These Highlights Do Not Include All The Information Needed To Use Daxxify Safely And Effectively. See Full Prescribing Information For Daxxify.
3aaa6e14-a3f7-4fb2-b9f9-d3a9c3ae1f74
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
The effects of all botulinum toxin products, including DAXXIFY, may spread from the area of injection to produce symptoms consistent with botulinum toxin effects. These symptoms have been reported hours to weeks after injection. Swallowing and breathing difficulties can be life-threatening, and there have been reports of death. The risk of symptoms is probably greatest in children treated for spasticity, an unapproved use for DAXXIFY, but symptoms can also occur in adults, particularly in those patients who have an underlying condition that would predispose them to these symptoms [see Warnings and Precautions (5.1) ].
Indications and Usage
DAXXIFY is an acetylcholine release inhibitor and neuromuscular-blocking agent indicated for: Temporary improvement in the appearance of moderate to severe glabellar lines associated with corrugator and/ or procerus muscle activity in adult patients. ( 1.1 ) The treatment of cervical dystonia in adult patients. ( 1.2 )
Dosage and Administration
Glabellar Lines : the recommended dose is 0.1 mL (8 Units) by intramuscular injection into each of five sites, for a total dose of 40 Units. ( 2.2 ) Cervical Dystonia : the recommended dose is 125 Units to 250 Units given intramuscularly as a divided dose among affected muscles. ( 2.3 )
Warnings and Precautions
The potency units of DAXXIFY are not interchangeable with other preparations of other botulinum toxin products. ( 5.2 , 11) Spread of toxin effects, swallowing and breathing difficulties can lead to death. Seek immediate medical attention if respiratory, speech, or swallowing difficulties occur. ( 5.1 ) Adverse event reports have been received involving the cardiovascular system with botulinum toxin products, some with fatal outcomes. Use caution when administering to patients with pre-existing cardiovascular disease. ( 5.5 ) Concomitant neuromuscular disorder may exacerbate clinical effects of treatment. ( 5.6 ) Use with caution in patients with compromised respiratory function or dysphagia. ( 5.7 ) Potential serious adverse reactions after administration of DAXXIFY for unapproved use. ( 5.3 )
Contraindications
DAXXIFY is contraindicated in: patients with known hypersensitivity to any botulinum toxin preparation, DAXXIFY, or any of the components in the DAXXIFY formulation [see Warnings and Precautions (5.4) ]. the presence of infection at the proposed injection sites.
Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling: Spread of Toxin Effect [see Warnings and Precautions (5.1) ] Hypersensitivity Reactions [see Warnings and Precautions (5.4) ] Cardiovascular System Adverse Reactions [see Warnings and Precautions (5.5) ] Increased Neuromuscular Compromise in Patients with Pre-Existing Neuromuscular Disorders [see Warnings and Precautions (5.6) ] Dysphagia and Breathing Difficulties [see Warnings and Precautions (5.7) ] Ophthalmic Adverse Reactions in Patients Treated for Glabellar Lines [see Warnings and Precautions (5.9) ]
Drug Interactions
No formal drug interaction studies have been conducted with DAXXIFY. However, the potential for certain drugs to potentiate the effects of DAXXIFY warrants consideration given the potential risks involved and should be used with caution. Aminoglycosides or other agents interfering with neuromuscular transmission Anticholinergic drugs Botulinum neurotoxin products Muscle relaxants
Medication Information
Warnings and Precautions
The potency units of DAXXIFY are not interchangeable with other preparations of other botulinum toxin products. ( 5.2 , 11) Spread of toxin effects, swallowing and breathing difficulties can lead to death. Seek immediate medical attention if respiratory, speech, or swallowing difficulties occur. ( 5.1 ) Adverse event reports have been received involving the cardiovascular system with botulinum toxin products, some with fatal outcomes. Use caution when administering to patients with pre-existing cardiovascular disease. ( 5.5 ) Concomitant neuromuscular disorder may exacerbate clinical effects of treatment. ( 5.6 ) Use with caution in patients with compromised respiratory function or dysphagia. ( 5.7 ) Potential serious adverse reactions after administration of DAXXIFY for unapproved use. ( 5.3 )
Indications and Usage
DAXXIFY is an acetylcholine release inhibitor and neuromuscular-blocking agent indicated for: Temporary improvement in the appearance of moderate to severe glabellar lines associated with corrugator and/ or procerus muscle activity in adult patients. ( 1.1 ) The treatment of cervical dystonia in adult patients. ( 1.2 )
Dosage and Administration
Glabellar Lines : the recommended dose is 0.1 mL (8 Units) by intramuscular injection into each of five sites, for a total dose of 40 Units. ( 2.2 ) Cervical Dystonia : the recommended dose is 125 Units to 250 Units given intramuscularly as a divided dose among affected muscles. ( 2.3 )
Contraindications
DAXXIFY is contraindicated in: patients with known hypersensitivity to any botulinum toxin preparation, DAXXIFY, or any of the components in the DAXXIFY formulation [see Warnings and Precautions (5.4) ]. the presence of infection at the proposed injection sites.
Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling: Spread of Toxin Effect [see Warnings and Precautions (5.1) ] Hypersensitivity Reactions [see Warnings and Precautions (5.4) ] Cardiovascular System Adverse Reactions [see Warnings and Precautions (5.5) ] Increased Neuromuscular Compromise in Patients with Pre-Existing Neuromuscular Disorders [see Warnings and Precautions (5.6) ] Dysphagia and Breathing Difficulties [see Warnings and Precautions (5.7) ] Ophthalmic Adverse Reactions in Patients Treated for Glabellar Lines [see Warnings and Precautions (5.9) ]
Drug Interactions
No formal drug interaction studies have been conducted with DAXXIFY. However, the potential for certain drugs to potentiate the effects of DAXXIFY warrants consideration given the potential risks involved and should be used with caution. Aminoglycosides or other agents interfering with neuromuscular transmission Anticholinergic drugs Botulinum neurotoxin products Muscle relaxants
Description
The effects of all botulinum toxin products, including DAXXIFY, may spread from the area of injection to produce symptoms consistent with botulinum toxin effects. These symptoms have been reported hours to weeks after injection. Swallowing and breathing difficulties can be life-threatening, and there have been reports of death. The risk of symptoms is probably greatest in children treated for spasticity, an unapproved use for DAXXIFY, but symptoms can also occur in adults, particularly in those patients who have an underlying condition that would predispose them to these symptoms [see Warnings and Precautions (5.1) ].
Section 42229-5
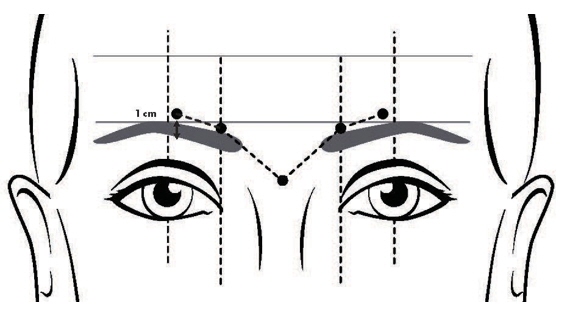
Recommended Dosage for Glabellar Lines
The total recommended dose is 40 Units per treatment session divided into five equal intramuscular injections of 8 Units each (two injections in each corrugator muscle and one injection in the procerus muscle).
