These Highlights Do Not Include All The Information Needed To Use Priftin®
3a64fb70-b85e-43d9-8bcd-7e893f568ae1
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions, Paradoxical Drug Reactions ( 5.5 ) 01/2026
Indications and Usage
PRIFTIN is a rifamycin antimycobacterial drug indicated in patients 12 years of age and older for the treatment of active pulmonary tuberculosis (TB) caused by Mycobacterium tuberculosis in combination with one or more antituberculosis (anti-TB) drugs to which the isolate is susceptible. ( 1.1 ) PRIFTIN is indicated for the treatment of latent tuberculosis infection (LTBI) caused by M. tuberculosis in combination with isoniazid in patients 2 years of age and older at high risk of progression to TB disease. ( 1.2 ) See Full Prescribing Information for Limitations of Use. ( 1.1 , 1.2 )
Dosage and Administration
Active pulmonary tuberculosis: PRIFTIN should be used in regimens consisting of an initial 2 month phase followed by a 4 month continuation phase. ( 2.1 ) Initial phase (2 Months): 600 mg twice weekly for two months as directly observed therapy (DOT), with no less than 72 hours between doses, in combination with other antituberculosis drugs. ( 2.1 ) Continuation phase (4 Months): 600 mg once weekly for 4 months as directly observed therapy with isoniazid or another appropriate antituberculosis agent. ( 2.1 ) Latent tuberculosis infection: PRIFTIN should be administered in combination with isoniazid once weekly for 12 weeks as directly observed therapy. ( 2.2 ) Adults and pediatric patients ≥12 years: PRIFTIN (based on weight, see table below) and isoniazid 15 mg/kg (900 mg maximum). ( 2.2 ) Pediatric patients 2 to 11 years: PRIFTIN (based on weight, see table below) and isoniazid 25 mg/kg (900 mg maximum). ( 2.2 ) Weight range PRIFTIN dose Number of PRIFTIN tablets 10–14 kg 300 mg 2 14.1–25 kg 450 mg 3 25.1–32 kg 600 mg 4 32.1–50 kg 750 mg 5 >50 kg 900 mg 6 For Latent Tuberculosis Infection, the maximum recommended dose of PRIFTIN is 900 mg once weekly for 12 weeks. ( 2.2 ) Take with food. Tablets may be crushed and added to semi-solid food. ( 2.3 )
Warnings and Precautions
Hepatotoxicity: Monitor for symptoms of liver injury and discontinue PRIFTIN if signs or symptoms or liver injury occur. ( 5.1 ) Hypersensitivity: Discontinue PRIFTIN if signs or symptoms of hypersensitivity reaction occur. ( 5.2 ) Severe cutaneous adverse reactions: Discontinue PRIFTIN at the first appearance of skin rash, mucosal lesions, or any other sign of hypersensitivity. ( 5.3 ) Relapse in the treatment of active pulmonary tuberculosis: Do not use as a once-weekly continuation phase regimen with isoniazid in HIV-infected patients. Monitor for signs or symptoms of relapse in patients with cavitary lesions or bilateral disease. ( 5.4 , 14.1 ) Paradoxical Drug Reactions: If worsening of symptoms or signs occur during antimycobacterial treatment, consider paradoxical drug reaction in the differential diagnosis, and monitor or treat accordingly. ( 5.5 ) Drug Interactions: May interact with drugs metabolized by CYP450. ( 5.6 , 7.1 , 7.4 ) Discoloration of body fluids: May permanently stain contact lenses or dentures red-orange. ( 5.7 ) Clostridioides difficile –associated diarrhea: Evaluate if diarrhea occurs. ( 5.8 ) Porphyria: Avoid use in patients with porphyria. ( 5.9 )
Contraindications
Known hypersensitivity to any rifamycin. ( 4.1 )
Adverse Reactions
Severe cutaneous adverse reactions (SCARs) such as Stevens-Johnson syndrome (SJS) and drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome have been reported in association with the use of rifapentine (PRIFTIN) treatment regimens in patients with active and latent tuberculosis. Discontinue PRIFTIN at the first appearance of skin rash, mucosal lesions, or any other sign of hypersensitivity [see Patient Counseling Information (17) ] .
Drug Interactions
Rifapentine is an inducer of CYP450 enzymes. Concomitant use of rifapentine with other drugs metabolized by these enzymes, such as protease inhibitors, certain reverse transcriptase inhibitors, and hormonal contraception may cause a significant decrease in plasma concentrations and loss of therapeutic effect [see Drug Interactions (7.1 , 7.2 , 7.3 , 7.4) and Clinical Pharmacology (12.3) ] .
Medication Information
Warnings and Precautions
Hepatotoxicity: Monitor for symptoms of liver injury and discontinue PRIFTIN if signs or symptoms or liver injury occur. ( 5.1 ) Hypersensitivity: Discontinue PRIFTIN if signs or symptoms of hypersensitivity reaction occur. ( 5.2 ) Severe cutaneous adverse reactions: Discontinue PRIFTIN at the first appearance of skin rash, mucosal lesions, or any other sign of hypersensitivity. ( 5.3 ) Relapse in the treatment of active pulmonary tuberculosis: Do not use as a once-weekly continuation phase regimen with isoniazid in HIV-infected patients. Monitor for signs or symptoms of relapse in patients with cavitary lesions or bilateral disease. ( 5.4 , 14.1 ) Paradoxical Drug Reactions: If worsening of symptoms or signs occur during antimycobacterial treatment, consider paradoxical drug reaction in the differential diagnosis, and monitor or treat accordingly. ( 5.5 ) Drug Interactions: May interact with drugs metabolized by CYP450. ( 5.6 , 7.1 , 7.4 ) Discoloration of body fluids: May permanently stain contact lenses or dentures red-orange. ( 5.7 ) Clostridioides difficile –associated diarrhea: Evaluate if diarrhea occurs. ( 5.8 ) Porphyria: Avoid use in patients with porphyria. ( 5.9 )
Indications and Usage
PRIFTIN is a rifamycin antimycobacterial drug indicated in patients 12 years of age and older for the treatment of active pulmonary tuberculosis (TB) caused by Mycobacterium tuberculosis in combination with one or more antituberculosis (anti-TB) drugs to which the isolate is susceptible. ( 1.1 ) PRIFTIN is indicated for the treatment of latent tuberculosis infection (LTBI) caused by M. tuberculosis in combination with isoniazid in patients 2 years of age and older at high risk of progression to TB disease. ( 1.2 ) See Full Prescribing Information for Limitations of Use. ( 1.1 , 1.2 )
Dosage and Administration
Active pulmonary tuberculosis: PRIFTIN should be used in regimens consisting of an initial 2 month phase followed by a 4 month continuation phase. ( 2.1 ) Initial phase (2 Months): 600 mg twice weekly for two months as directly observed therapy (DOT), with no less than 72 hours between doses, in combination with other antituberculosis drugs. ( 2.1 ) Continuation phase (4 Months): 600 mg once weekly for 4 months as directly observed therapy with isoniazid or another appropriate antituberculosis agent. ( 2.1 ) Latent tuberculosis infection: PRIFTIN should be administered in combination with isoniazid once weekly for 12 weeks as directly observed therapy. ( 2.2 ) Adults and pediatric patients ≥12 years: PRIFTIN (based on weight, see table below) and isoniazid 15 mg/kg (900 mg maximum). ( 2.2 ) Pediatric patients 2 to 11 years: PRIFTIN (based on weight, see table below) and isoniazid 25 mg/kg (900 mg maximum). ( 2.2 ) Weight range PRIFTIN dose Number of PRIFTIN tablets 10–14 kg 300 mg 2 14.1–25 kg 450 mg 3 25.1–32 kg 600 mg 4 32.1–50 kg 750 mg 5 >50 kg 900 mg 6 For Latent Tuberculosis Infection, the maximum recommended dose of PRIFTIN is 900 mg once weekly for 12 weeks. ( 2.2 ) Take with food. Tablets may be crushed and added to semi-solid food. ( 2.3 )
Contraindications
Known hypersensitivity to any rifamycin. ( 4.1 )
Adverse Reactions
Severe cutaneous adverse reactions (SCARs) such as Stevens-Johnson syndrome (SJS) and drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome have been reported in association with the use of rifapentine (PRIFTIN) treatment regimens in patients with active and latent tuberculosis. Discontinue PRIFTIN at the first appearance of skin rash, mucosal lesions, or any other sign of hypersensitivity [see Patient Counseling Information (17) ] .
Drug Interactions
Rifapentine is an inducer of CYP450 enzymes. Concomitant use of rifapentine with other drugs metabolized by these enzymes, such as protease inhibitors, certain reverse transcriptase inhibitors, and hormonal contraception may cause a significant decrease in plasma concentrations and loss of therapeutic effect [see Drug Interactions (7.1 , 7.2 , 7.3 , 7.4) and Clinical Pharmacology (12.3) ] .
Description
Warnings and Precautions, Paradoxical Drug Reactions ( 5.5 ) 01/2026
Section 42229-5
Limitations of Use
Do not use PRIFTIN monotherapy in either the initial or the continuation phases of active antituberculous treatment.
PRIFTIN should not be used once weekly in the continuation phase regimen in combination with isoniazid (INH) in HIV-infected patients with active pulmonary tuberculosis because of a higher rate of failure and/or relapse with rifampin (RIF)-resistant organisms [see Warnings and Precautions (5.4) and Clinical Studies (14.1)].
PRIFTIN has not been studied as part of the initial phase treatment regimen in HIV-infected patients with active pulmonary tuberculosis.
