84484-001
3c4a3b41-9183-3835-e063-6394a90aea61
34390-5
HUMAN OTC DRUG LABEL
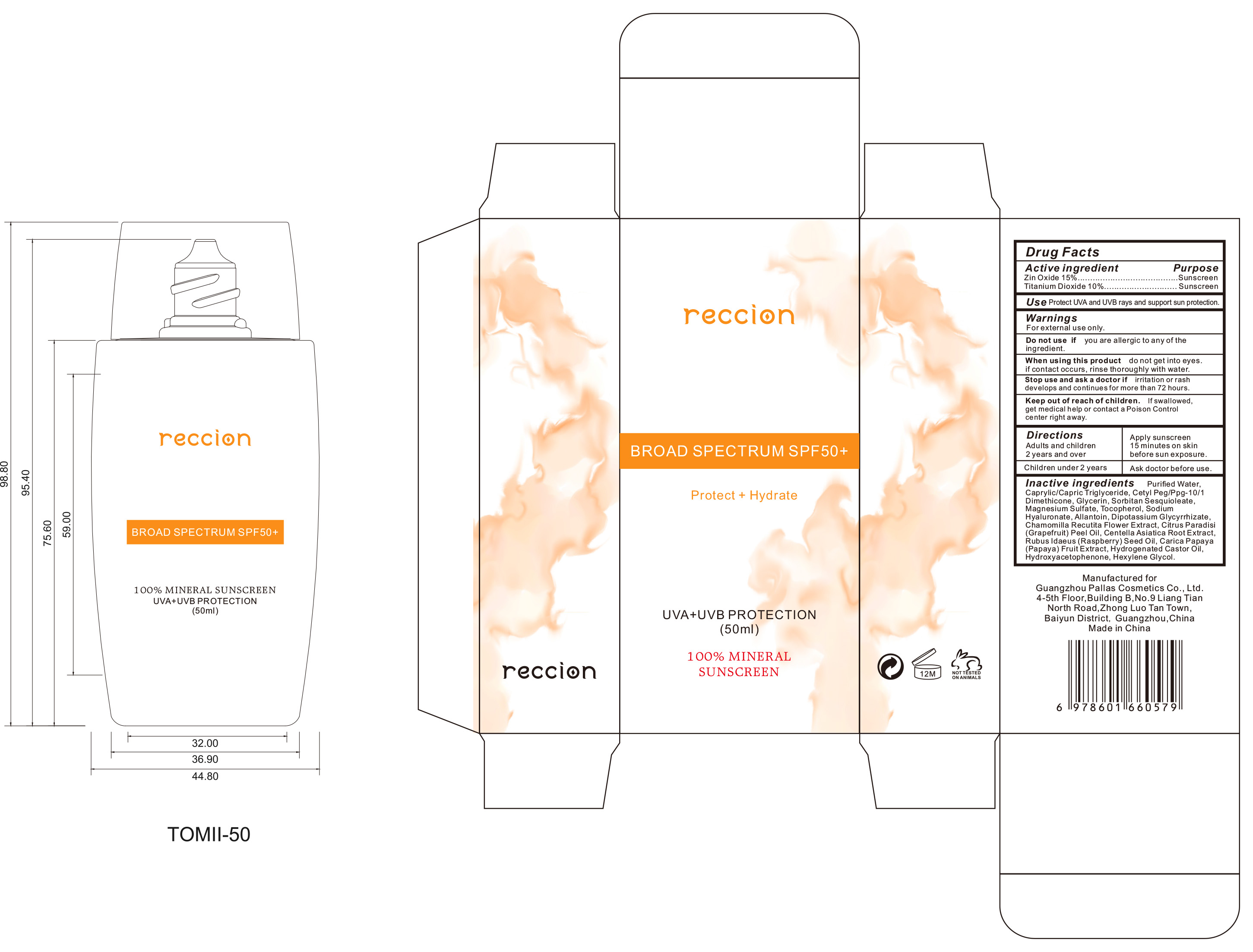
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Zin Oxide 1 5.% Titanium Dioxide 10%
Purpose
Sunscreen
Medication Information
Purpose
Sunscreen
Description
Zin Oxide 1 5.% Titanium Dioxide 10%
Use
Use Protect UVA and UVB rays and support sun protection.
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control
center right away.
Section 50566-9
Stop use and ask a doctor if irrita tion or rash develops and continues for more than 72 hours.
Section 50567-7
When using this product do not get into eyes. if contact occurs, rinse thoroughly with water.
Warnings
For external use only.
Directions
Adults and children 2 years and over:Apply sunscreen 15 minutes on skin before sun exposure.
Children under2 years: Ask doctor before use.
Do Not Use
DO not use if you are allergic to any of the ingredient.
Other Information
No
Active Ingredient(s)
Zin Oxide 1 5.%
Titanium Dioxide 10%
Inactive Ingredients
Purified Water, Caprylic/Capric Triglyceride, Cety| Peg/Ppg-10/1 Dimethicone, Glycerin, Sorbitan Sesquioleate,
Magnesium Sulfate, Tocopherol, Sodium Hyaluronate, Allantoin, Dipotassium Glycyrrhizate,
Chamomilla Recutita Flower E xtract, Citrus Paradisi (Grapefruit) Peel Oil, Centella Asiatica Root Extract,
Rubus ldaeus (Raspberry) Seed Oil, Carica Papaya (Papaya) Fruit Extract, Hydrogenated Castor Oil,
Hydroxyacetophenone, Hexylene Glycol.
Package Label Principal Display Panel
Structured Label Content
Use
Use Protect UVA and UVB rays and support sun protection.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control
center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if irrita tion or rash develops and continues for more than 72 hours.
Section 50567-7 (50567-7)
When using this product do not get into eyes. if contact occurs, rinse thoroughly with water.
Purpose
Sunscreen
Warnings
For external use only.
Directions
Adults and children 2 years and over:Apply sunscreen 15 minutes on skin before sun exposure.
Children under2 years: Ask doctor before use.
Do Not Use (Do not use)
DO not use if you are allergic to any of the ingredient.
Other Information (Other information)
No
Active Ingredient(s)
Zin Oxide 1 5.%
Titanium Dioxide 10%
Inactive Ingredients (Inactive ingredients)
Purified Water, Caprylic/Capric Triglyceride, Cety| Peg/Ppg-10/1 Dimethicone, Glycerin, Sorbitan Sesquioleate,
Magnesium Sulfate, Tocopherol, Sodium Hyaluronate, Allantoin, Dipotassium Glycyrrhizate,
Chamomilla Recutita Flower E xtract, Citrus Paradisi (Grapefruit) Peel Oil, Centella Asiatica Root Extract,
Rubus ldaeus (Raspberry) Seed Oil, Carica Papaya (Papaya) Fruit Extract, Hydrogenated Castor Oil,
Hydroxyacetophenone, Hexylene Glycol.
Package Label Principal Display Panel (Package Label - Principal Display Panel)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:47.870331 · Updated: 2026-03-14T23:09:45.619768