Section 42231-1
| MEDICATION GUIDE DAXXIFY®(dax'-i-fye) daxibotulinumtoxinA-lanm for injection, for intramuscular use |
|||
|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 11/2023 | ||
|
What is the most important information I should know about DAXXIFY? DAXXIFY may cause serious side effects that can be life-threatening. Call your healthcare provider or get medical help right away if you have any of these problems after treatment with DAXXIFY:
|
|||
|
|
||
| These symptoms can happen hours, days, or weeks after you receive an injection of DAXXIFY. These problems could make it unsafe for you to drive a car or do other dangerous activities. See " What should I avoid while receiving DAXXIFY?" |
|||
|
What is DAXXIFY?
DAXXIFY is a prescription medicine for adults that is injected into muscles and used:
|
|||
Do not receive DAXXIFY if you:
|
|||
Before receiving DAXXIFY, tell your healthcare provider about all of your medical conditions, including if you:
Using DAXXIFY with certain other medicines may cause serious side effects.Do not start any new medicines until you have told your healthcare provider that you have received DAXXIFY in the past. Especially tell your healthcare provider if you:
|
|||
How will I receive DAXXIFY?
|
|||
|
What should I avoid while receiving DAXXIFY?
DAXXIFY may cause loss of strength or general muscle weakness, blurred vision, or drooping eyelids within hours to weeks of receiving DAXXIFY.If this happens, do not drive a car, operate machinery, or do other dangerous activities.See " What is the most important information I should know about DAXXIFY?" |
|||
What are the possible side effects of DAXXIFY? DAXXIFY may cause serious side effects, including:
|
|||
|
|
||
| The most common side effects of DAXXIFY in people with cervical dystonia include: | |||
|
|
|
|
| These are not all the possible side effects of DAXXIFY. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Revance Therapeutics, Inc. at 1-877-373-8669. |
|||
|
General information about the safe and effective use of DAXXIFY.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about DAXXIFY that is written for health professionals. |
|||
|
What are the ingredients in DAXXIFY? Active ingredient:daxibotulinumtoxinA-lanm Inactive ingredients:L-histidine, L-histidine HCl monohydrate, polysorbate 20, RTP004 peptide, trehalose dihydrate Manufactured by: Revance Therapeutics, Inc. Newark, CA 94560 U.S. License Number 2101 © 2023 Revance Therapeutics, Inc. All trademarks are the property of their respective owners. MG761127-4.0 |
Section 43683-2
Section 44425-7
Storage and Handling
Unopened DAXXIFY vials should be stored at room temperature 20°C to 25°C (68°F to 77°F) or refrigerated at 2°C to 8° C (36°F to 46°F) in the original carton to protect from light.
10 Overdosage
Excessive doses of DAXXIFY may be expected to produce neuromuscular weakness with a variety of symptoms. Respiratory support may be required where excessive doses cause paralysis of the respiratory muscles. In the event of overdose, the patient should be medically monitored for symptoms or excessive muscle weakness or muscle paralysis[seeWarnings and Precautions (5.1),(5.7)]. Symptomatic treatment may be necessary.
Symptoms of overdose are not likely to be present immediately following injection. Should accidental injection or oral ingestion occur, the person should be medically supervised for several weeks for signs and symptoms of excessive muscle weakness or paralysis.
In the event of overdose, antitoxin raised against botulinum toxin is available from the Centers for Disease Control and Prevention (CDC) in Atlanta, GA. However, the antitoxin will not reverse any botulinum toxin-induced effects already apparent by the time of antitoxin administration. In the event of suspected or actual cases of botulinum toxin poisoning, please contact your local or state Health Department to process a request for antitoxin through the CDC. If you do not receive a response within 30 minutes, please contact the CDC directly at 1-770-488-7100.
11 Description
DaxibotulinumtoxinA-lanm is an acetylcholine release inhibitor and neuromuscular blocking agent. DaxibotulinumtoxinA-lanm is a 150 kDa botulinum toxin without accessory proteins purified from the bacteriumClostridium botulinumtype A.
DAXXIFY (daxibotulinumtoxinA-lanm) for injection is supplied as a sterile, preservative-free, white to off- white lyophilized powder in a single-dose vial for intramuscular use after reconstitution. DAXXIFY is formulated with a 35 amino acid peptide excipient (RTP004) that prevents surface adsorption and promotes thermal stability of DAXXIFY. Each vial contains 50 Units or 100 Units of daxibotulinumtoxinA-lanm, L- histidine (0.14 mg), L-histidine HCl monohydrate (0.65 mg), polysorbate 20 (0.1 mg), RTP004 peptide (11.7 mcg), and trehalose dihydrate (36 mg).
8.4 Pediatric Use
Safety and effectiveness of DAXXIFY in patients less than 18 years of age have not been established.
1.1 Glabellar Lines
DAXXIFY is indicated for the temporary improvement in the appearance of moderate to severe glabellar lines associated with corrugator and/or procerus muscle activity in adult patients.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of DAXXIFY.
4 Contraindications
DAXXIFY is contraindicated in:
- patients with known hypersensitivity to any botulinum toxin preparation, DAXXIFY, or any of the components in the DAXXIFY formulation[seeWarnings and Precautions (5.4)].
- the presence of infection at the proposed injection sites.
6 Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
- Spread of Toxin Effect[seeWarnings and Precautions (5.1)]
- Hypersensitivity Reactions[seeWarnings and Precautions (5.4)]
- Cardiovascular System Adverse Reactions[seeWarnings and Precautions (5.5)]
- Increased Neuromuscular Compromise in Patients with Pre-Existing Neuromuscular Disorders[seeWarnings and Precautions (5.6)]
- Dysphagia and Breathing Difficulties[seeWarnings and Precautions (5.7)]
- Ophthalmic Adverse Reactions in Patients Treated for Glabellar Lines[seeWarnings and Precautions (5.9)]
7 Drug Interactions
No formal drug interaction studies have been conducted with DAXXIFY.
However, the potential for certain drugs to potentiate the effects of DAXXIFY warrants consideration given the potential risks involved and should be used with caution.
- Aminoglycosides or other agents interfering with neuromuscular transmission
- Anticholinergic drugs
- Botulinum neurotoxin products
- Muscle relaxants
14.1 Glabellar Lines
Two randomized, double-blind, multi-center, placebo-controlled clinical trials, Studies GL-1 and GL-2, were conducted to evaluate DAXXIFY for use in the temporary improvement of moderate-to-severe glabellar lines in adults. The 2 trials enrolled a total of 609 subjects (≥18 years old) with glabellar lines of at least moderate severity at maximum frown. A total of 405 subjects were randomized and 406 were treated with 40 Units of DAXXIFY and 204 subjects were randomized and 203 were treated with an equal volume of placebo. Subjects were excluded if they had eyelid ptosis, deep dermal scarring, excessive dermatochalasis, or an inability to lessen glabellar lines by physically spreading them apart. Enrolled subjects were 21 to 75 years old (with a mean age of 50 years), and predominantly female (87%) and Caucasian (86%). The total dose was delivered in 5 equally divided intramuscular injections of 8 Units each to specific sites in the glabella (Figure 1). Subjects were followed for at least 24 weeks after treatment.
