Drug Facts
3a73ee4b-c2a4-496f-be90-9f78c8046062
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Naloxone hydrochloride 4 mg
Purpose
Emergency treatment of opioid overdose
Medication Information
Purpose
Emergency treatment of opioid overdose
Description
Naloxone hydrochloride 4 mg
Uses
- to “revive” someone during an overdose from many prescription pain medications or street drugs such as heroin
- this medicine can save a life
Section 42229-5
| Safe to Use Even if Opioids are Not Present |
*This product is not affiliated with, manufactured by, or produced by
the makers or owners of NARCAN®
Manufactured For: Teva Pharmaceuticals USA, Inc.
Parsippany, NJ 07054
Rev. A 1/2024
Warning
-
When using this product some people may experience
symptoms when they wake up, such as, shaking, sweating,
nausea, or feeling angry. This is to be expected.
Directions
|
|
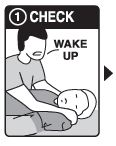
Step 1: CHECK if you suspect an overdose:
|
|
Step 2: GIVE 1st dose in the nose
|
|
|
Step 3: CALL
|
|
|
Step 4: WATCH & GIVE
|
|
|
Step 5: STAY
|
Questions?
1-888-838-2872 between 9 am and 5 pm ET, Monday-Friday.
www.tevausa.com/our-products/tevagenerics
Other Information
- Store below 77°F (25°C).
- do not freeze
- avoid excessive heat above 40°C (104°F)
- protect from light
- the product is packaged in individually-sealed blisters.
Do not use if the blister is open or torn, or if the device appears damaged.
Inactive Ingredients
benzalkonium chloride, edetate disodium, sodium chloride buffered with sodium hydroxide/hydrochloric acid, water
Active Ingredient (in Each Spray)
Naloxone hydrochloride 4 mg
Package/label Principal Display Panel – Carton Label
Original Prescription Strength
Easy to Use
Can Save a Life
Compare to the active
ingredient in NARCAN®*
NDC 0480-3478-68
Naloxone Hydrochloride
Nasal Spray
4 mg
Emergency Treatment of
Opioid Overdose
Designed to Rapidly Reverse the
Effects of a Life-Threatening
Opioid Emergency
For use in nose only
2 Single-Dose Nasal Spray Devices
0.003 fl oz (0.1 mL) each
Structured Label Content
Uses
- to “revive” someone during an overdose from many prescription pain medications or street drugs such as heroin
- this medicine can save a life
Section 42229-5 (42229-5)
| Safe to Use Even if Opioids are Not Present |
*This product is not affiliated with, manufactured by, or produced by
the makers or owners of NARCAN®
Manufactured For: Teva Pharmaceuticals USA, Inc.
Parsippany, NJ 07054
Rev. A 1/2024
Purpose
Emergency treatment of opioid overdose
Warning
-
When using this product some people may experience
symptoms when they wake up, such as, shaking, sweating,
nausea, or feeling angry. This is to be expected.
Directions
|
|
Step 1: CHECK if you suspect an overdose:
|
|
Step 2: GIVE 1st dose in the nose
|
|
|
Step 3: CALL
|
|
|
Step 4: WATCH & GIVE
|
|
|
Step 5: STAY
|
Questions?
1-888-838-2872 between 9 am and 5 pm ET, Monday-Friday.
www.tevausa.com/our-products/tevagenerics
Other Information (Other information)
- Store below 77°F (25°C).
- do not freeze
- avoid excessive heat above 40°C (104°F)
- protect from light
- the product is packaged in individually-sealed blisters.
Do not use if the blister is open or torn, or if the device appears damaged.
Inactive Ingredients
benzalkonium chloride, edetate disodium, sodium chloride buffered with sodium hydroxide/hydrochloric acid, water
Active Ingredient (in Each Spray) (Active ingredient (in each spray))
Naloxone hydrochloride 4 mg
Package/label Principal Display Panel – Carton Label (Package/Label Principal Display Panel – Carton Label)
Original Prescription Strength
Easy to Use
Can Save a Life
Compare to the active
ingredient in NARCAN®*
NDC 0480-3478-68
Naloxone Hydrochloride
Nasal Spray
4 mg
Emergency Treatment of
Opioid Overdose
Designed to Rapidly Reverse the
Effects of a Life-Threatening
Opioid Emergency
For use in nose only
2 Single-Dose Nasal Spray Devices
0.003 fl oz (0.1 mL) each
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:41.527161 · Updated: 2026-03-14T23:03:53.608631