85966-001 extra strength nail fungus treatment
3abc0892-e52b-a40f-e063-6294a90a0e35
34390-5
HUMAN OTC DRUG LABEL
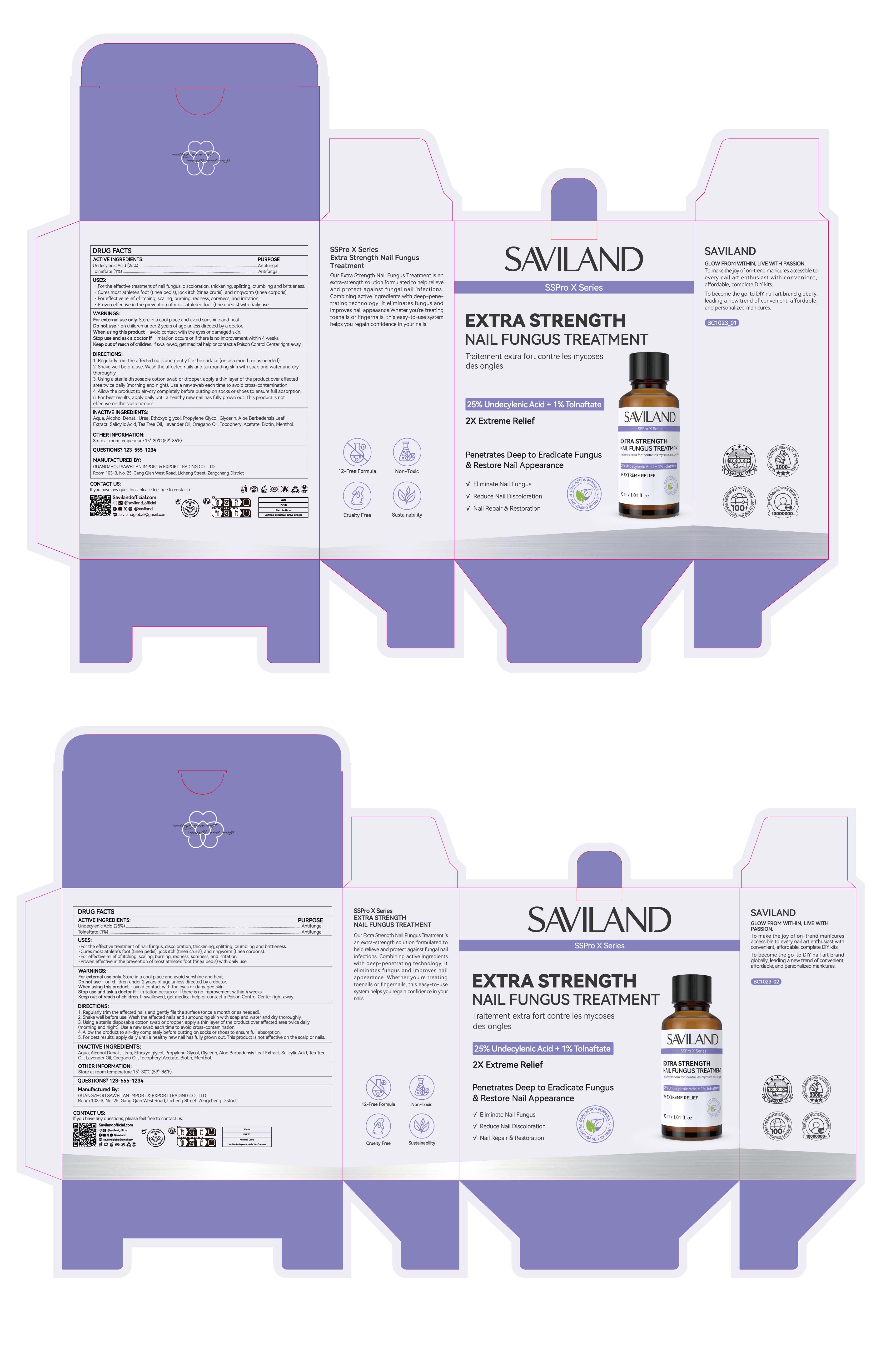
Drug Facts
Composition & Product
Identifiers & Packaging
Description
UNDECYLENIC ACID 25% TOLNAFTATE 1%
Medication Information
Warnings and Precautions
For external use only, Store in a cool place and avoid sunshine and heat.
