Doxazosin
3ab222b7-74cc-5343-e063-6294a90a9616
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Doxazosin is an alpha 1 adrenergic antagonist indicated for: Signs and symptoms of Benign Prostatic Hyperplasia (BPH) Treatment of Hypertension
Dosage and Administration
For the treatment of BPH: Initiate therapy at 1 mg once daily. Dose maybe titrated at 1 to 2-week intervals, up to 8 mg once daily. ( 2.2 ) For the treatment hypertension: Initiate therapy at 1 mg once daily. Dose may be titrated as needed, up to 16 mg once daily. ( 2.3 )
Contraindications
The use of Doxazosin tablet is contraindicated in patients with a hypersensitivity to doxazosin, other quinazolines (e.g., prazosin, terazosin), or any of its components.
Warnings and Precautions
Postural hypotension with or without syncope may occur. ( 5.1 ) Risk of Intraoperative Floppy Iris Syndrome during cataract surgery. ( 5.2 ) Screen for the presence of prostate cancer prior to treatment for BPH and at regular intervals afterwards. ( 5.3 )
Adverse Reactions
The most commonly reported adverse reactions from clinical trials are Fatigue, malaise, hypotension, and dizziness. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Greenstone LLC Professional Information Services at 1-800-438-1985 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Strong cytochrome P450 (CYP) 3A inhibitors may increase exposure to doxazosin and increased risk of hypotension. ( 7.1 ) Concomitant administration of Doxazosin with a phosphodiesterase-5 (PDE-5) inhibitor can result in additive blood pressure lowering effects and symptomatic hypotension. ( 7.2 )
How Supplied
Doxazosin is available as tablets for oral administration. Each tablet contains Doxazosin mesylate equivalent to 1 mg (white), 2 mg (yellow or white), 4 mg (orange or white) or 8 mg (green or white) of doxazosin as the free base. NDC and Pack Size Strength Description NDC 59762-2310-6 (Bottle of 100) 1 mg White, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "1 mg" on the other side. NDC 59762-2410-7 (Bottle of 100) 1 mg White, round tablet engraved "CN1" on one side and "Pfizer" on the other side. NDC 59762-2320-6 (Bottle of 100) 2 mg Yellow, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "2 mg" on the other side. NDC 59762-2420-7 (Bottle of 100) 2 mg White, capsule shaped tablet with break score and engraved "CN2" on one side and "Pfizer" on the other side. NDC 59762-2340-6 (Bottle of 100) 4 mg Orange, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "4 mg" on the other side. NDC 59762-2440-7 (Bottle of 100) 4 mg White, diamond shaped tablet with break score and engraved "CN4" on one side and "Pfizer" on the other side. NDC 59762-2380-6 (Bottle of 100) 8 mg Green, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "8 mg" on the other side. NDC 59762-2480-7 (Bottle of 100) 8 mg White, capsule shaped tablet with break score and engraved "CN8" on one side and "Pfizer" on the other side.
Storage and Handling
Doxazosin is available as tablets for oral administration. Each tablet contains Doxazosin mesylate equivalent to 1 mg (white), 2 mg (yellow or white), 4 mg (orange or white) or 8 mg (green or white) of doxazosin as the free base. NDC and Pack Size Strength Description NDC 59762-2310-6 (Bottle of 100) 1 mg White, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "1 mg" on the other side. NDC 59762-2410-7 (Bottle of 100) 1 mg White, round tablet engraved "CN1" on one side and "Pfizer" on the other side. NDC 59762-2320-6 (Bottle of 100) 2 mg Yellow, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "2 mg" on the other side. NDC 59762-2420-7 (Bottle of 100) 2 mg White, capsule shaped tablet with break score and engraved "CN2" on one side and "Pfizer" on the other side. NDC 59762-2340-6 (Bottle of 100) 4 mg Orange, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "4 mg" on the other side. NDC 59762-2440-7 (Bottle of 100) 4 mg White, diamond shaped tablet with break score and engraved "CN4" on one side and "Pfizer" on the other side. NDC 59762-2380-6 (Bottle of 100) 8 mg Green, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "8 mg" on the other side. NDC 59762-2480-7 (Bottle of 100) 8 mg White, capsule shaped tablet with break score and engraved "CN8" on one side and "Pfizer" on the other side.
Description
Doxazosin is an alpha 1 adrenergic antagonist indicated for: Signs and symptoms of Benign Prostatic Hyperplasia (BPH) Treatment of Hypertension
Medication Information
Warnings and Precautions
Postural hypotension with or without syncope may occur. ( 5.1 ) Risk of Intraoperative Floppy Iris Syndrome during cataract surgery. ( 5.2 ) Screen for the presence of prostate cancer prior to treatment for BPH and at regular intervals afterwards. ( 5.3 )
Indications and Usage
Doxazosin is an alpha 1 adrenergic antagonist indicated for: Signs and symptoms of Benign Prostatic Hyperplasia (BPH) Treatment of Hypertension
Dosage and Administration
For the treatment of BPH: Initiate therapy at 1 mg once daily. Dose maybe titrated at 1 to 2-week intervals, up to 8 mg once daily. ( 2.2 ) For the treatment hypertension: Initiate therapy at 1 mg once daily. Dose may be titrated as needed, up to 16 mg once daily. ( 2.3 )
Contraindications
The use of Doxazosin tablet is contraindicated in patients with a hypersensitivity to doxazosin, other quinazolines (e.g., prazosin, terazosin), or any of its components.
