These Highlights Do Not Include All The Information Needed To Use Flyrcado™ Safely And Effectively. See Full Prescribing Information For Flyrcado.
3bb6caf8-ac98-46c7-a9f0-961e66032705
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
FLYRCADO is indicated for positron emission tomography (PET) myocardial perfusion imaging (MPI) under rest or stress (pharmacologic or exercise) in adult patients with known or suspected coronary artery disease (CAD) to evaluate for myocardial ischemia and infarction.
Indications and Usage
FLYRCADO is indicated for positron emission tomography (PET) myocardial perfusion imaging (MPI) under rest or stress (pharmacologic or exercise) in adult patients with known or suspected coronary artery disease (CAD) to evaluate for myocardial ischemia and infarction.
Dosage and Administration
Administer FLYRCADO via intravenous injection. ( 2.2 ) When rest and stress imaging are conducted on the same day, the recommended administered activities are ( 2.2 ): Rest imaging: 93 MBq to 111 MBq (2.5 mCi to 3 mCi) Pharmacologic stress imaging: 222 MBq to 241 MBq (6 mCi to 6.5 mCi) Exercise stress imaging: 333 MBq to 352 MBq (9 mCi to 9.5 mCi) When rest and stress imaging are conducted over two days, the recommended rest and stress administered activities, for both pharmacologic and exercise stress, are 93 MBq to 111 MBq (2.5 mCi to 3 mCi). ( 2.2 ) See full prescribing information for radiation safety, preparation, administration, imaging, and radiation dosimetry information. ( 2.1 , 2.2 , 2.3 , 2.4 )
Warnings and Precautions
Risk associated with exercise or pharmacologic stress: Serious adverse reactions such as myocardial infarction, arrhythmia, hypotension, broncho-constriction, stroke, and seizures may occur. Perform stress testing in the setting where cardiac resuscitation equipment and trained staff are readily available. When pharmacologic stress is selected, perform the procedure in accordance with the pharmacologic stress agent's prescribing information. ( 5.1 ) Radiation risks: Ensure safe handling to minimize radiation exposure to patients and health care providers. ( 5.2 )
Contraindications
None.
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Risks Associated with Exercise or Pharmacologic Stress [see Warnings and Precautions (5.1) ]
Medication Information
Warnings and Precautions
Risk associated with exercise or pharmacologic stress: Serious adverse reactions such as myocardial infarction, arrhythmia, hypotension, broncho-constriction, stroke, and seizures may occur. Perform stress testing in the setting where cardiac resuscitation equipment and trained staff are readily available. When pharmacologic stress is selected, perform the procedure in accordance with the pharmacologic stress agent's prescribing information. ( 5.1 ) Radiation risks: Ensure safe handling to minimize radiation exposure to patients and health care providers. ( 5.2 )
Indications and Usage
FLYRCADO is indicated for positron emission tomography (PET) myocardial perfusion imaging (MPI) under rest or stress (pharmacologic or exercise) in adult patients with known or suspected coronary artery disease (CAD) to evaluate for myocardial ischemia and infarction.
Dosage and Administration
Administer FLYRCADO via intravenous injection. ( 2.2 ) When rest and stress imaging are conducted on the same day, the recommended administered activities are ( 2.2 ): Rest imaging: 93 MBq to 111 MBq (2.5 mCi to 3 mCi) Pharmacologic stress imaging: 222 MBq to 241 MBq (6 mCi to 6.5 mCi) Exercise stress imaging: 333 MBq to 352 MBq (9 mCi to 9.5 mCi) When rest and stress imaging are conducted over two days, the recommended rest and stress administered activities, for both pharmacologic and exercise stress, are 93 MBq to 111 MBq (2.5 mCi to 3 mCi). ( 2.2 ) See full prescribing information for radiation safety, preparation, administration, imaging, and radiation dosimetry information. ( 2.1 , 2.2 , 2.3 , 2.4 )
Contraindications
None.
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Risks Associated with Exercise or Pharmacologic Stress [see Warnings and Precautions (5.1) ]
Description
FLYRCADO is indicated for positron emission tomography (PET) myocardial perfusion imaging (MPI) under rest or stress (pharmacologic or exercise) in adult patients with known or suspected coronary artery disease (CAD) to evaluate for myocardial ischemia and infarction.
Section 42229-5
Recommended Dosage
- The recommended activity for each type of examination is presented in Table 1.
- Administer two doses by intravenous injection, one for rest imaging and one for stress imaging, using either pharmacologic or exercise stress, in either a 1-day or 2-day protocol.
- If performing a combined exercise/pharmacologic stress protocol, administer the recommended activity for pharmacologic stress.
- When rest and stress imaging are performed on the same day, the recommended minimum stress activity is 2-fold the rest activity for pharmacologic stress and 3-fold the rest activity for exercise stress to provide adequate image quality and obscure residual signal from the first acquisition.
- The recommended maximum total volume administered in one day is 6.1 mL.
| Length of Protocol | Activity for Rest Imaging | Activity for Stress Imaging | |
|---|---|---|---|
| Pharmacologic | Exercise | ||
| 1 day | 93 MBq to 111 MBq (2.5 mCi to 3 mCi) |
222 MBq to 241 MBq (6 mCi to 6.5 mCi) |
333 MBq to 352 MBq (9 mCi to 9.5 mCi) |
| 2 days | 93 MBq to 111 MBq (2.5 mCi to 3 mCi) |
93 MBq to 111 MBq (2.5 mCi to 3 mCi) |
93 MBq to 111 MBq (2.5 mCi to 3 mCi) |
Section 44425-7
Storage and Handling
Store FLYRCADO at 2°C to 30°C (36°F to 86°F); excursions to -20°C (-4°F) (up to 2 hours) or to 50°C (122°F) (up to 8 hours) may be permitted. Store FLYRCADO within radiation shielding. The product does not contain a preservative.
Do not use and discard FLYRCADO 8 hours after end of synthesis or when the activity falls below the radioactivity requirement at the time of injection, whichever is earlier. The expiration date and time are provided on the shield label.
Dispose of the product in accordance with all federal, state, and local laws and institutional requirements.
This preparation is for use by persons licensed by the Nuclear Regulatory Commission or the relevant regulatory authority of an Agreement State.