Efficacy was determined through the assessment by investigators and subjects of frown wrinkle severity at maximum frown using a 4-point scale (0 = none, 1 = mild, 2 = moderate, 3 = severe). The primary efficacy endpoint (treatment success) was defined as achieving a score of 0 or 1 (none or mild) and an improvement of at least 2 points from baseline for both the investigator's and subject's assessments at Week 4. The percentages of subjects with treatment success at Week 4 are presented in Table 5.
| STUDY GL-1 | STUDY GL-2 | |||||
|---|---|---|---|---|---|---|
| DAXXIFY (N=201) n (%) |
Placebo (N=102) n (%) |
Treatment Difference and 95% Confidence Interval | DAXXIFY (N=205) n (%) |
Placebo (N=101) n (%) |
Treatment Difference and 95% Confidence Interval | |
| Treatment Success A score of 0 or 1 (none or mild) and ≥ 2-grade improvement from baseline on both the investigator and subject assessment.
|
148 (74%) | 0 (0%) | 74% (68%, 80%) | 152 (74%) | 0 (0%) | 74% (68%, 80%) |
| Individual Components | ||||||
| Investigator Assessment | 172 (86%) | 1 (1%) | --- | 187 (92%) | 2 (2%) | --- |
| Subject Assessment | 152 (76%) | 0 (0%) | --- | 156 (77%) | 0 (0%) | --- |
Figure 2 shows the proportion of subjects, over 36 weeks, rated as 0 or 1 (none or mild) by the investigator, 0 or 1 by the subject, and 0 or 1 with at least a 2-point improvement from their baseline based on both the investigator and subject ratings. Subjects were followed through at least Week 24 and then were discontinued from the study when both the investigator and subject scores returned to baseline. Subjects who returned to baseline levels prior to Week 36 were counted as non-responders following study discontinuation.
FIGURE 2: Proportion of subjects rated as 0 or 1 (none or mild) by investigator, 0 or 1 by the subject, and 0 or 1 with at least 2-point improvement from their baseline (based on both the investigator and subject ratings) in Study GL-1 and Study GL-2.
| Treatment success is defined as a score of 0 or 1 (none or mild) and ≥ 2-grade improvement from baseline on both the investigator and subject assessment. |
1.2 Cervical Dystonia
DAXXIFY is indicated for the treatment of cervical dystonia in adult patients.
12.2 Pharmacodynamics
No formal pharmacodynamics studies have been conducted with DAXXIFY.
12.3 Pharmacokinetics
Using currently available analytical technology, it is not possible to detect DAXXIFY in the peripheral blood following intramuscular injection at the recommended dose.
14.2 Cervical Dystonia
The efficacy of DAXXIFY was evaluated in a randomized, double-blind, placebo-controlled, multi-center trial in a total of 301 patients (NCT03608397). The mean age of patients was 58 years, 65% were women, and 96% were White. At study baseline, 84% of patients had previously received a botulinum toxin as treatment for cervical dystonia. Patients had a clinical diagnosis of cervical dystonia with baseline Toronto Western Spasmodic Torticollis Rating Scale (TWSTRS) total score ≥ 20, TWSTRS severity score ≥15, TWSTRS disability score ≥3, and TWSTRS pain score ≥1. For patients who had previously received a botulinum toxin treatment for cervical dystonia, the trial required that ≥14 weeks had passed since the most recent botulinum toxin administration.
Patients were randomized (3:3:1) to receive a single administration of 2.5 mL of either DAXXIFY 125 Units (n = 125), DAXXIFY 250 Units (n = 130), or placebo (n = 46), divided amongst the affected muscles as selected by the investigator. Table 6 indicates the treated muscles, along with the number of patients treated and DAXXIFY units.
| Unilateral Muscle Injected | DAXXIFY 125 Units | DAXXIFY 250 Units | ||
|---|---|---|---|---|
| Number of Patients | Median Units (min, max) |
Number of Patients | Median Units (min, max) |
|
| Levator Scapulae | 106 | 20 (10, 30) | 105 | 50 (20, 60) |
| Longissimus Capitis and Cervices | 45 | 15 (10, 30) | 58 | 40 (20, 60) |
| Scalenus Complex | 55 | 15 (10, 15) | 44 | 30 (20, 30) |
| Splenius Capitis | 120 | 25 (10, 50) | 127 | 50 (20, 100) |
| Splenius Cervices | 65 | 20 (10, 50) | 71 | 40 (20, 100) |
| Sternocleidomastoid | 115 | 25 (10, 25) | 121 | 50 (20, 50) |
| Trapezius | 105 | 20 (15, 40) | 105 | 40 (30, 80) |
The primary efficacy endpoint was the mean change in the TWSTRS total score from baseline averaged over weeks 4 and 6. TWSTRS evaluates the severity of dystonia, patient-perceived disability from dystonia, and pain, with a range of possible scores from 0 to 85. The mean change from baseline in the total TWSTRS score was significantly greater for both dosage groups of DAXXIFY than for placebo (Table 7).
| TWSTRS Assessment | Placebo (N = 46) |
DAXI 125 Units (N = 125) |
DAXI 250 Units (N = 130) |
|---|---|---|---|
| Baseline mean | 45.3 | 43.1 | 42.6 |
| Least squares mean change from baseline | -4.3 | -12.7 | -10.9 |
| Least squares mean difference from placebo (95% CI) |
-8.4 (-12.2, -4.6) | -6.6 (-10.4, -2.8) | |
| p-value | <0.0001 | 0.0007 |
A similar pattern of significant improvement versus placebo was observed in the clinician global impression of change (CGIC) and patient global impression of change (PGIC) scales.
1 Indications and Usage
DAXXIFY is an acetylcholine release inhibitor and neuromuscular-blocking agent indicated for:
12.1 Mechanism of Action
DAXXIFY blocks neuromuscular transmission at the neuromuscular junction by inhibiting the release of acetylcholine. This inhibition occurs as the neurotoxin cleaves SNAP-25, a protein necessary for the successful docking and release of acetylcholine vesicles within nerve endings. Recovery of neurotransmission occurs gradually as the neuromuscular junction recovers from SNAP-25 cleavage and as new nerve endings are formed.
5 Warnings and Precautions
- The potency units of DAXXIFY are not interchangeable with other preparations of other botulinum toxin products. (5.2,11)
- Spread of toxin effects, swallowing and breathing difficulties can lead to death. Seek immediate medical attention if respiratory, speech, or swallowing difficulties occur. (5.1)
- Adverse event reports have been received involving the cardiovascular system with botulinum toxin products, some with fatal outcomes. Use caution when administering to patients with pre-existing cardiovascular disease. (5.5)
- Concomitant neuromuscular disorder may exacerbate clinical effects of treatment. (5.6)
- Use with caution in patients with compromised respiratory function or dysphagia. (5.7)
- Potential serious adverse reactions after administration of DAXXIFY for unapproved use. (5.3)
5.1 Spread of Toxin Effect
Postmarketing safety data from other approved botulinum toxins suggest that botulinum toxin effects may be observed beyond the site of local injection. The symptoms are consistent with the mechanism of action of botulinum toxin and may include asthenia, generalized muscle weakness, diplopia, blurred vision, ptosis, dysphagia, dysphonia, dysarthria, urinary incontinence, and breathing difficulties. These symptoms have been reported hours to weeks after injection. Swallowing and breathing difficulties can be life-threatening, and there have been reports of death related to the spread of toxin effects. The risk of symptoms is greatest in children treated for spasticity, an unapproved use for DAXXIFY, but symptoms can occur in adults treated for spasticity and other conditions, and particularly in those patients who have underlying conditions that would predispose them to these symptoms. In unapproved uses, including upper limb spasticity in children, and approved indications, symptoms consistent with spread of toxin effect have been reported at doses comparable to, or lower than, the maximum recommended total dose. Patients or caregivers should be advised to seek immediate medical care if swallowing, speech, or respiratory difficulties occur.
