These Highlights Do Not Include All The Information Needed To Use Tranylcypromine Tablets Safely And Effectively. See Full Prescribing Information For Tranylcypromine Tablets.
3c14a558-2df4-4d93-97dd-640dec6ee785
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: SUICIDAL THOUGHTS AND BEHAVIORS and HYPERTENSIVE CRISIS WITH SIGNIFICANT TYRAMINE USE See full prescribing information for complete boxed warning. • Increased risk of suicidal thoughts and behavior in pediatric and young adult patients taking antidepressants. Closely monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors. Tranylcypromine tablets are not approved for use in pediatric patients. ( 5.1 , 8.4 ) • Excessive consumption of foods or beverages with significant tyramine content or certain drugs can precipitate hypertensive crisis. Monitor blood pressure, allow for medication free intervals, and advise patients to avoid foods and beverages with high tyramine content. ( 5.2 , 7.1 , 7.2 )
Indications and Usage
Tranylcypromine tablets are indicated for the treatment of major depressive disorder (MDD) in adult patients who have not responded adequately to other antidepressants. Tranylcypromine tablets are not indicated for the initial treatment of MDD due to the potential for serious adverse reactions and drug interactions, and the need for dietary restrictions [see Contraindications (4) , Warnings and Precautions (5) , and Drug Interactions (7) ] .
Dosage and Administration
• Recommended daily dosage is 30 mg in divided doses ( 2.1 ) • If no adequate response, increase dosage in increments of 10 mg per day every 1 to 3 weeks to a maximum dosage of 30 mg twice daily (60 mg per day). Consider more gradual dosage increases in patients at risk for hypotension ( 2.1 ) • Consider discontinuing tranylcypromine tablets therapy gradually because of the risk for withdrawal effects ( 2.3 , 5.8 , 9.3 ) • Switching from or to other MAOIs or other antidepressants: See full prescribing information for instructions ( 2.2 , 7.1 )
Warnings and Precautions
• Activation of Mania/Hypomania : May be precipitated by antidepressant treatment in patients with bipolar disorder. Screen patients prior to treatment ( 5.4 ) • Hypotension (including syncope) : Monitor patients and adjust tranylcypromine tablets dosage or concomitant medication as necessary ( 5.5 ) • Hypotension and Hypertension during Anesthesia and Perioperative Care : If possible, discontinue tranylcypromine tablets prior to elective surgery ( 5.6 ) • Hepatitis and Elevated Liver Enzymes : Monitor accordingly ( 5.10 )
Contraindications
• Concomitant use or use in rapid succession with other MAOIs; selective serotonin reuptake inhibitors; serotonin and norepinephrine reuptake inhibitors; tricyclic antidepressants; sympathomimetic drugs; and numerous other drugs. See Full Prescribing Information for the full list of contraindicated products ( 4.1 , 7.1 ) • Pheochromocytoma, other catecholamine-releasing paraganglioma ( 4.2 )
Adverse Reactions
Although excretion of tranylcypromine tablets are rapid, inhibition of MAO may persist up to 10 days following discontinuation. This should be taken into account when considering the use of potentially interacting substances or the consumption of tyramine-rich food or beverages [see Drug Interactions (7.2) ] , or when interpreting adverse reactions observed after discontinuation of tranylcypromine tablets. Care should be taken to differentiate symptoms of persistent MAO inhibition from withdrawal symptoms [see Drug Abuse and Dependence (9.3) ] .
Drug Interactions
Tables 3 and 4 lists drug classes and individual products, respectively, with a potential for interaction with tranylcypromine tablets, describes the predominant observed or anticipated risks, and provides advice on concomitant use. Given serious adverse reactions with multiple agents, patients should avoid taking over-the-counter medications or dietary supplements without prior consultation with a healthcare provider able to provide advice on the potential for interactions.
Storage and Handling
Tranylcypromine Tablets, USP are available as round, red, film-coated tablets debossed with "10" on one side and plain on the other side, containing tranylcypromine sulfate equivalent to 10 mg of tranylcypromine. They are supplied in bottles of 100 tablets with a desiccant. • 10 mg, bottles of 100 tablets: NDC 43547-655-10
How Supplied
Tranylcypromine Tablets, USP are available as round, red, film-coated tablets debossed with "10" on one side and plain on the other side, containing tranylcypromine sulfate equivalent to 10 mg of tranylcypromine. They are supplied in bottles of 100 tablets with a desiccant. • 10 mg, bottles of 100 tablets: NDC 43547-655-10
Medication Information
Warnings and Precautions
• Activation of Mania/Hypomania : May be precipitated by antidepressant treatment in patients with bipolar disorder. Screen patients prior to treatment ( 5.4 ) • Hypotension (including syncope) : Monitor patients and adjust tranylcypromine tablets dosage or concomitant medication as necessary ( 5.5 ) • Hypotension and Hypertension during Anesthesia and Perioperative Care : If possible, discontinue tranylcypromine tablets prior to elective surgery ( 5.6 ) • Hepatitis and Elevated Liver Enzymes : Monitor accordingly ( 5.10 )
Indications and Usage
Tranylcypromine tablets are indicated for the treatment of major depressive disorder (MDD) in adult patients who have not responded adequately to other antidepressants. Tranylcypromine tablets are not indicated for the initial treatment of MDD due to the potential for serious adverse reactions and drug interactions, and the need for dietary restrictions [see Contraindications (4) , Warnings and Precautions (5) , and Drug Interactions (7) ] .
Dosage and Administration
• Recommended daily dosage is 30 mg in divided doses ( 2.1 ) • If no adequate response, increase dosage in increments of 10 mg per day every 1 to 3 weeks to a maximum dosage of 30 mg twice daily (60 mg per day). Consider more gradual dosage increases in patients at risk for hypotension ( 2.1 ) • Consider discontinuing tranylcypromine tablets therapy gradually because of the risk for withdrawal effects ( 2.3 , 5.8 , 9.3 ) • Switching from or to other MAOIs or other antidepressants: See full prescribing information for instructions ( 2.2 , 7.1 )
Contraindications
• Concomitant use or use in rapid succession with other MAOIs; selective serotonin reuptake inhibitors; serotonin and norepinephrine reuptake inhibitors; tricyclic antidepressants; sympathomimetic drugs; and numerous other drugs. See Full Prescribing Information for the full list of contraindicated products ( 4.1 , 7.1 ) • Pheochromocytoma, other catecholamine-releasing paraganglioma ( 4.2 )
Adverse Reactions
Although excretion of tranylcypromine tablets are rapid, inhibition of MAO may persist up to 10 days following discontinuation. This should be taken into account when considering the use of potentially interacting substances or the consumption of tyramine-rich food or beverages [see Drug Interactions (7.2) ] , or when interpreting adverse reactions observed after discontinuation of tranylcypromine tablets. Care should be taken to differentiate symptoms of persistent MAO inhibition from withdrawal symptoms [see Drug Abuse and Dependence (9.3) ] .
Drug Interactions
Tables 3 and 4 lists drug classes and individual products, respectively, with a potential for interaction with tranylcypromine tablets, describes the predominant observed or anticipated risks, and provides advice on concomitant use. Given serious adverse reactions with multiple agents, patients should avoid taking over-the-counter medications or dietary supplements without prior consultation with a healthcare provider able to provide advice on the potential for interactions.
