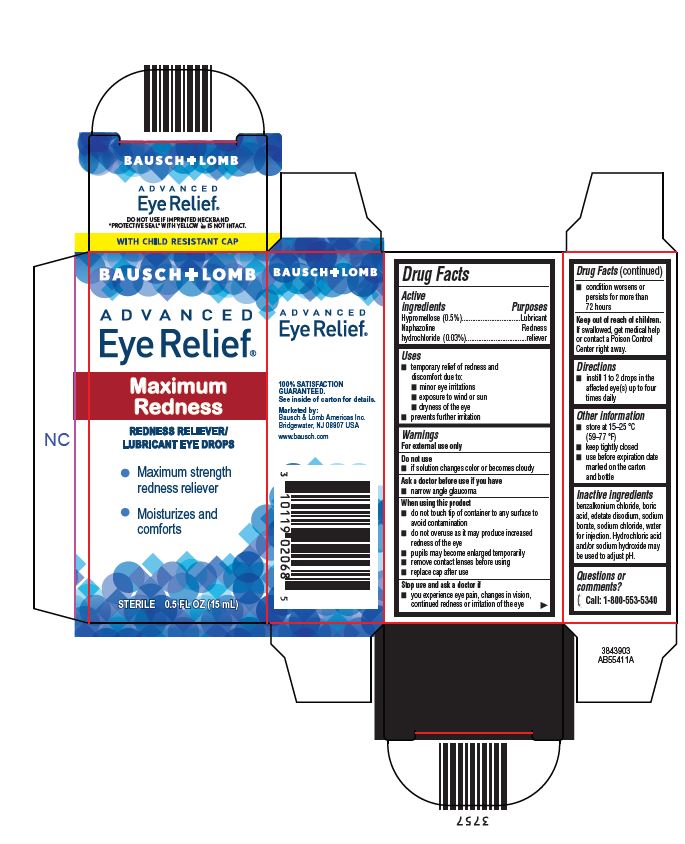
Drug Facts
3c11285a-7e86-4b47-b1ee-e7d59858087d
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Hypromellose (0.5%) Naphazoline hydrochloride (0.03%)
Purpose
Lubricant Redness reliever
Medication Information
Purpose
Lubricant
Redness reliever
Description
Hypromellose (0.5%) Naphazoline hydrochloride (0.03%)
Uses
- temporary relief of redness and discomfort due to:
- minor eye irritations
- exposure to wind or sun
- dryness of the eye
- prevents further irritation
Warnings
For external use only
Directions
- instill 1 to 2 drops in the affected eye(s) up to four times daily
Do Not Use
- if solution changes color or becomes cloudy
Other Information
- store at 15–25 °C (59–77 °F)
- keep tightly closed
- use before expiration date marked on the carton and bottle
Active Ingredients
Hypromellose (0.5%)
Naphazoline hydrochloride (0.03%)
Inactive Ingredients
benzalkonium chloride, boric acid, edetate disodium, sodium borate, sodium chloride, water for injection. Hydrochloric acid and/or sodium hydroxide may be used to adjust pH.
Questions Or Comments?
[phone icon] Call: 1-800-553-5340
When Using This Product
- do not touch tip of container to any surface to avoid contamination
- do not overuse as it may produce increased redness of the eye
- pupils may become enlarged temporarily
- remove contact lenses before using
- replace cap after use
Stop Use and Ask A Doctor If
- you experience eye pain, changes in vision, continued redness or irritation of the eye
- condition worsens or persists for more than 72 hours
Keep Out of Reach of Children
If swallowed, get medical help or contact a Poison Control Center right away.
Ask A Doctor Before Use If You Have
- narrow angle glaucoma
Package/label Principal Display Panel
BAUSCH + LOMB
ADVANCED
Eye Relief
®
Maximum
Redness
REDNESS RELIEVER/
LUBRICANT EYE DROPS
- Maximum strength
redness reliever - Moisturizes and
comforts
STERILE 0.5 FL OZ (15 mL)
3843903
AB55411A
Structured Label Content
Uses
- temporary relief of redness and discomfort due to:
- minor eye irritations
- exposure to wind or sun
- dryness of the eye
- prevents further irritation
Purpose
Lubricant
Redness reliever
Warnings
For external use only
Directions
- instill 1 to 2 drops in the affected eye(s) up to four times daily
Do Not Use (Do not use)
- if solution changes color or becomes cloudy
Other Information (Other information)
- store at 15–25 °C (59–77 °F)
- keep tightly closed
- use before expiration date marked on the carton and bottle
Active Ingredients (Active ingredients)
Hypromellose (0.5%)
Naphazoline hydrochloride (0.03%)
Inactive Ingredients (Inactive ingredients)
benzalkonium chloride, boric acid, edetate disodium, sodium borate, sodium chloride, water for injection. Hydrochloric acid and/or sodium hydroxide may be used to adjust pH.
Questions Or Comments? (Questions or comments?)
[phone icon] Call: 1-800-553-5340
When Using This Product (When using this product)
- do not touch tip of container to any surface to avoid contamination
- do not overuse as it may produce increased redness of the eye
- pupils may become enlarged temporarily
- remove contact lenses before using
- replace cap after use
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- you experience eye pain, changes in vision, continued redness or irritation of the eye
- condition worsens or persists for more than 72 hours
Keep Out of Reach of Children (Keep out of reach of children)
If swallowed, get medical help or contact a Poison Control Center right away.
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- narrow angle glaucoma
Package/label Principal Display Panel (Package/Label Principal Display Panel)
BAUSCH + LOMB
ADVANCED
Eye Relief
®
Maximum
Redness
REDNESS RELIEVER/
LUBRICANT EYE DROPS
- Maximum strength
redness reliever - Moisturizes and
comforts
STERILE 0.5 FL OZ (15 mL)
3843903
AB55411A
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:37.492535 · Updated: 2026-03-14T23:03:48.236290