Adverse Reactions
The most commonly reported adverse reactions from clinical trials are Fatigue, malaise, hypotension, and dizziness. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Greenstone LLC Professional Information Services at 1-800-438-1985 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Strong cytochrome P450 (CYP) 3A inhibitors may increase exposure to doxazosin and increased risk of hypotension. ( 7.1 ) Concomitant administration of Doxazosin with a phosphodiesterase-5 (PDE-5) inhibitor can result in additive blood pressure lowering effects and symptomatic hypotension. ( 7.2 )
Storage and Handling
Doxazosin is available as tablets for oral administration. Each tablet contains Doxazosin mesylate equivalent to 1 mg (white), 2 mg (yellow or white), 4 mg (orange or white) or 8 mg (green or white) of doxazosin as the free base. NDC and Pack Size Strength Description NDC 59762-2310-6 (Bottle of 100) 1 mg White, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "1 mg" on the other side. NDC 59762-2410-7 (Bottle of 100) 1 mg White, round tablet engraved "CN1" on one side and "Pfizer" on the other side. NDC 59762-2320-6 (Bottle of 100) 2 mg Yellow, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "2 mg" on the other side. NDC 59762-2420-7 (Bottle of 100) 2 mg White, capsule shaped tablet with break score and engraved "CN2" on one side and "Pfizer" on the other side. NDC 59762-2340-6 (Bottle of 100) 4 mg Orange, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "4 mg" on the other side. NDC 59762-2440-7 (Bottle of 100) 4 mg White, diamond shaped tablet with break score and engraved "CN4" on one side and "Pfizer" on the other side. NDC 59762-2380-6 (Bottle of 100) 8 mg Green, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "8 mg" on the other side. NDC 59762-2480-7 (Bottle of 100) 8 mg White, capsule shaped tablet with break score and engraved "CN8" on one side and "Pfizer" on the other side.
How Supplied
Doxazosin is available as tablets for oral administration. Each tablet contains Doxazosin mesylate equivalent to 1 mg (white), 2 mg (yellow or white), 4 mg (orange or white) or 8 mg (green or white) of doxazosin as the free base. NDC and Pack Size Strength Description NDC 59762-2310-6 (Bottle of 100) 1 mg White, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "1 mg" on the other side. NDC 59762-2410-7 (Bottle of 100) 1 mg White, round tablet engraved "CN1" on one side and "Pfizer" on the other side. NDC 59762-2320-6 (Bottle of 100) 2 mg Yellow, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "2 mg" on the other side. NDC 59762-2420-7 (Bottle of 100) 2 mg White, capsule shaped tablet with break score and engraved "CN2" on one side and "Pfizer" on the other side. NDC 59762-2340-6 (Bottle of 100) 4 mg Orange, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "4 mg" on the other side. NDC 59762-2440-7 (Bottle of 100) 4 mg White, diamond shaped tablet with break score and engraved "CN4" on one side and "Pfizer" on the other side. NDC 59762-2380-6 (Bottle of 100) 8 mg Green, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "8 mg" on the other side. NDC 59762-2480-7 (Bottle of 100) 8 mg White, capsule shaped tablet with break score and engraved "CN8" on one side and "Pfizer" on the other side.
Description
Doxazosin is an alpha 1 adrenergic antagonist indicated for: Signs and symptoms of Benign Prostatic Hyperplasia (BPH) Treatment of Hypertension
Section 42229-5
Benign Prostatic Hyperplasia (BPH)
The incidence of adverse events has been ascertained from worldwide clinical trials in 965 BPH patients. The incidence rates presented below (Table 2) are based on combined data from seven placebo-controlled trials involving once-daily administration of Doxazosin tablet in doses of 1 to 16 mg in hypertensives and 0.5 to 8 mg in normotensives. Adverse reactions occurring more than 1% more frequently in BPH patients treated with Doxazosin vs placebo are summarized in Table 1.
| BODY SYSTEM |
Doxazosin
N=665 |
Placebo
N=300 |
|---|---|---|
| NERVOUS SYSTEM DISORDERS | ||
|
Dizziness Includes vertigo
|
15.6% |
9.0% |
|
Somnolence |
3.0% |
1.0% |
|
CARDIAC DISORDERS |
||
|
Hypotension |
1.7% |
0% |
|
RESPIRATORY, THORACIC AND MEDIASTINAL DISORDERS |
||
|
Dyspnoea |
2.6% |
0.3% |
|
GASTROINTESTINAL DISORDERS |
||
|
Dry Mouth |
1.4% |
0.3% |
|
GENERAL DISORDERS AND ADMINISTRATION SITE CONDITIONS |
||
|
Fatigue |
8.0% |
1.7% |
|
Oedema |
2.7% |
0.7% |
Other adverse reactions occurring less than 1% more frequently in BPH patients treated with Doxazosin vs placebo but plausibly related to Doxazosin include: palpitations.
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration | |||
| Revised: 12/2020 | |||
|
PATIENT INFORMATION
|
|||
|
What is Doxazosin? Doxazosin is a prescription medicine that contains Doxazosin and is called an "alpha-blocker". Doxazosin is used to treat:
It is not known if Doxazosin is safe and effective in children. |
|||
|
Who should not take Doxazosin? Do not take Doxazosin if you:
|
|||
|
What should I tell my healthcare provider before taking Doxazosin? Before taking Doxazosin, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. Doxazosin may affect the way other medicines work, and other medicines may affect the way Doxazosin works causing side effects. Especially tell your healthcare provider if you take:
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
|
How should I take Doxazosin?
|
|||
|
What should I avoid while taking Doxazosin? Do not drive or perform any hazardous task until at least 24 hours after you have taken Doxazosin if you are taking:
|
|||
|
What are the possible side effects of Doxazosin? Doxazosin may cause serious side effects, including:
The most common side effects of Doxazosin are: |
|||
|
|
||
|
Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of Doxazosin. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
General information about the safe and effective use of Doxazosin. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Doxazosin for a condition for which it was not prescribed. Do not give Doxazosin to other people, even if they have the same symptoms you have. It may harm them. This Patient Information leaflet summarizes the most important information about Doxazosin. For more information, ask your healthcare provider. You can ask your healthcare provider or pharmacist for information that is written for healthcare professionals. |
|||
|
What are the ingredients in Doxazosin? The size, shape and appearance of the tablet that you receive may vary, all the tablets have the same active ingredient, and this will not affect the way that the medicine works. You can identify the tablet that you have from the following information. |
|||
|
Strength |
Either |
Or |
|
|
1 mg Tablet |
White, round tablet marked "CN1" on one side and "Pfizer" on the other side. |
White, capsule shaped tablet marked "Cardura" on one side, scored and marked "1 mg" on the other side. |
|
|
2 mg Tablet |
White, capsule shaped tablet with break score and marked "CN2" on one side and "Pfizer" on the other side. |
Yellow, capsule shaped tablet marked "Cardura" on one side, scored and marked "2 mg" on the other side. |
|
|
4 mg Tablet |
White, diamond shaped tablet with break score and marked "CN4" on one side and "Pfizer" on the other side. |
Orange, capsule shaped tablet marked "Cardura" on one side, scored and marked "4 mg" on the other side. |
|
|
8 mg Tablet |
White, capsule shaped tablet with break score and marked "CN8" on one side and "Pfizer" on the other side. |
Green, capsule shaped tablet marked "Cardura" on one side, scored and marked "8 mg" on the other side. |
|
|
Active ingredient:Doxazosin Inactive ingredients:microcrystalline cellulose, lactose, sodium starch glycolate, magnesium stearate and sodium lauryl sulfate. The 2 mg yellow tablet contains D & C yellow 10 and FD & C yellow 6; the 4 mg orange tablet contains FD & C yellow 6; the 8 mg green tablet contains FD & C blue 2 and D & C yellow 10. LAB-0720-4.0 For more information, go to www.greenstonellc.com or call 1-800-438-1985. |
Section 44425-7
Recommended Storage: Store at 25°C (77°F); excursions permitted from 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
5.4 Priapism
Alpha1 antagonists, including doxazosin, have been associated with priapism (painful penile erection, sustained for hours and unrelieved by sexual intercourse or masturbation). This condition can lead to permanent impotence if not promptly treated.