Storage and Handling
Tranylcypromine Tablets, USP are available as round, red, film-coated tablets debossed with "10" on one side and plain on the other side, containing tranylcypromine sulfate equivalent to 10 mg of tranylcypromine. They are supplied in bottles of 100 tablets with a desiccant. • 10 mg, bottles of 100 tablets: NDC 43547-655-10
How Supplied
Tranylcypromine Tablets, USP are available as round, red, film-coated tablets debossed with "10" on one side and plain on the other side, containing tranylcypromine sulfate equivalent to 10 mg of tranylcypromine. They are supplied in bottles of 100 tablets with a desiccant. • 10 mg, bottles of 100 tablets: NDC 43547-655-10
Description
WARNING: SUICIDAL THOUGHTS AND BEHAVIORS and HYPERTENSIVE CRISIS WITH SIGNIFICANT TYRAMINE USE See full prescribing information for complete boxed warning. • Increased risk of suicidal thoughts and behavior in pediatric and young adult patients taking antidepressants. Closely monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors. Tranylcypromine tablets are not approved for use in pediatric patients. ( 5.1 , 8.4 ) • Excessive consumption of foods or beverages with significant tyramine content or certain drugs can precipitate hypertensive crisis. Monitor blood pressure, allow for medication free intervals, and advise patients to avoid foods and beverages with high tyramine content. ( 5.2 , 7.1 , 7.2 )
Section 42229-5
Suicidal Thoughts and Behaviors
Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and for emergence of suicidal thoughts and behaviors [see Warnings and Precautions (5.1)] . Tranylcypromine tablets are not approved for use in pediatric patients [see Use in Specific Populations (8.4)] .
Section 42231-1
|
||||||
|
||||||
|
|
|||||
|
||||||
|
|
|||||
|
A hypertensive crisis can also happen if you take tranylcypromine tablets with certain other medicines. See, " Who should not take tranylcypromine tablets?" |
||||||
|
||||||
|
||||||
|
Do not take tranylcypromine tablets if you:
Ask your healthcare provider or pharmacist if you are not sure if you take any of these medicines.
|
||||||
|
Before taking tranylcypromine tablets, tell your healthcare provider about all your medical conditions, including if you:
|
||||||
|
How should I take tranylcypromine tablets?
|
||||||
|
What should I avoid while taking tranylcypromine tablets?
|
||||||
|
Type of Food and Drink that contain Tyramine |
||||||
|
Meat, Poultry, and Fish |
|
|||||
|
Vegetables |
|
|||||
|
Dairy (milk products) |
|
|||||
|
Drinks |
|
|||||
|
Other |
|
|||||
|
||||||
|
||||||
|
|
|||||
|
If you have any of these symptoms, call your healthcare provider or go to the nearest hospital emergency room right away. |
||||||
|
||||||
|
|
|||||
|
||||||
|
|
|
||||
The most common side effects of tranylcypromine tablets include: |
||||||
|
|
|||||
|
||||||
|
How do I store tranylcypromine tablets?
Keep tranylcypromine tablets and all medicines out of the reach of children. |
||||||
For more information contact Solco Healthcare at 866-931-9829 or at www.solcohealthcare.com. |
||||||
Distributed by: This Medication Guide has been approved by the U.S. Food and Drug Administration L7152 Rev. 10/2022 |
||||||
Section 43683-2
Section 44425-7
Store between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature]. Dispense in a tight, light-resistant container.
9.2 Abuse
Abuse of tranylcypromine tablets has been reported. Some of these patients had a history of previous substance abuse.
The potential for abuse and the increased risk of serious adverse reactions with higher doses should be taken into account when considering the use of tranylcypromine tablets for patients at increased risk for substance abuse.
5.11 Seizures
Seizures have been reported with tranylcypromine tablets withdrawal after abuse, and with overdose. Patients at risk for seizures should be monitored accordingly.
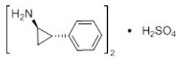
11 Description
Tranylcypromine sulfate, the active ingredient of Tranylcypromine Tablets, USP, is a non-hydrazine MAOI. The chemical name is (±)- trans-2-phenylcyclopropylamine sulfate (2:1). The molecular formula is (C 9H 11N) 2∙H 2SO 4 and its molecular weight is 364.46. The structural formula is:
Tranylcypromine film-coated tablets are intended for oral administration. Each round, red tablet is debossed on one side with "10" and plain on the other side, and contains tranylcypromine sulfate equivalent to 10 mg of tranylcypromine.
Inactive ingredients consist of microcrystalline cellulose, pregelatinized starch, colloidal silicon dioxide, magnesium stearate, polyvinyl alcohol, polyethylene glycol, talc, FD&C Red No. 40-Aluminum Lake, titanium dioxide, and carmine.
9.3 Dependence
Dependence, evidenced by precipitation of withdrawal effects following abrupt discontinuation of tranylcypromine tablets has been reported. Reported withdrawal effects included delirium (even with low daily doses), restlessness, anxiety, confusion, hallucinations, headache, weakness, diarrhea, and/or rapid relapse into depression. Thrombocytopenia and liver enzyme increases have also been observed in association with tranylcypromine tablets withdrawal from high doses [see Overdosage (10.1)]
Withdrawal effects have appeared within 1 to 3 days of discontinuation and have persisted for several weeks after discontinuation. The use of daily doses greater than recommended and longer duration of use appear to be associated with a higher risk of withdrawal effects.
Monitor for withdrawal effects for at least 1 week after discontinuation. Consider discontinuing tranylcypromine tablets therapy by slow, gradual dose reduction [see Dosage and Administration (2.3)] .
5.5 Hypotension
Hypotension, including postural hypotension, has been observed during therapy with tranylcypromine tablets. At doses above 30 mg daily, postural hypotension is a major adverse reaction and may result in syncope. Symptoms of postural hypotension are seen most commonly, but not exclusively, in patients with pre-existing hypertension. Blood pressure usually returns rapidly to pretreatment levels upon discontinuation of tranylcypromine tablets.
Dosage increases should be made more gradually in patients with a tendency toward hypotension and/or postural hypotension (e.g., elderly patients) [see Dosage and Administration (2.2) and Use in Specific Populations (8.5)] . Such patients should be closely observed for postural changes in blood pressure throughout treatment. Also, when tranylcypromine tablets are used concomitantly with other agents known to cause hypotension, the possibility of additive hypotensive effects should be considered [see Drug Interactions (7.1)] . Postural hypotension may be relieved by having patients lie down until blood pressure returns to normal.
8.4 Pediatric Use
Safety and effectiveness of tranylcypromine tablets in the pediatric population have not been established. All risks associated with the use of tranylcypromine tablets, including the risk of suicidal thoughts and behavior, apply to adults and pediatric patients [see Boxed Warning and Warnings and Precautions (5)] .
8.5 Geriatric Use
Older patients may be at greater risk of postural hypotension and other serious adverse reactions [see Warnings and Precautions (5)] . In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
4 Contraindications
-
•Concomitant use or use in rapid succession with other MAOIs; selective serotonin reuptake inhibitors; serotonin and norepinephrine reuptake inhibitors; tricyclic antidepressants; sympathomimetic drugs; and numerous other drugs. See Full Prescribing Information for the full list of contraindicated products ( 4.1, 7.1)
-
•Pheochromocytoma, other catecholamine-releasing paraganglioma ( 4.2)
5.10 Hepatotoxicity
Hepatitis and elevated aminotransferases have been reported in association with tranylcypromine tablets administration. Patients should be monitored accordingly. Tranylcypromine tablets should be discontinued in patients who develop signs and symptoms of hepatotoxicity.
Sedation has occurred in tranylcypromine tablets-treated patients with cirrhosis. Patients with cirrhosis receiving tranylcypromine tablets should be monitored for possible increased risks of central nervous system adverse reactions, such as excessive drowsiness.
6 Adverse Reactions
The following adverse reactions are described in greater detail in other sections:
-
•Suicidal thoughts and behaviors [see Warnings and Precautions (5.1)]
-
•Hypertensive crisis and hypertension [see Warnings and Precautions (5.2)]
-
•Serotonin syndrome [see Warnings and Precautions (5.3)]
-
•Activation of mania/hypomania [see Warnings and Precautions (5.4)]
-
•Hypotension [see Warnings and Precautions (5.5)]
-
•Hypotension and hypertension during anesthesia and perioperative care [see Warnings and Precautions (5.6)]
-
•Discontinuation syndrome [see Warnings and Precautions (5.8)]
-
•Persistence of MAO inhibition after discontinuation [see Warnings and Precautions (5.9)]
-
•Hepatotoxicity [see Warnings and Precautions (5.10)]
-
•Seizures [see Warnings and Precautions (5.11)]
-
•Hypoglycemia in diabetic patients [see Warnings and Precautions (5.12)]
-
•Aggravation of coexisting symptoms of depression [see Warnings and Precautions (5.13)]
-
•Adverse effects on the ability to drive and operate machinery [see Warnings and Precautions (5.14)]
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Based on clinical trial data, the most common adverse reactions to tranylcypromine were dry mouth, dizziness, insomnia, sedation, and headache (>30%) and overexcitement, constipation, blurred vision, and tremor (>10%).