Section 42231-1
| Medication Guide PRIFTIN (prif - tin) (rifapentine) Tablets |
|||||
|---|---|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | |||||
| Revised: December 2025 | |||||
| Read this Medication Guide before you start taking PRIFTIN and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment. | |||||
|
What is the most important information I should know about PRIFTIN? |
|||||
PRIFTIN may cause serious side effects, including:
|
|||||
|
|
||||
|
|||||
|
|
||||
| Signs and symptoms of a flu-like reaction may include: | |||||
|
|
|
|||
|
|||||
|
|
|
|||
| What is PRIFTIN? | |||||
PRIFTIN is a prescription medicine used with other anti-tuberculosis (TB) medicines to:
|
|||||
PRIFTIN should not be used:
|
|||||
| PRIFTIN is safe and effective in children older than 2 years of age who have inactive (latent TB), but it is not known if PRIFTIN is safe and effective for use in the treatment of active TB in children under 12 years of age. | |||||
Who should not take PRIFTIN?
|
|||||
| What should I tell my doctor before taking PRIFTIN? | |||||
Before taking PRIFTIN, tell your doctor about all of your medical conditions, including if you:
|
|||||
| Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |||||
| Using PRIFTIN with other medicines may affect each other causing serious side effects. PRIFTIN may affect the way other medicines work, and other medicines may affect how PRIFTIN works. Especially tell your doctor if you take medicines to treat HIV infection or oral contraceptives. | |||||
| Ask your doctor or pharmacist for a list of these medicines if you are not sure. | |||||
| Know the medicines you take. Keep a list of them to show your doctor or pharmacist when you get a new medicine. | |||||
How should I take PRIFTIN?
|
|||||
| What are possible side effects of PRIFTIN? | |||||
PRIFTIN may cause serious side effects, including:
|
|||||
| The most common side effects of PRIFTIN include: allergic reactions and flu-like symptoms; abnormalities such as low red blood cells, low white blood cells, coughing up blood, cough, excessive number of platelets in the blood, increased sweating, high liver function tests, back pain, rash, decreased appetite, joint pain, increased blood urea, and headache. | |||||
| Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of PRIFTIN. For more information, ask your doctor or pharmacist. | |||||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||||
How should I store PRIFTIN?
|
|||||
| General information about the safe and effective use of PRIFTIN. | |||||
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use PRIFTIN for a condition for which it was not prescribed. Do not give PRIFTIN to other people, even if they have the same symptoms you have. It may harm them. | |||||
| This Medication Guide summarizes the most important information about PRIFTIN. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about PRIFTIN that is written for healthcare professionals. | |||||
| What are the ingredients in PRIFTIN? | |||||
| Active ingredient: rifapentine | |||||
| Inactive ingredients: calcium stearate, disodium EDTA, FD&C Blue No. 2 aluminum lake, hydroxypropyl cellulose, hypromellose USP, microcrystalline cellulose, polyethylene glycol, pregelatinized starch, propylene glycol, sodium ascorbate, sodium lauryl sulfate, sodium starch glycolate, synthetic red iron oxide, and titanium dioxide | |||||
| Manufactured by: sanofi-aventis U.S. LLC, Morristown, NJ 07960 For more information, go to www.sanofi.us or call 1-800-633-1610, and select option 1. |
Section 43683-2
| Warnings and Precautions, Paradoxical Drug Reactions (5.5) | 01/2026 |
Section 44425-7
Storage
Store at 25°C (77°F); excursions permitted 15–30°C (59–86°F) (see USP Controlled Room Temperature). Protect from excessive heat and humidity.
10 Overdosage
While there is no experience with the treatment of acute overdose with PRIFTIN, clinical experience with rifamycins suggests that gastric lavage to evacuate gastric contents (within a few hours of overdose), followed by instillation of an activated charcoal slurry into the stomach, may help adsorb any remaining drug from the gastrointestinal tract.
Rifapentine and 25-desacetyl rifapentine are 97.7% and 93.2% plasma protein bound, respectively. Rifapentine and related compounds excreted in urine account for only 17% of the administered dose, therefore, neither hemodialysis nor forced diuresis is expected to enhance the systemic elimination of unchanged rifapentine from the body of a patient with PRIFTIN overdose.
5.9 Porphyria
Porphyria has been reported in patients receiving rifampin, attributed to induction of delta amino levulinic acid synthetase. Because PRIFTIN may have similar enzyme induction properties, avoid the use of PRIFTIN in patients with porphyria.
11 Description
PRIFTIN (rifapentine) for oral administration contains 150 mg of the active ingredient rifapentine per tablet.
The 150 mg tablets also contain, as inactive ingredients: calcium stearate, disodium EDTA, FD&C Blue No. 2 aluminum lake, hydroxypropyl cellulose, hypromellose USP, microcrystalline cellulose, polyethylene glycol, pregelatinized starch, propylene glycol, sodium ascorbate, sodium lauryl sulfate, sodium starch glycolate, synthetic red iron oxide, and titanium dioxide.
Rifapentine is a rifamycin derivative antimicrobial and has a similar profile of microbiological activity to rifampicin. The molecular weight is 877.04.
The molecular formula is C47H64N4O12.
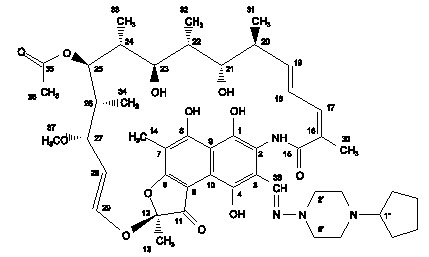
The chemical name for rifapentine is rifamycin, 3-[[(4-cyclopentyl-1-piperazinyl)imino]methyl]- or 3-[N-(4-cyclopentyl - 1-piperazinyl)formimidoyl] rifamycin or 5,6,9,17,19,21-hexahydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-8-[N-(4-cyclopentyl-l-piperazinyl)-formimidoyl]-2,7-(epoxypentadeca[1,11,13]trienimino)naphtho[2,1-b]furan-1,11(2H)-dione 21-acetate. It has the following structure:
8.4 Pediatric Use
The safety and effectiveness of PRIFTIN in the treatment of active pulmonary tuberculosis have not been established in pediatric patients under the age of 12.
The safety and effectiveness of PRIFTIN in combination with isoniazid once-weekly regimen has been evaluated in pediatric patients (2 to 17 years of age) for the treatment of latent tuberculosis infection. In clinical studies, the safety profile in pediatric patients was similar to that observed in adult patients [see Adverse Reactions (6.1) and Clinical Studies (14.2)].
In a pharmacokinetic study conducted in 2 to 11-year-old pediatric patients with latent tuberculosis infection, PRIFTIN was administered once weekly based on weight (15 mg/kg to 30 mg/kg, up to a maximum of 900 mg). Exposures (AUC) in pediatric patients 2 to 11 years old with latent tuberculosis infection were higher (average 31%) than those observed in adults receiving PRIFTIN 900 mg once weekly [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
8.5 Geriatric Use
Clinical studies with PRIFTIN did not include sufficient numbers of subjects aged 65 years and over to determine whether they respond differently from younger subjects. In a pharmacokinetic study with PRIFTIN, no substantial differences in the pharmacokinetics of rifapentine and 25-desacetyl metabolite were observed in the elderly compared to younger adults [see Clinical Pharmacology (12.3)].
2.3 Administration
Take PRIFTIN with meals. Administration of PRIFTIN with a meal increases oral bioavailability and may reduce the incidence of gastrointestinal upset, nausea, and/or vomiting [see Clinical Pharmacology (12.3)].
For patients who cannot swallow tablets, the tablets may be crushed and added to a small amount of semi-solid food, all of which should be consumed immediately [see Clinical Pharmacology (12.3)].
5.1 Hepatotoxicity
Elevations of liver transaminases may occur in patients receiving PRIFTIN [see Adverse Reactions (6.1)]. Patients on PRIFTIN should be monitored for symptoms of liver injury.
Patients with abnormal liver tests and/or liver disease or patients initiating treatment for active pulmonary tuberculosis should only be given PRIFTIN in cases of necessity and under strict medical supervision. In such patients, obtain serum transaminase levels prior to therapy and every 2 to 4 weeks while on therapy. Discontinue PRIFTIN if evidence of liver injury occurs.
4 Contraindications
Known hypersensitivity to any rifamycin. (4.1)
6 Adverse Reactions
The following serious and otherwise important adverse drug reactions are discussed in greater detail in other sections of labeling:
- Hepatotoxicity [see Warnings and Precautions (5.1)]
- Hypersensitivity [see Contraindications (4.1) and Warnings and Precautions (5.2)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.3)]
- Paradoxical Drug Reactions [see Warnings and Precautions (5.5)]
- Discoloration of Body Fluids [see Warnings and Precautions (5.7)]
- Clostridioides Difficile–Associated Diarrhea [see Warnings and Precautions (5.8)]
- Porphyria [see Warnings and Precautions (5.9)]
7 Drug Interactions
- Protease Inhibitors and Reverse Transcriptase Inhibitors. (5.2, 7.1)
- Hormonal Contraceptives: Use an effective non-hormonal method of contraception or add a barrier method of contraception during treatment with PRIFTIN. (7.3)
- May increase metabolism and decrease the activity of drugs metabolized by cytochrome P450 3A4 and 2C8/9. Dosage adjustments may be necessary if given concomitantly. (7.4)
4.1 Hypersensitivity
PRIFTIN is contraindicated in patients with a history of hypersensitivity to rifamycins.
12.3 Pharmacokinetics
When oral doses of PRIFTIN were administered once daily or once every 72 hours to healthy volunteers for 10 days, single dose AUC (0–∞) of rifapentine was similar to its steady-state AUCss (0–24h) or AUCss (0–72h) values, suggesting no significant auto-induction effect on steady-state pharmacokinetics of rifapentine. Steady-state conditions were achieved by day 10 following daily administration of PRIFTIN 600 mg. No plasma accumulation of rifapentine and 25-desacetyl rifapentine (active metabolite) is expected after once weekly administration of PRIFTIN.
