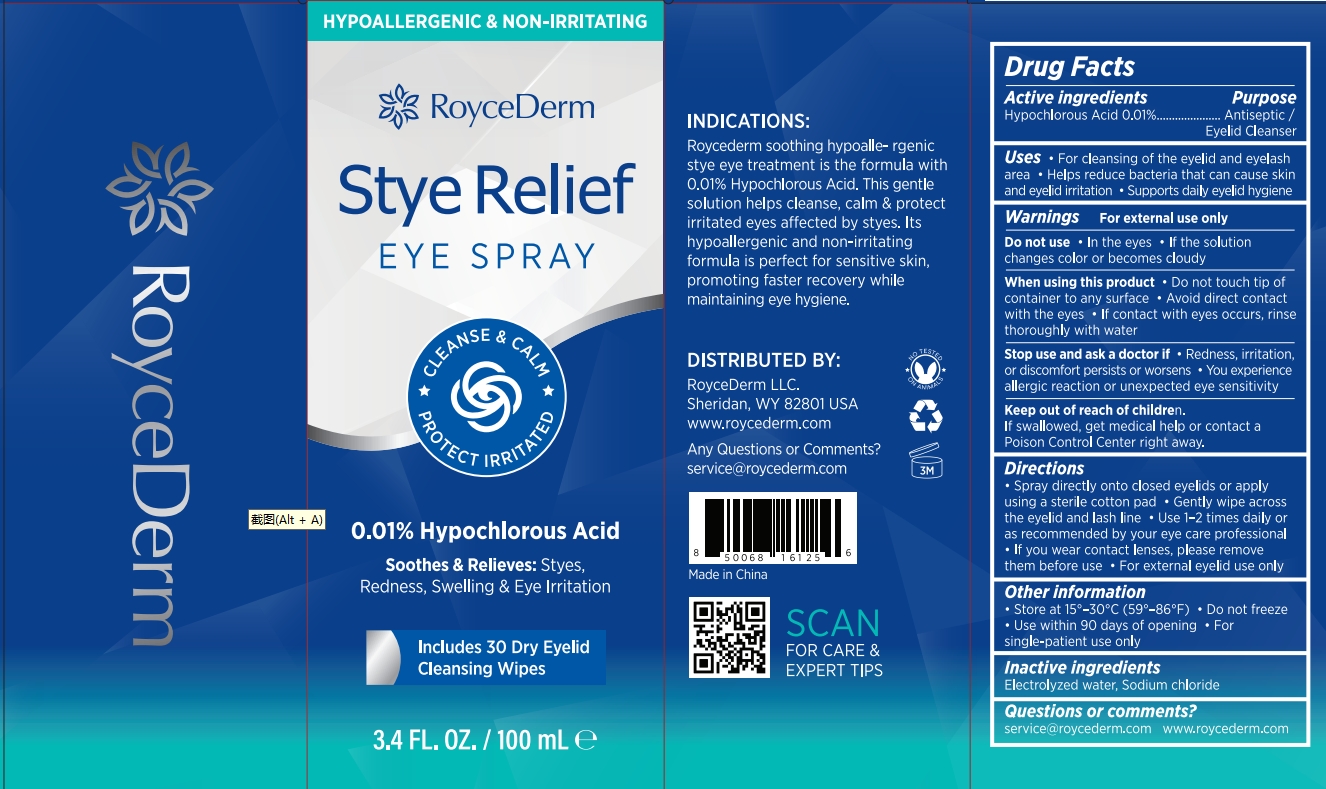
Initial Drug Listing - Roycederm Stye Relief Eye Spray
3afa5e69-c285-fd73-e063-6294a90a93f9
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Hypochlorous Acid 0.01%
Medication Information
Warnings and Precautions
For external use only
Indications and Usage
- Spray directly onto closed eyelids or applyusing a sterile cotton pad
- Gently wipe acrossthe eyelid and lash line
- Use 1-2 times daily oras recommended by your eye care professional
- If you wear contact lenses, please removethem before use
- For external eyelid use only
Dosage and Administration
For cleansing of the eyelid and eyelash
area
Helps reduce bacteria that can cause skin
and eyelid irritation
Supports daily eyelid hygiene
Description
Hypochlorous Acid 0.01%
Section 50565-1
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
- Redness, irritation,or discomfort persists or worsens
- You experience allergic reaction or unexpected eye sensitivity
Section 50567-7
- Do not touch tip of container to any surface
- Avoid direct contact with the eyes
- If contact with eyes occurs, rinse thoroughly with water
Section 50569-3
- Redness, irritation, or discomfort persists or worsens
- You experience allergic reaction or unexpected eye sensitivity
Section 50570-1
In the eyes · If the solution changes color or becomes cloudy
Section 51727-6
Electrolyzed water, Sodium chloride
Section 51945-4
Section 53413-1
- [email protected]
- www.roycederm.com
Section 55105-1
Antiseptic / Eyelid Cleanser
Section 55106-9
Hypochlorous Acid 0.01%
Section 60561-8
- Store at 15°-30°C(59°-86°F)
- Do not freeze
- Use within 90 days of opening
- Forsingle-patient use only
Structured Label Content
Indications and Usage (34067-9)
- Spray directly onto closed eyelids or applyusing a sterile cotton pad
- Gently wipe acrossthe eyelid and lash line
- Use 1-2 times daily oras recommended by your eye care professional
- If you wear contact lenses, please removethem before use
- For external eyelid use only
Dosage and Administration (34068-7)
For cleansing of the eyelid and eyelash
area
Helps reduce bacteria that can cause skin
and eyelid irritation
Supports daily eyelid hygiene
Warnings and Precautions (34071-1)
For external use only
Section 50565-1 (50565-1)
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
- Redness, irritation,or discomfort persists or worsens
- You experience allergic reaction or unexpected eye sensitivity
Section 50567-7 (50567-7)
- Do not touch tip of container to any surface
- Avoid direct contact with the eyes
- If contact with eyes occurs, rinse thoroughly with water
Section 50569-3 (50569-3)
- Redness, irritation, or discomfort persists or worsens
- You experience allergic reaction or unexpected eye sensitivity
Section 50570-1 (50570-1)
In the eyes · If the solution changes color or becomes cloudy
Section 51727-6 (51727-6)
Electrolyzed water, Sodium chloride
Section 51945-4 (51945-4)
Section 53413-1 (53413-1)
- [email protected]
- www.roycederm.com
Section 55105-1 (55105-1)
Antiseptic / Eyelid Cleanser
Section 55106-9 (55106-9)
Hypochlorous Acid 0.01%
Section 60561-8 (60561-8)
- Store at 15°-30°C(59°-86°F)
- Do not freeze
- Use within 90 days of opening
- Forsingle-patient use only
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:43.359971 · Updated: 2026-03-14T23:09:39.646164