The following adverse reactions have been identified in clinical trials or during postapproval use of tranylcypromine tablets:
Blood and lymphatic system disorders: agranulocytosis, leukopenia, thrombocytopenia, anemia
Endocrine disorders: impaired water excretion compatible with the syndrome of inappropriate secretion of antidiuretic hormone (SIADH)
Metabolism and nutrition disorders: significant anorexia, weight gain
Psychiatric disorders: excessive stimulation/overexcitement, manic symptoms/hypomania, agitation, insomnia, anxiety, confusion, disorientation, loss of libido
Nervous system disorders: dizziness, restlessness/akathisia, akinesia, ataxia, myoclonic jerks, tremor, hyperreflexia, muscle spasm, paresthesia, numbness, memory loss, sedation, drowsiness, dysgeusia, headaches (without blood pressure elevation)
Eye disorders: blurred vision, nystagmus
Ear and labyrinth disorders: tinnitus
Cardiac disorders: tachycardia, palpitations
Vascular disorders: hypertensive crisis, hypertension, hypotension (including postural hypotension with syncope)
Gastrointestinal disorders: diarrhea, constipation, nausea, abdominal pain, dry mouth, fissuring in corner of mouth
Hepatobiliary disorders: hepatitis, elevated aminotransferases
Skin and subcutaneous tissue disorders: localized scleroderma, flare-up of cystic acne, urticaria, rash, alopecia, sweating
Renal and urinary disorders: urinary retention, urinary incontinence, urinary frequency
Reproductive system and breast disorders: impotence, delayed ejaculation
General disorders and administration site conditions: edema, chills, weakness, fatigue/lethargy
7 Drug Interactions
See Full Prescribing Information for a list of products, foods and beverages that can interact with tranylcypromine tablets ( 7)
12.2 Pharmacodynamics
Although tranylcypromine is eliminated in 24 hours, recovery MAO activity takes up to 3 to 5 days [see Warnings and Precautions (5.9)] .
Bottle Label Oct 2022
2.1 Recommended Dosage
Tranylcypromine tablets are for oral use. The recommended dosage is 30 mg per day (in divided doses). If patients do not have an adequate response, increase the dosage in increments of 10 mg per day every 1 to 3 weeks to a maximum 30 mg twice daily (60 mg per day). Dosage increases should be made more gradually in patients at risk for hypotension (e.g., geriatric patients) [see Warnings and Precautions (5.5) ] .
5.3 Serotonin Syndrome
The development of a potentially life-threatening serotonin syndrome has been reported with MAOIs when used concomitantly with other serotonergic drugs. Such drugs include SSRIs, SNRIs, tricyclic antidepressants, triptans, fentanyl, lithium, tramadol, tryptophan, buspirone, St. John's wort, S-adenosyl-L-methionine (SAM-e), and other MAOIs used to treat nonpsychiatric disorders (such as linezolid or intravenous methylene blue).
Manifestations of the serotonin syndrome may include mental status changes (e.g., agitation, hallucinations, delirium, coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia; with possible rapid fluctuations of vital signs), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). Fatal outcome of serotonin syndrome has been reported, including in patients who had been treated with tranylcypromine tablets. In some cases of an interaction between tranylcypromine tablets and SSRIs or SNRIs, the features of the syndrome resembled neuroleptic malignant syndrome.
The concomitant use, or use in rapid succession, of tranylcypromine tablets with other serotonergic drugs is contraindicated. However, there may be circumstances when treatment with other serotonergic substances (such as linezolid or intravenous methylene blue) is necessary and cannot be delayed. In such cases, tranylcypromine tablets must be discontinued as soon as possible before initiating treatment with the other agent.
Treatment with tranylcypromine tablets and any concomitant serotonergic agents should be discontinued immediately if the above events occur, and supportive symptomatic treatment should be initiated.
1 Indications and Usage
Tranylcypromine tablets are indicated for the treatment of major depressive disorder (MDD) in adult patients who have not responded adequately to other antidepressants. Tranylcypromine tablets are not indicated for the initial treatment of MDD due to the potential for serious adverse reactions and drug interactions, and the need for dietary restrictions [see Contraindications (4), Warnings and Precautions (5), and Drug Interactions (7)] .
12.1 Mechanism of Action
The mechanism of action of tranylcypromine tablets as an antidepressant is not fully understood, but is presumed to be linked to potentiation of monoamine neurotransmitter activity in the central nervous system (CNS) resulting from its irreversible inhibition of the enzyme monoamine oxidase (MAO).
10.2 Overdosage Management
There are no specific antidotes for tranylcypromine tablets. For current information on the management of poisoning or overdosage, contact a poison control center at 1-800-222-1222.
Abrupt withdrawal of tranylcypromine tablets following overdosage can precipitate withdrawal symptoms, including delirium [see Warnings and Precautions (5.9) and Drug Abuse and Dependence (9.3)] .
Medical management should normally consist of general supportive measures, close observation of vital signs, and steps to counteract specific manifestations as they occur [see Warnings and Precautions (5)] .The toxic effects of tranylcypromine tablets may be delayed or prolonged following the last dose of the drug [ see Clinical Pharmacology (12.2)] . Therefore, the patient should be closely observed for at least 1 week.
Data on the dialyzability of tranylcypromine are lacking.
5 Warnings and Precautions
-
•Activation of Mania/Hypomania: May be precipitated by antidepressant treatment in patients with bipolar disorder. Screen patients prior to treatment ( 5.4)
-
•Hypotension (including syncope): Monitor patients and adjust tranylcypromine tablets dosage or concomitant medication as necessary ( 5.5)
-
•Hypotension and Hypertension during Anesthesia and Perioperative Care: If possible, discontinue tranylcypromine tablets prior to elective surgery ( 5.6)
-
•Hepatitis and Elevated Liver Enzymes: Monitor accordingly ( 5.10)
2 Dosage and Administration
-
•Recommended daily dosage is 30 mg in divided doses ( 2.1)
-
•If no adequate response, increase dosage in increments of 10 mg per day every 1 to 3 weeks to a maximum dosage of 30 mg twice daily (60 mg per day). Consider more gradual dosage increases in patients at risk for hypotension ( 2.1)
-
•Consider discontinuing tranylcypromine tablets therapy gradually because of the risk for withdrawal effects ( 2.3, 5.8, 9.3)
-
•Switching from or to other MAOIs or other antidepressants: See full prescribing information for instructions ( 2.2, 7.1)
2.3 Discontinuing Treatment
Withdrawal effects, including delirium, have been reported with abrupt discontinuation of tranylcypromine tablets therapy. Higher daily doses and longer duration of use appear to be associated with a higher risk of withdrawal effects. Consider discontinuing tranylcypromine tablets therapy by slow, gradual dosage reduction [see Warnings and Precautions (5.8) and Drug Abuse and Dependence (9.3)] .
3 Dosage Forms and Strengths
Tablets containing tranylcypromine sulfate equivalent to 10 mg tranylcypromine are round, red, film-coated, and debossed on one side with "10" and plain on the other side.
5.8 Discontinuation Syndrome
Abrupt discontinuation or dosage reduction of tranylcypromine tablets has been associated with the appearance of new symptoms that include dizziness, nausea, headache, irritability, insomnia, diarrhea, anxiety, fatigue, abnormal dreams, and hyperhidrosis. In general, discontinuation events occurred more frequently with longer duration of therapy.