10 Overdosage
Experience with Doxazosin tablet overdosage is limited. Two adolescents, who each intentionally ingested 40 mg Doxazosin tablet with diclofenac or acetaminophen, were treated with gastric lavage with activated charcoal and made full recoveries. A two-year-old child who accidently ingested 4 mg Doxazosin tablet was treated with gastric lavage and remained normotensive during the five-hour emergency room observation period. A six-month-old child accidentally received a crushed 1 mg tablet of Doxazosin tablet and was reported to have been drowsy. A 32-year-old female with chronic renal failure, epilepsy, and depression intentionally ingested 60 mg Doxazosin tablet (blood level = 0.9 mcg/mL; normal values in hypertensives = 0.02 mcg/mL); death was attributed to a grand mal seizure resulting from hypotension. A 39-year-old female who ingested 70 mg Doxazosin tablet, alcohol, and Dalmane ®(flurazepam) developed hypotension which responded to fluid therapy.
The oral LD 50of doxazosin is greater than 1000 mg/kg in mice and rats. The most likely manifestation of overdosage would be hypotension, for which the usual treatment would be intravenous infusion of fluid. As doxazosin is highly protein bound, dialysis would not be indicated.
11 Description
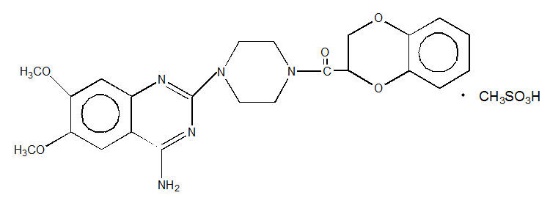
Doxazosin is a quinazoline compound that is a selective inhibitor of the alpha 1subtype of alpha-adrenergic receptors. The chemical name of Doxazosin is 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-(1,4-benzodioxan-2-ylcarbonyl) piperazine methanesulfonate. The empirical formula for Doxazosin is C 23H 25N 5O 5∙ CH 4O 3S and the molecular weight is 547.6. It has the following structure:
Doxazosin is freely soluble in dimethylsulfoxide, soluble in dimethylformamide, slightly soluble in methanol, ethanol, and water (0.8% at 25°C), and very slightly soluble in acetone and methylene chloride. Doxazosin tablet is available as colored tablets for oral use and contains doxazosin mesylate equivalent to 1 mg (white), 2 mg (yellow or white), 4 mg (orange or white) and 8 mg (green or white) of doxazosin as the free base.
The inactive ingredients for all tablets are microcrystalline cellulose, lactose, sodium starch glycolate, magnesium stearate and sodium lauryl sulfate. The 2 mg yellow tablet contains D & C yellow 10 and FD & C yellow 6; the 4 mg orange tablet contains FD & C yellow 6; the 8 mg green tablet contains FD & C blue 2 and D & C yellow 10.
1.2 Hypertension
Doxazosin tablet is indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes, including this drug.
Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than one drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program's Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC).
Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly.
Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal.
Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy.
Doxazosin tablet may be used alone or in combination with other antihypertensives.
2.3 Hypertension
The initial dosage of Doxazosin tablet is 1 mg given once daily. Daily dosage may be doubled up 16 mg once daily, as needed, to achieve the desired reduction in blood pressure.
14.2 Hypertension
In a pooled analysis of placebo-controlled hypertension studies with about 300 hypertensive patients per treatment group, doxazosin, at doses of 1 to 16 mg given once daily, lowered blood pressure at 24 hours by about 10/8 mmHg compared to placebo in the standing position and about 9/5 mmHg in the supine position. Peak blood pressure effects (1–6 hours) were larger by about 50–75% (i.e., trough values were about 55–70% of peak effect), with the larger peak-trough differences seen in systolic pressures. There was no apparent difference in the blood pressure response of Caucasians and blacks or of patients above and below age 65. In the same patient population, patients receiving Doxazosin gained a mean of 0.6 kg compared to a mean loss of 0.1 kg for placebo patients.
| PLACEBO(N=85) | Doxazosin(N=183) | |||
|---|---|---|---|---|
|
Sitting BP (mmHg) |
Baseline |
Chang e |
Baseline |
Chang e |
|
Systolic |
128.4 |
–1.4 |
128.8 |
–4.9 p ≤0.05 compared to placebo
|
|
Diastolic |
79.2 |
–1.2 |
79.6 |
–2.4 |
|
Standing BP (mmHg) |
Baseline |
Chang e |
Baseline |
Chang e |
|
Systolic |
128.5 |
–0.6 |
128.5 |
–5.3 |
|
Diastolic |
80.5 |
–0.7 |
80.4 |
–2.6 |
8.4 Pediatric Use
The safety and effectiveness of Doxazosin have not been established in children.
4 Contraindications
The use of Doxazosin tablet is contraindicated in patients with a hypersensitivity to doxazosin, other quinazolines (e.g., prazosin, terazosin), or any of its components.
