3c414e9b-6c40-2c8d-e063-6394a90af4a6
34390-5
HUMAN OTC DRUG LABEL
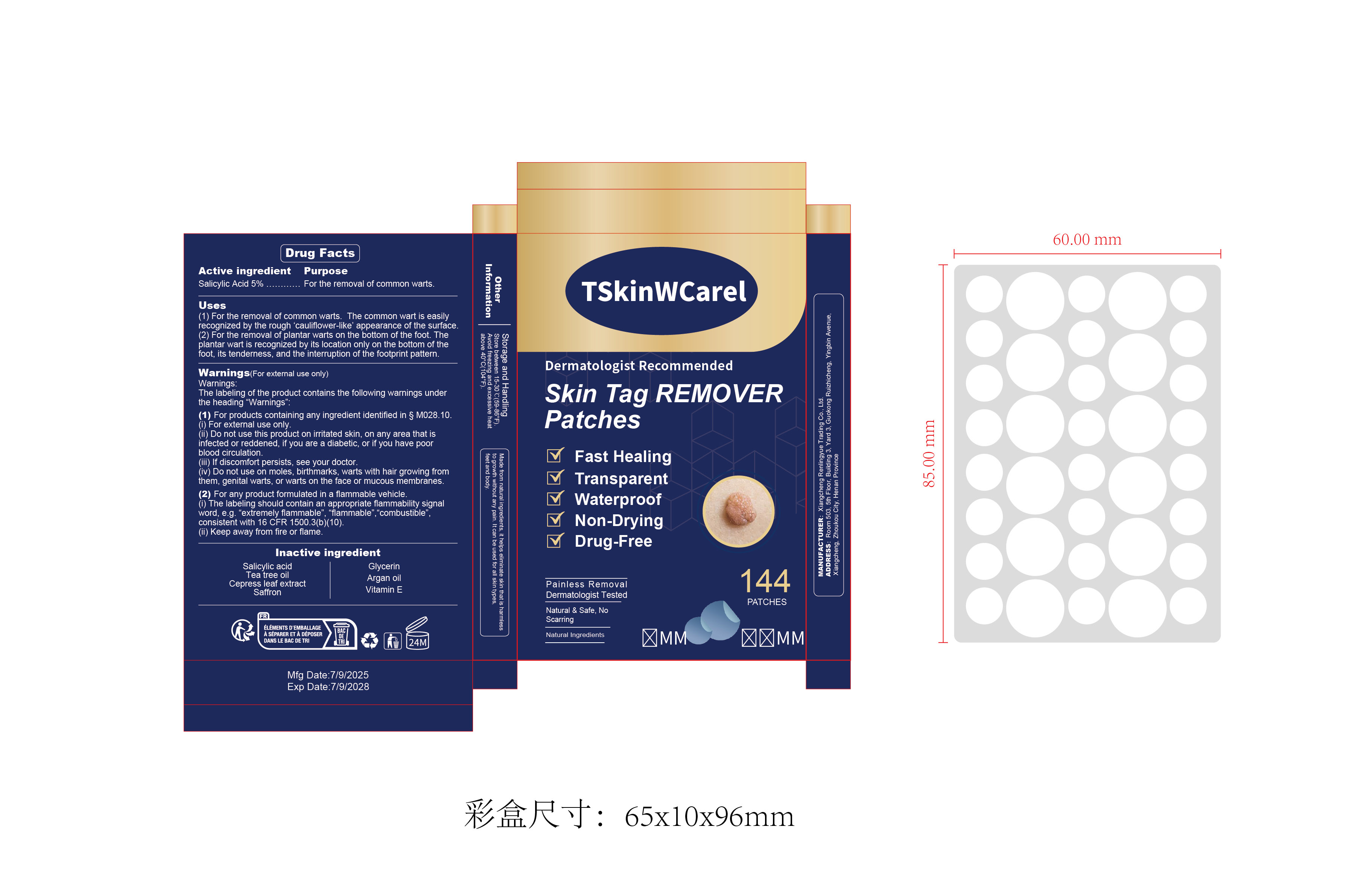
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Salicylic Acid 5% For the removal of common warts.
Medication Information
Warnings and Precautions
Warnings.The labeling of the product contains the following warnings underthe heading “Warnings”
(1)For products containing any ingredient identified in S MO28.10.(i)For external use only.(ii) Do not use this product on irritated skin, on any area that isinfected or reddened, if you are a diabetic, or if you have poorblood circulation.
(iii) lf discomfort persists, see your doctor.(iv) Do not use on moles, birthmarks, warts with hair growing fromthem, genital warts, or warts on the face or mucous membranes.
(2) For any product formulated in a flammable vehicle.(i)The labeling should contain an appropriate flammability signalword, e.g.“extremely flammable",“flammable".“combustible”consistent with 16 CFR 1500.3(b)(10).(ii) Keep away from fire or flame.
Indications and Usage
For the removal of plantar warts on the bottom of the foot. The
plantar wart is recognized by its location only on the bottom of the
foot, its tenderness, and the interruption of the footprint pattern.
Dosage and Administration
(1)For the removal of common warts. The common wart is easily
recognized by the rough 'cauliflower-like’ appearance of the surface.
Description
Salicylic Acid 5% For the removal of common warts.
Section 50565-1
KEEP OUT OF CHILDREN
Section 50570-1
Do not use this product on irritated skin, on any area that is
infected or reddened, if you are a diabetic, or if you have poor
blood circulation.
Section 51727-6
Salicylic acidTea tree oilCepress leaf extractSaffron
GlycerinArgan oilVitamin E
Section 51945-4
Section 55105-1
Fast HealingTransparentWaterproofNon-DryingDrug-Free
Section 55106-9
Salicylic Acid 5%
For the removal of common warts.
Structured Label Content
Indications and Usage (34067-9)
For the removal of plantar warts on the bottom of the foot. The
plantar wart is recognized by its location only on the bottom of the
foot, its tenderness, and the interruption of the footprint pattern.
Dosage and Administration (34068-7)
(1)For the removal of common warts. The common wart is easily
recognized by the rough 'cauliflower-like’ appearance of the surface.
Warnings and Precautions (34071-1)
Warnings.The labeling of the product contains the following warnings underthe heading “Warnings”
(1)For products containing any ingredient identified in S MO28.10.(i)For external use only.(ii) Do not use this product on irritated skin, on any area that isinfected or reddened, if you are a diabetic, or if you have poorblood circulation.
(iii) lf discomfort persists, see your doctor.(iv) Do not use on moles, birthmarks, warts with hair growing fromthem, genital warts, or warts on the face or mucous membranes.
(2) For any product formulated in a flammable vehicle.(i)The labeling should contain an appropriate flammability signalword, e.g.“extremely flammable",“flammable".“combustible”consistent with 16 CFR 1500.3(b)(10).(ii) Keep away from fire or flame.
Section 50565-1 (50565-1)
KEEP OUT OF CHILDREN
Section 50570-1 (50570-1)
Do not use this product on irritated skin, on any area that is
infected or reddened, if you are a diabetic, or if you have poor
blood circulation.
Section 51727-6 (51727-6)
Salicylic acidTea tree oilCepress leaf extractSaffron
GlycerinArgan oilVitamin E
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Fast HealingTransparentWaterproofNon-DryingDrug-Free
Section 55106-9 (55106-9)
Salicylic Acid 5%
For the removal of common warts.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:45.506534 · Updated: 2026-03-14T23:09:42.602872