2 Dosage and Administration
3 Dosage Forms and Strengths
For injection: 50 Units or 100 Units of white to off-white lyophilized powder in single-dose vial for reconstitution with sterile preservative-free 0.9% Sodium Chloride Injection, USP.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The most common side effects from treatment with DAXXIFY usually occur within one to two weeks after injection and, while generally transient, may have a duration of several weeks or months.
5.4 Hypersensitivity Reactions
Serious and/or immediate hypersensitivity reactions have been reported for botulinum toxin products. These reactions include anaphylaxis, serum sickness, urticaria, soft tissue edema, and dyspnea. If such a reaction occurs, discontinue further injection of DAXXIFY and immediately institute appropriate medical therapy. The use of DAXXIFY in patients with a known hypersensitivity to any botulinum toxin preparation, DAXXIFY, or any of its formulation components could lead to a life-threatening allergic reaction[seeContraindications (4)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.4 Preparation and Dilution Technique
DAXXIFY is supplied in single-dose 50 Unit and 100 Unit vials. Prior to intramuscular injection, reconstitute each vial of DAXXIFY with the required amount of sterile, preservative-free 0.9% Sodium Chloride Injection, USP to obtain a reconstituted solution at the appropriate concentration described in Tables 1 and 2.
| Indication | Diluent Preservative-free 0.9% Sodium Chloride Injection, USP Added to 50 Unit Vial |
Resulting Dose in Units per 0.1 mL |
|---|---|---|
| Glabellar Lines, Adults | 0.6 mL | 8 Units |
| Indication | Diluent Preservative-free 0.9% Sodium Chloride Injection, USP Added to 100 Unit Vials |
Resulting Dose in Units per 0.1 mL |
|---|---|---|
| Glabellar Lines, Adults | 1.2 mL | 8 Units |
| Cervical Dystonia, Adults | 1 mL or 2 mL |
10 Units or 5 Units |
Slowly inject the diluent into the vial. Discard the vial if a vacuum does not pull the diluent into the vial. Dispose of any unused diluent. Gently mix DAXXIFY with 0.9% Sodium Chloride Injection, USP by rotating the vial.
Reconstituted DAXXIFY solution is clear to slightly opalescent and colorless and free of particulate matter. Inspect visually the reconstituted DAXXIFY for particulate matter and discoloration prior to administration. Do not use if the solution is cloudy or discolored or contains flakes or particles.
Administer DAXXIFY within 72 hours after reconstitution. During this time period, store unused reconstituted DAXXIFY in a refrigerator between 2°C to 8°C (36°F to 46°F) and protected from light. Do not freeze reconstituted DAXXIFY. Dispose of any unused DAXXIFY.
Warning: Distant Spread of Toxin Effect
The effects of all botulinum toxin products, including DAXXIFY, may spread from the area of injection to produce symptoms consistent with botulinum toxin effects. These symptoms have been reported hours to weeks after injection. Swallowing and breathing difficulties can be life-threatening, and there have been reports of death. The risk of symptoms is probably greatest in children treated for spasticity, an unapproved use for DAXXIFY, but symptoms can also occur in adults, particularly in those patients who have an underlying condition that would predispose them to these symptoms[seeWarnings and Precautions (5.1)].
5.7 Dysphagia and Breathing Difficulties
Treatment with botulinum toxin products, including DAXXIFY, can result in swallowing or breathing difficulties. These reactions can occur within hours to weeks after injection with botulinum toxin. Patients with pre-existing swallowing or breathing difficulties may be more susceptible to these complications. In most cases, this is a consequence of weakening of muscles in the area of injection that are involved in breathing or swallowing. When distant effects occur, additional respiratory mechanisms may be involved[seeWarnings and Precautions (5.3)].
Deaths as a complication of severe dysphagia have been reported after treatment with botulinum toxin products. Dysphagia may persist for several months. Aspiration may result from severe dysphagia and is a particular risk when treating patients in whom swallowing or respiratory function is already compromised. Treatment with botulinum toxins, including DAXXIFY, may weaken neck muscles that serve as accessory muscles of ventilation. This may result in a critical loss of breathing capacity in patients with respiratory disorders who may have become dependent upon these accessory muscles. There have been postmarketing reports from other botulinum toxin products of serious breathing difficulties, including respiratory failure.
Patients treated with botulinum toxin may require immediate medical attention should they develop problems with swallowing, speech, or respiratory disorders.
2.1 Important Administration Instructions
The potency units of DAXXIFY for injection are specific to the preparation and test method utilized. They are not interchangeable with other preparations of botulinum toxin products, and, therefore, units of biological activity of DAXXIFY cannot be compared to, or converted into, units of any other botulinum toxin products assessed with any other specific test method[seeWarnings and Precautions (5.2)andDescription (11)].
The safe and effective use of DAXXIFY depends upon proper storage of the product, selection of the correct dose, and proper reconstitution and administration techniques.
DAXXIFY should be administered no more frequently than every three months for any indication. Consideration of the cumulative dose is necessary when treating adult patients with DAXXIFY. Physicians should be aware of whether patients are receiving treatment with other botulinum toxin products for other indications.
Reconstituted DAXXIFY is intended for intramuscular injection only.
After reconstitution, only use each DAXXIFY vial for only one injection session and for only one patient. Discard any remaining solution in vial immediately after administration.
Reconstitution instructions are provided specifically for the 50 Unit and the 100 Unit vials (Table 1; Table 2).
5.5 Cardiovascular System Adverse Reactions
There have been reports following administration of botulinum toxins of adverse events involving the cardiovascular system, including arrhythmia and myocardial infarction, some with fatal outcomes. Some of these patients had risk factors, including pre-existing cardiovascular disease. Use caution when administering to patients with pre-existing cardiovascular disease.
2.3 Recommended Dosage for Cervical Dystonia
The recommended dose of DAXXIFY for the treatment of cervical dystonia ranges from 125 Units to 250 Units given intramuscularly as a divided dose among affected muscles. In patients previously treated with another botulinum toxin, their past dose, response to treatment, duration of effect, and adverse event history should be taken into consideration when determining the initial DAXXIFY dose. A description of the average DAXXIFY dose and percentage of total dose injected into specific muscles in the pivotal clinical trials can be found in Section 14. Limiting the dose injected into the sternocleidomastoid muscle may reduce the occurrence of dysphagia.