8.4 Pediatric Use
Safety and effectiveness of FLYRCADO in pediatric patients have not been established.
8.5 Geriatric Use
Of 1,600 subjects in clinical studies of FLYRCADO, 720 (45%) were 65 years of age and over and 181 (11%) were 75 years of age or older. No overall differences in safety or effectiveness of FLYRCADO have been observed between patients 65 years of age and older and younger adult patients.
4 Contraindications
None.
5.2 Radiation Risks
FLYRCADO contributes to a patient's overall long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk of cancer. Ensure safe handling to minimize radiation exposure to patients and health care providers [see Dosage and Administration (2.1, 2.4)]. Advise patients to hydrate before and after administration and to void frequently after administration.
6 Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling:
- Risks Associated with Exercise or Pharmacologic Stress [see Warnings and Precautions (5.1)]
12.2 Pharmacodynamics
The relationship between flurpiridaz F 18 plasma concentrations and successful imaging was not explored in clinical trials.
12.3 Pharmacokinetics
The pharmacokinetics of flurpiridaz F 18 were evaluated in healthy subjects. Blood radioactivity peaked at 2.3 minutes with blood clearance followed by a rise and plateau at around 3% of the 296 MBq (8 mCi) flurpiridaz F 18 intravenous dose until 7 hours post administration. The fluorine-18 radioactivity in blood during the first 15-minutes post administration was associated with flurpiridaz while it was associated with flurpiridaz metabolites thereafter.
1 Indications and Usage
FLYRCADO is indicated for positron emission tomography (PET) myocardial perfusion imaging (MPI) under rest or stress (pharmacologic or exercise) in adult patients with known or suspected coronary artery disease (CAD) to evaluate for myocardial ischemia and infarction.
11.3 External Radiation
The point source air-kerma rate constant for fluorine-18 is 3.74E-17 Gy m2/(Bq s); this coefficient was formerly defined as the specific gamma-ray constant of 5.7 R/hr/mCi at 1 cm. The first half-value thickness of lead (Pb) for fluorine-18 gamma rays is approximately 6 mm. The relative reduction of radiation emitted by fluorine-18 that results from various thicknesses of lead shielding is shown in Table 5. The use of about 8 cm of Pb will decrease the radiation transmission (i.e., exposure) by a factor of about 10,000.
| Shielding Thickness cm of Lead (Pb) | Coefficient of Attenuation |
|---|---|
| 0.6 | 0.5 |
| 2 | 0.1 |
| 4 | 0.01 |
| 6 | 0.001 |
| 8 | 0.0001 |
2.4 Radiation Dosimetry
Table 2 shows the estimated radiation absorbed doses per unit of injected activity.
| Organ | Absorbed Dose per Unit of Administered Activity (mGy/MBq) |
||
|---|---|---|---|
| Rest | Pharmacologic Stress The pharmacologic stress agent used was adenosine.
|
Exercise Stress | |
| Adrenals | 0.016 | 0.016 | 0.014 |
| Bone surfaces | 0.019 | 0.019 | 0.02 |
| Brain | 0.025 | 0.022 | 0.011 |
| Breasts | 0.009 | 0.009 | 0.01 |
| Gallbladder Wall | 0.017 | 0.018 | 0.015 |
| Gastrointestinal Tract | |||
| Stomach Wall | 0.04 | 0.033 | 0.024 |
| Small Intestine Wall | 0.013 | 0.012 | 0.014 |
| Upper Large Intestine Wall | 0.013 | 0.012 | 0.014 |
| Lower Large Intestine Wall | 0.012 | 0.011 | 0.014 |
| Heart Wall | 0.048 | 0.09 | 0.039 |
| Kidneys | 0.066 | 0.057 | 0.027 |
| Liver | 0.039 | 0.044 | 0.015 |
| Lungs | 0.011 | 0.012 | 0.012 |
| Muscle | 0.01 | 0.01 | 0.012 |
| Ovaries | 0.012 | 0.012 | 0.014 |
| Pancreas | 0.016 | 0.016 | 0.015 |
| Red Marrow | 0.016 | 0.018 | 0.015 |
| Salivary Glands | 0.035 | 0.076 | 0.007 |
| Skin | 0.008 | 0.008 | 0.009 |
| Spleen | 0.016 | 0.012 | 0.013 |
| Testes | 0.009 | 0.009 | 0.011 |
| Thymus | 0.011 | 0.012 | 0.013 |
| Thyroid | 0.032 | 0.036 | 0.014 |
| Urinary Bladder Wall | 0.023 | 0.021 | 0.016 |
| Uterus | 0.012 | 0.012 | 0.014 |
| Total Body | 0.012 | 0.012 | 0.012 |
| Effective Dose (mSv/MBq) | 0.019 | 0.019 | 0.015 |
The whole-body effective dose resulting from the administration of maximal activity of FLYRCADO of 111 MBq at rest, 241 MBq during pharmacological stress, and 352 MBq during exercise stress is, respectively, 2.1 mSv, 4.6 mSv, and 5.3 mSv. Under the same conditions, the absorbed dose to the target organ (heart wall) is 5.3 mGy, 22 mGy, and 14 mGy for each administered activity, respectively.
The use of a CT scan to calculate attenuation correction for the reconstruction of FLYRCADO PET images (as done in PET/CT imaging) will add radiation exposure.
12.1 Mechanism of Action
Flurpiridaz F 18 is an analog of the mitochondrial complex 1 (MC-1) inhibitor, pyridaben. Flurpiridaz F 18 is extracted by the myocardium proportional to the blood flow and binds to heart tissue that has biologically active mitochondria. Therefore, radioactivity in viable myocardium is higher than in infarcted tissue.