There have been spontaneous reports of adverse reactions occurring upon discontinuation of MAOIs, particularly when abrupt, including dysphoric mood, irritability, agitation, dizziness, sensory disturbances (e.g. paresthesia, such as electric shock sensations), anxiety, confusion, headache, lethargy, emotional lability, insomnia, hypomania, tinnitus, and seizures. While these reactions are generally self-limiting, there have been reports of prolonged discontinuation symptoms.
Patients should be monitored for these symptoms when discontinuing treatment with tranylcypromine tablets. A gradual reduction in the dose rather than abrupt cessation is recommended whenever possible [see Dosage and Administration (2.3) and Adverse Reactions (6)] .
17 Patient Counseling Information
Advise the patient to read FDA-approved patient labeling (Medication Guide).
4.1 Combination With Certain Drugs
Concomitant use of tranylcypromine tablets or use in rapid succession with the products in Table 1 is contraindicated. Such use may cause severe or life-threatening reactions such as hypertensive crises or serotonin syndrome [see Drug Interactions (7.1) ] . Medication-free periods between administration of tranylcypromine tablets and contraindicated agents are recommended [see Dosage and Administration (2.2) and Drug Interactions (7.1) ] .
|
Drug Classes |
||
|
Non-selective H1 receptor antagonists |
||
|
Antidepressants including but not limited to:
|
||
|
Amphetamines and methylphenidates and derivatives |
||
|
Sympathomimetic products (e.g., cold, hay fever or weight-reducing products that contain vasoconstrictors such as pseudoephedrine, phenylephrine, and ephedrine; or dietary supplements that contain sympathomimetics) |
||
|
Triptans |
||
|
Individual Drugs (not included in the above classes) |
||
|
buspirone |
levodopa |
s-adenosyl-L-methionine (SAM-e) |
|
carbamazepine |
meperidine |
tapentadol |
|
cyclobenzaprine |
methyldopa |
tetrabenazine |
|
dextromethorphan |
milnacipran |
tryptophan |
|
dopamine |
rasagiline |
|
|
hydroxytryptophan |
reserpine |
16 How Supplied/storage and Handling
Tranylcypromine Tablets, USP are available as round, red, film-coated tablets debossed with "10" on one side and plain on the other side, containing tranylcypromine sulfate equivalent to 10 mg of tranylcypromine. They are supplied in bottles of 100 tablets with a desiccant.
-
•10 mg, bottles of 100 tablets: NDC 43547-655-10
5.4 Activation of Mania Or Hypomania
In patients with bipolar disorder, treating a depressive episode with tranylcypromine tablets or another antidepressant may precipitate a mixed/manic episode. Prior to initiating treatment with tranylcypromine tablets, screen patients for any personal or family history of bipolar disorder, mania, or hypomania.
5.12 Hypoglycemia in Diabetic Patients
Some MAOIs have contributed to hypoglycemic episodes in diabetic patients receiving insulin or other blood-glucose-lowering agents. Monitor blood glucose in patients receiving both tranylcypromine tablets and blood-glucose-lowering agents. A reduction of the dosage of such agents may be necessary [see Drug Interactions (7.1)].
7.2 Tyramine Containing Foods and Beverages
Tranylcypromine tablets inhibits intestinal MAO, which is responsible for the catabolism of tyramine in food and beverages. As a result of this inhibition, large amounts of tyramine may enter the systemic circulation and precipitate a sudden elevation in blood pressure or hypertensive crisis [see Warnings and Precautions (5.2)] . Instruct tranylcypromine tablets-treated patients to avoid foods and beverages with significant tyramine content during treatment with tranylcypromine tablets or within 2 weeks of stopping treatment (see Table 5 for a list of food and beverages containing significant amounts of tyramine).
| Class of Food or Beverage | Tyramine-Rich Foods and Beverages to Avoid | Acceptable Foods and Drinks, Containing No or Little Tyramine |
|---|---|---|
|
Meat, Poultry, and Fish |
Air dried, aged and fermented meats, sausages and salamis (including cacciatore, hard salami and mortadella); pickled herring; and any spoiled or improperly stored meat, poultry, and fish (e.g., foods that have undergone changes in coloration, odor, or become moldy); spoiled or improperly stored animal livers |
Fresh meat, poultry, and fish, including fresh processed meats (e.g., lunch meats, hot dogs, breakfast sausage, and cooked sliced ham) |
|
Vegetables |
Broad bean pods (fava bean pods) |
All other vegetables |
|
Dairy |
Aged cheeses |
Processed cheeses, mozzarella, ricotta cheese, cottage cheese, and yogurt |
|
Beverages |
All varieties of tap beer and beers that have not been pasteurized so as to allow for ongoing fermentation and excessive amounts of caffeine. |
Concomitant use of alcohol with tranylcypromine tablets is not recommended. (Bottled and canned beers and wines contain little or no tyramine.) |
|
Other |
Concentrated yeast extract (e.g., Marmite), sauerkraut, most soybean products (including soy sauce and tofu), OTC supplements containing tyramine, and chocolate |
Brewer's yeast, baker's yeast, soy milk, commercial chain restaurant pizzas prepared with cheeses low in tyramine |
7.1 Clinically Significant Drug Interactions
Tables 3 and 4 lists drug classes and individual products, respectively, with a potential for interaction with tranylcypromine tablets, describes the predominant observed or anticipated risks, and provides advice on concomitant use. Given serious adverse reactions with multiple agents, patients should avoid taking over-the-counter medications or dietary supplements without prior consultation with a healthcare provider able to provide advice on the potential for interactions.
5.13 Aggravation of Coexisting Symptoms of Depression
Tranylcypromine tablets may aggravate coexisting symptoms in depression, such as anxiety and agitation.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenesis, mutagenesis, or fertility impairment studies were conducted.
5.7 Need for Emergency Treatment With Contraindicated Drugs
If in the absence of therapeutic alternatives emergency treatment with a contraindicated product (e.g., linezolid, intravenous methylene blue, direct-acting sympathomimetic drugs such as epinephrine) becomes necessary and cannot be delayed, discontinue tranylcypromine tablets as soon as possible before initiating treatment with the other product and monitor closely for adverse reactions [see Drug Interactions (7.1)] .
10.1 Overdosage Symptoms, Signs, and Laboratory Abnormalities
Overdose of tranylcypromine tablets can cause the adverse reactions generally associated with tranylcypromine tablets administration [see Warnings and Precautions (5), Adverse Reactions (6) and Drug Interactions (7.1)] . However, these reactions may be more severe, including fatal reactions. Effects reported with overdosage of tranylcypromine tablets and/or other MAOIs include:
-
•Insomnia, restlessness, and anxiety, progressing in severe cases to agitation, mental confusion, and incoherence; delirium; seizures
-
•Hypotension, dizziness, weakness, and drowsiness, progressing in severe cases to extreme dizziness and shock
-
•Hypertension with severe headache and other symptoms/complications
-
•Twitching or myoclonic fibrillation of skeletal muscles, with hyperpyrexia, sometimes progressing to generalized rigidity and coma
4.2 Pheochromocytoma and Catecholamine Releasing Paragangliomas
Tranylcypromine tablets are contraindicated in the presence of pheochromocytoma or other catecholamine-releasing paragangliomas because such tumors secrete pressor substances and can lead to hypertensive crisis [see Warnings and Precautions (5.3)] .
5.14 Adverse Effects On the Ability to Drive and Operate Machinery
Some tranylcypromine tablets adverse reactions (e.g., hypotension, faintness, drowsiness, confusion, disorientation) can impair a patient's ability to operate machinery or use an automobile. Patients should be cautioned about operating hazardous machinery, including automobiles, until they are reasonably certain that tranylcypromine tablets therapy does not impair their ability to engage in such activities.
