3cba84ea-5ee0-7ee3-e063-6294a90af11b
34390-5
HUMAN OTC DRUG LABEL
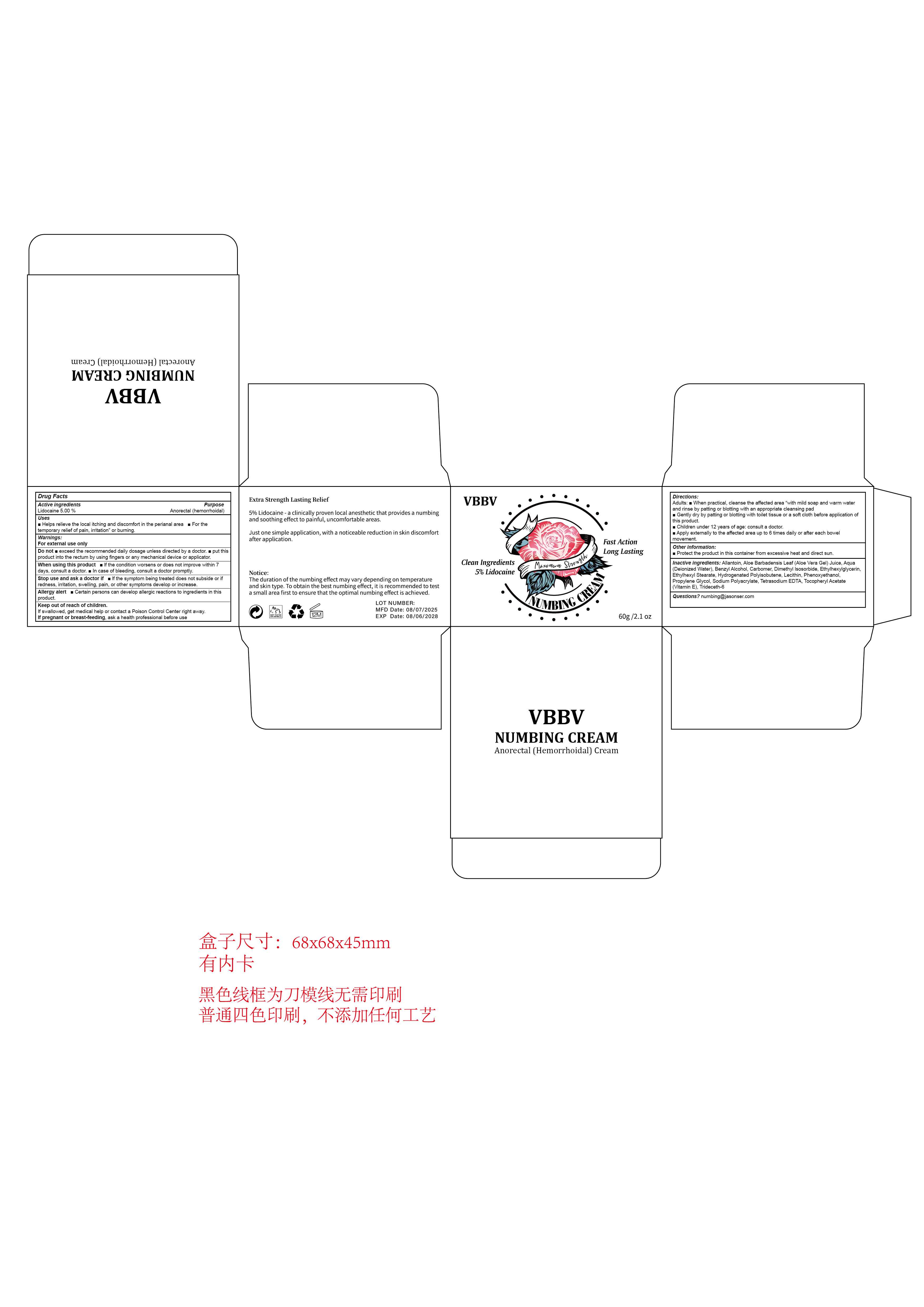
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 5.00 % Anorectal (hemorrhoidal)
Medication Information
Warnings and Precautions
Warnings:For external use only
Indications and Usage
When using this product m lf the condition worsens or does not improve within 7days, consult a doctor. m In case of bleeding, consult a doctor promptly.
Dosage and Administration
Directions:
Adults: m When practical, cleanse the affected area “with mild soap and warm waterand rinse by patting or blotting with an appropriate cleansing padGently dry by patting or blotting with toilet tissue or a soft cloth before application ofthis product.
Children under 12 years of age: consult a doctor.Apply externally to the affected area up to 6 times daily or after each bowelmovement.
Description
Lidocaine 5.00 % Anorectal (hemorrhoidal)
Section 50565-1
Keep out of reach of children.
lf swallowed, get medical help or contact a Poison Control Center right away.If pregnant or breast-feeding, ask a health professional before use
Section 51727-6
Inactive ingredients: Allantoin, Aloe Barbadensis Leaf (Aloe Vera Gel) Juice, Aqua(Deionized Water), Benzy Alcohol, Carbomer, Dimethyl lsosorbide, Ethylhexylglycerin,.Ethylhexyl Stearate, Hydrogenated Polyisobutene, Lecithin, Phenoxyethanol,Propylene Glycol, Sodium Polyacrylate, Tetrasodium EDTA, Tocopheryl Acetate(Vitamin E), Trideceth-6
Section 51945-4
Section 55105-1
Helps relieve the local itching and discomfort in the perianal area m For thetemporary relief of pain, irritation" or burning.
Section 55106-9
Lidocaine 5.00 %
Anorectal (hemorrhoidal)
Section 60561-8
Other information:Protect the product in this container from excessive heat and direct sun.
Structured Label Content
Indications and Usage (34067-9)
When using this product m lf the condition worsens or does not improve within 7days, consult a doctor. m In case of bleeding, consult a doctor promptly.
Dosage and Administration (34068-7)
Directions:
Adults: m When practical, cleanse the affected area “with mild soap and warm waterand rinse by patting or blotting with an appropriate cleansing padGently dry by patting or blotting with toilet tissue or a soft cloth before application ofthis product.
Children under 12 years of age: consult a doctor.Apply externally to the affected area up to 6 times daily or after each bowelmovement.
Warnings and Precautions (34071-1)
Warnings:For external use only
Section 50565-1 (50565-1)
Keep out of reach of children.
lf swallowed, get medical help or contact a Poison Control Center right away.If pregnant or breast-feeding, ask a health professional before use
Section 51727-6 (51727-6)
Inactive ingredients: Allantoin, Aloe Barbadensis Leaf (Aloe Vera Gel) Juice, Aqua(Deionized Water), Benzy Alcohol, Carbomer, Dimethyl lsosorbide, Ethylhexylglycerin,.Ethylhexyl Stearate, Hydrogenated Polyisobutene, Lecithin, Phenoxyethanol,Propylene Glycol, Sodium Polyacrylate, Tetrasodium EDTA, Tocopheryl Acetate(Vitamin E), Trideceth-6
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Helps relieve the local itching and discomfort in the perianal area m For thetemporary relief of pain, irritation" or burning.
Section 55106-9 (55106-9)
Lidocaine 5.00 %
Anorectal (hemorrhoidal)
Section 60561-8 (60561-8)
Other information:Protect the product in this container from excessive heat and direct sun.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:48.862587 · Updated: 2026-03-14T23:09:49.100948