5 Warnings and Precautions
- Risk associated with exercise or pharmacologic stress: Serious adverse reactions such as myocardial infarction, arrhythmia, hypotension, broncho-constriction, stroke, and seizures may occur. Perform stress testing in the setting where cardiac resuscitation equipment and trained staff are readily available. When pharmacologic stress is selected, perform the procedure in accordance with the pharmacologic stress agent's prescribing information. (5.1)
- Radiation risks: Ensure safe handling to minimize radiation exposure to patients and health care providers. (5.2)
2 Dosage and Administration
- Administer FLYRCADO via intravenous injection. (2.2)
- When rest and stress imaging are conducted on the same day, the recommended administered activities are (2.2):
- Rest imaging: 93 MBq to 111 MBq (2.5 mCi to 3 mCi)
- Pharmacologic stress imaging: 222 MBq to 241 MBq (6 mCi to 6.5 mCi)
- Exercise stress imaging: 333 MBq to 352 MBq (9 mCi to 9.5 mCi)
- When rest and stress imaging are conducted over two days, the recommended rest and stress administered activities, for both pharmacologic and exercise stress, are 93 MBq to 111 MBq (2.5 mCi to 3 mCi). (2.2)
- See full prescribing information for radiation safety, preparation, administration, imaging, and radiation dosimetry information. (2.1, 2.2, 2.3, 2.4)
3 Dosage Forms and Strengths
Injection: 190 MBq/mL to 2,050 MBq/mL (5 mCi/mL to 55 mCi/mL) of flurpiridaz F 18 at end of synthesis as a clear, colorless to yellow solution in a shielded multiple-dose vial with up to 30 mL fill volume.
11.1 Chemical Characteristics
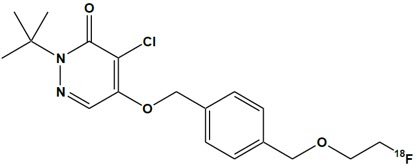
FLYRCADO (flurpiridaz F 18) injection is a radioactive diagnostic drug for intravenous use. The molecular formula of flurpiridaz F 18 is C18H22Cl18FN2O3, the molecular mass is 367.8, and the structural formula is:
Chemically, flurpiridaz F 18 is 2-tert-butyl-4-chloro-5-[[4-(2-(18F)fluoranylethoxymethyl)phenyl]methoxy]pyridazin-3-one.
FLYRCADO is a sterile, preservative-free, non-pyrogenic, clear, colorless to yellow radioactive solution. Each mL contains 190 MBq to 2,050 MBq (5 mCi to 55 mCi) of flurpiridaz F 18 at end of synthesis, up to 2.3 mcg flurpiridaz, and the following inactive ingredients: 45 mg hydroxypropyl-β-cyclodextrin (as a solubilizer and co-radiostabilizer), 35 mg L-(+)-ascorbic acid (as a radiostabilizer), 8.2 mg sodium hydroxide, and 55.2 mg anhydrous ethanol, in water for injection. The pH of the solution is between 5.5 and 8.
11.2 Physical Characteristics
Fluorine-18 decays by positron (β+) emission and has a half-life of 109.8 minutes. The principal photons useful for diagnostic imaging are the 511 keV gamma photons, resulting from the interaction of the emitted positron with an electron. Principal emission data for fluorine-18 are shown in Table 4.
| Radiation/Emission | % per Disintegration | Mean Energy (keV) |
|---|---|---|
| Positron | 96.7 | 249.8 |
| Gamma | 193.5 | 511 |
8 Use in Specific Populations
Lactation: Temporarily discontinue breastfeeding. A lactating woman should pump and discard breastmilk for at least 8 hours after FLYRCADO administration. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of FLYRCADO was evaluated in 1,600 subjects in clinical studies, including 1,575 (98%) subjects with known or suspected coronary artery disease and 25 (2%) healthy subjects. All 1,600 subjects were dosed under rest conditions, with a mean dose of 102 MBq (2.8 mCi) FLYRCADO. A total of 1,568 (98%) subjects were also dosed under stress (exercise or pharmacologic) conditions, with a mean activity of 252 MBq (6.8 mCi) FLYRCADO by intravenous route. The demographic characteristics of the study population were 31% female, mean age 62 years (range 19 years to 90 years), 81% White, 11% Black or African American, 1% Asian, and 7% other or unreported race, and 10% Hispanic or Latino, 81% Not Hispanic or Latino, and 9% unreported ethnicity.
Stress testing procedures are associated with serious adverse reactions [see Warnings and Precautions (5.1)]. Adverse reactions occurring in ≥2% subjects receiving FLYRCADO during PET MPI under rest and stress (pharmacologic or exercise) are presented in Table 3.
| Adverse Reaction | FLYRCADO PET MPI Under Rest and Stress (Pharmacologic or Exercise) N=1,600 Includes 32 subjects who received only one dose at rest.
% |
|---|---|
| Dyspnea | 17 |
| Headache | 15 |
| Angina pectoris | 10 |
| Chest pain | 8 |
| Fatigue | 7 |
| ST segment changes | 6 |
| Flushing | 5 |
| Nausea | 4 |
| Abdominal pain | 4 |
| Dizziness | 4 |
| Arrhythmia | 4 |
Adverse reactions occurring during FLYRCADO PET MPI under rest and stress (pharmacologic or exercise) in <2% of subjects included diarrhea, palpitations, back pain, cardiac conduction disturbance, rash, dysgeusia, cough, hypotension, anxiety, vomiting, pruritus, bronchospasm, dry mouth, blood pressure elevation, syncope, and wheezing.
14.1 Overview of Clinical Studies
The safety and effectiveness of FLYRCADO were evaluated in two prospective, multicenter, open-label clinical studies in adults with either suspected coronary artery disease (CAD) (Study 1: NCT03354273) or known or suspected CAD (Study 2: NCT01347710).
Subjects received two injections of FLYRCADO: one at rest and one during stress [see Dosage and Administration (2.2, 2.3)]. For the FLYRCADO stress injection, subjects received either a pharmacologic stress agent or engaged in exercise stress. PET myocardial perfusion imaging (MPI) was performed at both rest and stress using cardiac gating and low-dose CT attenuation correction. Subjects also received rest and stress SPECT MPI using technetium Tc 99m sestamibi or technetium Tc 99m tetrofosmin on a different day from the PET MPI. The stress modality was to be the same for PET and SPECT.
Stress and rest images were displayed side-by-side for the assessment of perfusion and wall motion abnormalities. Three qualified readers, blinded to clinical data, performed independent assessment of each subject's rest and stress images, with each recording an overall qualitative diagnosis of normal, ischemia, ischemia plus scar, or scar. For analyses of sensitivity and specificity, normal was considered image negative and all other diagnoses were considered image positive.