The pharmacokinetic parameters of rifapentine and 25-desacetyl rifapentine on day 10 following oral administration of 600 mg PRIFTIN every 72 hours to healthy volunteers are described in Table 5.
| Parameter | Rifapentine | 25-desacetyl Rifapentine |
|---|---|---|
| Mean ± SD (n=12) | ||
| Cmax (µg/mL) | 15.05 ± 4.62 | 6.26 ± 2.06 |
| AUC (0–72h) (µg∙h/mL) | 319.54 ± 91.52 | 215.88 ± 85.96 |
| T1/2 (h) | 13.19 ± 1.38 | 13.35 ± 2.67 |
| Tmax (h) | 4.83 ± 1.80 | 11.25 ± 2.73 |
| Cl/F (L/h) | 2.03 ± 0.60 | -- |
The pharmacokinetic parameters of rifapentine and 25-desacetyl rifapentine following single-dose oral administration of 900 mg PRIFTIN in combination with 900 mg isoniazid in fed conditions are described in Table 6.
| Parameter | Rifapentine | 25-desacetyl Rifapentine |
|---|---|---|
| Cmax (µg/mL) | 25.8 ± 5.83 | 13.3 ± 4.83 |
| AUC (µg∙h/mL) | 817 ± 128 | 601 ± 187 |
| T1/2(h) | 16.6 ± 5.02 | 17.5 ± 7.42 |
| Tmax (h) Median (Min–Max).
|
8 (3–10) | 24 (10–36) |
| Cl/F (L/h) | 1.13 ± 0.174 | NA Not Applicable.
|
5.6 Drug Interactions
Rifapentine is an inducer of CYP450 enzymes. Concomitant use of rifapentine with other drugs metabolized by these enzymes, such as protease inhibitors, certain reverse transcriptase inhibitors, and hormonal contraception may cause a significant decrease in plasma concentrations and loss of therapeutic effect [see Drug Interactions (7.1, 7.2, 7.3, 7.4) and Clinical Pharmacology (12.3)].
7.5 Other Interactions
The conversion of PRIFTIN to 25-desacetyl rifapentine is mediated by an esterase enzyme. There is minimal potential for PRIFTIN metabolism to be inhibited or induced by another drug, based upon the characteristics of the esterase enzymes.
Since PRIFTIN is highly bound to albumin, drug displacement interactions may also occur [see Clinical Pharmacology (12.3)].
1 Indications and Usage
- PRIFTIN is a rifamycin antimycobacterial drug indicated in patients 12 years of age and older for the treatment of active pulmonary tuberculosis (TB) caused by Mycobacterium tuberculosis in combination with one or more antituberculosis (anti-TB) drugs to which the isolate is susceptible. (1.1)
- PRIFTIN is indicated for the treatment of latent tuberculosis infection (LTBI) caused by M. tuberculosis in combination with isoniazid in patients 2 years of age and older at high risk of progression to TB disease. (1.2)
- See Full Prescribing Information for Limitations of Use. (1.1, 1.2)
12.1 Mechanism of Action
Rifapentine, a cyclopentyl rifamycin, is an antimycobacterial agent [see Microbiology (12.4)].
5 Warnings and Precautions
- Hepatotoxicity: Monitor for symptoms of liver injury and discontinue PRIFTIN if signs or symptoms or liver injury occur. (5.1)
- Hypersensitivity: Discontinue PRIFTIN if signs or symptoms of hypersensitivity reaction occur. (5.2)
- Severe cutaneous adverse reactions: Discontinue PRIFTIN at the first appearance of skin rash, mucosal lesions, or any other sign of hypersensitivity. (5.3)
- Relapse in the treatment of active pulmonary tuberculosis: Do not use as a once-weekly continuation phase regimen with isoniazid in HIV-infected patients. Monitor for signs or symptoms of relapse in patients with cavitary lesions or bilateral disease. (5.4, 14.1)
- Paradoxical Drug Reactions: If worsening of symptoms or signs occur during antimycobacterial treatment, consider paradoxical drug reaction in the differential diagnosis, and monitor or treat accordingly. (5.5)
- Drug Interactions: May interact with drugs metabolized by CYP450. (5.6, 7.1, 7.4)
- Discoloration of body fluids: May permanently stain contact lenses or dentures red-orange. (5.7)
- Clostridioides difficile–associated diarrhea: Evaluate if diarrhea occurs. (5.8)
- Porphyria: Avoid use in patients with porphyria. (5.9)
2 Dosage and Administration
-
Active pulmonary tuberculosis: PRIFTIN should be used in regimens consisting of an initial 2 month phase followed by a 4 month continuation phase. (2.1)
Initial phase (2 Months): 600 mg twice weekly for two months as directly observed therapy (DOT), with no less than 72 hours between doses, in combination with other antituberculosis drugs. (2.1)
Continuation phase (4 Months): 600 mg once weekly for 4 months as directly observed therapy with isoniazid or another appropriate antituberculosis agent. (2.1) -
Latent tuberculosis infection: PRIFTIN should be administered in combination with isoniazid once weekly for 12 weeks as directly observed therapy. (2.2)
Adults and pediatric patients ≥12 years: PRIFTIN (based on weight, see table below) and isoniazid 15 mg/kg (900 mg maximum). (2.2)
Pediatric patients 2 to 11 years: PRIFTIN (based on weight, see table below) and isoniazid 25 mg/kg (900 mg maximum). (2.2)
Weight range PRIFTIN dose Number of PRIFTIN tablets 10–14 kg 300 mg 2 14.1–25 kg 450 mg 3 25.1–32 kg 600 mg 4 32.1–50 kg 750 mg 5 >50 kg 900 mg 6
For Latent Tuberculosis Infection, the maximum recommended dose of PRIFTIN is 900 mg once weekly for 12 weeks. (2.2) - Take with food. Tablets may be crushed and added to semi-solid food. (2.3)
7.3 Hormonal Contraceptives
PRIFTIN may reduce the effectiveness of hormonal contraceptives. Patients using hormonal contraception should be advised to use an alternative non-hormonal contraceptive method or add a barrier method of contraception during treatment with PRIFTIN [see Use in Specific Populations (8.3) and Clinical Pharmacology (12.3)].
3 Dosage Forms and Strengths
PRIFTIN is supplied as 150 mg round normal convex dark-pink film-coated tablets debossed "F" on one side of tablet.
5.8 Clostridioides Difficile
Clostridioides difficile–associated diarrhea (CDAD) has been reported with the use of nearly all systemic antibacterial agents, including PRIFTIN, with severity ranging from mild diarrhea to fatal colitis. Treatment with antibacterial agents can alter the normal flora of the colon and may permit overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary because CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, discontinue antibacterial use not directed against C. difficile if possible. Institute appropriate measures such as fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation as clinically indicated.
6.2 Postmarketing Experience
The following adverse reactions have been identified from postmarketing surveillance of rifapentine. Because these reactions are reported from a population of unknown size, it is not always possible to estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations
- Pregnancy: Based on animal data, may cause fetal harm. (8.1)
- Lactation: Monitor infants exposed to PRIFTIN through breast milk for irritability, prolonged unexplained crying, yellowing of the eyes, changes in color of the urine or stool. (8.2)
- Pediatrics: Safety and effectiveness in treating active pulmonary tuberculosis in pediatric patients under the age of 12 years have not been established. (8.4)
5.5 Paradoxical Drug Reactions
Paradoxical drug reactions are characterized by the recurrence or appearance of new symptoms or physical and radiological signs in a patient who had previously shown improvement with appropriate antimycobacterial treatment, in the absence of disease relapse, poor treatment compliance, drug resistance, side effects of treatment, or secondary infection/diagnosis. Paradoxical drug reactions have been reported with antimycobacterial therapy, including PRIFTIN, within the first few weeks or months of initiation of tuberculosis therapy [see Adverse Reactions (6.2)].
Paradoxical drug reactions are often transient and should not be misinterpreted as failure to respond to treatment. If worsening of symptoms or signs occurs during antimycobacterial treatment, consider paradoxical drug reaction in the differential diagnosis, and monitor or treat accordingly.
Advise patients to seek medical advice immediately if their symptoms of tuberculosis worsen or reappear.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.7 Discoloration of Body Fluids
PRIFTIN may produce a red-orange discoloration of body tissues and/or fluids (e.g., skin, teeth, tongue, urine, feces, saliva, sputum, tears, sweat, and cerebrospinal fluid). Contact lenses or dentures may become permanently stained.
1.1 Active Pulmonary Tuberculosis
PRIFTIN® (rifapentine) is indicated in adults and pediatric patients 12 years and older for the treatment of active pulmonary tuberculosis (TB) caused by Mycobacterium tuberculosis. PRIFTIN must always be used in combination with one or more antituberculosis (anti-TB) drugs to which the isolate is susceptible [see Dosage and Administration (2.1) and Clinical Studies (14.1)].
1.2 Latent Tuberculosis Infection
PRIFTIN is indicated in adults and pediatric patients 2 years and older for the treatment of latent tuberculosis infection caused by Mycobacterium tuberculosis in patients at high risk of progression to tuberculosis disease (including those in close contact with active tuberculosis patients, recent conversion to a positive tuberculin skin test, HIV-infected patients, or those with pulmonary fibrosis on radiograph) [see Clinical Studies (14.2)].
17 Patient Counseling Information
Advise patient to read FDA-approved patient labeling (Medication Guide).
7.4 Cytochrome P450 3a4 and 2c8/9
Rifapentine is an inducer of cytochromes P450 3A4 and P450 2C8/9. Therefore, PRIFTIN may increase the metabolism of other coadministered drugs that are metabolized by these enzymes. Induction of enzyme activities by PRIFTIN occurred within 4 days after the first dose. Enzyme activities returned to baseline levels 14 days after discontinuing PRIFTIN.
Rifampin has been reported to accelerate the metabolism and may reduce the activity of the following drugs; hence, PRIFTIN may also increase the metabolism and decrease the activity of these drugs. Dosage adjustments of the drugs in Table 4 or of other drugs metabolized by cytochrome P450 3A4 or P450 2C8/9 may be necessary if they are given concurrently with PRIFTIN.
| Drug Class | Examples of Drugs Within Class |
|---|---|
| Antiarrhythmics | Disopyramide, mexiletine, quinidine, tocainide |
| Antibiotics | Chloramphenicol, clarithromycin, dapsone, doxycycline; Fluoroquinolones (such as ciprofloxacin) |
| Oral Anticoagulants | Warfarin |
| Anticonvulsants | Phenytoin |
| Antimalarials | Quinine |
| Azole Antifungals | Fluconazole, itraconazole, ketoconazole |
| Antipsychotics | Haloperidol |
| Barbiturates | Phenobarbital |
| Benzodiazepines | Diazepam |
| Beta-Blockers | Propranolol |
| Calcium Channel Blockers | Diltiazem, nifedipine, verapamil |
| Cardiac Glycoside Preparations | Digoxin |
| Corticosteroids | Prednisone |
| Fibrates | Clofibrate |
| Oral Hypoglycemics | Sulfonylureas (e.g., glyburide, glipizide) |
| Hormonal Contraceptives/Progestins | Ethinyl estradiol, levonorgestrel |
| Immunosuppressants | Cyclosporine, tacrolimus |
| Methylxanthines | Theophylline |
| Narcotic analgesics | Methadone |
| Phosphodiesterase-5 (PDE-5) Inhibitors | Sildenafil |
| Thyroid preparations | Levothyroxine |
| Tricyclic antidepressants | Amitriptyline, nortriptyline |
14.1 Active Pulmonary Tuberculosis
PRIFTIN was studied in two randomized, open-label controlled clinical trials in the treatment of active pulmonary tuberculosis.