Principal Display Panel 100 Units Vial Carton
daxibotulinumtoxinA-lanm
DAXXIFY®
FOR INJECTION
100
units/vial
REVANCE®
ATTENTION:
Each patient is required to
receive the enclosed
Medication Guide.
Rx only
1 Single-Dose Vial
For intramuscular use
5.3 Serious Adverse Reactions With Unapproved Use
Serious adverse reactions, including excessive weakness, dysphagia, and aspiration pneumonia, with some adverse reactions associated with fatal outcomes, have been reported in patients who received botulinum toxin injections for unapproved uses. In these cases, the adverse reactions may have resulted from the administration of botulinum toxin products to the site of injection and/or adjacent structures. In some cases, patients had pre-existing dysphagia or other significant disabilities. There is insufficient information to identify factors associated with an increased risk for adverse reactions associated with the unapproved uses of botulinum toxin products.
5.8 Facial Anatomy in the Treatment of Glabellar Lines
Use caution when administering DAXXIFY to patients with surgical alterations to the facial anatomy, marked facial asymmetry, excessive dermatochalasis, deep dermal scarring, thick sebaceous skin, inflammation at the injection site(s), pre-existing eyelid or eyebrow ptosis, when excessive weakness or atrophy is present in the target muscles, or the inability to substantially lessen glabellar lines even by physically spreading them apart.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate the carcinogenic potential of DAXXIFY.
Genotoxicity studies have not been conducted for DAXXIFY.
In fertility and early embryonic development studies in rats, intramuscular administration of 3, 10, or 20 Units/kg in males and 3, 10, or 30 Units/kg in females to either male or female rats, respectively, prior to mating and during mating (7 doses, 1 week apart for males; and 4 doses, 1 week apart for females) to untreated animals, caused reduced fertility at doses associated with paternal or maternal toxicity as indicated by reductions in body weight gain and food intake. No effects on fertility were noted at 3 Units/kg in males or at 10 Units/kg in females, which are approximately 4 and 15 times, respectively, the MRHD.
5.2 Lack of Interchangeability Between Botulinum Toxin Products
The potency units of DAXXIFY are specific to the preparation and assay method utilized. They are not interchangeable with other preparations of botulinum toxin products; therefore, units of biological activity of DAXXIFY cannot be compared to or converted to units of any other botulinum toxin products assessed with any other specific assay method[seeDescription (11)].
5.9 Ophthalmic Adverse Reactions in Patients Treated for Glabellar Lines
Dry eye has been reported with the use of botulinum toxin products in the treatment of glabellar lines. Reduced tear production, reduced blinking, and corneal disorders may occur with use of botulinum toxins, including DAXXIFY. If symptoms of dry eye (e.g., eye irritation, photophobia, or visual changes) persist, consider referring patient to an ophthalmologist.
5.6 Increased Neuromuscular Compromise in Patients With Pre Existing Neuromuscular Disorders
Monitor patients with peripheral motor neuropathic diseases, amyotrophic lateral sclerosis, or neuromuscular junctional disorders (e.g., myasthenia gravis or Lambert-Eaton syndrome) for increased neuromuscular compromise following botulinum toxin treatment. Patients with neuromuscular disorders may be at increased risk of clinically significant effects, including generalized muscle weakness, diplopia, ptosis, dysphonia, dysarthria, severe dysphagia, and respiratory compromise from typical doses of DAXXIFY.
Structured Label Content
Section 42229-5 (42229-5)
Recommended Dosage for Glabellar Lines
The total recommended dose is 40 Units per treatment session divided into five equal intramuscular injections of 8 Units each (two injections in each corrugator muscle and one injection in the procerus muscle).
Section 42231-1 (42231-1)
| MEDICATION GUIDE DAXXIFY®(dax'-i-fye) daxibotulinumtoxinA-lanm for injection, for intramuscular use |
|||
|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 11/2023 | ||
|
What is the most important information I should know about DAXXIFY? DAXXIFY may cause serious side effects that can be life-threatening. Call your healthcare provider or get medical help right away if you have any of these problems after treatment with DAXXIFY:
|
|||
|
|
||
| These symptoms can happen hours, days, or weeks after you receive an injection of DAXXIFY. These problems could make it unsafe for you to drive a car or do other dangerous activities. See " What should I avoid while receiving DAXXIFY?" |
|||
|
What is DAXXIFY?
DAXXIFY is a prescription medicine for adults that is injected into muscles and used:
|
|||
Do not receive DAXXIFY if you:
|
|||
Before receiving DAXXIFY, tell your healthcare provider about all of your medical conditions, including if you:
Using DAXXIFY with certain other medicines may cause serious side effects.Do not start any new medicines until you have told your healthcare provider that you have received DAXXIFY in the past. Especially tell your healthcare provider if you:
|
|||
How will I receive DAXXIFY?
|
|||
|
What should I avoid while receiving DAXXIFY?
DAXXIFY may cause loss of strength or general muscle weakness, blurred vision, or drooping eyelids within hours to weeks of receiving DAXXIFY.If this happens, do not drive a car, operate machinery, or do other dangerous activities.See " What is the most important information I should know about DAXXIFY?" |
|||
What are the possible side effects of DAXXIFY? DAXXIFY may cause serious side effects, including:
|
|||
|
|
||
| The most common side effects of DAXXIFY in people with cervical dystonia include: | |||
|
|
|
|
| These are not all the possible side effects of DAXXIFY. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Revance Therapeutics, Inc. at 1-877-373-8669. |
|||
|
General information about the safe and effective use of DAXXIFY.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about DAXXIFY that is written for health professionals. |
|||
|
What are the ingredients in DAXXIFY? Active ingredient:daxibotulinumtoxinA-lanm Inactive ingredients:L-histidine, L-histidine HCl monohydrate, polysorbate 20, RTP004 peptide, trehalose dihydrate Manufactured by: Revance Therapeutics, Inc. Newark, CA 94560 U.S. License Number 2101 © 2023 Revance Therapeutics, Inc. All trademarks are the property of their respective owners. MG761127-4.0 |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage and Handling
Unopened DAXXIFY vials should be stored at room temperature 20°C to 25°C (68°F to 77°F) or refrigerated at 2°C to 8° C (36°F to 46°F) in the original carton to protect from light.
10 Overdosage (10 OVERDOSAGE)
Excessive doses of DAXXIFY may be expected to produce neuromuscular weakness with a variety of symptoms. Respiratory support may be required where excessive doses cause paralysis of the respiratory muscles. In the event of overdose, the patient should be medically monitored for symptoms or excessive muscle weakness or muscle paralysis[seeWarnings and Precautions (5.1),(5.7)]. Symptomatic treatment may be necessary.
Symptoms of overdose are not likely to be present immediately following injection. Should accidental injection or oral ingestion occur, the person should be medically supervised for several weeks for signs and symptoms of excessive muscle weakness or paralysis.
In the event of overdose, antitoxin raised against botulinum toxin is available from the Centers for Disease Control and Prevention (CDC) in Atlanta, GA. However, the antitoxin will not reverse any botulinum toxin-induced effects already apparent by the time of antitoxin administration. In the event of suspected or actual cases of botulinum toxin poisoning, please contact your local or state Health Department to process a request for antitoxin through the CDC. If you do not receive a response within 30 minutes, please contact the CDC directly at 1-770-488-7100.