2.3 Image Acquisition Instructions
For rest and stress imaging, image reconstruction should include attenuation correction.
14.2 Suspected Coronary Artery Disease
Study 1 evaluated the sensitivity and specificity of FLYRCADO PET MPI for the detection of significant CAD in subjects with suspected CAD who were scheduled for invasive coronary angiography (ICA).
A total of 578 subjects were evaluable for effectiveness, having rest and stress imaging and evaluable truth standard data. Subjects ranged in age from 26 years to 88 years, with a mean age of 64 years. A total of 188 (33%) were female, and 473 (82%) were White, 35 (6%) were Black or African American, 6 (1%) were Asian, and 64 (11%) were other races or not reported. In addition, 79 subjects (14%) reported Hispanic/Latino ethnicity. Pharmacologic stress was performed in 83% of subjects and exercise stress in 17% of subjects.
The sensitivity and specificity of FLYRCADO PET MPI for detection of significant CAD, defined as the presence of significant stenosis in at least one major epicardial coronary artery or major branch by quantitative coronary angiography (QCA) are reported in Table 6. Results for both ≥50% stenosis and a secondary analysis using ≥70% stenosis as the threshold for significant CAD are shown.
| ≥50% Stenosis Reference Standard | ≥70% Stenosis Reference Standard | |||
|---|---|---|---|---|
| Reader | Sensitivity (95% CI) N=249 |
Specificity (95% CI) N=329 |
Sensitivity (95% CI) N=127 |
Specificity (95% CI) N=449 |
| Abbreviations: CI = confidence interval, MPI = myocardial perfusion imaging | ||||
| Reader 1 | 77% (72%, 82%) |
66% (61%, 71%) |
91% (86%, 96%) |
58% (54%, 63%) |
| Reader 2 | 74% (68%, 79%) |
70% (65%, 75%) |
87% (82%, 93%) |
62% (58%, 67%) |
| Reader 3 | 89% (85%, 93%) |
53% (47%, 58%) |
97% (94%, 100%) |
44% (39%, 49%) |
From a blinded re-evaluation of 60 randomly selected PET MPI images presented during the main reading sessions, intra-reader kappa ranged from 0.71 to 0.93 for the three readers.
Using a 50% stenosis threshold, sensitivity of SPECT MPI was 61% to 76% for the three readers, with the lower bound of the 95% confidence intervals ranging from 55% to 70%, and specificity of SPECT MPI was 51% to 65%, with the lower bound of the 95% confidence intervals ranging from 46% to 60%.
2.1 Radiation Safety – Drug Handling
Handle FLYRCADO with appropriate safety measures to minimize radiation exposure [see Warnings and Precautions (5.2)]. Use waterproof gloves and effective shielding, including lead-glass syringe shields, when handling and administering FLYRCADO.
Radioactive drugs should be used by or under the control of healthcare providers who are qualified by specific training and experience in the safe use and handling of radionuclides, and whose experience and training have been approved by the appropriate governmental agency authorized to license the use of radionuclides.
Principal Display Panel 30 Ml Vial Label
NDC 0407-8787-01
Multiple-Dose Vial
Non-Pyrogenic
Sterile
Flyrcado™
(flurpiridaz F 18) injection
190 MBq/mL to 2,050 MBq/mL
(5 mCi/mL to 55 mCi/mL) at end of synthesis
Diagnostic - For Intravenous Use Only
CAUTION
RADIOACTIVE
MATERIAL
Total: ______MBq (____mCi) in_____mL at _____:_____ on date ___________
Concentration: ____MBq/mL (____mCi/mL) Exp Date_________ Exp Time__________
Batch No.: _______________ Date: ________________ Vial No.: __________
Store FLYRCADO at 2°C to 30°C (36°F to 86°F); excursions to -20°C (-4°F) (up to 2 hours) or to 50°C
(122°F) (up to 8 hours) may be permitted.
Recommended Dosage: See Prescribing Information
Distributed by GE Healthcare Inc., Arlington Heights, IL 60004, U.S.A.
Rx ONLY
200112-0B
Principal Display Panel 30 Ml Shield Label
NDC 0407-8787-01
Non-Pyrogenic
Sterile
Flyrcado™
(flurpiridaz F 18) injection
CAUTION
RADIOACTIVE
MATERIAL
Multiple-Dose Vial
190 MBq/mL to 2,050 MBq/mL
(5 mCi/mL to 55 mCi/mL) at end of synthesis
Diagnostic - For Intravenous Use Only
Total _____MBq (___mCi) in ____mL at____:____ on date________
Batch No.:_____________ Exp. Date:_______ Exp. Time:____:____
Concentration:_______MBq/mL (____mCi/mL) Vial No.: ____________
Each mL contains up to 2.3 micrograms flurpiridaz, 45 mg hydroxypropyl-
β-cyclodextrin, 35 mg L-(+)-ascorbic acid, 8.2 mg sodium hydroxide, and
55.2 mg anhydrous ethanol in water for injection.
Store Flyrcado at 2°C to 30°C (36°F to 86°F); excursions to -20°C (-4°F) (up
to 2 hours) or to 50°C (122°F) (up to 8 hours) may be permitted.
Recommended Dosage: See Prescribing Information
Distributed by GE Healthcare Inc., Arlington Heights, IL 60004, U.S.A.
Rx ONLY
200113-0B
14.3 Known Or Suspected Coronary Artery Disease
Study 2 evaluated the sensitivity and specificity of FLYRCADO PET MPI for the detection of significant CAD in subjects with known or suspected CAD who had ICA without intervention within 60 days prior to imaging or were scheduled for ICA.
A total of 755 subjects were evaluable for effectiveness, having rest and stress imaging for FLYRCADO PET and evaluable truth standard data. Subjects ranged in age from 36 years to 90 years, with a mean age of 63 years. A total of 235 (31%) were female, and 619 (82%) were White, 101 (13%) were Black or African American, 8 (1%) were Asian, and 24 (4%) were other races or not reported. Pharmacologic stress was performed in 71% of subjects and exercise stress in 29% of subjects.