The first trial was an open-label, prospective, parallel group, active-controlled trial in HIV-negative patients with active pulmonary tuberculosis. The population mostly comprised Black (approximately 60%) or multiracial (approximately 31%) patients. Treatment groups were comparable for age and sex and consisted primarily of male subjects with a mean age of 37 ± 11 years. In the initial 2-month phase of treatment, 361 patients received PRIFTIN 600 mg twice a week in combination with daily isoniazid, pyrazinamide, and ethambutol and 361 subjects received rifampin 600 mg in combination with isoniazid, pyrazinamide and ethambutol all administered daily. The doses of the companion drugs were the same in both treatment groups during the initial phase: isoniazid 300 mg, pyrazinamide 2000 mg, and ethambutol 1200 mg. For patients weighing less than 50 kg, the doses of rifampin (450 mg), pyrazinamide (1500 mg) and ethambutol (800 mg) were reduced. Ethambutol was discontinued when isoniazid and rifampin susceptibility testing results were confirmed. During the 4-month continuation phase, 317 patients in the PRIFTIN group continued to receive PRIFTIN 600 mg dosed once weekly with isoniazid 300 mg and 304 patients in the rifampin group received twice weekly rifampin and isoniazid 900 mg. For patients weighing less than 50 kg, the doses of rifampin (450 mg) and isoniazid (600 mg) were reduced. Both treatment groups received pyridoxine (Vitamin B6) over the 6-month treatment period. Treatment was directly observed. 65/361 (18%) of patients in the PRIFTIN group and 34/361 (9%) in the rifampin group received overdoses of one or more of the administered study medications during the initial or continuation phase of treatment. Seven of these patients had adverse reactions reported with the overdose (5 in the PRIFTIN group and 2 in the rifampin group).
Table 8 below contains assessments of sputum conversion at end of treatment (6 months) and relapse rates at the end of follow-up (24 months).
| PRIFTIN Combination Treatment % and (n/N All data for patients with confirmed susceptible M. tuberculosis (PRIFTIN combination treatment, N=286; rifampin combination treatment, N=283). ) |
Rifampin Combination Treatment % and (n/N) | |
|---|---|---|
| Status at End of 6 months of Treatment | ||
| Converted | 87% (248/286) | 80% (226/283) |
| Not Converted | 1% (4/286) | 3% (8/283) |
| Lost to Follow-up | 12% (34/286) | 17% (49/283) |
|
Status Through 24 Month Follow-up
Twenty-two (22) deaths occurred during the study; 11 in each treatment group.
|
||
| Relapsed | 12% (29/248) | 7% (15/226) |
| Sputum Negative | 57% (142/248) | 64% (145/226) |
| Lost to Follow-up | 31% (77/248) | 29% (66/226) |
Risk of relapse was greater in the group treated with the PRIFTIN combination. Higher relapse rates were associated with a lower rate of compliance as well as a failure to convert sputum cultures at the end of the initial 2-month treatment phase. Relapse rates were also higher for males in both regimens. Relapse in the PRIFTIN group was not associated with development of monoresistance to rifampin.
The second trial was randomized, open-label performed in 1075 HIV-negative and HIV-positive patients with active pulmonary tuberculosis. Patients with culture-positive, drug-susceptible pulmonary tuberculosis who had completed the initial 2-month phase of treatment with 4 drugs (rifampin, isoniazid, pyrazinamide, and either ethambutol or streptomycin) under direct observation were randomly assigned to receive either PRIFTIN 600 mg and isoniazid 15 mg/kg (max 900 mg) once weekly or rifampin 10 mg/kg (max 600 mg) and isoniazid 15 mg/kg (max 900 mg) twice weekly for the 4 month continuation phase. Study drugs were given under direct observation therapy in both groups.
In the PRIFTIN group, 502 HIV-negative and 36 HIV-positive patients were randomized and in the rifampin group 502 HIV-negative and 35 HIV-positive patients were randomized to treatment. Enrollment of HIV-infected patients was stopped when 4 of 36 patients in the PRIFTIN combination group relapsed with isolates that were rifampin resistant.
Table 9 below contains assessments of sputum conversion at the end of treatment (6 months total: 2 months of initial and 4 months of randomized continuation treatment) and relapse rates at the end of follow-up (24 months) in all HIV-negative patients randomized to treatment. Positive culture was based on either one sputum sample with >10 colonies on solid media OR at least 2 positive sputum samples on liquid or solid media. However, only one sputum sample was collected at each visit in a majority of patients.
| PRIFTIN Combination Treatment % (n/N) | Rifampin Combination Treatment % (n/N) | |
|---|---|---|
| Status at End of 4 Months Continuation Phase | ||
| Treatment Response Treatment response was defined as subjects who had two negative sputum cultures after 16 doses of rifampin and isoniazid or after 8 doses of PRIFTIN and isoniazid, and remained sputum negative through the end of continuation phase therapy.
|
93.8% (471/502) | 91% (457/502) |
| Not Converted | 1% (5/502) | 1.2% (6/502) |
| Did Not Complete Treatment Due to drug toxic effects, non-adherence, withdrawal of consent, receipt of non-study regimen, other.
|
4.2% (21/502) | 7% (35/502) |
| Deaths | 1% (5/502) | 0.8% (4/502) |
| Status Through 24 Month Follow-up: | ||
| Relapsed | 8.7% (41/471) | 4.8% (22/457) |
| Sputum Negative | 79.4% (374/471) | 80.1% (366/457) |
| Lost to Follow-up | 7.9% (37/471) | 9.8% (45/457) |
| Deaths | 4% (19/471) | 5.3% (24/457) |
In HIV-negative patients, higher relapse rates were seen in patients with a positive sputum culture at 2 months (i.e., at the time of study randomization), cavitation on chest x-ray, and bilateral pulmonary involvement.
Sixty-one HIV-positive patients were assessed for relapse. The rates of relapse were 16.7% (5/30) in the PRIFTIN group and 9.7% (3/31) in the rifampin group. In HIV-positive patients, 4 of the 5 relapses in the PRIFTIN combination group involved M. tuberculosis strains with rifampin monoresistance. No relapse strain in the twice weekly rifampin/isoniazid group acquired drug resistance.
The death rate among all study participants did not differ between the two treatment groups.
14.2 Latent Tuberculosis Infection
A multicenter, prospective, open-label, randomized, active-controlled trial compared the effectiveness of 12 weekly doses of PRIFTIN in combination with isoniazid (3RPT/INH arm) administered by directly observed therapy to 9 months of self-administered daily isoniazid (9INH arm). The trial enrolled patients two years of age or older with positive tuberculin skin test and at high risk for progression to tuberculosis disease. Enrolled patients included those having close contact with a patient with active tuberculosis disease, recent (within two years) conversion to a positive tuberculin skin test, HIV-infection, or fibrosis on chest radiograph. PRIFTIN was dosed by weight, for a maximum of 900 mg weekly. Isoniazid mg/kg dose was determined by age, for a maximum of 900 mg weekly in the 3RPT/INH arm and 300 mg daily in the 9INH arm [see Dosage and Administration (2.2)].
The outcome measure was the development of active tuberculosis disease, defined as culture-confirmed tuberculosis in adults and culture-confirmed or clinical tuberculosis in pediatric patients less than 18 years of age, at 33 months after trial enrollment. Patients who were found after enrollment to be ineligible because they had active tuberculosis disease, were contacts of a source case with culture-negative or drug-resistant tuberculosis disease cases or no information regarding susceptibility of M. tuberculosis, and young pediatric patients lacking a positive TST on initial and repeat testing were excluded from the analysis.
Active tuberculosis disease developed in 5 of 3074 randomized patients in the 3RPT/INH group (0.16%) versus 10 of 3074 patients in 9INH group (0.32%), for a difference in cumulative rates of 0.17%, 95% CI (-0.43, 0.09) (Table 10).
| Outcome | 3RPT/INH (n=3074) | 9INH (n=3074) | Difference Rate in the 3RPT/INH group minus the rate in the 9INH group. , 95% CI |
|---|---|---|---|
| Tuberculosis n (%) | 5 (0.16) | 10 (0.32) | -0.16 (-0.42, 0.01) |
| Cumulative TB Rate (%) | 0.17 | 0.35 | -0.17 (-0.43, 0.09) |
| Deaths | 22 (0.72) | 35 (1.14) | -0.42 (-0.91, 0.06) |
| Lost to Follow-Up | 320 (10.41) | 357 (11.61) | -1.20 (-2.77, -0.36) |
The proportion of patients completing treatment was 81.2% in the 3RPT/INH group and 68.3% in the 9INH group for a difference (3RPT/INH-9INH) of 12.8% 95% CI (10.7, 15.0).
In the 9INH treatment group, two of the thirteen culture-confirmed cases were found to be isoniazid-monoresistant. In the 3RPT/INH treatment group, one of the seven cases was rifampin-resistant, isoniazid-susceptible M. bovis infection.
5.3 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions (SCARs) such as Stevens-Johnson syndrome (SJS) and drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome have been reported in association with the use of rifapentine (PRIFTIN) treatment regimens in patients with active and latent tuberculosis. Discontinue PRIFTIN at the first appearance of skin rash, mucosal lesions, or any other sign of hypersensitivity [see Patient Counseling Information (17)].
7.6 Interactions With Laboratory Tests
Therapeutic concentrations of rifampin have been shown to inhibit standard microbiological assays for serum folate and Vitamin B12. Similar drug-laboratory interactions should be considered for PRIFTIN; thus, alternative assay methods should be considered.
