and HSV-2) virus.
12.1 Mechanism of Action
Acyclovir is an antiviral drug [see Microbiology (12.4)].
5.3 Risk of Contamination
This product is sterile when packaged. Patients should be advised to not allow the tip of the container to touch any surface, as this may contaminate the ointment. If pain develops, or if redness, itching, or inflammation becomes aggravated, the patient should be advised to consult a physician.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
5.1 Topical Ophthalmic Use
AVACLYR is indicated for topical ophthalmic use.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended dosing regimen is to apply a 1 cm ribbon of ointment in the lower cul-de-sac of the affected eye 5 times per day (approximately every 3 hours while awake) until the corneal ulcer heals and then a 1 cm ribbon 3 times per day for 7 days.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
AVACLYR is an ophthalmic ointment containing 3% acyclovir.
5.2 Avoidance of Contact Lenses
Patients should not wear contact lenses if they have signs or symptoms of herpetic keratitis or during the course of therapy with AVACLYR.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
AVACLYR is available in a 3.5 g aluminum tube with a white high density polyethylene cap as a clear, colorless, sterile ophthalmic ointment for topical use containing 3% acyclovir active drug. Each tube is packaged in an individual carton.
3.5 g tube (NDC 48102-028-35)
Store at 15°C to 25°C (59°F to 77°F).
Principal Display Panel 30 Mg Tube Label (Principal Display Panel - 30 mg Tube Label)
FERA
NDC 48102-028-35
Avaclyr®
(Acyclovir
Ophthalmic
Ointment) 3%
Each gram contains:
acyclovir USP 30 mg
in an ointment base
of white petrolatum.
For Topical
Ophthalmic Use.
Sterile
3.5g Net Wt. Rx only
Lot:
Principal Display Panel 30 Mg Carton Label (Principal Display Panel - 30 mg Carton Label)
FERA
NDC 48102-028-35
Avaclyr®
(Acyclovir
Ophthalmic
Ointment) 3%
For
Topical
Ophthalmic
Use.
STERILE
3.5g Net Wt. Rx only
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:59.596810 · Updated: 2026-03-14T22:45:11.004418