The sensitivity and specificity of FLYRCADO PET MPI for the detection of significant CAD, defined as the presence of significant stenosis in at least one major epicardial coronary artery or major branch by QCA or as history of myocardial infarction are reported in Table 7. Results for both ≥50% stenosis and a secondary analysis using a threshold of ≥70% stenosis for significant CAD are shown.
| ≥50% Stenosis or Confirmed MI Reference Standard | ≥70% Stenosis or Confirmed MI Reference Standard | |||
|---|---|---|---|---|
| Reader | Sensitivity (95% CI) N=352 |
Specificity (95% CI) N=403 |
Sensitivity (95% CI) N=245 |
Specificity (95% CI) N=510 |
| Abbreviations: CI = confidence interval, MI = myocardial infarction, MPI = myocardial perfusion imaging | ||||
| Reader 1 | 73% (68%, 78%) |
73% (68%, 77%) |
82% (77%, 87%) |
68% (63%, 71%) |
| Reader 2 | 63% (57%, 67%) |
86% (82%, 89%) |
72% (66%, 78%) |
80% (77%, 83%) |
| Reader 3 | 77% (72%, 80%) |
66% (62%, 71%) |
85% (80%, 89%) |
61% (57%, 66%) |
From a blinded re-evaluation of a randomly selected 10% of PET MPI images presented during the main reading sessions, intra-reader agreement ranged from 90% to 95% for the three readers.
Using a 50% stenosis threshold, sensitivity of SPECT MPI was 43% to 58% for the three readers, with the lower bound of the 95% confidence intervals ranging from 38% to 53%, and specificity for SPECT MPI was 80% to 92%, with the lower bound of the 95% confidence intervals ranging from 76% to 89%.
5.1 Risks Associated With Exercise Or Pharmacologic Stress
Patients evaluated with exercise or pharmacologic stress may experience serious adverse reactions such as myocardial infarction, arrhythmia, hypotension, bronchoconstriction, stroke, and seizure. Perform stress testing in the setting where cardiac resuscitation equipment and trained staff are readily available. When pharmacologic stress is selected as an alternative to exercise, perform the procedure in accordance with the pharmacologic stress agent's prescribing information.
Structured Label Content
Section 42229-5 (42229-5)
Recommended Dosage
- The recommended activity for each type of examination is presented in Table 1.
- Administer two doses by intravenous injection, one for rest imaging and one for stress imaging, using either pharmacologic or exercise stress, in either a 1-day or 2-day protocol.
- If performing a combined exercise/pharmacologic stress protocol, administer the recommended activity for pharmacologic stress.
- When rest and stress imaging are performed on the same day, the recommended minimum stress activity is 2-fold the rest activity for pharmacologic stress and 3-fold the rest activity for exercise stress to provide adequate image quality and obscure residual signal from the first acquisition.
- The recommended maximum total volume administered in one day is 6.1 mL.
| Length of Protocol | Activity for Rest Imaging | Activity for Stress Imaging | |
|---|---|---|---|
| Pharmacologic | Exercise | ||
| 1 day | 93 MBq to 111 MBq (2.5 mCi to 3 mCi) |
222 MBq to 241 MBq (6 mCi to 6.5 mCi) |
333 MBq to 352 MBq (9 mCi to 9.5 mCi) |
| 2 days | 93 MBq to 111 MBq (2.5 mCi to 3 mCi) |
93 MBq to 111 MBq (2.5 mCi to 3 mCi) |
93 MBq to 111 MBq (2.5 mCi to 3 mCi) |
Section 44425-7 (44425-7)
Storage and Handling
Store FLYRCADO at 2°C to 30°C (36°F to 86°F); excursions to -20°C (-4°F) (up to 2 hours) or to 50°C (122°F) (up to 8 hours) may be permitted. Store FLYRCADO within radiation shielding. The product does not contain a preservative.
Do not use and discard FLYRCADO 8 hours after end of synthesis or when the activity falls below the radioactivity requirement at the time of injection, whichever is earlier. The expiration date and time are provided on the shield label.
Dispose of the product in accordance with all federal, state, and local laws and institutional requirements.
This preparation is for use by persons licensed by the Nuclear Regulatory Commission or the relevant regulatory authority of an Agreement State.
8.4 Pediatric Use
Safety and effectiveness of FLYRCADO in pediatric patients have not been established.
8.5 Geriatric Use
Of 1,600 subjects in clinical studies of FLYRCADO, 720 (45%) were 65 years of age and over and 181 (11%) were 75 years of age or older. No overall differences in safety or effectiveness of FLYRCADO have been observed between patients 65 years of age and older and younger adult patients.
4 Contraindications (4 CONTRAINDICATIONS)
None.
5.2 Radiation Risks
FLYRCADO contributes to a patient's overall long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk of cancer. Ensure safe handling to minimize radiation exposure to patients and health care providers [see Dosage and Administration (2.1, 2.4)]. Advise patients to hydrate before and after administration and to void frequently after administration.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described elsewhere in the labeling:
- Risks Associated with Exercise or Pharmacologic Stress [see Warnings and Precautions (5.1)]
12.2 Pharmacodynamics
The relationship between flurpiridaz F 18 plasma concentrations and successful imaging was not explored in clinical trials.
12.3 Pharmacokinetics
The pharmacokinetics of flurpiridaz F 18 were evaluated in healthy subjects. Blood radioactivity peaked at 2.3 minutes with blood clearance followed by a rise and plateau at around 3% of the 296 MBq (8 mCi) flurpiridaz F 18 intravenous dose until 7 hours post administration. The fluorine-18 radioactivity in blood during the first 15-minutes post administration was associated with flurpiridaz while it was associated with flurpiridaz metabolites thereafter.
1 Indications and Usage (1 INDICATIONS AND USAGE)
FLYRCADO is indicated for positron emission tomography (PET) myocardial perfusion imaging (MPI) under rest or stress (pharmacologic or exercise) in adult patients with known or suspected coronary artery disease (CAD) to evaluate for myocardial ischemia and infarction.