5.2 Hypersensitivity and Related Reactions
Hypersensitivity reactions may occur in patients receiving PRIFTIN. Signs and symptoms of these reactions may include hypotension, urticaria, angioedema, acute bronchospasm, conjunctivitis, thrombocytopenia, neutropenia or flu-like syndrome (weakness, fatigue, muscle pain, nausea, vomiting, headache, fever, chills, aches, rash, itching, sweats, dizziness, shortness of breath, chest pain, cough, syncope, palpitations). There have been reports of anaphylaxis [see Patient Counseling Information (17)].
Monitor patients receiving PRIFTIN therapy for signs and/or symptoms of hypersensitivity reactions. If these symptoms occur, administer supportive measures and discontinue PRIFTIN.
2.1 Dosage in Active Pulmonary Tuberculosis
PRIFTIN is only recommended for the treatment of active pulmonary tuberculosis caused by drug-susceptible organisms as part of regimens consisting of a 2-month initial phase followed by a 4-month continuation phase.
PRIFTIN should not be used in the treatment of active pulmonary tuberculosis caused by rifampin-resistant strains.
2.2 Dosage in Latent Tuberculosis Infection
PRIFTIN should be administered once weekly in combination with isoniazid for 12 weeks as directly observed therapy.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Hepatocellular carcinomas were increased in male NMRI mice (Harlan Winklemann) which were treated orally with rifapentine for two years at or above doses of 5 mg/kg/day (0.04 times the recommended human dose based on body surface area conversions). In a two year rat study, there was an increase in nasal cavity adenomas in Wistar rats treated orally with rifapentine at 40 mg/kg/day (0.6 times human dose based on body surface area conversions).
Rifapentine was negative in the following genotoxicity tests: in vitro gene mutation assay in bacteria (Ames test); in vitro point mutation test in Aspergillus nidulans; in vitro gene conversion assay in Saccharomyces cerevisiae; host-mediated (mouse) gene conversion assay with Saccharomyces cerevisiae; in vitro Chinese hamster ovary cell/hypoxanthine-guanine phosphoribosyltransferase (CHO/HGPRT) forward mutation assay; in vitro chromosomal aberration assay utilizing rat lymphocytes; and in vivo mouse bone marrow micronucleus assay.
The 25-desacetyl metabolite of rifapentine was positive in the in vitro mammalian chromosome aberration test in V79 Chinese hamster cells, but was negative in the in vitro gene mutation assay in bacteria (Ames test), the in vitro Chinese hamster ovary cell/hypoxanthine-guanine phosphoribosyltransferase (CHO/HGPRT) forward mutation assay, and the in vivo mouse bone marrow micronucleus assay. Fertility and reproductive performance were not affected by oral administration of rifapentine to male and female rats at doses of up to 20 mg/kg/day (one-third of the human dose based on body surface area conversions).
Principal Display Panel 150 Mg Tablet Blister Pack Carton
NDC 0088-2102-24
PRIFTIN®
RIFAPENTINE
150 mg per tablet
Dispense with Medication Guide
24 Tablets
Three 8-Tablet
Blister Strips
sanofi
7.1 Protease Inhibitors and Reverse Transcriptase Inhibitors
Rifapentine is an inducer of CYP450 enzymes. Concomitant use of PRIFTIN with other drugs metabolized by these enzymes, such as protease inhibitors and certain reverse transcriptase inhibitors, may cause a significant decrease in plasma concentrations and loss of therapeutic effect of the protease inhibitor or reverse transcriptase inhibitor [see Warnings and Precautions (5.6) and Clinical Pharmacology (12.3)].
5.4 Relapse in the Treatment of Active Pulmonary Tuberculosis
PRIFTIN has not been evaluated as part of the initial phase treatment regimen in HIV-infected patients with active pulmonary TB.
Do not use PRIFTIN as a once-weekly continuation phase regimen in HIV-infected patients with active pulmonary tuberculosis because of a higher rate of failure and/or relapse with rifampin-resistant organisms [see Clinical Studies (14.1)].
Higher relapse rates may occur in patients with cavitary pulmonary lesions and/or positive sputum cultures after the initial phase of active tuberculosis treatment and in patients with evidence of bilateral pulmonary disease. Monitor for signs and symptoms of TB relapse in these patients [see Clinical Studies (14.1)].
Poor adherence to therapy is associated with high relapse rate. Emphasize the importance of compliance with therapy [see Patient Counseling Information (17)].
7.2 Fixed Dose Combination of Efavirenz, Emtricitabine, and Tenofovir
Once-weekly coadministration of 900 mg PRIFTIN with the antiretroviral fixed-dose combination of efavirenz 600 mg, emtricitabine 200 mg and tenofovir disoproxil fumarate 300 mg in HIV-infected patients did not result in any substantial change in steady state exposures of efavirenz, emtricitabine, and tenofovir. No clinically significant change in CD4 cell counts or viral loads were noted [see Clinical Pharmacology (12.3)].
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
Do not use PRIFTIN monotherapy in either the initial or the continuation phases of active antituberculous treatment.
PRIFTIN should not be used once weekly in the continuation phase regimen in combination with isoniazid (INH) in HIV-infected patients with active pulmonary tuberculosis because of a higher rate of failure and/or relapse with rifampin (RIF)-resistant organisms [see Warnings and Precautions (5.4) and Clinical Studies (14.1)].
PRIFTIN has not been studied as part of the initial phase treatment regimen in HIV-infected patients with active pulmonary tuberculosis.
Section 42231-1 (42231-1)
| Medication Guide PRIFTIN (prif - tin) (rifapentine) Tablets |
|||||
|---|---|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | |||||
| Revised: December 2025 | |||||
| Read this Medication Guide before you start taking PRIFTIN and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment. | |||||
|
What is the most important information I should know about PRIFTIN? |
|||||
PRIFTIN may cause serious side effects, including:
|
|||||
|
|
||||
|
|||||
|
|
||||
| Signs and symptoms of a flu-like reaction may include: | |||||
|
|
|
|||
|
|||||
|
|
|
|||
| What is PRIFTIN? | |||||
PRIFTIN is a prescription medicine used with other anti-tuberculosis (TB) medicines to:
|
|||||
PRIFTIN should not be used:
|
|||||
| PRIFTIN is safe and effective in children older than 2 years of age who have inactive (latent TB), but it is not known if PRIFTIN is safe and effective for use in the treatment of active TB in children under 12 years of age. | |||||
Who should not take PRIFTIN?
|
|||||
| What should I tell my doctor before taking PRIFTIN? | |||||
Before taking PRIFTIN, tell your doctor about all of your medical conditions, including if you:
|
|||||
| Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |||||
| Using PRIFTIN with other medicines may affect each other causing serious side effects. PRIFTIN may affect the way other medicines work, and other medicines may affect how PRIFTIN works. Especially tell your doctor if you take medicines to treat HIV infection or oral contraceptives. | |||||
| Ask your doctor or pharmacist for a list of these medicines if you are not sure. | |||||
| Know the medicines you take. Keep a list of them to show your doctor or pharmacist when you get a new medicine. | |||||
How should I take PRIFTIN?
|
|||||
| What are possible side effects of PRIFTIN? | |||||
PRIFTIN may cause serious side effects, including:
|
|||||
| The most common side effects of PRIFTIN include: allergic reactions and flu-like symptoms; abnormalities such as low red blood cells, low white blood cells, coughing up blood, cough, excessive number of platelets in the blood, increased sweating, high liver function tests, back pain, rash, decreased appetite, joint pain, increased blood urea, and headache. | |||||
| Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of PRIFTIN. For more information, ask your doctor or pharmacist. | |||||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||||
How should I store PRIFTIN?
|
|||||
| General information about the safe and effective use of PRIFTIN. | |||||
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use PRIFTIN for a condition for which it was not prescribed. Do not give PRIFTIN to other people, even if they have the same symptoms you have. It may harm them. | |||||
| This Medication Guide summarizes the most important information about PRIFTIN. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about PRIFTIN that is written for healthcare professionals. | |||||
| What are the ingredients in PRIFTIN? | |||||
| Active ingredient: rifapentine | |||||
| Inactive ingredients: calcium stearate, disodium EDTA, FD&C Blue No. 2 aluminum lake, hydroxypropyl cellulose, hypromellose USP, microcrystalline cellulose, polyethylene glycol, pregelatinized starch, propylene glycol, sodium ascorbate, sodium lauryl sulfate, sodium starch glycolate, synthetic red iron oxide, and titanium dioxide | |||||
| Manufactured by: sanofi-aventis U.S. LLC, Morristown, NJ 07960 For more information, go to www.sanofi.us or call 1-800-633-1610, and select option 1. |
Section 43683-2 (43683-2)
| Warnings and Precautions, Paradoxical Drug Reactions (5.5) | 01/2026 |
Section 44425-7 (44425-7)
Storage
Store at 25°C (77°F); excursions permitted 15–30°C (59–86°F) (see USP Controlled Room Temperature). Protect from excessive heat and humidity.
10 Overdosage (10 OVERDOSAGE)
While there is no experience with the treatment of acute overdose with PRIFTIN, clinical experience with rifamycins suggests that gastric lavage to evacuate gastric contents (within a few hours of overdose), followed by instillation of an activated charcoal slurry into the stomach, may help adsorb any remaining drug from the gastrointestinal tract.
Rifapentine and 25-desacetyl rifapentine are 97.7% and 93.2% plasma protein bound, respectively. Rifapentine and related compounds excreted in urine account for only 17% of the administered dose, therefore, neither hemodialysis nor forced diuresis is expected to enhance the systemic elimination of unchanged rifapentine from the body of a patient with PRIFTIN overdose.
5.9 Porphyria
Porphyria has been reported in patients receiving rifampin, attributed to induction of delta amino levulinic acid synthetase. Because PRIFTIN may have similar enzyme induction properties, avoid the use of PRIFTIN in patients with porphyria.
11 Description (11 DESCRIPTION)
PRIFTIN (rifapentine) for oral administration contains 150 mg of the active ingredient rifapentine per tablet.
The 150 mg tablets also contain, as inactive ingredients: calcium stearate, disodium EDTA, FD&C Blue No. 2 aluminum lake, hydroxypropyl cellulose, hypromellose USP, microcrystalline cellulose, polyethylene glycol, pregelatinized starch, propylene glycol, sodium ascorbate, sodium lauryl sulfate, sodium starch glycolate, synthetic red iron oxide, and titanium dioxide.
Rifapentine is a rifamycin derivative antimicrobial and has a similar profile of microbiological activity to rifampicin. The molecular weight is 877.04.