11 Description (11 DESCRIPTION)
DaxibotulinumtoxinA-lanm is an acetylcholine release inhibitor and neuromuscular blocking agent. DaxibotulinumtoxinA-lanm is a 150 kDa botulinum toxin without accessory proteins purified from the bacteriumClostridium botulinumtype A.
DAXXIFY (daxibotulinumtoxinA-lanm) for injection is supplied as a sterile, preservative-free, white to off- white lyophilized powder in a single-dose vial for intramuscular use after reconstitution. DAXXIFY is formulated with a 35 amino acid peptide excipient (RTP004) that prevents surface adsorption and promotes thermal stability of DAXXIFY. Each vial contains 50 Units or 100 Units of daxibotulinumtoxinA-lanm, L- histidine (0.14 mg), L-histidine HCl monohydrate (0.65 mg), polysorbate 20 (0.1 mg), RTP004 peptide (11.7 mcg), and trehalose dihydrate (36 mg).
8.4 Pediatric Use
Safety and effectiveness of DAXXIFY in patients less than 18 years of age have not been established.
1.1 Glabellar Lines
DAXXIFY is indicated for the temporary improvement in the appearance of moderate to severe glabellar lines associated with corrugator and/or procerus muscle activity in adult patients.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of DAXXIFY.
4 Contraindications (4 CONTRAINDICATIONS)
DAXXIFY is contraindicated in:
- patients with known hypersensitivity to any botulinum toxin preparation, DAXXIFY, or any of the components in the DAXXIFY formulation[seeWarnings and Precautions (5.4)].
- the presence of infection at the proposed injection sites.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
- Spread of Toxin Effect[seeWarnings and Precautions (5.1)]
- Hypersensitivity Reactions[seeWarnings and Precautions (5.4)]
- Cardiovascular System Adverse Reactions[seeWarnings and Precautions (5.5)]
- Increased Neuromuscular Compromise in Patients with Pre-Existing Neuromuscular Disorders[seeWarnings and Precautions (5.6)]
- Dysphagia and Breathing Difficulties[seeWarnings and Precautions (5.7)]
- Ophthalmic Adverse Reactions in Patients Treated for Glabellar Lines[seeWarnings and Precautions (5.9)]
7 Drug Interactions (7 DRUG INTERACTIONS)
No formal drug interaction studies have been conducted with DAXXIFY.
However, the potential for certain drugs to potentiate the effects of DAXXIFY warrants consideration given the potential risks involved and should be used with caution.
- Aminoglycosides or other agents interfering with neuromuscular transmission
- Anticholinergic drugs
- Botulinum neurotoxin products
- Muscle relaxants
14.1 Glabellar Lines
Two randomized, double-blind, multi-center, placebo-controlled clinical trials, Studies GL-1 and GL-2, were conducted to evaluate DAXXIFY for use in the temporary improvement of moderate-to-severe glabellar lines in adults. The 2 trials enrolled a total of 609 subjects (≥18 years old) with glabellar lines of at least moderate severity at maximum frown. A total of 405 subjects were randomized and 406 were treated with 40 Units of DAXXIFY and 204 subjects were randomized and 203 were treated with an equal volume of placebo. Subjects were excluded if they had eyelid ptosis, deep dermal scarring, excessive dermatochalasis, or an inability to lessen glabellar lines by physically spreading them apart. Enrolled subjects were 21 to 75 years old (with a mean age of 50 years), and predominantly female (87%) and Caucasian (86%). The total dose was delivered in 5 equally divided intramuscular injections of 8 Units each to specific sites in the glabella (Figure 1). Subjects were followed for at least 24 weeks after treatment.
Efficacy was determined through the assessment by investigators and subjects of frown wrinkle severity at maximum frown using a 4-point scale (0 = none, 1 = mild, 2 = moderate, 3 = severe). The primary efficacy endpoint (treatment success) was defined as achieving a score of 0 or 1 (none or mild) and an improvement of at least 2 points from baseline for both the investigator's and subject's assessments at Week 4. The percentages of subjects with treatment success at Week 4 are presented in Table 5.
| STUDY GL-1 | STUDY GL-2 | |||||
|---|---|---|---|---|---|---|
| DAXXIFY (N=201) n (%) |
Placebo (N=102) n (%) |
Treatment Difference and 95% Confidence Interval | DAXXIFY (N=205) n (%) |
Placebo (N=101) n (%) |
Treatment Difference and 95% Confidence Interval | |
| Treatment Success A score of 0 or 1 (none or mild) and ≥ 2-grade improvement from baseline on both the investigator and subject assessment.
|
148 (74%) | 0 (0%) | 74% (68%, 80%) | 152 (74%) | 0 (0%) | 74% (68%, 80%) |
| Individual Components | ||||||
| Investigator Assessment | 172 (86%) | 1 (1%) | --- | 187 (92%) | 2 (2%) | --- |
| Subject Assessment | 152 (76%) | 0 (0%) | --- | 156 (77%) | 0 (0%) | --- |
Figure 2 shows the proportion of subjects, over 36 weeks, rated as 0 or 1 (none or mild) by the investigator, 0 or 1 by the subject, and 0 or 1 with at least a 2-point improvement from their baseline based on both the investigator and subject ratings. Subjects were followed through at least Week 24 and then were discontinued from the study when both the investigator and subject scores returned to baseline. Subjects who returned to baseline levels prior to Week 36 were counted as non-responders following study discontinuation.
FIGURE 2: Proportion of subjects rated as 0 or 1 (none or mild) by investigator, 0 or 1 by the subject, and 0 or 1 with at least 2-point improvement from their baseline (based on both the investigator and subject ratings) in Study GL-1 and Study GL-2.
| Treatment success is defined as a score of 0 or 1 (none or mild) and ≥ 2-grade improvement from baseline on both the investigator and subject assessment. |
1.2 Cervical Dystonia
DAXXIFY is indicated for the treatment of cervical dystonia in adult patients.
12.2 Pharmacodynamics
No formal pharmacodynamics studies have been conducted with DAXXIFY.
12.3 Pharmacokinetics
Using currently available analytical technology, it is not possible to detect DAXXIFY in the peripheral blood following intramuscular injection at the recommended dose.
14.2 Cervical Dystonia
The efficacy of DAXXIFY was evaluated in a randomized, double-blind, placebo-controlled, multi-center trial in a total of 301 patients (NCT03608397). The mean age of patients was 58 years, 65% were women, and 96% were White. At study baseline, 84% of patients had previously received a botulinum toxin as treatment for cervical dystonia. Patients had a clinical diagnosis of cervical dystonia with baseline Toronto Western Spasmodic Torticollis Rating Scale (TWSTRS) total score ≥ 20, TWSTRS severity score ≥15, TWSTRS disability score ≥3, and TWSTRS pain score ≥1. For patients who had previously received a botulinum toxin treatment for cervical dystonia, the trial required that ≥14 weeks had passed since the most recent botulinum toxin administration.