5.1 Suicidal Thoughts and Behaviors in Adolescents and Young Adults
In pooled analyses of placebo-controlled trials of antidepressant drugs (SSRIs and other antidepressant classes) that included approximately 77,000 adult patients and 4,500 pediatric patients, the incidence of suicidal thoughts and behaviors in antidepressant-treated patients age 24 years and younger was greater than in placebo-treated patients. There was considerable variation in risk of suicidal thoughts and behaviors among drugs, but there was an increased risk identified in young patients for most drugs studied. There were differences in absolute risk of suicidal thoughts and behaviors across the different indications, with the highest incidence in patients with MDD. The drug-placebo differences in the number of cases of suicidal thoughts and behaviors per 1000 patients treated are provided in Table 2.
| Age Range | Drug-Placebo Difference in Number of Patients of Suicidal Thoughts or Behaviors per 1000 Patients Treated |
|---|---|
|
Increases Compared to Placebo |
|
|
<18 years old |
14 additional patients |
|
18-24 years old |
5 additional patients |
|
Decreases Compared to Placebo |
|
|
25-64 years old |
1 fewer patient |
|
≥65 years old |
6 fewer patients |
It is unknown whether the risk of suicidal thoughts and behaviors in children, adolescents, and young adults extends to longer-term use, i.e., beyond four months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with MDD that antidepressants delay the recurrence of depression and that depression itself is a risk factor for suicidal thoughts and behaviors.
Monitor all antidepressant-treated patients for any indication for clinical worsening and emergence of suicidal thoughts and behaviors, especially during the initial few months of drug therapy, and at times of dosage changes. Counsel family members or caregivers of patients to monitor for changes in behavior and to alert the healthcare provider. Consider changing the therapeutic regimen, including possibly discontinuing tranylcypromine tablets, in patients whose depression is persistently worse, or who are experiencing emergent suicidal thoughts or behaviors.
5.6 Hypotension and Hypertension During Anesthesia and Perioperative Care
It is recommended that tranylcypromine tablets be discontinued at least 10 days prior to elective surgery. If this is not possible, for general anesthesia, regional and local anesthesia, and perioperative care avoid the use of agents that are contraindicated for concomitant use with tranylcypromine tablets. Carefully consider the risk of agents and techniques that increase the risk for hypotension (e.g., epidural or spinal anesthesia) or other adverse reactions to tranylcypromine tablets (e.g., hypertension associated with the use of vasoconstrictors in local anesthetics).
Warning: Suicidal Thoughts and Behaviors and Hypertensive Crisis With Significant Tyramine Use
WARNING: SUICIDAL THOUGHTS AND BEHAVIORS and HYPERTENSIVE CRISIS WITH SIGNIFICANT TYRAMINE USE
See full prescribing information for complete boxed warning.
-
•Increased risk of suicidal thoughts and behavior in pediatric and young adult patients taking antidepressants. Closely monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors. Tranylcypromine tablets are not approved for use in pediatric patients. ( 5.1, 8.4)
-
•Excessive consumption of foods or beverages with significant tyramine content or certain drugs can precipitate hypertensive crisis. Monitor blood pressure, allow for medication free intervals, and advise patients to avoid foods and beverages with high tyramine content. ( 5.2, 7.1, 7.2)
2.4 Screen for Bipolar Disorder and Elevated Blood Pressure Prior to Starting Tranylcypromine Tablets
Prior to initiating treatment with tranylcypromine tablets:
-
•Screen patients for a history of mania [see Warnings and Precautions (5.4)] .
-
•Measure blood pressure [see Warnings and Precautions (5.2, 5.5)] .
5.9 Risk of Clinically Significant Adverse Reactions Due to Persistence of Mao Inhibition After Discontinuation
Although excretion of tranylcypromine tablets are rapid, inhibition of MAO may persist up to 10 days following discontinuation. This should be taken into account when considering the use of potentially interacting substances or the consumption of tyramine-rich food or beverages [see Drug Interactions (7.2)] , or when interpreting adverse reactions observed after discontinuation of tranylcypromine tablets. Care should be taken to differentiate symptoms of persistent MAO inhibition from withdrawal symptoms [see Drug Abuse and Dependence (9.3)] .
Structured Label Content
Section 42229-5 (42229-5)
Suicidal Thoughts and Behaviors
Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and for emergence of suicidal thoughts and behaviors [see Warnings and Precautions (5.1)] . Tranylcypromine tablets are not approved for use in pediatric patients [see Use in Specific Populations (8.4)] .
Section 42231-1 (42231-1)
|
||||||
|
||||||
|
|
|||||
|
||||||
|
|
|||||
|
A hypertensive crisis can also happen if you take tranylcypromine tablets with certain other medicines. See, " Who should not take tranylcypromine tablets?" |
||||||
|
||||||
|
||||||
|
Do not take tranylcypromine tablets if you:
Ask your healthcare provider or pharmacist if you are not sure if you take any of these medicines.
|
||||||
|
Before taking tranylcypromine tablets, tell your healthcare provider about all your medical conditions, including if you:
|
||||||
|
How should I take tranylcypromine tablets?
|
||||||
|
What should I avoid while taking tranylcypromine tablets?
|
||||||
|
Type of Food and Drink that contain Tyramine |
||||||
|
Meat, Poultry, and Fish |
|
|||||
|
Vegetables |
|
|||||
|
Dairy (milk products) |
|
|||||
|
Drinks |
|
|||||
|
Other |
|
|||||
|
||||||
|
||||||
|
|
|||||
|
If you have any of these symptoms, call your healthcare provider or go to the nearest hospital emergency room right away. |
||||||
|
||||||
|
|
|||||
|
||||||
|
|
|
||||
The most common side effects of tranylcypromine tablets include: |
||||||
|
|
|||||
|
||||||
|
How do I store tranylcypromine tablets?
Keep tranylcypromine tablets and all medicines out of the reach of children. |
||||||
For more information contact Solco Healthcare at 866-931-9829 or at www.solcohealthcare.com. |
||||||
Distributed by: This Medication Guide has been approved by the U.S. Food and Drug Administration L7152 Rev. 10/2022 |
||||||
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature]. Dispense in a tight, light-resistant container.
9.2 Abuse
Abuse of tranylcypromine tablets has been reported. Some of these patients had a history of previous substance abuse.
The potential for abuse and the increased risk of serious adverse reactions with higher doses should be taken into account when considering the use of tranylcypromine tablets for patients at increased risk for substance abuse.
5.11 Seizures
Seizures have been reported with tranylcypromine tablets withdrawal after abuse, and with overdose. Patients at risk for seizures should be monitored accordingly.
11 Description (11 DESCRIPTION)
Tranylcypromine sulfate, the active ingredient of Tranylcypromine Tablets, USP, is a non-hydrazine MAOI. The chemical name is (±)- trans-2-phenylcyclopropylamine sulfate (2:1). The molecular formula is (C 9H 11N) 2∙H 2SO 4 and its molecular weight is 364.46. The structural formula is:
Tranylcypromine film-coated tablets are intended for oral administration. Each round, red tablet is debossed on one side with "10" and plain on the other side, and contains tranylcypromine sulfate equivalent to 10 mg of tranylcypromine.
Inactive ingredients consist of microcrystalline cellulose, pregelatinized starch, colloidal silicon dioxide, magnesium stearate, polyvinyl alcohol, polyethylene glycol, talc, FD&C Red No. 40-Aluminum Lake, titanium dioxide, and carmine.
9.3 Dependence
Dependence, evidenced by precipitation of withdrawal effects following abrupt discontinuation of tranylcypromine tablets has been reported. Reported withdrawal effects included delirium (even with low daily doses), restlessness, anxiety, confusion, hallucinations, headache, weakness, diarrhea, and/or rapid relapse into depression. Thrombocytopenia and liver enzyme increases have also been observed in association with tranylcypromine tablets withdrawal from high doses [see Overdosage (10.1)]
Withdrawal effects have appeared within 1 to 3 days of discontinuation and have persisted for several weeks after discontinuation. The use of daily doses greater than recommended and longer duration of use appear to be associated with a higher risk of withdrawal effects.