The molecular formula is C47H64N4O12.
The chemical name for rifapentine is rifamycin, 3-[[(4-cyclopentyl-1-piperazinyl)imino]methyl]- or 3-[N-(4-cyclopentyl - 1-piperazinyl)formimidoyl] rifamycin or 5,6,9,17,19,21-hexahydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-8-[N-(4-cyclopentyl-l-piperazinyl)-formimidoyl]-2,7-(epoxypentadeca[1,11,13]trienimino)naphtho[2,1-b]furan-1,11(2H)-dione 21-acetate. It has the following structure:
8.4 Pediatric Use
The safety and effectiveness of PRIFTIN in the treatment of active pulmonary tuberculosis have not been established in pediatric patients under the age of 12.
The safety and effectiveness of PRIFTIN in combination with isoniazid once-weekly regimen has been evaluated in pediatric patients (2 to 17 years of age) for the treatment of latent tuberculosis infection. In clinical studies, the safety profile in pediatric patients was similar to that observed in adult patients [see Adverse Reactions (6.1) and Clinical Studies (14.2)].
In a pharmacokinetic study conducted in 2 to 11-year-old pediatric patients with latent tuberculosis infection, PRIFTIN was administered once weekly based on weight (15 mg/kg to 30 mg/kg, up to a maximum of 900 mg). Exposures (AUC) in pediatric patients 2 to 11 years old with latent tuberculosis infection were higher (average 31%) than those observed in adults receiving PRIFTIN 900 mg once weekly [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
8.5 Geriatric Use
Clinical studies with PRIFTIN did not include sufficient numbers of subjects aged 65 years and over to determine whether they respond differently from younger subjects. In a pharmacokinetic study with PRIFTIN, no substantial differences in the pharmacokinetics of rifapentine and 25-desacetyl metabolite were observed in the elderly compared to younger adults [see Clinical Pharmacology (12.3)].
2.3 Administration
Take PRIFTIN with meals. Administration of PRIFTIN with a meal increases oral bioavailability and may reduce the incidence of gastrointestinal upset, nausea, and/or vomiting [see Clinical Pharmacology (12.3)].
For patients who cannot swallow tablets, the tablets may be crushed and added to a small amount of semi-solid food, all of which should be consumed immediately [see Clinical Pharmacology (12.3)].
5.1 Hepatotoxicity
Elevations of liver transaminases may occur in patients receiving PRIFTIN [see Adverse Reactions (6.1)]. Patients on PRIFTIN should be monitored for symptoms of liver injury.
Patients with abnormal liver tests and/or liver disease or patients initiating treatment for active pulmonary tuberculosis should only be given PRIFTIN in cases of necessity and under strict medical supervision. In such patients, obtain serum transaminase levels prior to therapy and every 2 to 4 weeks while on therapy. Discontinue PRIFTIN if evidence of liver injury occurs.
4 Contraindications (4 CONTRAINDICATIONS)
Known hypersensitivity to any rifamycin. (4.1)
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious and otherwise important adverse drug reactions are discussed in greater detail in other sections of labeling:
- Hepatotoxicity [see Warnings and Precautions (5.1)]
- Hypersensitivity [see Contraindications (4.1) and Warnings and Precautions (5.2)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.3)]
- Paradoxical Drug Reactions [see Warnings and Precautions (5.5)]
- Discoloration of Body Fluids [see Warnings and Precautions (5.7)]
- Clostridioides Difficile–Associated Diarrhea [see Warnings and Precautions (5.8)]
- Porphyria [see Warnings and Precautions (5.9)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Protease Inhibitors and Reverse Transcriptase Inhibitors. (5.2, 7.1)
- Hormonal Contraceptives: Use an effective non-hormonal method of contraception or add a barrier method of contraception during treatment with PRIFTIN. (7.3)
- May increase metabolism and decrease the activity of drugs metabolized by cytochrome P450 3A4 and 2C8/9. Dosage adjustments may be necessary if given concomitantly. (7.4)
4.1 Hypersensitivity
PRIFTIN is contraindicated in patients with a history of hypersensitivity to rifamycins.
12.3 Pharmacokinetics
When oral doses of PRIFTIN were administered once daily or once every 72 hours to healthy volunteers for 10 days, single dose AUC (0–∞) of rifapentine was similar to its steady-state AUCss (0–24h) or AUCss (0–72h) values, suggesting no significant auto-induction effect on steady-state pharmacokinetics of rifapentine. Steady-state conditions were achieved by day 10 following daily administration of PRIFTIN 600 mg. No plasma accumulation of rifapentine and 25-desacetyl rifapentine (active metabolite) is expected after once weekly administration of PRIFTIN.
The pharmacokinetic parameters of rifapentine and 25-desacetyl rifapentine on day 10 following oral administration of 600 mg PRIFTIN every 72 hours to healthy volunteers are described in Table 5.
| Parameter | Rifapentine | 25-desacetyl Rifapentine |
|---|---|---|
| Mean ± SD (n=12) | ||
| Cmax (µg/mL) | 15.05 ± 4.62 | 6.26 ± 2.06 |
| AUC (0–72h) (µg∙h/mL) | 319.54 ± 91.52 | 215.88 ± 85.96 |
| T1/2 (h) | 13.19 ± 1.38 | 13.35 ± 2.67 |
| Tmax (h) | 4.83 ± 1.80 | 11.25 ± 2.73 |
| Cl/F (L/h) | 2.03 ± 0.60 | -- |
The pharmacokinetic parameters of rifapentine and 25-desacetyl rifapentine following single-dose oral administration of 900 mg PRIFTIN in combination with 900 mg isoniazid in fed conditions are described in Table 6.
| Parameter | Rifapentine | 25-desacetyl Rifapentine |
|---|---|---|
| Cmax (µg/mL) | 25.8 ± 5.83 | 13.3 ± 4.83 |
| AUC (µg∙h/mL) | 817 ± 128 | 601 ± 187 |
| T1/2(h) | 16.6 ± 5.02 | 17.5 ± 7.42 |
| Tmax (h) Median (Min–Max).
|
8 (3–10) | 24 (10–36) |
| Cl/F (L/h) | 1.13 ± 0.174 | NA Not Applicable.
|
5.6 Drug Interactions
Rifapentine is an inducer of CYP450 enzymes. Concomitant use of rifapentine with other drugs metabolized by these enzymes, such as protease inhibitors, certain reverse transcriptase inhibitors, and hormonal contraception may cause a significant decrease in plasma concentrations and loss of therapeutic effect [see Drug Interactions (7.1, 7.2, 7.3, 7.4) and Clinical Pharmacology (12.3)].
7.5 Other Interactions
The conversion of PRIFTIN to 25-desacetyl rifapentine is mediated by an esterase enzyme. There is minimal potential for PRIFTIN metabolism to be inhibited or induced by another drug, based upon the characteristics of the esterase enzymes.
Since PRIFTIN is highly bound to albumin, drug displacement interactions may also occur [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
- PRIFTIN is a rifamycin antimycobacterial drug indicated in patients 12 years of age and older for the treatment of active pulmonary tuberculosis (TB) caused by Mycobacterium tuberculosis in combination with one or more antituberculosis (anti-TB) drugs to which the isolate is susceptible. (1.1)
- PRIFTIN is indicated for the treatment of latent tuberculosis infection (LTBI) caused by M. tuberculosis in combination with isoniazid in patients 2 years of age and older at high risk of progression to TB disease. (1.2)
- See Full Prescribing Information for Limitations of Use. (1.1, 1.2)
12.1 Mechanism of Action
Rifapentine, a cyclopentyl rifamycin, is an antimycobacterial agent [see Microbiology (12.4)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hepatotoxicity: Monitor for symptoms of liver injury and discontinue PRIFTIN if signs or symptoms or liver injury occur. (5.1)
- Hypersensitivity: Discontinue PRIFTIN if signs or symptoms of hypersensitivity reaction occur. (5.2)
- Severe cutaneous adverse reactions: Discontinue PRIFTIN at the first appearance of skin rash, mucosal lesions, or any other sign of hypersensitivity. (5.3)
- Relapse in the treatment of active pulmonary tuberculosis: Do not use as a once-weekly continuation phase regimen with isoniazid in HIV-infected patients. Monitor for signs or symptoms of relapse in patients with cavitary lesions or bilateral disease. (5.4, 14.1)
- Paradoxical Drug Reactions: If worsening of symptoms or signs occur during antimycobacterial treatment, consider paradoxical drug reaction in the differential diagnosis, and monitor or treat accordingly. (5.5)
- Drug Interactions: May interact with drugs metabolized by CYP450. (5.6, 7.1, 7.4)
- Discoloration of body fluids: May permanently stain contact lenses or dentures red-orange. (5.7)
- Clostridioides difficile–associated diarrhea: Evaluate if diarrhea occurs. (5.8)
- Porphyria: Avoid use in patients with porphyria. (5.9)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
Active pulmonary tuberculosis: PRIFTIN should be used in regimens consisting of an initial 2 month phase followed by a 4 month continuation phase. (2.1)
Initial phase (2 Months): 600 mg twice weekly for two months as directly observed therapy (DOT), with no less than 72 hours between doses, in combination with other antituberculosis drugs. (2.1)
Continuation phase (4 Months): 600 mg once weekly for 4 months as directly observed therapy with isoniazid or another appropriate antituberculosis agent. (2.1) -
Latent tuberculosis infection: PRIFTIN should be administered in combination with isoniazid once weekly for 12 weeks as directly observed therapy. (2.2)
Adults and pediatric patients ≥12 years: PRIFTIN (based on weight, see table below) and isoniazid 15 mg/kg (900 mg maximum). (2.2)
Pediatric patients 2 to 11 years: PRIFTIN (based on weight, see table below) and isoniazid 25 mg/kg (900 mg maximum). (2.2)
Weight range PRIFTIN dose Number of PRIFTIN tablets 10–14 kg 300 mg 2 14.1–25 kg 450 mg 3 25.1–32 kg 600 mg 4 32.1–50 kg 750 mg 5 >50 kg 900 mg 6
For Latent Tuberculosis Infection, the maximum recommended dose of PRIFTIN is 900 mg once weekly for 12 weeks. (2.2) - Take with food. Tablets may be crushed and added to semi-solid food. (2.3)
7.3 Hormonal Contraceptives
PRIFTIN may reduce the effectiveness of hormonal contraceptives. Patients using hormonal contraception should be advised to use an alternative non-hormonal contraceptive method or add a barrier method of contraception during treatment with PRIFTIN [see Use in Specific Populations (8.3) and Clinical Pharmacology (12.3)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
PRIFTIN is supplied as 150 mg round normal convex dark-pink film-coated tablets debossed "F" on one side of tablet.