Patients were randomized (3:3:1) to receive a single administration of 2.5 mL of either DAXXIFY 125 Units (n = 125), DAXXIFY 250 Units (n = 130), or placebo (n = 46), divided amongst the affected muscles as selected by the investigator. Table 6 indicates the treated muscles, along with the number of patients treated and DAXXIFY units.
| Unilateral Muscle Injected | DAXXIFY 125 Units | DAXXIFY 250 Units | ||
|---|---|---|---|---|
| Number of Patients | Median Units (min, max) |
Number of Patients | Median Units (min, max) |
|
| Levator Scapulae | 106 | 20 (10, 30) | 105 | 50 (20, 60) |
| Longissimus Capitis and Cervices | 45 | 15 (10, 30) | 58 | 40 (20, 60) |
| Scalenus Complex | 55 | 15 (10, 15) | 44 | 30 (20, 30) |
| Splenius Capitis | 120 | 25 (10, 50) | 127 | 50 (20, 100) |
| Splenius Cervices | 65 | 20 (10, 50) | 71 | 40 (20, 100) |
| Sternocleidomastoid | 115 | 25 (10, 25) | 121 | 50 (20, 50) |
| Trapezius | 105 | 20 (15, 40) | 105 | 40 (30, 80) |
The primary efficacy endpoint was the mean change in the TWSTRS total score from baseline averaged over weeks 4 and 6. TWSTRS evaluates the severity of dystonia, patient-perceived disability from dystonia, and pain, with a range of possible scores from 0 to 85. The mean change from baseline in the total TWSTRS score was significantly greater for both dosage groups of DAXXIFY than for placebo (Table 7).
| TWSTRS Assessment | Placebo (N = 46) |
DAXI 125 Units (N = 125) |
DAXI 250 Units (N = 130) |
|---|---|---|---|
| Baseline mean | 45.3 | 43.1 | 42.6 |
| Least squares mean change from baseline | -4.3 | -12.7 | -10.9 |
| Least squares mean difference from placebo (95% CI) |
-8.4 (-12.2, -4.6) | -6.6 (-10.4, -2.8) | |
| p-value | <0.0001 | 0.0007 |
A similar pattern of significant improvement versus placebo was observed in the clinician global impression of change (CGIC) and patient global impression of change (PGIC) scales.
1 Indications and Usage (1 INDICATIONS AND USAGE)
DAXXIFY is an acetylcholine release inhibitor and neuromuscular-blocking agent indicated for:
12.1 Mechanism of Action
DAXXIFY blocks neuromuscular transmission at the neuromuscular junction by inhibiting the release of acetylcholine. This inhibition occurs as the neurotoxin cleaves SNAP-25, a protein necessary for the successful docking and release of acetylcholine vesicles within nerve endings. Recovery of neurotransmission occurs gradually as the neuromuscular junction recovers from SNAP-25 cleavage and as new nerve endings are formed.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- The potency units of DAXXIFY are not interchangeable with other preparations of other botulinum toxin products. (5.2,11)
- Spread of toxin effects, swallowing and breathing difficulties can lead to death. Seek immediate medical attention if respiratory, speech, or swallowing difficulties occur. (5.1)
- Adverse event reports have been received involving the cardiovascular system with botulinum toxin products, some with fatal outcomes. Use caution when administering to patients with pre-existing cardiovascular disease. (5.5)
- Concomitant neuromuscular disorder may exacerbate clinical effects of treatment. (5.6)
- Use with caution in patients with compromised respiratory function or dysphagia. (5.7)
- Potential serious adverse reactions after administration of DAXXIFY for unapproved use. (5.3)
5.1 Spread of Toxin Effect
Postmarketing safety data from other approved botulinum toxins suggest that botulinum toxin effects may be observed beyond the site of local injection. The symptoms are consistent with the mechanism of action of botulinum toxin and may include asthenia, generalized muscle weakness, diplopia, blurred vision, ptosis, dysphagia, dysphonia, dysarthria, urinary incontinence, and breathing difficulties. These symptoms have been reported hours to weeks after injection. Swallowing and breathing difficulties can be life-threatening, and there have been reports of death related to the spread of toxin effects. The risk of symptoms is greatest in children treated for spasticity, an unapproved use for DAXXIFY, but symptoms can occur in adults treated for spasticity and other conditions, and particularly in those patients who have underlying conditions that would predispose them to these symptoms. In unapproved uses, including upper limb spasticity in children, and approved indications, symptoms consistent with spread of toxin effect have been reported at doses comparable to, or lower than, the maximum recommended total dose. Patients or caregivers should be advised to seek immediate medical care if swallowing, speech, or respiratory difficulties occur.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For injection: 50 Units or 100 Units of white to off-white lyophilized powder in single-dose vial for reconstitution with sterile preservative-free 0.9% Sodium Chloride Injection, USP.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The most common side effects from treatment with DAXXIFY usually occur within one to two weeks after injection and, while generally transient, may have a duration of several weeks or months.
5.4 Hypersensitivity Reactions
Serious and/or immediate hypersensitivity reactions have been reported for botulinum toxin products. These reactions include anaphylaxis, serum sickness, urticaria, soft tissue edema, and dyspnea. If such a reaction occurs, discontinue further injection of DAXXIFY and immediately institute appropriate medical therapy. The use of DAXXIFY in patients with a known hypersensitivity to any botulinum toxin preparation, DAXXIFY, or any of its formulation components could lead to a life-threatening allergic reaction[seeContraindications (4)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.4 Preparation and Dilution Technique
DAXXIFY is supplied in single-dose 50 Unit and 100 Unit vials. Prior to intramuscular injection, reconstitute each vial of DAXXIFY with the required amount of sterile, preservative-free 0.9% Sodium Chloride Injection, USP to obtain a reconstituted solution at the appropriate concentration described in Tables 1 and 2.
| Indication | Diluent Preservative-free 0.9% Sodium Chloride Injection, USP Added to 50 Unit Vial |
Resulting Dose in Units per 0.1 mL |
|---|---|---|
| Glabellar Lines, Adults | 0.6 mL | 8 Units |
| Indication | Diluent Preservative-free 0.9% Sodium Chloride Injection, USP Added to 100 Unit Vials |
Resulting Dose in Units per 0.1 mL |
|---|---|---|
| Glabellar Lines, Adults | 1.2 mL | 8 Units |
| Cervical Dystonia, Adults | 1 mL or 2 mL |
10 Units or 5 Units |
Slowly inject the diluent into the vial. Discard the vial if a vacuum does not pull the diluent into the vial. Dispose of any unused diluent. Gently mix DAXXIFY with 0.9% Sodium Chloride Injection, USP by rotating the vial.
Reconstituted DAXXIFY solution is clear to slightly opalescent and colorless and free of particulate matter. Inspect visually the reconstituted DAXXIFY for particulate matter and discoloration prior to administration. Do not use if the solution is cloudy or discolored or contains flakes or particles.
Administer DAXXIFY within 72 hours after reconstitution. During this time period, store unused reconstituted DAXXIFY in a refrigerator between 2°C to 8°C (36°F to 46°F) and protected from light. Do not freeze reconstituted DAXXIFY. Dispose of any unused DAXXIFY.