Monitor for withdrawal effects for at least 1 week after discontinuation. Consider discontinuing tranylcypromine tablets therapy by slow, gradual dose reduction [see Dosage and Administration (2.3)] .
5.5 Hypotension
Hypotension, including postural hypotension, has been observed during therapy with tranylcypromine tablets. At doses above 30 mg daily, postural hypotension is a major adverse reaction and may result in syncope. Symptoms of postural hypotension are seen most commonly, but not exclusively, in patients with pre-existing hypertension. Blood pressure usually returns rapidly to pretreatment levels upon discontinuation of tranylcypromine tablets.
Dosage increases should be made more gradually in patients with a tendency toward hypotension and/or postural hypotension (e.g., elderly patients) [see Dosage and Administration (2.2) and Use in Specific Populations (8.5)] . Such patients should be closely observed for postural changes in blood pressure throughout treatment. Also, when tranylcypromine tablets are used concomitantly with other agents known to cause hypotension, the possibility of additive hypotensive effects should be considered [see Drug Interactions (7.1)] . Postural hypotension may be relieved by having patients lie down until blood pressure returns to normal.
8.4 Pediatric Use
Safety and effectiveness of tranylcypromine tablets in the pediatric population have not been established. All risks associated with the use of tranylcypromine tablets, including the risk of suicidal thoughts and behavior, apply to adults and pediatric patients [see Boxed Warning and Warnings and Precautions (5)] .
8.5 Geriatric Use
Older patients may be at greater risk of postural hypotension and other serious adverse reactions [see Warnings and Precautions (5)] . In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
4 Contraindications (4 CONTRAINDICATIONS)
-
•Concomitant use or use in rapid succession with other MAOIs; selective serotonin reuptake inhibitors; serotonin and norepinephrine reuptake inhibitors; tricyclic antidepressants; sympathomimetic drugs; and numerous other drugs. See Full Prescribing Information for the full list of contraindicated products ( 4.1, 7.1)
-
•Pheochromocytoma, other catecholamine-releasing paraganglioma ( 4.2)
5.10 Hepatotoxicity
Hepatitis and elevated aminotransferases have been reported in association with tranylcypromine tablets administration. Patients should be monitored accordingly. Tranylcypromine tablets should be discontinued in patients who develop signs and symptoms of hepatotoxicity.
Sedation has occurred in tranylcypromine tablets-treated patients with cirrhosis. Patients with cirrhosis receiving tranylcypromine tablets should be monitored for possible increased risks of central nervous system adverse reactions, such as excessive drowsiness.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described in greater detail in other sections:
-
•Suicidal thoughts and behaviors [see Warnings and Precautions (5.1)]
-
•Hypertensive crisis and hypertension [see Warnings and Precautions (5.2)]
-
•Serotonin syndrome [see Warnings and Precautions (5.3)]
-
•Activation of mania/hypomania [see Warnings and Precautions (5.4)]
-
•Hypotension [see Warnings and Precautions (5.5)]
-
•Hypotension and hypertension during anesthesia and perioperative care [see Warnings and Precautions (5.6)]
-
•Discontinuation syndrome [see Warnings and Precautions (5.8)]
-
•Persistence of MAO inhibition after discontinuation [see Warnings and Precautions (5.9)]
-
•Hepatotoxicity [see Warnings and Precautions (5.10)]
-
•Seizures [see Warnings and Precautions (5.11)]
-
•Hypoglycemia in diabetic patients [see Warnings and Precautions (5.12)]
-
•Aggravation of coexisting symptoms of depression [see Warnings and Precautions (5.13)]
-
•Adverse effects on the ability to drive and operate machinery [see Warnings and Precautions (5.14)]
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Based on clinical trial data, the most common adverse reactions to tranylcypromine were dry mouth, dizziness, insomnia, sedation, and headache (>30%) and overexcitement, constipation, blurred vision, and tremor (>10%).
The following adverse reactions have been identified in clinical trials or during postapproval use of tranylcypromine tablets:
Blood and lymphatic system disorders: agranulocytosis, leukopenia, thrombocytopenia, anemia
Endocrine disorders: impaired water excretion compatible with the syndrome of inappropriate secretion of antidiuretic hormone (SIADH)
Metabolism and nutrition disorders: significant anorexia, weight gain
Psychiatric disorders: excessive stimulation/overexcitement, manic symptoms/hypomania, agitation, insomnia, anxiety, confusion, disorientation, loss of libido
Nervous system disorders: dizziness, restlessness/akathisia, akinesia, ataxia, myoclonic jerks, tremor, hyperreflexia, muscle spasm, paresthesia, numbness, memory loss, sedation, drowsiness, dysgeusia, headaches (without blood pressure elevation)
Eye disorders: blurred vision, nystagmus
Ear and labyrinth disorders: tinnitus
Cardiac disorders: tachycardia, palpitations
Vascular disorders: hypertensive crisis, hypertension, hypotension (including postural hypotension with syncope)
Gastrointestinal disorders: diarrhea, constipation, nausea, abdominal pain, dry mouth, fissuring in corner of mouth
Hepatobiliary disorders: hepatitis, elevated aminotransferases
Skin and subcutaneous tissue disorders: localized scleroderma, flare-up of cystic acne, urticaria, rash, alopecia, sweating
Renal and urinary disorders: urinary retention, urinary incontinence, urinary frequency
Reproductive system and breast disorders: impotence, delayed ejaculation
General disorders and administration site conditions: edema, chills, weakness, fatigue/lethargy
7 Drug Interactions (7 DRUG INTERACTIONS)
See Full Prescribing Information for a list of products, foods and beverages that can interact with tranylcypromine tablets ( 7)
12.2 Pharmacodynamics
Although tranylcypromine is eliminated in 24 hours, recovery MAO activity takes up to 3 to 5 days [see Warnings and Precautions (5.9)] .
Bottle Label Oct 2022
2.1 Recommended Dosage
Tranylcypromine tablets are for oral use. The recommended dosage is 30 mg per day (in divided doses). If patients do not have an adequate response, increase the dosage in increments of 10 mg per day every 1 to 3 weeks to a maximum 30 mg twice daily (60 mg per day). Dosage increases should be made more gradually in patients at risk for hypotension (e.g., geriatric patients) [see Warnings and Precautions (5.5) ] .
5.3 Serotonin Syndrome
The development of a potentially life-threatening serotonin syndrome has been reported with MAOIs when used concomitantly with other serotonergic drugs. Such drugs include SSRIs, SNRIs, tricyclic antidepressants, triptans, fentanyl, lithium, tramadol, tryptophan, buspirone, St. John's wort, S-adenosyl-L-methionine (SAM-e), and other MAOIs used to treat nonpsychiatric disorders (such as linezolid or intravenous methylene blue).
Manifestations of the serotonin syndrome may include mental status changes (e.g., agitation, hallucinations, delirium, coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia; with possible rapid fluctuations of vital signs), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). Fatal outcome of serotonin syndrome has been reported, including in patients who had been treated with tranylcypromine tablets. In some cases of an interaction between tranylcypromine tablets and SSRIs or SNRIs, the features of the syndrome resembled neuroleptic malignant syndrome.
The concomitant use, or use in rapid succession, of tranylcypromine tablets with other serotonergic drugs is contraindicated. However, there may be circumstances when treatment with other serotonergic substances (such as linezolid or intravenous methylene blue) is necessary and cannot be delayed. In such cases, tranylcypromine tablets must be discontinued as soon as possible before initiating treatment with the other agent.
Treatment with tranylcypromine tablets and any concomitant serotonergic agents should be discontinued immediately if the above events occur, and supportive symptomatic treatment should be initiated.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Tranylcypromine tablets are indicated for the treatment of major depressive disorder (MDD) in adult patients who have not responded adequately to other antidepressants. Tranylcypromine tablets are not indicated for the initial treatment of MDD due to the potential for serious adverse reactions and drug interactions, and the need for dietary restrictions [see Contraindications (4), Warnings and Precautions (5), and Drug Interactions (7)] .