5.8 Clostridioides Difficile
Clostridioides difficile–associated diarrhea (CDAD) has been reported with the use of nearly all systemic antibacterial agents, including PRIFTIN, with severity ranging from mild diarrhea to fatal colitis. Treatment with antibacterial agents can alter the normal flora of the colon and may permit overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary because CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, discontinue antibacterial use not directed against C. difficile if possible. Institute appropriate measures such as fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation as clinically indicated.
6.2 Postmarketing Experience
The following adverse reactions have been identified from postmarketing surveillance of rifapentine. Because these reactions are reported from a population of unknown size, it is not always possible to estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy: Based on animal data, may cause fetal harm. (8.1)
- Lactation: Monitor infants exposed to PRIFTIN through breast milk for irritability, prolonged unexplained crying, yellowing of the eyes, changes in color of the urine or stool. (8.2)
- Pediatrics: Safety and effectiveness in treating active pulmonary tuberculosis in pediatric patients under the age of 12 years have not been established. (8.4)
5.5 Paradoxical Drug Reactions
Paradoxical drug reactions are characterized by the recurrence or appearance of new symptoms or physical and radiological signs in a patient who had previously shown improvement with appropriate antimycobacterial treatment, in the absence of disease relapse, poor treatment compliance, drug resistance, side effects of treatment, or secondary infection/diagnosis. Paradoxical drug reactions have been reported with antimycobacterial therapy, including PRIFTIN, within the first few weeks or months of initiation of tuberculosis therapy [see Adverse Reactions (6.2)].
Paradoxical drug reactions are often transient and should not be misinterpreted as failure to respond to treatment. If worsening of symptoms or signs occurs during antimycobacterial treatment, consider paradoxical drug reaction in the differential diagnosis, and monitor or treat accordingly.
Advise patients to seek medical advice immediately if their symptoms of tuberculosis worsen or reappear.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.7 Discoloration of Body Fluids
PRIFTIN may produce a red-orange discoloration of body tissues and/or fluids (e.g., skin, teeth, tongue, urine, feces, saliva, sputum, tears, sweat, and cerebrospinal fluid). Contact lenses or dentures may become permanently stained.
1.1 Active Pulmonary Tuberculosis
PRIFTIN® (rifapentine) is indicated in adults and pediatric patients 12 years and older for the treatment of active pulmonary tuberculosis (TB) caused by Mycobacterium tuberculosis. PRIFTIN must always be used in combination with one or more antituberculosis (anti-TB) drugs to which the isolate is susceptible [see Dosage and Administration (2.1) and Clinical Studies (14.1)].
1.2 Latent Tuberculosis Infection
PRIFTIN is indicated in adults and pediatric patients 2 years and older for the treatment of latent tuberculosis infection caused by Mycobacterium tuberculosis in patients at high risk of progression to tuberculosis disease (including those in close contact with active tuberculosis patients, recent conversion to a positive tuberculin skin test, HIV-infected patients, or those with pulmonary fibrosis on radiograph) [see Clinical Studies (14.2)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patient to read FDA-approved patient labeling (Medication Guide).
7.4 Cytochrome P450 3a4 and 2c8/9 (7.4 Cytochrome P450 3A4 and 2C8/9)
Rifapentine is an inducer of cytochromes P450 3A4 and P450 2C8/9. Therefore, PRIFTIN may increase the metabolism of other coadministered drugs that are metabolized by these enzymes. Induction of enzyme activities by PRIFTIN occurred within 4 days after the first dose. Enzyme activities returned to baseline levels 14 days after discontinuing PRIFTIN.
Rifampin has been reported to accelerate the metabolism and may reduce the activity of the following drugs; hence, PRIFTIN may also increase the metabolism and decrease the activity of these drugs. Dosage adjustments of the drugs in Table 4 or of other drugs metabolized by cytochrome P450 3A4 or P450 2C8/9 may be necessary if they are given concurrently with PRIFTIN.
| Drug Class | Examples of Drugs Within Class |
|---|---|
| Antiarrhythmics | Disopyramide, mexiletine, quinidine, tocainide |
| Antibiotics | Chloramphenicol, clarithromycin, dapsone, doxycycline; Fluoroquinolones (such as ciprofloxacin) |
| Oral Anticoagulants | Warfarin |
| Anticonvulsants | Phenytoin |
| Antimalarials | Quinine |
| Azole Antifungals | Fluconazole, itraconazole, ketoconazole |
| Antipsychotics | Haloperidol |
| Barbiturates | Phenobarbital |
| Benzodiazepines | Diazepam |
| Beta-Blockers | Propranolol |
| Calcium Channel Blockers | Diltiazem, nifedipine, verapamil |
| Cardiac Glycoside Preparations | Digoxin |
| Corticosteroids | Prednisone |
| Fibrates | Clofibrate |
| Oral Hypoglycemics | Sulfonylureas (e.g., glyburide, glipizide) |
| Hormonal Contraceptives/Progestins | Ethinyl estradiol, levonorgestrel |
| Immunosuppressants | Cyclosporine, tacrolimus |
| Methylxanthines | Theophylline |
| Narcotic analgesics | Methadone |
| Phosphodiesterase-5 (PDE-5) Inhibitors | Sildenafil |
| Thyroid preparations | Levothyroxine |
| Tricyclic antidepressants | Amitriptyline, nortriptyline |
14.1 Active Pulmonary Tuberculosis
PRIFTIN was studied in two randomized, open-label controlled clinical trials in the treatment of active pulmonary tuberculosis.
The first trial was an open-label, prospective, parallel group, active-controlled trial in HIV-negative patients with active pulmonary tuberculosis. The population mostly comprised Black (approximately 60%) or multiracial (approximately 31%) patients. Treatment groups were comparable for age and sex and consisted primarily of male subjects with a mean age of 37 ± 11 years. In the initial 2-month phase of treatment, 361 patients received PRIFTIN 600 mg twice a week in combination with daily isoniazid, pyrazinamide, and ethambutol and 361 subjects received rifampin 600 mg in combination with isoniazid, pyrazinamide and ethambutol all administered daily. The doses of the companion drugs were the same in both treatment groups during the initial phase: isoniazid 300 mg, pyrazinamide 2000 mg, and ethambutol 1200 mg. For patients weighing less than 50 kg, the doses of rifampin (450 mg), pyrazinamide (1500 mg) and ethambutol (800 mg) were reduced. Ethambutol was discontinued when isoniazid and rifampin susceptibility testing results were confirmed. During the 4-month continuation phase, 317 patients in the PRIFTIN group continued to receive PRIFTIN 600 mg dosed once weekly with isoniazid 300 mg and 304 patients in the rifampin group received twice weekly rifampin and isoniazid 900 mg. For patients weighing less than 50 kg, the doses of rifampin (450 mg) and isoniazid (600 mg) were reduced. Both treatment groups received pyridoxine (Vitamin B6) over the 6-month treatment period. Treatment was directly observed. 65/361 (18%) of patients in the PRIFTIN group and 34/361 (9%) in the rifampin group received overdoses of one or more of the administered study medications during the initial or continuation phase of treatment. Seven of these patients had adverse reactions reported with the overdose (5 in the PRIFTIN group and 2 in the rifampin group).
Table 8 below contains assessments of sputum conversion at end of treatment (6 months) and relapse rates at the end of follow-up (24 months).
| PRIFTIN Combination Treatment % and (n/N All data for patients with confirmed susceptible M. tuberculosis (PRIFTIN combination treatment, N=286; rifampin combination treatment, N=283). ) |
Rifampin Combination Treatment % and (n/N) | |
|---|---|---|
| Status at End of 6 months of Treatment | ||
| Converted | 87% (248/286) | 80% (226/283) |
| Not Converted | 1% (4/286) | 3% (8/283) |
| Lost to Follow-up | 12% (34/286) | 17% (49/283) |
|
Status Through 24 Month Follow-up
Twenty-two (22) deaths occurred during the study; 11 in each treatment group.
|
||
| Relapsed | 12% (29/248) | 7% (15/226) |
| Sputum Negative | 57% (142/248) | 64% (145/226) |
| Lost to Follow-up | 31% (77/248) | 29% (66/226) |
Risk of relapse was greater in the group treated with the PRIFTIN combination. Higher relapse rates were associated with a lower rate of compliance as well as a failure to convert sputum cultures at the end of the initial 2-month treatment phase. Relapse rates were also higher for males in both regimens. Relapse in the PRIFTIN group was not associated with development of monoresistance to rifampin.
The second trial was randomized, open-label performed in 1075 HIV-negative and HIV-positive patients with active pulmonary tuberculosis. Patients with culture-positive, drug-susceptible pulmonary tuberculosis who had completed the initial 2-month phase of treatment with 4 drugs (rifampin, isoniazid, pyrazinamide, and either ethambutol or streptomycin) under direct observation were randomly assigned to receive either PRIFTIN 600 mg and isoniazid 15 mg/kg (max 900 mg) once weekly or rifampin 10 mg/kg (max 600 mg) and isoniazid 15 mg/kg (max 900 mg) twice weekly for the 4 month continuation phase. Study drugs were given under direct observation therapy in both groups.
In the PRIFTIN group, 502 HIV-negative and 36 HIV-positive patients were randomized and in the rifampin group 502 HIV-negative and 35 HIV-positive patients were randomized to treatment. Enrollment of HIV-infected patients was stopped when 4 of 36 patients in the PRIFTIN combination group relapsed with isolates that were rifampin resistant.