11.3 External Radiation
The point source air-kerma rate constant for fluorine-18 is 3.74E-17 Gy m2/(Bq s); this coefficient was formerly defined as the specific gamma-ray constant of 5.7 R/hr/mCi at 1 cm. The first half-value thickness of lead (Pb) for fluorine-18 gamma rays is approximately 6 mm. The relative reduction of radiation emitted by fluorine-18 that results from various thicknesses of lead shielding is shown in Table 5. The use of about 8 cm of Pb will decrease the radiation transmission (i.e., exposure) by a factor of about 10,000.
| Shielding Thickness cm of Lead (Pb) | Coefficient of Attenuation |
|---|---|
| 0.6 | 0.5 |
| 2 | 0.1 |
| 4 | 0.01 |
| 6 | 0.001 |
| 8 | 0.0001 |
2.4 Radiation Dosimetry
Table 2 shows the estimated radiation absorbed doses per unit of injected activity.
| Organ | Absorbed Dose per Unit of Administered Activity (mGy/MBq) |
||
|---|---|---|---|
| Rest | Pharmacologic Stress The pharmacologic stress agent used was adenosine.
|
Exercise Stress | |
| Adrenals | 0.016 | 0.016 | 0.014 |
| Bone surfaces | 0.019 | 0.019 | 0.02 |
| Brain | 0.025 | 0.022 | 0.011 |
| Breasts | 0.009 | 0.009 | 0.01 |
| Gallbladder Wall | 0.017 | 0.018 | 0.015 |
| Gastrointestinal Tract | |||
| Stomach Wall | 0.04 | 0.033 | 0.024 |
| Small Intestine Wall | 0.013 | 0.012 | 0.014 |
| Upper Large Intestine Wall | 0.013 | 0.012 | 0.014 |
| Lower Large Intestine Wall | 0.012 | 0.011 | 0.014 |
| Heart Wall | 0.048 | 0.09 | 0.039 |
| Kidneys | 0.066 | 0.057 | 0.027 |
| Liver | 0.039 | 0.044 | 0.015 |
| Lungs | 0.011 | 0.012 | 0.012 |
| Muscle | 0.01 | 0.01 | 0.012 |
| Ovaries | 0.012 | 0.012 | 0.014 |
| Pancreas | 0.016 | 0.016 | 0.015 |
| Red Marrow | 0.016 | 0.018 | 0.015 |
| Salivary Glands | 0.035 | 0.076 | 0.007 |
| Skin | 0.008 | 0.008 | 0.009 |
| Spleen | 0.016 | 0.012 | 0.013 |
| Testes | 0.009 | 0.009 | 0.011 |
| Thymus | 0.011 | 0.012 | 0.013 |
| Thyroid | 0.032 | 0.036 | 0.014 |
| Urinary Bladder Wall | 0.023 | 0.021 | 0.016 |
| Uterus | 0.012 | 0.012 | 0.014 |
| Total Body | 0.012 | 0.012 | 0.012 |
| Effective Dose (mSv/MBq) | 0.019 | 0.019 | 0.015 |
The whole-body effective dose resulting from the administration of maximal activity of FLYRCADO of 111 MBq at rest, 241 MBq during pharmacological stress, and 352 MBq during exercise stress is, respectively, 2.1 mSv, 4.6 mSv, and 5.3 mSv. Under the same conditions, the absorbed dose to the target organ (heart wall) is 5.3 mGy, 22 mGy, and 14 mGy for each administered activity, respectively.
The use of a CT scan to calculate attenuation correction for the reconstruction of FLYRCADO PET images (as done in PET/CT imaging) will add radiation exposure.
12.1 Mechanism of Action
Flurpiridaz F 18 is an analog of the mitochondrial complex 1 (MC-1) inhibitor, pyridaben. Flurpiridaz F 18 is extracted by the myocardium proportional to the blood flow and binds to heart tissue that has biologically active mitochondria. Therefore, radioactivity in viable myocardium is higher than in infarcted tissue.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Risk associated with exercise or pharmacologic stress: Serious adverse reactions such as myocardial infarction, arrhythmia, hypotension, broncho-constriction, stroke, and seizures may occur. Perform stress testing in the setting where cardiac resuscitation equipment and trained staff are readily available. When pharmacologic stress is selected, perform the procedure in accordance with the pharmacologic stress agent's prescribing information. (5.1)
- Radiation risks: Ensure safe handling to minimize radiation exposure to patients and health care providers. (5.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Administer FLYRCADO via intravenous injection. (2.2)
- When rest and stress imaging are conducted on the same day, the recommended administered activities are (2.2):
- Rest imaging: 93 MBq to 111 MBq (2.5 mCi to 3 mCi)
- Pharmacologic stress imaging: 222 MBq to 241 MBq (6 mCi to 6.5 mCi)
- Exercise stress imaging: 333 MBq to 352 MBq (9 mCi to 9.5 mCi)
- When rest and stress imaging are conducted over two days, the recommended rest and stress administered activities, for both pharmacologic and exercise stress, are 93 MBq to 111 MBq (2.5 mCi to 3 mCi). (2.2)
- See full prescribing information for radiation safety, preparation, administration, imaging, and radiation dosimetry information. (2.1, 2.2, 2.3, 2.4)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 190 MBq/mL to 2,050 MBq/mL (5 mCi/mL to 55 mCi/mL) of flurpiridaz F 18 at end of synthesis as a clear, colorless to yellow solution in a shielded multiple-dose vial with up to 30 mL fill volume.
11.1 Chemical Characteristics
FLYRCADO (flurpiridaz F 18) injection is a radioactive diagnostic drug for intravenous use. The molecular formula of flurpiridaz F 18 is C18H22Cl18FN2O3, the molecular mass is 367.8, and the structural formula is:
Chemically, flurpiridaz F 18 is 2-tert-butyl-4-chloro-5-[[4-(2-(18F)fluoranylethoxymethyl)phenyl]methoxy]pyridazin-3-one.
FLYRCADO is a sterile, preservative-free, non-pyrogenic, clear, colorless to yellow radioactive solution. Each mL contains 190 MBq to 2,050 MBq (5 mCi to 55 mCi) of flurpiridaz F 18 at end of synthesis, up to 2.3 mcg flurpiridaz, and the following inactive ingredients: 45 mg hydroxypropyl-β-cyclodextrin (as a solubilizer and co-radiostabilizer), 35 mg L-(+)-ascorbic acid (as a radiostabilizer), 8.2 mg sodium hydroxide, and 55.2 mg anhydrous ethanol, in water for injection. The pH of the solution is between 5.5 and 8.