12.1 Mechanism of Action
The mechanism of action of tranylcypromine tablets as an antidepressant is not fully understood, but is presumed to be linked to potentiation of monoamine neurotransmitter activity in the central nervous system (CNS) resulting from its irreversible inhibition of the enzyme monoamine oxidase (MAO).
10.2 Overdosage Management
There are no specific antidotes for tranylcypromine tablets. For current information on the management of poisoning or overdosage, contact a poison control center at 1-800-222-1222.
Abrupt withdrawal of tranylcypromine tablets following overdosage can precipitate withdrawal symptoms, including delirium [see Warnings and Precautions (5.9) and Drug Abuse and Dependence (9.3)] .
Medical management should normally consist of general supportive measures, close observation of vital signs, and steps to counteract specific manifestations as they occur [see Warnings and Precautions (5)] .The toxic effects of tranylcypromine tablets may be delayed or prolonged following the last dose of the drug [ see Clinical Pharmacology (12.2)] . Therefore, the patient should be closely observed for at least 1 week.
Data on the dialyzability of tranylcypromine are lacking.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Activation of Mania/Hypomania: May be precipitated by antidepressant treatment in patients with bipolar disorder. Screen patients prior to treatment ( 5.4)
-
•Hypotension (including syncope): Monitor patients and adjust tranylcypromine tablets dosage or concomitant medication as necessary ( 5.5)
-
•Hypotension and Hypertension during Anesthesia and Perioperative Care: If possible, discontinue tranylcypromine tablets prior to elective surgery ( 5.6)
-
•Hepatitis and Elevated Liver Enzymes: Monitor accordingly ( 5.10)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Recommended daily dosage is 30 mg in divided doses ( 2.1)
-
•If no adequate response, increase dosage in increments of 10 mg per day every 1 to 3 weeks to a maximum dosage of 30 mg twice daily (60 mg per day). Consider more gradual dosage increases in patients at risk for hypotension ( 2.1)
-
•Consider discontinuing tranylcypromine tablets therapy gradually because of the risk for withdrawal effects ( 2.3, 5.8, 9.3)
-
•Switching from or to other MAOIs or other antidepressants: See full prescribing information for instructions ( 2.2, 7.1)
2.3 Discontinuing Treatment
Withdrawal effects, including delirium, have been reported with abrupt discontinuation of tranylcypromine tablets therapy. Higher daily doses and longer duration of use appear to be associated with a higher risk of withdrawal effects. Consider discontinuing tranylcypromine tablets therapy by slow, gradual dosage reduction [see Warnings and Precautions (5.8) and Drug Abuse and Dependence (9.3)] .
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets containing tranylcypromine sulfate equivalent to 10 mg tranylcypromine are round, red, film-coated, and debossed on one side with "10" and plain on the other side.
5.8 Discontinuation Syndrome
Abrupt discontinuation or dosage reduction of tranylcypromine tablets has been associated with the appearance of new symptoms that include dizziness, nausea, headache, irritability, insomnia, diarrhea, anxiety, fatigue, abnormal dreams, and hyperhidrosis. In general, discontinuation events occurred more frequently with longer duration of therapy.
There have been spontaneous reports of adverse reactions occurring upon discontinuation of MAOIs, particularly when abrupt, including dysphoric mood, irritability, agitation, dizziness, sensory disturbances (e.g. paresthesia, such as electric shock sensations), anxiety, confusion, headache, lethargy, emotional lability, insomnia, hypomania, tinnitus, and seizures. While these reactions are generally self-limiting, there have been reports of prolonged discontinuation symptoms.
Patients should be monitored for these symptoms when discontinuing treatment with tranylcypromine tablets. A gradual reduction in the dose rather than abrupt cessation is recommended whenever possible [see Dosage and Administration (2.3) and Adverse Reactions (6)] .
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read FDA-approved patient labeling (Medication Guide).
4.1 Combination With Certain Drugs (4.1 Combination with Certain Drugs)
Concomitant use of tranylcypromine tablets or use in rapid succession with the products in Table 1 is contraindicated. Such use may cause severe or life-threatening reactions such as hypertensive crises or serotonin syndrome [see Drug Interactions (7.1) ] . Medication-free periods between administration of tranylcypromine tablets and contraindicated agents are recommended [see Dosage and Administration (2.2) and Drug Interactions (7.1) ] .
|
Drug Classes |
||
|
Non-selective H1 receptor antagonists |
||
|
Antidepressants including but not limited to:
|
||
|
Amphetamines and methylphenidates and derivatives |
||
|
Sympathomimetic products (e.g., cold, hay fever or weight-reducing products that contain vasoconstrictors such as pseudoephedrine, phenylephrine, and ephedrine; or dietary supplements that contain sympathomimetics) |
||
|
Triptans |
||
|
Individual Drugs (not included in the above classes) |
||
|
buspirone |
levodopa |
s-adenosyl-L-methionine (SAM-e) |
|
carbamazepine |
meperidine |
tapentadol |
|
cyclobenzaprine |
methyldopa |
tetrabenazine |
|
dextromethorphan |
milnacipran |
tryptophan |
|
dopamine |
rasagiline |
|
|
hydroxytryptophan |
reserpine |
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Tranylcypromine Tablets, USP are available as round, red, film-coated tablets debossed with "10" on one side and plain on the other side, containing tranylcypromine sulfate equivalent to 10 mg of tranylcypromine. They are supplied in bottles of 100 tablets with a desiccant.
-
•10 mg, bottles of 100 tablets: NDC 43547-655-10
5.4 Activation of Mania Or Hypomania (5.4 Activation of Mania or Hypomania)
In patients with bipolar disorder, treating a depressive episode with tranylcypromine tablets or another antidepressant may precipitate a mixed/manic episode. Prior to initiating treatment with tranylcypromine tablets, screen patients for any personal or family history of bipolar disorder, mania, or hypomania.
5.12 Hypoglycemia in Diabetic Patients
Some MAOIs have contributed to hypoglycemic episodes in diabetic patients receiving insulin or other blood-glucose-lowering agents. Monitor blood glucose in patients receiving both tranylcypromine tablets and blood-glucose-lowering agents. A reduction of the dosage of such agents may be necessary [see Drug Interactions (7.1)].
7.2 Tyramine Containing Foods and Beverages (7.2 Tyramine-Containing Foods and Beverages)
Tranylcypromine tablets inhibits intestinal MAO, which is responsible for the catabolism of tyramine in food and beverages. As a result of this inhibition, large amounts of tyramine may enter the systemic circulation and precipitate a sudden elevation in blood pressure or hypertensive crisis [see Warnings and Precautions (5.2)] . Instruct tranylcypromine tablets-treated patients to avoid foods and beverages with significant tyramine content during treatment with tranylcypromine tablets or within 2 weeks of stopping treatment (see Table 5 for a list of food and beverages containing significant amounts of tyramine).
| Class of Food or Beverage | Tyramine-Rich Foods and Beverages to Avoid | Acceptable Foods and Drinks, Containing No or Little Tyramine |
|---|---|---|
|
Meat, Poultry, and Fish |
Air dried, aged and fermented meats, sausages and salamis (including cacciatore, hard salami and mortadella); pickled herring; and any spoiled or improperly stored meat, poultry, and fish (e.g., foods that have undergone changes in coloration, odor, or become moldy); spoiled or improperly stored animal livers |
Fresh meat, poultry, and fish, including fresh processed meats (e.g., lunch meats, hot dogs, breakfast sausage, and cooked sliced ham) |
|
Vegetables |
Broad bean pods (fava bean pods) |
All other vegetables |
|
Dairy |
Aged cheeses |
Processed cheeses, mozzarella, ricotta cheese, cottage cheese, and yogurt |
|
Beverages |
All varieties of tap beer and beers that have not been pasteurized so as to allow for ongoing fermentation and excessive amounts of caffeine. |
Concomitant use of alcohol with tranylcypromine tablets is not recommended. (Bottled and canned beers and wines contain little or no tyramine.) |
|
Other |
Concentrated yeast extract (e.g., Marmite), sauerkraut, most soybean products (including soy sauce and tofu), OTC supplements containing tyramine, and chocolate |
Brewer's yeast, baker's yeast, soy milk, commercial chain restaurant pizzas prepared with cheeses low in tyramine |
7.1 Clinically Significant Drug Interactions (7.1 Clinically-Significant Drug Interactions)
Tables 3 and 4 lists drug classes and individual products, respectively, with a potential for interaction with tranylcypromine tablets, describes the predominant observed or anticipated risks, and provides advice on concomitant use. Given serious adverse reactions with multiple agents, patients should avoid taking over-the-counter medications or dietary supplements without prior consultation with a healthcare provider able to provide advice on the potential for interactions.