5.3 Prostate Cancer
Carcinoma of the prostate causes many of the symptoms associated with BPH and the two disorders frequently co-exist. Carcinoma of the prostate should therefore be ruled out prior to commencing therapy with Doxazosin tablet for the treatment of BPH.
6 Adverse Reactions
The most commonly reported adverse reactions from clinical trials are Fatigue, malaise, hypotension, and dizziness. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Greenstone LLC Professional Information Services at 1-800-438-1985 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
5.2 Cataract Surgery
Intraoperative Floppy Iris Syndrome (IFIS) has been observed during cataract surgery in some patients on or previously treated with alpha1 blockers. This variant of small pupil syndrome is characterized by the combination of a flaccid iris that billows in response to intraoperative irrigation currents, progressive intraoperative miosis despite preoperative dilation with standard mydriatic drugs, and potential prolapse of the iris toward the phacoemulsification incisions. The patient's surgeon should be prepared for possible modifications to their surgical technique, such as the utilization of iris hooks, iris dilator rings, or viscoelastic substances. There does not appear to be a benefit of stopping alpha1 blocker therapy prior to cataract surgery.
7.1 Cyp 3a Inhibitors
In vitrostudies suggest that doxazosin is a substrate of CYP 3A4. Strong CYP3A inhibitors may increase exposure to doxazosin. Monitor blood pressure and for symptoms of hypotension when Doxazosin is used concomitantly with strong CYP3A inhibitors [ see Clinical Pharmacology (12.3) ].
2.1 Dosing Information
Following the initial dose and with each dose increase of Doxazosin, monitor blood pressure for at least 6 hours following administration. If Doxazosin tablet administration is discontinued for several days, therapy should be restarted using the initial dosing regimen.
8.6 Hepatic Impairment
Doxazosin is extensively metabolized in the liver. Hepatic impairment is expected to increase exposure to doxazosin. Use of Doxazosin in patients with severe hepatic impairment (Child-Pugh Class C) is not recommended. Monitor blood pressure and for symptoms of hypotension in patients with lesser degrees of hepatic impairment (Child-Pugh Class A and B) [ see Clinical Pharmacology (12.3) ].
1 Indications and Usage
Doxazosin is an alpha 1adrenergic antagonist indicated for:
- Signs and symptoms of Benign Prostatic Hyperplasia (BPH)
- Treatment of Hypertension
Repackaging Information
Please reference the HOW SUPPLIED section listed above for a description of individual drug products listed below. This drug product has been received by Aphena Pharma Solutions - Tennessee, LLC in a manufacturer or distributor packaged configuration and repackaged in full compliance with all applicable cGMP regulations. The package configurations available from Aphena are listed below:
4mg
NDC 71610-927-53, Bottles of 60 Tablets
NDC 71610-927-60, Bottles of 90 Tablets
NDC 71610-927-80, Bottles of 180 Tablets
Store between 20°-25°C (68°-77°F). See USP Controlled Room Temperature. Dispense in a tight light-resistant container as defined by USP. Keep this and all drugs out of the reach of children.
Repackaged by:
Cookeville, TN 38506
20250724AMH
5.1 Postural Hypotension
Postural hypotension with or without symptoms (e.g., dizziness) may develop within a few hours following administration of Doxazosin. However, infrequently, symptomatic postural hypotension has also been reported later than a few hours after dosing. As with other alpha-blockers, there is a potential for syncope, especially after the initial dose or after an increase in dosage strength. Advise patient how to avoid symptoms resulting from postural hypotension and what measures to take should they develop.
Concomitant administration of Doxazosin with a PDE-5 inhibitor can result in additive blood pressure lowering effects and symptomatic hypotension.
5 Warnings and Precautions
2 Dosage and Administration
3 Dosage Forms and Strengths
Tablets: 1 mg (white), 2 mg (yellow or white), 4 mg (orange or white) or 8 mg (green or white).
Each tablet contains doxazosin mesylate equivalent to 1 mg, 2 mg, 4 mg or 8 mg doxazosin (free base).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Doxazosin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
In post-marketing experience, the following additional adverse reactions have been reported:
Blood and Lymphatic System Disorders:leukopenia, thrombocytopenia;
Immune System Disorders:allergic reaction;
Nervous System Disorders:hypoesthesia;
Eye Disorders:Intraoperative Floppy Iris Syndrome [see Warnings and precautions (5.4)] ;
Cardiac Disorders:bradycardia;
Respiratory, Thoracic and Mediastinal Disorders:bronchospasm aggravated;
Gastrointestinal Disorders:vomiting;
Hepatobiliary Disorders:cholestasis, hepatitis cholestatic;
Skin and Subcutaneous Tissue Disorders:urticaria;
Musculoskeletal and Connective Tissue Disorders:muscle cramps, muscle weakness;
Renal and Urinary Disorders:hematuria, micturition disorder, micturition frequency, nocturia;
Reproductive System and Breast Disorders:gynecomastia, priapism.
Principal Display Panel 4mg
NDC 71610-927 - Doxazosin 4mg Tablets - Rx Only
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
2.2 Benign Prostatic Hyperplasia
The recommended initial dosage of Doxazosin tablet is 1 mg given once daily either in the morning or evening.
Depending on the individual patient's urodynamics and BPH symptomatology, the dose may be titrated at 1 to 2 week intervals to 2 mg, and thereafter to 4 mg and 8 mg once daily. The maximum recommended dose for BPH is 8 mg once daily.
Routinely monitor blood pressure in these patients.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
Doxazosin is available as tablets for oral administration. Each tablet contains Doxazosin mesylate equivalent to 1 mg (white), 2 mg (yellow or white), 4 mg (orange or white) or 8 mg (green or white) of doxazosin as the free base.
| NDC and Pack Size | Strength | Description |
|---|---|---|
|
NDC 59762-2310-6 (Bottle of 100) |
1 mg |
White, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "1 mg" on the other side. |
|
NDC 59762-2410-7 (Bottle of 100) |
1 mg |
White, round tablet engraved "CN1" on one side and "Pfizer" on the other side. |
|
NDC 59762-2320-6 (Bottle of 100) |
2 mg |
Yellow, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "2 mg" on the other side. |
|
NDC 59762-2420-7 (Bottle of 100) |
2 mg |
White, capsule shaped tablet with break score and engraved "CN2" on one side and "Pfizer" on the other side. |
|
NDC 59762-2340-6 (Bottle of 100) |
4 mg |
Orange, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "4 mg" on the other side. |
|
NDC 59762-2440-7 (Bottle of 100) |
4 mg |
White, diamond shaped tablet with break score and engraved "CN4" on one side and "Pfizer" on the other side. |
|
NDC 59762-2380-6 (Bottle of 100) |
8 mg |
Green, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "8 mg" on the other side. |
|
NDC 59762-2480-7 (Bottle of 100) |
8 mg |
White, capsule shaped tablet with break score and engraved "CN8" on one side and "Pfizer" on the other side. |
1.1 Benign Prostatic Hyperplasia (bph)
Doxazosin tablet is indicated for the treatment of the signs and symptoms of BPH.