Table 9 below contains assessments of sputum conversion at the end of treatment (6 months total: 2 months of initial and 4 months of randomized continuation treatment) and relapse rates at the end of follow-up (24 months) in all HIV-negative patients randomized to treatment. Positive culture was based on either one sputum sample with >10 colonies on solid media OR at least 2 positive sputum samples on liquid or solid media. However, only one sputum sample was collected at each visit in a majority of patients.
| PRIFTIN Combination Treatment % (n/N) | Rifampin Combination Treatment % (n/N) | |
|---|---|---|
| Status at End of 4 Months Continuation Phase | ||
| Treatment Response Treatment response was defined as subjects who had two negative sputum cultures after 16 doses of rifampin and isoniazid or after 8 doses of PRIFTIN and isoniazid, and remained sputum negative through the end of continuation phase therapy.
|
93.8% (471/502) | 91% (457/502) |
| Not Converted | 1% (5/502) | 1.2% (6/502) |
| Did Not Complete Treatment Due to drug toxic effects, non-adherence, withdrawal of consent, receipt of non-study regimen, other.
|
4.2% (21/502) | 7% (35/502) |
| Deaths | 1% (5/502) | 0.8% (4/502) |
| Status Through 24 Month Follow-up: | ||
| Relapsed | 8.7% (41/471) | 4.8% (22/457) |
| Sputum Negative | 79.4% (374/471) | 80.1% (366/457) |
| Lost to Follow-up | 7.9% (37/471) | 9.8% (45/457) |
| Deaths | 4% (19/471) | 5.3% (24/457) |
In HIV-negative patients, higher relapse rates were seen in patients with a positive sputum culture at 2 months (i.e., at the time of study randomization), cavitation on chest x-ray, and bilateral pulmonary involvement.
Sixty-one HIV-positive patients were assessed for relapse. The rates of relapse were 16.7% (5/30) in the PRIFTIN group and 9.7% (3/31) in the rifampin group. In HIV-positive patients, 4 of the 5 relapses in the PRIFTIN combination group involved M. tuberculosis strains with rifampin monoresistance. No relapse strain in the twice weekly rifampin/isoniazid group acquired drug resistance.
The death rate among all study participants did not differ between the two treatment groups.
14.2 Latent Tuberculosis Infection
A multicenter, prospective, open-label, randomized, active-controlled trial compared the effectiveness of 12 weekly doses of PRIFTIN in combination with isoniazid (3RPT/INH arm) administered by directly observed therapy to 9 months of self-administered daily isoniazid (9INH arm). The trial enrolled patients two years of age or older with positive tuberculin skin test and at high risk for progression to tuberculosis disease. Enrolled patients included those having close contact with a patient with active tuberculosis disease, recent (within two years) conversion to a positive tuberculin skin test, HIV-infection, or fibrosis on chest radiograph. PRIFTIN was dosed by weight, for a maximum of 900 mg weekly. Isoniazid mg/kg dose was determined by age, for a maximum of 900 mg weekly in the 3RPT/INH arm and 300 mg daily in the 9INH arm [see Dosage and Administration (2.2)].
The outcome measure was the development of active tuberculosis disease, defined as culture-confirmed tuberculosis in adults and culture-confirmed or clinical tuberculosis in pediatric patients less than 18 years of age, at 33 months after trial enrollment. Patients who were found after enrollment to be ineligible because they had active tuberculosis disease, were contacts of a source case with culture-negative or drug-resistant tuberculosis disease cases or no information regarding susceptibility of M. tuberculosis, and young pediatric patients lacking a positive TST on initial and repeat testing were excluded from the analysis.
Active tuberculosis disease developed in 5 of 3074 randomized patients in the 3RPT/INH group (0.16%) versus 10 of 3074 patients in 9INH group (0.32%), for a difference in cumulative rates of 0.17%, 95% CI (-0.43, 0.09) (Table 10).
| Outcome | 3RPT/INH (n=3074) | 9INH (n=3074) | Difference Rate in the 3RPT/INH group minus the rate in the 9INH group. , 95% CI |
|---|---|---|---|
| Tuberculosis n (%) | 5 (0.16) | 10 (0.32) | -0.16 (-0.42, 0.01) |
| Cumulative TB Rate (%) | 0.17 | 0.35 | -0.17 (-0.43, 0.09) |
| Deaths | 22 (0.72) | 35 (1.14) | -0.42 (-0.91, 0.06) |
| Lost to Follow-Up | 320 (10.41) | 357 (11.61) | -1.20 (-2.77, -0.36) |
The proportion of patients completing treatment was 81.2% in the 3RPT/INH group and 68.3% in the 9INH group for a difference (3RPT/INH-9INH) of 12.8% 95% CI (10.7, 15.0).
In the 9INH treatment group, two of the thirteen culture-confirmed cases were found to be isoniazid-monoresistant. In the 3RPT/INH treatment group, one of the seven cases was rifampin-resistant, isoniazid-susceptible M. bovis infection.
5.3 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions (SCARs) such as Stevens-Johnson syndrome (SJS) and drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome have been reported in association with the use of rifapentine (PRIFTIN) treatment regimens in patients with active and latent tuberculosis. Discontinue PRIFTIN at the first appearance of skin rash, mucosal lesions, or any other sign of hypersensitivity [see Patient Counseling Information (17)].
7.6 Interactions With Laboratory Tests (7.6 Interactions with Laboratory Tests)
Therapeutic concentrations of rifampin have been shown to inhibit standard microbiological assays for serum folate and Vitamin B12. Similar drug-laboratory interactions should be considered for PRIFTIN; thus, alternative assay methods should be considered.
5.2 Hypersensitivity and Related Reactions
Hypersensitivity reactions may occur in patients receiving PRIFTIN. Signs and symptoms of these reactions may include hypotension, urticaria, angioedema, acute bronchospasm, conjunctivitis, thrombocytopenia, neutropenia or flu-like syndrome (weakness, fatigue, muscle pain, nausea, vomiting, headache, fever, chills, aches, rash, itching, sweats, dizziness, shortness of breath, chest pain, cough, syncope, palpitations). There have been reports of anaphylaxis [see Patient Counseling Information (17)].
Monitor patients receiving PRIFTIN therapy for signs and/or symptoms of hypersensitivity reactions. If these symptoms occur, administer supportive measures and discontinue PRIFTIN.
2.1 Dosage in Active Pulmonary Tuberculosis
PRIFTIN is only recommended for the treatment of active pulmonary tuberculosis caused by drug-susceptible organisms as part of regimens consisting of a 2-month initial phase followed by a 4-month continuation phase.
PRIFTIN should not be used in the treatment of active pulmonary tuberculosis caused by rifampin-resistant strains.
2.2 Dosage in Latent Tuberculosis Infection
PRIFTIN should be administered once weekly in combination with isoniazid for 12 weeks as directly observed therapy.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Hepatocellular carcinomas were increased in male NMRI mice (Harlan Winklemann) which were treated orally with rifapentine for two years at or above doses of 5 mg/kg/day (0.04 times the recommended human dose based on body surface area conversions). In a two year rat study, there was an increase in nasal cavity adenomas in Wistar rats treated orally with rifapentine at 40 mg/kg/day (0.6 times human dose based on body surface area conversions).
Rifapentine was negative in the following genotoxicity tests: in vitro gene mutation assay in bacteria (Ames test); in vitro point mutation test in Aspergillus nidulans; in vitro gene conversion assay in Saccharomyces cerevisiae; host-mediated (mouse) gene conversion assay with Saccharomyces cerevisiae; in vitro Chinese hamster ovary cell/hypoxanthine-guanine phosphoribosyltransferase (CHO/HGPRT) forward mutation assay; in vitro chromosomal aberration assay utilizing rat lymphocytes; and in vivo mouse bone marrow micronucleus assay.
The 25-desacetyl metabolite of rifapentine was positive in the in vitro mammalian chromosome aberration test in V79 Chinese hamster cells, but was negative in the in vitro gene mutation assay in bacteria (Ames test), the in vitro Chinese hamster ovary cell/hypoxanthine-guanine phosphoribosyltransferase (CHO/HGPRT) forward mutation assay, and the in vivo mouse bone marrow micronucleus assay. Fertility and reproductive performance were not affected by oral administration of rifapentine to male and female rats at doses of up to 20 mg/kg/day (one-third of the human dose based on body surface area conversions).
Principal Display Panel 150 Mg Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 150 mg Tablet Blister Pack Carton)
NDC 0088-2102-24
PRIFTIN®
RIFAPENTINE
150 mg per tablet
Dispense with Medication Guide
24 Tablets
Three 8-Tablet
Blister Strips
sanofi
7.1 Protease Inhibitors and Reverse Transcriptase Inhibitors
Rifapentine is an inducer of CYP450 enzymes. Concomitant use of PRIFTIN with other drugs metabolized by these enzymes, such as protease inhibitors and certain reverse transcriptase inhibitors, may cause a significant decrease in plasma concentrations and loss of therapeutic effect of the protease inhibitor or reverse transcriptase inhibitor [see Warnings and Precautions (5.6) and Clinical Pharmacology (12.3)].
5.4 Relapse in the Treatment of Active Pulmonary Tuberculosis
PRIFTIN has not been evaluated as part of the initial phase treatment regimen in HIV-infected patients with active pulmonary TB.
Do not use PRIFTIN as a once-weekly continuation phase regimen in HIV-infected patients with active pulmonary tuberculosis because of a higher rate of failure and/or relapse with rifampin-resistant organisms [see Clinical Studies (14.1)].
Higher relapse rates may occur in patients with cavitary pulmonary lesions and/or positive sputum cultures after the initial phase of active tuberculosis treatment and in patients with evidence of bilateral pulmonary disease. Monitor for signs and symptoms of TB relapse in these patients [see Clinical Studies (14.1)].
Poor adherence to therapy is associated with high relapse rate. Emphasize the importance of compliance with therapy [see Patient Counseling Information (17)].
7.2 Fixed Dose Combination of Efavirenz, Emtricitabine, and Tenofovir (7.2 Fixed-Dose Combination of Efavirenz, Emtricitabine, and Tenofovir)
Once-weekly coadministration of 900 mg PRIFTIN with the antiretroviral fixed-dose combination of efavirenz 600 mg, emtricitabine 200 mg and tenofovir disoproxil fumarate 300 mg in HIV-infected patients did not result in any substantial change in steady state exposures of efavirenz, emtricitabine, and tenofovir. No clinically significant change in CD4 cell counts or viral loads were noted [see Clinical Pharmacology (12.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:53:22.328688 · Updated: 2026-03-14T22:46:15.425828