5.13 Aggravation of Coexisting Symptoms of Depression
Tranylcypromine tablets may aggravate coexisting symptoms in depression, such as anxiety and agitation.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenesis, mutagenesis, or fertility impairment studies were conducted.
5.7 Need for Emergency Treatment With Contraindicated Drugs (5.7 Need for Emergency Treatment with Contraindicated Drugs)
If in the absence of therapeutic alternatives emergency treatment with a contraindicated product (e.g., linezolid, intravenous methylene blue, direct-acting sympathomimetic drugs such as epinephrine) becomes necessary and cannot be delayed, discontinue tranylcypromine tablets as soon as possible before initiating treatment with the other product and monitor closely for adverse reactions [see Drug Interactions (7.1)] .
10.1 Overdosage Symptoms, Signs, and Laboratory Abnormalities
Overdose of tranylcypromine tablets can cause the adverse reactions generally associated with tranylcypromine tablets administration [see Warnings and Precautions (5), Adverse Reactions (6) and Drug Interactions (7.1)] . However, these reactions may be more severe, including fatal reactions. Effects reported with overdosage of tranylcypromine tablets and/or other MAOIs include:
-
•Insomnia, restlessness, and anxiety, progressing in severe cases to agitation, mental confusion, and incoherence; delirium; seizures
-
•Hypotension, dizziness, weakness, and drowsiness, progressing in severe cases to extreme dizziness and shock
-
•Hypertension with severe headache and other symptoms/complications
-
•Twitching or myoclonic fibrillation of skeletal muscles, with hyperpyrexia, sometimes progressing to generalized rigidity and coma
4.2 Pheochromocytoma and Catecholamine Releasing Paragangliomas (4.2 Pheochromocytoma and Catecholamine-Releasing Paragangliomas)
Tranylcypromine tablets are contraindicated in the presence of pheochromocytoma or other catecholamine-releasing paragangliomas because such tumors secrete pressor substances and can lead to hypertensive crisis [see Warnings and Precautions (5.3)] .
5.14 Adverse Effects On the Ability to Drive and Operate Machinery (5.14 Adverse Effects on the Ability to Drive and Operate Machinery)
Some tranylcypromine tablets adverse reactions (e.g., hypotension, faintness, drowsiness, confusion, disorientation) can impair a patient's ability to operate machinery or use an automobile. Patients should be cautioned about operating hazardous machinery, including automobiles, until they are reasonably certain that tranylcypromine tablets therapy does not impair their ability to engage in such activities.
5.1 Suicidal Thoughts and Behaviors in Adolescents and Young Adults
In pooled analyses of placebo-controlled trials of antidepressant drugs (SSRIs and other antidepressant classes) that included approximately 77,000 adult patients and 4,500 pediatric patients, the incidence of suicidal thoughts and behaviors in antidepressant-treated patients age 24 years and younger was greater than in placebo-treated patients. There was considerable variation in risk of suicidal thoughts and behaviors among drugs, but there was an increased risk identified in young patients for most drugs studied. There were differences in absolute risk of suicidal thoughts and behaviors across the different indications, with the highest incidence in patients with MDD. The drug-placebo differences in the number of cases of suicidal thoughts and behaviors per 1000 patients treated are provided in Table 2.
| Age Range | Drug-Placebo Difference in Number of Patients of Suicidal Thoughts or Behaviors per 1000 Patients Treated |
|---|---|
|
Increases Compared to Placebo |
|
|
<18 years old |
14 additional patients |
|
18-24 years old |
5 additional patients |
|
Decreases Compared to Placebo |
|
|
25-64 years old |
1 fewer patient |
|
≥65 years old |
6 fewer patients |
It is unknown whether the risk of suicidal thoughts and behaviors in children, adolescents, and young adults extends to longer-term use, i.e., beyond four months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with MDD that antidepressants delay the recurrence of depression and that depression itself is a risk factor for suicidal thoughts and behaviors.
Monitor all antidepressant-treated patients for any indication for clinical worsening and emergence of suicidal thoughts and behaviors, especially during the initial few months of drug therapy, and at times of dosage changes. Counsel family members or caregivers of patients to monitor for changes in behavior and to alert the healthcare provider. Consider changing the therapeutic regimen, including possibly discontinuing tranylcypromine tablets, in patients whose depression is persistently worse, or who are experiencing emergent suicidal thoughts or behaviors.
5.6 Hypotension and Hypertension During Anesthesia and Perioperative Care (5.6 Hypotension and Hypertension during Anesthesia and Perioperative Care)
It is recommended that tranylcypromine tablets be discontinued at least 10 days prior to elective surgery. If this is not possible, for general anesthesia, regional and local anesthesia, and perioperative care avoid the use of agents that are contraindicated for concomitant use with tranylcypromine tablets. Carefully consider the risk of agents and techniques that increase the risk for hypotension (e.g., epidural or spinal anesthesia) or other adverse reactions to tranylcypromine tablets (e.g., hypertension associated with the use of vasoconstrictors in local anesthetics).
Warning: Suicidal Thoughts and Behaviors and Hypertensive Crisis With Significant Tyramine Use (WARNING: SUICIDAL THOUGHTS AND BEHAVIORS and HYPERTENSIVE CRISIS WITH SIGNIFICANT TYRAMINE USE)
WARNING: SUICIDAL THOUGHTS AND BEHAVIORS and HYPERTENSIVE CRISIS WITH SIGNIFICANT TYRAMINE USE
See full prescribing information for complete boxed warning.
-
•Increased risk of suicidal thoughts and behavior in pediatric and young adult patients taking antidepressants. Closely monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors. Tranylcypromine tablets are not approved for use in pediatric patients. ( 5.1, 8.4)
-
•Excessive consumption of foods or beverages with significant tyramine content or certain drugs can precipitate hypertensive crisis. Monitor blood pressure, allow for medication free intervals, and advise patients to avoid foods and beverages with high tyramine content. ( 5.2, 7.1, 7.2)
2.4 Screen for Bipolar Disorder and Elevated Blood Pressure Prior to Starting Tranylcypromine Tablets
Prior to initiating treatment with tranylcypromine tablets:
-
•Screen patients for a history of mania [see Warnings and Precautions (5.4)] .
-
•Measure blood pressure [see Warnings and Precautions (5.2, 5.5)] .
5.9 Risk of Clinically Significant Adverse Reactions Due to Persistence of Mao Inhibition After Discontinuation (5.9 Risk of Clinically Significant Adverse Reactions due to Persistence of MAO Inhibition after Discontinuation)
Although excretion of tranylcypromine tablets are rapid, inhibition of MAO may persist up to 10 days following discontinuation. This should be taken into account when considering the use of potentially interacting substances or the consumption of tyramine-rich food or beverages [see Drug Interactions (7.2)] , or when interpreting adverse reactions observed after discontinuation of tranylcypromine tablets. Care should be taken to differentiate symptoms of persistent MAO inhibition from withdrawal symptoms [see Drug Abuse and Dependence (9.3)] .
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:44:23.102143 · Updated: 2026-03-14T22:19:49.692010