13.2 Animal Toxicology and Pharmacology
An increased incidence of myocardial necrosis or fibrosis was observed in long-term (6–12 months) studies in rats and mice (exposure 8 times human AUC exposure in rats and somewhat equivalent to human C maxexposure in mice). Findings were not seen at lower doses. In dogs no cardiotoxicity was observed following 12 months of oral dosing at doses that resulted in maximum plasma concentrations (C max)14 times the C maxexposure in humans receiving a 12 mg/day therapeutic dose or in Wistar rats at C maxexposures 15 times human C maxexposure. There is no evidence that similar lesions occur in humans.
14.1 Benign Prostatic Hyperplasia (bph)
The efficacy of Doxazosin tablet was evaluated extensively in over 900 patients with BPH in double-blind, placebo-controlled trials. Doxazosin tablet treatment was superior to placebo in improving patient symptoms and urinary flow rate. Significant relief with Doxazosin tablet was seen as early as one week into the treatment regimen, with Doxazosin tablet -treated patients (N=173) showing a significant (p<0.01) increase in maximum flow rate of 0.8 mL/sec compared to a decrease of 0.5 mL/sec in the placebo group (N=41). In long-term studies, improvement was maintained for up to 2 years of treatment. In 66–71% of patients, improvements above baseline were seen in both symptoms and maximum urinary flow rate.
In three placebo-controlled studies of 14–16 weeks' duration, obstructive symptoms (hesitation, intermittency, dribbling, weak urinary stream, incomplete emptying of the bladder) and irritative symptoms (nocturia, daytime frequency, urgency, burning) of BPH were evaluated at each visit by patient-assessed symptom questionnaires. The bothersomeness of symptoms was measured with a modified Boyarsky questionnaire. Symptom severity/frequency was assessed using a modified Boyarsky questionnaire or an AUA-based questionnaire. Uroflowmetric evaluations were performed at times of peak (2–6 hours post-dose) and/or trough (24 hours post-dose) plasma concentrations of Doxazosin tablet.
The results from the three placebo-controlled studies (N=609) showing significant efficacy with 4 mg and 8 mg doxazosin are summarized in Table 3. In all three studies, Doxazosin tablet resulted in statistically significant relief of obstructive and irritative symptoms compared to placebo. Statistically significant improvements of 2.3–3.3 mL/sec in maximum flow rate were seen with Doxazosin tablet in Studies 1 and 2, compared to 0.1–0.7 mL/sec with placebo.
|
Table 3 |
In one fixed-dose study (Study 2), Doxazosin tablet therapy (4 to 8 mg, once daily) resulted in a significant and sustained improvement in maximum urinary flow rate of 2.3–3.3 mL/sec (Table 3) compared to placebo (0.1 mL/sec). In this study, the only study in which weekly evaluations were made, significant improvement with Doxazosin tablet vs. placebo was seen after one week. The proportion of patients who responded with a maximum flow rate improvement of ≥3 mL/sec was significantly larger with Doxazosin tablet (34–42%) than placebo (13–17%). A significantly greater improvement was also seen in average flow rate with Doxazosin tablet (1.6 mL/sec) than with placebo (0.2 mL/sec). The onset and time course of symptom relief and increased urinary flow from Study 1 are illustrated in Figure 1.
|
Figure 1 – Study 1 |
7.2 Phosphodiesterase 5 (pde 5) Inhibitors
Concomitant administration of Doxazosin with a phosphodiesterase-5 (PDE-5) inhibitor can result in additive blood pressure lowering effects and symptomatic hypotension. Monitor blood pressure and for symptoms of hypotension [ see Warnings and Precautions (5.1) ].
Structured Label Content
Section 42229-5 (42229-5)
Benign Prostatic Hyperplasia (BPH)
The incidence of adverse events has been ascertained from worldwide clinical trials in 965 BPH patients. The incidence rates presented below (Table 2) are based on combined data from seven placebo-controlled trials involving once-daily administration of Doxazosin tablet in doses of 1 to 16 mg in hypertensives and 0.5 to 8 mg in normotensives. Adverse reactions occurring more than 1% more frequently in BPH patients treated with Doxazosin vs placebo are summarized in Table 1.
| BODY SYSTEM |
Doxazosin
N=665 |
Placebo
N=300 |
|---|---|---|
| NERVOUS SYSTEM DISORDERS | ||
|
Dizziness Includes vertigo
|
15.6% |
9.0% |
|
Somnolence |
3.0% |
1.0% |
|
CARDIAC DISORDERS |
||
|
Hypotension |
1.7% |
0% |
|
RESPIRATORY, THORACIC AND MEDIASTINAL DISORDERS |
||
|
Dyspnoea |
2.6% |
0.3% |
|
GASTROINTESTINAL DISORDERS |
||
|
Dry Mouth |
1.4% |
0.3% |
|
GENERAL DISORDERS AND ADMINISTRATION SITE CONDITIONS |
||
|
Fatigue |
8.0% |
1.7% |
|
Oedema |
2.7% |
0.7% |
Other adverse reactions occurring less than 1% more frequently in BPH patients treated with Doxazosin vs placebo but plausibly related to Doxazosin include: palpitations.