11.2 Physical Characteristics
Fluorine-18 decays by positron (β+) emission and has a half-life of 109.8 minutes. The principal photons useful for diagnostic imaging are the 511 keV gamma photons, resulting from the interaction of the emitted positron with an electron. Principal emission data for fluorine-18 are shown in Table 4.
| Radiation/Emission | % per Disintegration | Mean Energy (keV) |
|---|---|---|
| Positron | 96.7 | 249.8 |
| Gamma | 193.5 | 511 |
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Temporarily discontinue breastfeeding. A lactating woman should pump and discard breastmilk for at least 8 hours after FLYRCADO administration. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of FLYRCADO was evaluated in 1,600 subjects in clinical studies, including 1,575 (98%) subjects with known or suspected coronary artery disease and 25 (2%) healthy subjects. All 1,600 subjects were dosed under rest conditions, with a mean dose of 102 MBq (2.8 mCi) FLYRCADO. A total of 1,568 (98%) subjects were also dosed under stress (exercise or pharmacologic) conditions, with a mean activity of 252 MBq (6.8 mCi) FLYRCADO by intravenous route. The demographic characteristics of the study population were 31% female, mean age 62 years (range 19 years to 90 years), 81% White, 11% Black or African American, 1% Asian, and 7% other or unreported race, and 10% Hispanic or Latino, 81% Not Hispanic or Latino, and 9% unreported ethnicity.
Stress testing procedures are associated with serious adverse reactions [see Warnings and Precautions (5.1)]. Adverse reactions occurring in ≥2% subjects receiving FLYRCADO during PET MPI under rest and stress (pharmacologic or exercise) are presented in Table 3.
| Adverse Reaction | FLYRCADO PET MPI Under Rest and Stress (Pharmacologic or Exercise) N=1,600 Includes 32 subjects who received only one dose at rest.
% |
|---|---|
| Dyspnea | 17 |
| Headache | 15 |
| Angina pectoris | 10 |
| Chest pain | 8 |
| Fatigue | 7 |
| ST segment changes | 6 |
| Flushing | 5 |
| Nausea | 4 |
| Abdominal pain | 4 |
| Dizziness | 4 |
| Arrhythmia | 4 |
Adverse reactions occurring during FLYRCADO PET MPI under rest and stress (pharmacologic or exercise) in <2% of subjects included diarrhea, palpitations, back pain, cardiac conduction disturbance, rash, dysgeusia, cough, hypotension, anxiety, vomiting, pruritus, bronchospasm, dry mouth, blood pressure elevation, syncope, and wheezing.
14.1 Overview of Clinical Studies
The safety and effectiveness of FLYRCADO were evaluated in two prospective, multicenter, open-label clinical studies in adults with either suspected coronary artery disease (CAD) (Study 1: NCT03354273) or known or suspected CAD (Study 2: NCT01347710).
Subjects received two injections of FLYRCADO: one at rest and one during stress [see Dosage and Administration (2.2, 2.3)]. For the FLYRCADO stress injection, subjects received either a pharmacologic stress agent or engaged in exercise stress. PET myocardial perfusion imaging (MPI) was performed at both rest and stress using cardiac gating and low-dose CT attenuation correction. Subjects also received rest and stress SPECT MPI using technetium Tc 99m sestamibi or technetium Tc 99m tetrofosmin on a different day from the PET MPI. The stress modality was to be the same for PET and SPECT.
Stress and rest images were displayed side-by-side for the assessment of perfusion and wall motion abnormalities. Three qualified readers, blinded to clinical data, performed independent assessment of each subject's rest and stress images, with each recording an overall qualitative diagnosis of normal, ischemia, ischemia plus scar, or scar. For analyses of sensitivity and specificity, normal was considered image negative and all other diagnoses were considered image positive.
2.3 Image Acquisition Instructions
For rest and stress imaging, image reconstruction should include attenuation correction.
14.2 Suspected Coronary Artery Disease
Study 1 evaluated the sensitivity and specificity of FLYRCADO PET MPI for the detection of significant CAD in subjects with suspected CAD who were scheduled for invasive coronary angiography (ICA).
A total of 578 subjects were evaluable for effectiveness, having rest and stress imaging and evaluable truth standard data. Subjects ranged in age from 26 years to 88 years, with a mean age of 64 years. A total of 188 (33%) were female, and 473 (82%) were White, 35 (6%) were Black or African American, 6 (1%) were Asian, and 64 (11%) were other races or not reported. In addition, 79 subjects (14%) reported Hispanic/Latino ethnicity. Pharmacologic stress was performed in 83% of subjects and exercise stress in 17% of subjects.
The sensitivity and specificity of FLYRCADO PET MPI for detection of significant CAD, defined as the presence of significant stenosis in at least one major epicardial coronary artery or major branch by quantitative coronary angiography (QCA) are reported in Table 6. Results for both ≥50% stenosis and a secondary analysis using ≥70% stenosis as the threshold for significant CAD are shown.
| ≥50% Stenosis Reference Standard | ≥70% Stenosis Reference Standard | |||
|---|---|---|---|---|
| Reader | Sensitivity (95% CI) N=249 |
Specificity (95% CI) N=329 |
Sensitivity (95% CI) N=127 |
Specificity (95% CI) N=449 |
| Abbreviations: CI = confidence interval, MPI = myocardial perfusion imaging | ||||
| Reader 1 | 77% (72%, 82%) |
66% (61%, 71%) |
91% (86%, 96%) |
58% (54%, 63%) |
| Reader 2 | 74% (68%, 79%) |
70% (65%, 75%) |
87% (82%, 93%) |
62% (58%, 67%) |
| Reader 3 | 89% (85%, 93%) |
53% (47%, 58%) |
97% (94%, 100%) |
44% (39%, 49%) |
From a blinded re-evaluation of 60 randomly selected PET MPI images presented during the main reading sessions, intra-reader kappa ranged from 0.71 to 0.93 for the three readers.
