3b387c89-25d5-f12b-e063-6294a90a5942
34390-5
HUMAN OTC DRUG LABEL
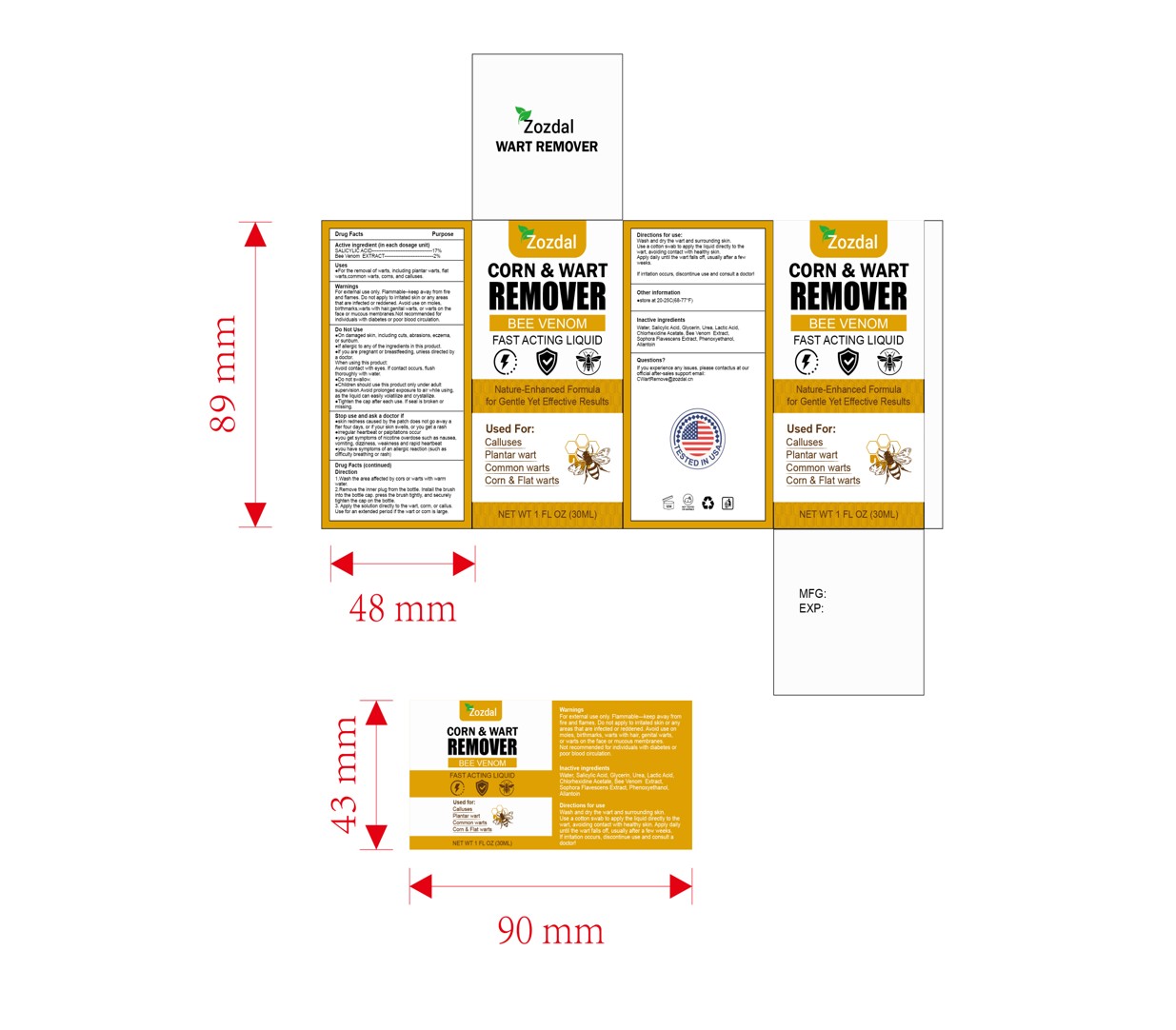
Drug Facts
Composition & Product
Identifiers & Packaging
Description
SALICYLIC ACID 17% Bee Venom EXTRACT 2%
Medication Information
Warnings and Precautions
WarningsFor extemal use only, Flammable-keep away from fireand flames.Do not apply to irritated skin or any areasthat are infected or reddened.Avoid usehair.genital warts.,or warts on theface ormucous membranes.Not recommended forindividuals with diabetes or poor blood circulation.
Indications and Usage
Direction
Wash he area aeced by ors or watswith wamwaler2.Removethe lmnerplug fiom the boe. hsathebnushino the boe cap.pes he knush ighy, and secureltghen hiecap onhe bole.3.Apythesoluion direclyto
the wart, corm, or callus,Use for an extended period if the wart or corn is large.
Dosage and Administration
Apply the solution directly to the wart, corm, or callus.Use for an extended period if the wart or corn is large.
Description
SALICYLIC ACID 17% Bee Venom EXTRACT 2%
Section 50565-1
Please keep the product out of reach of children. lf accidentally ingested, please call the emergency center immediately
Section 50566-9
stop use and aska docdor eskthnredhescaused by the patch does not o aay ater lour day,or yourshin snl,or yougeta rasheregular heatbeat or palpaons cuyougetsmiploms of noine overnose such as
nausea.vomiting,dizziness, weakness and rapid heartbeateyou have symptoms of an allergic reaction (such asdificully breathing or rash)
Section 51727-6
AQUA
GLYCERIN
LACTIC ACID
UREA
Chlorhexidine Acetate
SOPHORA FLAVESCENS EXTRACT
HYDROXYETHYLCELLULOSE
ALLANTOIN
PHENOXYETHANOL
Section 51945-4
Section 55105-1
wart, corn and callus remover
Section 55106-9
SALICYLIC ACID 17%
Bee Venom EXTRACT 2%
Structured Label Content
Indications and Usage (34067-9)
Direction
Wash he area aeced by ors or watswith wamwaler2.Removethe lmnerplug fiom the boe. hsathebnushino the boe cap.pes he knush ighy, and secureltghen hiecap onhe bole.3.Apythesoluion direclyto
the wart, corm, or callus,Use for an extended period if the wart or corn is large.
Dosage and Administration (34068-7)
Apply the solution directly to the wart, corm, or callus.Use for an extended period if the wart or corn is large.
Warnings and Precautions (34071-1)
WarningsFor extemal use only, Flammable-keep away from fireand flames.Do not apply to irritated skin or any areasthat are infected or reddened.Avoid usehair.genital warts.,or warts on theface ormucous membranes.Not recommended forindividuals with diabetes or poor blood circulation.
Section 50565-1 (50565-1)
Please keep the product out of reach of children. lf accidentally ingested, please call the emergency center immediately
Section 50566-9 (50566-9)
stop use and aska docdor eskthnredhescaused by the patch does not o aay ater lour day,or yourshin snl,or yougeta rasheregular heatbeat or palpaons cuyougetsmiploms of noine overnose such as
nausea.vomiting,dizziness, weakness and rapid heartbeateyou have symptoms of an allergic reaction (such asdificully breathing or rash)
Section 51727-6 (51727-6)
AQUA
GLYCERIN
LACTIC ACID
UREA
Chlorhexidine Acetate
SOPHORA FLAVESCENS EXTRACT
HYDROXYETHYLCELLULOSE
ALLANTOIN
PHENOXYETHANOL
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
wart, corn and callus remover
Section 55106-9 (55106-9)
SALICYLIC ACID 17%
Bee Venom EXTRACT 2%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:39.828624 · Updated: 2026-03-14T23:09:33.254922