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration | |||
| Revised: 12/2020 | |||
|
PATIENT INFORMATION
|
|||
|
What is Doxazosin? Doxazosin is a prescription medicine that contains Doxazosin and is called an "alpha-blocker". Doxazosin is used to treat:
It is not known if Doxazosin is safe and effective in children. |
|||
|
Who should not take Doxazosin? Do not take Doxazosin if you:
|
|||
|
What should I tell my healthcare provider before taking Doxazosin? Before taking Doxazosin, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. Doxazosin may affect the way other medicines work, and other medicines may affect the way Doxazosin works causing side effects. Especially tell your healthcare provider if you take:
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
|
How should I take Doxazosin?
|
|||
|
What should I avoid while taking Doxazosin? Do not drive or perform any hazardous task until at least 24 hours after you have taken Doxazosin if you are taking:
|
|||
|
What are the possible side effects of Doxazosin? Doxazosin may cause serious side effects, including:
The most common side effects of Doxazosin are: |
|||
|
|
||
|
Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of Doxazosin. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
General information about the safe and effective use of Doxazosin. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Doxazosin for a condition for which it was not prescribed. Do not give Doxazosin to other people, even if they have the same symptoms you have. It may harm them. This Patient Information leaflet summarizes the most important information about Doxazosin. For more information, ask your healthcare provider. You can ask your healthcare provider or pharmacist for information that is written for healthcare professionals. |
|||
|
What are the ingredients in Doxazosin? The size, shape and appearance of the tablet that you receive may vary, all the tablets have the same active ingredient, and this will not affect the way that the medicine works. You can identify the tablet that you have from the following information. |
|||
|
Strength |
Either |
Or |
|
|
1 mg Tablet |
White, round tablet marked "CN1" on one side and "Pfizer" on the other side. |
White, capsule shaped tablet marked "Cardura" on one side, scored and marked "1 mg" on the other side. |
|
|
2 mg Tablet |
White, capsule shaped tablet with break score and marked "CN2" on one side and "Pfizer" on the other side. |
Yellow, capsule shaped tablet marked "Cardura" on one side, scored and marked "2 mg" on the other side. |
|
|
4 mg Tablet |
White, diamond shaped tablet with break score and marked "CN4" on one side and "Pfizer" on the other side. |
Orange, capsule shaped tablet marked "Cardura" on one side, scored and marked "4 mg" on the other side. |
|
|
8 mg Tablet |
White, capsule shaped tablet with break score and marked "CN8" on one side and "Pfizer" on the other side. |
Green, capsule shaped tablet marked "Cardura" on one side, scored and marked "8 mg" on the other side. |
|
|
Active ingredient:Doxazosin Inactive ingredients:microcrystalline cellulose, lactose, sodium starch glycolate, magnesium stearate and sodium lauryl sulfate. The 2 mg yellow tablet contains D & C yellow 10 and FD & C yellow 6; the 4 mg orange tablet contains FD & C yellow 6; the 8 mg green tablet contains FD & C blue 2 and D & C yellow 10. LAB-0720-4.0 For more information, go to www.greenstonellc.com or call 1-800-438-1985. |
Section 44425-7 (44425-7)
Recommended Storage: Store at 25°C (77°F); excursions permitted from 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
5.4 Priapism
Alpha1 antagonists, including doxazosin, have been associated with priapism (painful penile erection, sustained for hours and unrelieved by sexual intercourse or masturbation). This condition can lead to permanent impotence if not promptly treated.
10 Overdosage (10 OVERDOSAGE)
Experience with Doxazosin tablet overdosage is limited. Two adolescents, who each intentionally ingested 40 mg Doxazosin tablet with diclofenac or acetaminophen, were treated with gastric lavage with activated charcoal and made full recoveries. A two-year-old child who accidently ingested 4 mg Doxazosin tablet was treated with gastric lavage and remained normotensive during the five-hour emergency room observation period. A six-month-old child accidentally received a crushed 1 mg tablet of Doxazosin tablet and was reported to have been drowsy. A 32-year-old female with chronic renal failure, epilepsy, and depression intentionally ingested 60 mg Doxazosin tablet (blood level = 0.9 mcg/mL; normal values in hypertensives = 0.02 mcg/mL); death was attributed to a grand mal seizure resulting from hypotension. A 39-year-old female who ingested 70 mg Doxazosin tablet, alcohol, and Dalmane ®(flurazepam) developed hypotension which responded to fluid therapy.
The oral LD 50of doxazosin is greater than 1000 mg/kg in mice and rats. The most likely manifestation of overdosage would be hypotension, for which the usual treatment would be intravenous infusion of fluid. As doxazosin is highly protein bound, dialysis would not be indicated.
11 Description (11 DESCRIPTION)
Doxazosin is a quinazoline compound that is a selective inhibitor of the alpha 1subtype of alpha-adrenergic receptors. The chemical name of Doxazosin is 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-(1,4-benzodioxan-2-ylcarbonyl) piperazine methanesulfonate. The empirical formula for Doxazosin is C 23H 25N 5O 5∙ CH 4O 3S and the molecular weight is 547.6. It has the following structure:
Doxazosin is freely soluble in dimethylsulfoxide, soluble in dimethylformamide, slightly soluble in methanol, ethanol, and water (0.8% at 25°C), and very slightly soluble in acetone and methylene chloride. Doxazosin tablet is available as colored tablets for oral use and contains doxazosin mesylate equivalent to 1 mg (white), 2 mg (yellow or white), 4 mg (orange or white) and 8 mg (green or white) of doxazosin as the free base.
The inactive ingredients for all tablets are microcrystalline cellulose, lactose, sodium starch glycolate, magnesium stearate and sodium lauryl sulfate. The 2 mg yellow tablet contains D & C yellow 10 and FD & C yellow 6; the 4 mg orange tablet contains FD & C yellow 6; the 8 mg green tablet contains FD & C blue 2 and D & C yellow 10.
1.2 Hypertension
Doxazosin tablet is indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes, including this drug.
Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than one drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program's Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC).
Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly.
Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal.
Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy.
Doxazosin tablet may be used alone or in combination with other antihypertensives.
2.3 Hypertension
The initial dosage of Doxazosin tablet is 1 mg given once daily. Daily dosage may be doubled up 16 mg once daily, as needed, to achieve the desired reduction in blood pressure.