Using a 50% stenosis threshold, sensitivity of SPECT MPI was 61% to 76% for the three readers, with the lower bound of the 95% confidence intervals ranging from 55% to 70%, and specificity of SPECT MPI was 51% to 65%, with the lower bound of the 95% confidence intervals ranging from 46% to 60%.
2.1 Radiation Safety – Drug Handling
Handle FLYRCADO with appropriate safety measures to minimize radiation exposure [see Warnings and Precautions (5.2)]. Use waterproof gloves and effective shielding, including lead-glass syringe shields, when handling and administering FLYRCADO.
Radioactive drugs should be used by or under the control of healthcare providers who are qualified by specific training and experience in the safe use and handling of radionuclides, and whose experience and training have been approved by the appropriate governmental agency authorized to license the use of radionuclides.
Principal Display Panel 30 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 30 mL Vial Label)
NDC 0407-8787-01
Multiple-Dose Vial
Non-Pyrogenic
Sterile
Flyrcado™
(flurpiridaz F 18) injection
190 MBq/mL to 2,050 MBq/mL
(5 mCi/mL to 55 mCi/mL) at end of synthesis
Diagnostic - For Intravenous Use Only
CAUTION
RADIOACTIVE
MATERIAL
Total: ______MBq (____mCi) in_____mL at _____:_____ on date ___________
Concentration: ____MBq/mL (____mCi/mL) Exp Date_________ Exp Time__________
Batch No.: _______________ Date: ________________ Vial No.: __________
Store FLYRCADO at 2°C to 30°C (36°F to 86°F); excursions to -20°C (-4°F) (up to 2 hours) or to 50°C
(122°F) (up to 8 hours) may be permitted.
Recommended Dosage: See Prescribing Information
Distributed by GE Healthcare Inc., Arlington Heights, IL 60004, U.S.A.
Rx ONLY
200112-0B
Principal Display Panel 30 Ml Shield Label (PRINCIPAL DISPLAY PANEL - 30 mL Shield Label)
NDC 0407-8787-01
Non-Pyrogenic
Sterile
Flyrcado™
(flurpiridaz F 18) injection
CAUTION
RADIOACTIVE
MATERIAL
Multiple-Dose Vial
190 MBq/mL to 2,050 MBq/mL
(5 mCi/mL to 55 mCi/mL) at end of synthesis
Diagnostic - For Intravenous Use Only
Total _____MBq (___mCi) in ____mL at____:____ on date________
Batch No.:_____________ Exp. Date:_______ Exp. Time:____:____
Concentration:_______MBq/mL (____mCi/mL) Vial No.: ____________
Each mL contains up to 2.3 micrograms flurpiridaz, 45 mg hydroxypropyl-
β-cyclodextrin, 35 mg L-(+)-ascorbic acid, 8.2 mg sodium hydroxide, and
55.2 mg anhydrous ethanol in water for injection.
Store Flyrcado at 2°C to 30°C (36°F to 86°F); excursions to -20°C (-4°F) (up
to 2 hours) or to 50°C (122°F) (up to 8 hours) may be permitted.
Recommended Dosage: See Prescribing Information
Distributed by GE Healthcare Inc., Arlington Heights, IL 60004, U.S.A.
Rx ONLY
200113-0B
14.3 Known Or Suspected Coronary Artery Disease (14.3 Known or Suspected Coronary Artery Disease)
Study 2 evaluated the sensitivity and specificity of FLYRCADO PET MPI for the detection of significant CAD in subjects with known or suspected CAD who had ICA without intervention within 60 days prior to imaging or were scheduled for ICA.
A total of 755 subjects were evaluable for effectiveness, having rest and stress imaging for FLYRCADO PET and evaluable truth standard data. Subjects ranged in age from 36 years to 90 years, with a mean age of 63 years. A total of 235 (31%) were female, and 619 (82%) were White, 101 (13%) were Black or African American, 8 (1%) were Asian, and 24 (4%) were other races or not reported. Pharmacologic stress was performed in 71% of subjects and exercise stress in 29% of subjects.
The sensitivity and specificity of FLYRCADO PET MPI for the detection of significant CAD, defined as the presence of significant stenosis in at least one major epicardial coronary artery or major branch by QCA or as history of myocardial infarction are reported in Table 7. Results for both ≥50% stenosis and a secondary analysis using a threshold of ≥70% stenosis for significant CAD are shown.
| ≥50% Stenosis or Confirmed MI Reference Standard | ≥70% Stenosis or Confirmed MI Reference Standard | |||
|---|---|---|---|---|
| Reader | Sensitivity (95% CI) N=352 |
Specificity (95% CI) N=403 |
Sensitivity (95% CI) N=245 |
Specificity (95% CI) N=510 |
| Abbreviations: CI = confidence interval, MI = myocardial infarction, MPI = myocardial perfusion imaging | ||||
| Reader 1 | 73% (68%, 78%) |
73% (68%, 77%) |
82% (77%, 87%) |
68% (63%, 71%) |
| Reader 2 | 63% (57%, 67%) |
86% (82%, 89%) |
72% (66%, 78%) |
80% (77%, 83%) |
| Reader 3 | 77% (72%, 80%) |
66% (62%, 71%) |
85% (80%, 89%) |
61% (57%, 66%) |
From a blinded re-evaluation of a randomly selected 10% of PET MPI images presented during the main reading sessions, intra-reader agreement ranged from 90% to 95% for the three readers.
Using a 50% stenosis threshold, sensitivity of SPECT MPI was 43% to 58% for the three readers, with the lower bound of the 95% confidence intervals ranging from 38% to 53%, and specificity for SPECT MPI was 80% to 92%, with the lower bound of the 95% confidence intervals ranging from 76% to 89%.
5.1 Risks Associated With Exercise Or Pharmacologic Stress (5.1 Risks Associated with Exercise or Pharmacologic Stress)
Patients evaluated with exercise or pharmacologic stress may experience serious adverse reactions such as myocardial infarction, arrhythmia, hypotension, bronchoconstriction, stroke, and seizure. Perform stress testing in the setting where cardiac resuscitation equipment and trained staff are readily available. When pharmacologic stress is selected as an alternative to exercise, perform the procedure in accordance with the pharmacologic stress agent's prescribing information.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:53.938208 · Updated: 2026-03-14T22:43:50.676219