Active Ingredient
UNDECYLENIC ACID 25%
TOLNAFTATE 1%
Indications and Usage
For the effective treatment of nail fungus, discoloration, thickening, splitting, crumbling and brittleness
Cures most athlete's foot (tinea pedis), jock itch (tinea cruris), and ringworm (tinea corporis).
For effective relief of itching, scaling, burning, redness, soreness, and irritation.
Proven effective in the prevention of most athlete's foot (tinea pedis) with daily use.
Dosage and Administration
1. Regularly trim the affected nails and gently file the surface (once a month or as needed).
2. Shake well before use. Wash the affected nails and surrounding skin with soap and water and dry thoroughly.
3. Using a sterile disposable cotton swab or dropper, apply a thin layer of the product over affectedarea twice daily (morning and night). Use a new swab each time to avoid cross-contamination.
4. Allow the product to air-dry completely before putting on socks or shoes to ensure full absorption.
5. For best results, apply daily until a healthy new nail has fully grown out. This product is noteffective on the scalp or nails.
Description
UNDECYLENIC ACID 25% TOLNAFTATE 1%
Section 50565-1
Keep out of reach of children. lf swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if . irritation occurs or if there is no improvement within 4 weeks.
Section 50567-7
When using this product · avoid contact with the eyes or damaged skin.
Section 50570-1
Do not use on children under 2 years of age unless directed by a doctor
Section 51727-6
UREA
ETHOXYDIGLYCOL
PROPYLENE GLYCOL
ALCOHOL DENAT.
TEA TREE OIL
OREGANO OIL
LAVENDER OIL
MENTHOL
ALOE BARBADENSIS LEAF EXTRACT
SALICYLIC ACID
BIOTIN/VITAMIN H
TOCOPHEROL ACETATE / VITAMIN E
AQUA
GLYCERIN
Section 51945-4
Section 55105-1
Antifungal
Structured Label Content
Warnings and Precautions (34071-1)
For external use only, Store in a cool place and avoid sunshine and heat.
Active Ingredient (55106-9)
UNDECYLENIC ACID 25%
TOLNAFTATE 1%
Indications and Usage (34067-9)
For the effective treatment of nail fungus, discoloration, thickening, splitting, crumbling and brittleness
Cures most athlete's foot (tinea pedis), jock itch (tinea cruris), and ringworm (tinea corporis).
For effective relief of itching, scaling, burning, redness, soreness, and irritation.
Proven effective in the prevention of most athlete's foot (tinea pedis) with daily use.
Dosage and Administration (34068-7)
1. Regularly trim the affected nails and gently file the surface (once a month or as needed).
2. Shake well before use. Wash the affected nails and surrounding skin with soap and water and dry thoroughly.
3. Using a sterile disposable cotton swab or dropper, apply a thin layer of the product over affectedarea twice daily (morning and night). Use a new swab each time to avoid cross-contamination.
4. Allow the product to air-dry completely before putting on socks or shoes to ensure full absorption.
5. For best results, apply daily until a healthy new nail has fully grown out. This product is noteffective on the scalp or nails.
Section 50565-1 (50565-1)
Keep out of reach of children. lf swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if . irritation occurs or if there is no improvement within 4 weeks.
Section 50567-7 (50567-7)
When using this product · avoid contact with the eyes or damaged skin.
Section 50570-1 (50570-1)
Do not use on children under 2 years of age unless directed by a doctor
Section 51727-6 (51727-6)
UREA
ETHOXYDIGLYCOL
PROPYLENE GLYCOL
ALCOHOL DENAT.
TEA TREE OIL
OREGANO OIL
LAVENDER OIL
MENTHOL
ALOE BARBADENSIS LEAF EXTRACT
SALICYLIC ACID
BIOTIN/VITAMIN H
TOCOPHEROL ACETATE / VITAMIN E
AQUA
GLYCERIN
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Antifungal
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:54.389880 · Updated: 2026-03-14T23:09:57.827589