14.2 Hypertension
In a pooled analysis of placebo-controlled hypertension studies with about 300 hypertensive patients per treatment group, doxazosin, at doses of 1 to 16 mg given once daily, lowered blood pressure at 24 hours by about 10/8 mmHg compared to placebo in the standing position and about 9/5 mmHg in the supine position. Peak blood pressure effects (1–6 hours) were larger by about 50–75% (i.e., trough values were about 55–70% of peak effect), with the larger peak-trough differences seen in systolic pressures. There was no apparent difference in the blood pressure response of Caucasians and blacks or of patients above and below age 65. In the same patient population, patients receiving Doxazosin gained a mean of 0.6 kg compared to a mean loss of 0.1 kg for placebo patients.
| PLACEBO(N=85) | Doxazosin(N=183) | |||
|---|---|---|---|---|
|
Sitting BP (mmHg) |
Baseline |
Chang e |
Baseline |
Chang e |
|
Systolic |
128.4 |
–1.4 |
128.8 |
–4.9 p ≤0.05 compared to placebo
|
|
Diastolic |
79.2 |
–1.2 |
79.6 |
–2.4 |
|
Standing BP (mmHg) |
Baseline |
Chang e |
Baseline |
Chang e |
|
Systolic |
128.5 |
–0.6 |
128.5 |
–5.3 |
|
Diastolic |
80.5 |
–0.7 |
80.4 |
–2.6 |
8.4 Pediatric Use
The safety and effectiveness of Doxazosin have not been established in children.
4 Contraindications (4 CONTRAINDICATIONS)
The use of Doxazosin tablet is contraindicated in patients with a hypersensitivity to doxazosin, other quinazolines (e.g., prazosin, terazosin), or any of its components.
5.3 Prostate Cancer
Carcinoma of the prostate causes many of the symptoms associated with BPH and the two disorders frequently co-exist. Carcinoma of the prostate should therefore be ruled out prior to commencing therapy with Doxazosin tablet for the treatment of BPH.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most commonly reported adverse reactions from clinical trials are Fatigue, malaise, hypotension, and dizziness. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Greenstone LLC Professional Information Services at 1-800-438-1985 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
5.2 Cataract Surgery
Intraoperative Floppy Iris Syndrome (IFIS) has been observed during cataract surgery in some patients on or previously treated with alpha1 blockers. This variant of small pupil syndrome is characterized by the combination of a flaccid iris that billows in response to intraoperative irrigation currents, progressive intraoperative miosis despite preoperative dilation with standard mydriatic drugs, and potential prolapse of the iris toward the phacoemulsification incisions. The patient's surgeon should be prepared for possible modifications to their surgical technique, such as the utilization of iris hooks, iris dilator rings, or viscoelastic substances. There does not appear to be a benefit of stopping alpha1 blocker therapy prior to cataract surgery.
7.1 Cyp 3a Inhibitors (7.1 CYP 3A Inhibitors)
In vitrostudies suggest that doxazosin is a substrate of CYP 3A4. Strong CYP3A inhibitors may increase exposure to doxazosin. Monitor blood pressure and for symptoms of hypotension when Doxazosin is used concomitantly with strong CYP3A inhibitors [ see Clinical Pharmacology (12.3) ].
2.1 Dosing Information
Following the initial dose and with each dose increase of Doxazosin, monitor blood pressure for at least 6 hours following administration. If Doxazosin tablet administration is discontinued for several days, therapy should be restarted using the initial dosing regimen.
8.6 Hepatic Impairment
Doxazosin is extensively metabolized in the liver. Hepatic impairment is expected to increase exposure to doxazosin. Use of Doxazosin in patients with severe hepatic impairment (Child-Pugh Class C) is not recommended. Monitor blood pressure and for symptoms of hypotension in patients with lesser degrees of hepatic impairment (Child-Pugh Class A and B) [ see Clinical Pharmacology (12.3) ].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Doxazosin is an alpha 1adrenergic antagonist indicated for:
- Signs and symptoms of Benign Prostatic Hyperplasia (BPH)
- Treatment of Hypertension
Repackaging Information (REPACKAGING INFORMATION)
Please reference the HOW SUPPLIED section listed above for a description of individual drug products listed below. This drug product has been received by Aphena Pharma Solutions - Tennessee, LLC in a manufacturer or distributor packaged configuration and repackaged in full compliance with all applicable cGMP regulations. The package configurations available from Aphena are listed below:
4mg
NDC 71610-927-53, Bottles of 60 Tablets
NDC 71610-927-60, Bottles of 90 Tablets
NDC 71610-927-80, Bottles of 180 Tablets
Store between 20°-25°C (68°-77°F). See USP Controlled Room Temperature. Dispense in a tight light-resistant container as defined by USP. Keep this and all drugs out of the reach of children.
Repackaged by:
Cookeville, TN 38506
20250724AMH
5.1 Postural Hypotension
Postural hypotension with or without symptoms (e.g., dizziness) may develop within a few hours following administration of Doxazosin. However, infrequently, symptomatic postural hypotension has also been reported later than a few hours after dosing. As with other alpha-blockers, there is a potential for syncope, especially after the initial dose or after an increase in dosage strength. Advise patient how to avoid symptoms resulting from postural hypotension and what measures to take should they develop.
Concomitant administration of Doxazosin with a PDE-5 inhibitor can result in additive blood pressure lowering effects and symptomatic hypotension.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 1 mg (white), 2 mg (yellow or white), 4 mg (orange or white) or 8 mg (green or white).
Each tablet contains doxazosin mesylate equivalent to 1 mg, 2 mg, 4 mg or 8 mg doxazosin (free base).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Doxazosin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
In post-marketing experience, the following additional adverse reactions have been reported:
Blood and Lymphatic System Disorders:leukopenia, thrombocytopenia;
Immune System Disorders:allergic reaction;
Nervous System Disorders:hypoesthesia;
Eye Disorders:Intraoperative Floppy Iris Syndrome [see Warnings and precautions (5.4)] ;
Cardiac Disorders:bradycardia;
Respiratory, Thoracic and Mediastinal Disorders:bronchospasm aggravated;
Gastrointestinal Disorders:vomiting;
Hepatobiliary Disorders:cholestasis, hepatitis cholestatic;
Skin and Subcutaneous Tissue Disorders:urticaria;
Musculoskeletal and Connective Tissue Disorders:muscle cramps, muscle weakness;
Renal and Urinary Disorders:hematuria, micturition disorder, micturition frequency, nocturia;
Reproductive System and Breast Disorders:gynecomastia, priapism.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Principal Display Panel 4mg (PRINCIPAL DISPLAY PANEL - 4mg)
NDC 71610-927 - Doxazosin 4mg Tablets - Rx Only
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
2.2 Benign Prostatic Hyperplasia
The recommended initial dosage of Doxazosin tablet is 1 mg given once daily either in the morning or evening.