Warning: Distant Spread of Toxin Effect (WARNING: DISTANT SPREAD OF TOXIN EFFECT)
The effects of all botulinum toxin products, including DAXXIFY, may spread from the area of injection to produce symptoms consistent with botulinum toxin effects. These symptoms have been reported hours to weeks after injection. Swallowing and breathing difficulties can be life-threatening, and there have been reports of death. The risk of symptoms is probably greatest in children treated for spasticity, an unapproved use for DAXXIFY, but symptoms can also occur in adults, particularly in those patients who have an underlying condition that would predispose them to these symptoms[seeWarnings and Precautions (5.1)].
5.7 Dysphagia and Breathing Difficulties
Treatment with botulinum toxin products, including DAXXIFY, can result in swallowing or breathing difficulties. These reactions can occur within hours to weeks after injection with botulinum toxin. Patients with pre-existing swallowing or breathing difficulties may be more susceptible to these complications. In most cases, this is a consequence of weakening of muscles in the area of injection that are involved in breathing or swallowing. When distant effects occur, additional respiratory mechanisms may be involved[seeWarnings and Precautions (5.3)].
Deaths as a complication of severe dysphagia have been reported after treatment with botulinum toxin products. Dysphagia may persist for several months. Aspiration may result from severe dysphagia and is a particular risk when treating patients in whom swallowing or respiratory function is already compromised. Treatment with botulinum toxins, including DAXXIFY, may weaken neck muscles that serve as accessory muscles of ventilation. This may result in a critical loss of breathing capacity in patients with respiratory disorders who may have become dependent upon these accessory muscles. There have been postmarketing reports from other botulinum toxin products of serious breathing difficulties, including respiratory failure.
Patients treated with botulinum toxin may require immediate medical attention should they develop problems with swallowing, speech, or respiratory disorders.
2.1 Important Administration Instructions
The potency units of DAXXIFY for injection are specific to the preparation and test method utilized. They are not interchangeable with other preparations of botulinum toxin products, and, therefore, units of biological activity of DAXXIFY cannot be compared to, or converted into, units of any other botulinum toxin products assessed with any other specific test method[seeWarnings and Precautions (5.2)andDescription (11)].
The safe and effective use of DAXXIFY depends upon proper storage of the product, selection of the correct dose, and proper reconstitution and administration techniques.
DAXXIFY should be administered no more frequently than every three months for any indication. Consideration of the cumulative dose is necessary when treating adult patients with DAXXIFY. Physicians should be aware of whether patients are receiving treatment with other botulinum toxin products for other indications.
Reconstituted DAXXIFY is intended for intramuscular injection only.
After reconstitution, only use each DAXXIFY vial for only one injection session and for only one patient. Discard any remaining solution in vial immediately after administration.
Reconstitution instructions are provided specifically for the 50 Unit and the 100 Unit vials (Table 1; Table 2).
5.5 Cardiovascular System Adverse Reactions
There have been reports following administration of botulinum toxins of adverse events involving the cardiovascular system, including arrhythmia and myocardial infarction, some with fatal outcomes. Some of these patients had risk factors, including pre-existing cardiovascular disease. Use caution when administering to patients with pre-existing cardiovascular disease.
2.3 Recommended Dosage for Cervical Dystonia
The recommended dose of DAXXIFY for the treatment of cervical dystonia ranges from 125 Units to 250 Units given intramuscularly as a divided dose among affected muscles. In patients previously treated with another botulinum toxin, their past dose, response to treatment, duration of effect, and adverse event history should be taken into consideration when determining the initial DAXXIFY dose. A description of the average DAXXIFY dose and percentage of total dose injected into specific muscles in the pivotal clinical trials can be found in Section 14. Limiting the dose injected into the sternocleidomastoid muscle may reduce the occurrence of dysphagia.
Principal Display Panel 100 Units Vial Carton (PRINCIPAL DISPLAY PANEL - 100 Units Vial Carton)
daxibotulinumtoxinA-lanm
DAXXIFY®
FOR INJECTION
100
units/vial
REVANCE®
ATTENTION:
Each patient is required to
receive the enclosed
Medication Guide.
Rx only
1 Single-Dose Vial
For intramuscular use
5.3 Serious Adverse Reactions With Unapproved Use (5.3 Serious Adverse Reactions with Unapproved Use)
Serious adverse reactions, including excessive weakness, dysphagia, and aspiration pneumonia, with some adverse reactions associated with fatal outcomes, have been reported in patients who received botulinum toxin injections for unapproved uses. In these cases, the adverse reactions may have resulted from the administration of botulinum toxin products to the site of injection and/or adjacent structures. In some cases, patients had pre-existing dysphagia or other significant disabilities. There is insufficient information to identify factors associated with an increased risk for adverse reactions associated with the unapproved uses of botulinum toxin products.
5.8 Facial Anatomy in the Treatment of Glabellar Lines
Use caution when administering DAXXIFY to patients with surgical alterations to the facial anatomy, marked facial asymmetry, excessive dermatochalasis, deep dermal scarring, thick sebaceous skin, inflammation at the injection site(s), pre-existing eyelid or eyebrow ptosis, when excessive weakness or atrophy is present in the target muscles, or the inability to substantially lessen glabellar lines even by physically spreading them apart.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate the carcinogenic potential of DAXXIFY.
Genotoxicity studies have not been conducted for DAXXIFY.
In fertility and early embryonic development studies in rats, intramuscular administration of 3, 10, or 20 Units/kg in males and 3, 10, or 30 Units/kg in females to either male or female rats, respectively, prior to mating and during mating (7 doses, 1 week apart for males; and 4 doses, 1 week apart for females) to untreated animals, caused reduced fertility at doses associated with paternal or maternal toxicity as indicated by reductions in body weight gain and food intake. No effects on fertility were noted at 3 Units/kg in males or at 10 Units/kg in females, which are approximately 4 and 15 times, respectively, the MRHD.
5.2 Lack of Interchangeability Between Botulinum Toxin Products (5.2 Lack of Interchangeability between Botulinum Toxin Products)
The potency units of DAXXIFY are specific to the preparation and assay method utilized. They are not interchangeable with other preparations of botulinum toxin products; therefore, units of biological activity of DAXXIFY cannot be compared to or converted to units of any other botulinum toxin products assessed with any other specific assay method[seeDescription (11)].
5.9 Ophthalmic Adverse Reactions in Patients Treated for Glabellar Lines
Dry eye has been reported with the use of botulinum toxin products in the treatment of glabellar lines. Reduced tear production, reduced blinking, and corneal disorders may occur with use of botulinum toxins, including DAXXIFY. If symptoms of dry eye (e.g., eye irritation, photophobia, or visual changes) persist, consider referring patient to an ophthalmologist.
5.6 Increased Neuromuscular Compromise in Patients With Pre Existing Neuromuscular Disorders (5.6 Increased Neuromuscular Compromise in Patients with Pre-Existing Neuromuscular Disorders)
Monitor patients with peripheral motor neuropathic diseases, amyotrophic lateral sclerosis, or neuromuscular junctional disorders (e.g., myasthenia gravis or Lambert-Eaton syndrome) for increased neuromuscular compromise following botulinum toxin treatment. Patients with neuromuscular disorders may be at increased risk of clinically significant effects, including generalized muscle weakness, diplopia, ptosis, dysphonia, dysarthria, severe dysphagia, and respiratory compromise from typical doses of DAXXIFY.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:01.156767 · Updated: 2026-03-14T22:42:38.249942