Depending on the individual patient's urodynamics and BPH symptomatology, the dose may be titrated at 1 to 2 week intervals to 2 mg, and thereafter to 4 mg and 8 mg once daily. The maximum recommended dose for BPH is 8 mg once daily.
Routinely monitor blood pressure in these patients.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Doxazosin is available as tablets for oral administration. Each tablet contains Doxazosin mesylate equivalent to 1 mg (white), 2 mg (yellow or white), 4 mg (orange or white) or 8 mg (green or white) of doxazosin as the free base.
| NDC and Pack Size | Strength | Description |
|---|---|---|
|
NDC 59762-2310-6 (Bottle of 100) |
1 mg |
White, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "1 mg" on the other side. |
|
NDC 59762-2410-7 (Bottle of 100) |
1 mg |
White, round tablet engraved "CN1" on one side and "Pfizer" on the other side. |
|
NDC 59762-2320-6 (Bottle of 100) |
2 mg |
Yellow, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "2 mg" on the other side. |
|
NDC 59762-2420-7 (Bottle of 100) |
2 mg |
White, capsule shaped tablet with break score and engraved "CN2" on one side and "Pfizer" on the other side. |
|
NDC 59762-2340-6 (Bottle of 100) |
4 mg |
Orange, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "4 mg" on the other side. |
|
NDC 59762-2440-7 (Bottle of 100) |
4 mg |
White, diamond shaped tablet with break score and engraved "CN4" on one side and "Pfizer" on the other side. |
|
NDC 59762-2380-6 (Bottle of 100) |
8 mg |
Green, capsule shaped tablet engraved "Cardura" on one side, scored and engraved "8 mg" on the other side. |
|
NDC 59762-2480-7 (Bottle of 100) |
8 mg |
White, capsule shaped tablet with break score and engraved "CN8" on one side and "Pfizer" on the other side. |
1.1 Benign Prostatic Hyperplasia (bph) (1.1 Benign Prostatic Hyperplasia (BPH))
Doxazosin tablet is indicated for the treatment of the signs and symptoms of BPH.
13.2 Animal Toxicology and Pharmacology
An increased incidence of myocardial necrosis or fibrosis was observed in long-term (6–12 months) studies in rats and mice (exposure 8 times human AUC exposure in rats and somewhat equivalent to human C maxexposure in mice). Findings were not seen at lower doses. In dogs no cardiotoxicity was observed following 12 months of oral dosing at doses that resulted in maximum plasma concentrations (C max)14 times the C maxexposure in humans receiving a 12 mg/day therapeutic dose or in Wistar rats at C maxexposures 15 times human C maxexposure. There is no evidence that similar lesions occur in humans.
14.1 Benign Prostatic Hyperplasia (bph) (14.1 Benign Prostatic Hyperplasia (BPH))
The efficacy of Doxazosin tablet was evaluated extensively in over 900 patients with BPH in double-blind, placebo-controlled trials. Doxazosin tablet treatment was superior to placebo in improving patient symptoms and urinary flow rate. Significant relief with Doxazosin tablet was seen as early as one week into the treatment regimen, with Doxazosin tablet -treated patients (N=173) showing a significant (p<0.01) increase in maximum flow rate of 0.8 mL/sec compared to a decrease of 0.5 mL/sec in the placebo group (N=41). In long-term studies, improvement was maintained for up to 2 years of treatment. In 66–71% of patients, improvements above baseline were seen in both symptoms and maximum urinary flow rate.
In three placebo-controlled studies of 14–16 weeks' duration, obstructive symptoms (hesitation, intermittency, dribbling, weak urinary stream, incomplete emptying of the bladder) and irritative symptoms (nocturia, daytime frequency, urgency, burning) of BPH were evaluated at each visit by patient-assessed symptom questionnaires. The bothersomeness of symptoms was measured with a modified Boyarsky questionnaire. Symptom severity/frequency was assessed using a modified Boyarsky questionnaire or an AUA-based questionnaire. Uroflowmetric evaluations were performed at times of peak (2–6 hours post-dose) and/or trough (24 hours post-dose) plasma concentrations of Doxazosin tablet.
The results from the three placebo-controlled studies (N=609) showing significant efficacy with 4 mg and 8 mg doxazosin are summarized in Table 3. In all three studies, Doxazosin tablet resulted in statistically significant relief of obstructive and irritative symptoms compared to placebo. Statistically significant improvements of 2.3–3.3 mL/sec in maximum flow rate were seen with Doxazosin tablet in Studies 1 and 2, compared to 0.1–0.7 mL/sec with placebo.
|
Table 3 |
In one fixed-dose study (Study 2), Doxazosin tablet therapy (4 to 8 mg, once daily) resulted in a significant and sustained improvement in maximum urinary flow rate of 2.3–3.3 mL/sec (Table 3) compared to placebo (0.1 mL/sec). In this study, the only study in which weekly evaluations were made, significant improvement with Doxazosin tablet vs. placebo was seen after one week. The proportion of patients who responded with a maximum flow rate improvement of ≥3 mL/sec was significantly larger with Doxazosin tablet (34–42%) than placebo (13–17%). A significantly greater improvement was also seen in average flow rate with Doxazosin tablet (1.6 mL/sec) than with placebo (0.2 mL/sec). The onset and time course of symptom relief and increased urinary flow from Study 1 are illustrated in Figure 1.
|
Figure 1 – Study 1 |
7.2 Phosphodiesterase 5 (pde 5) Inhibitors (7.2 Phosphodiesterase-5 (PDE-5) inhibitors)
Concomitant administration of Doxazosin with a phosphodiesterase-5 (PDE-5) inhibitor can result in additive blood pressure lowering effects and symptomatic hypotension. Monitor blood pressure and for symptoms of hypotension [ see Warnings and Precautions (5.1) ].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:30.011316 · Updated: 2026-03-14T22:29:58.342833