3a5b64a3-b19e-8b1a-e063-6294a90a6641
34390-5
HUMAN OTC DRUG LABEL
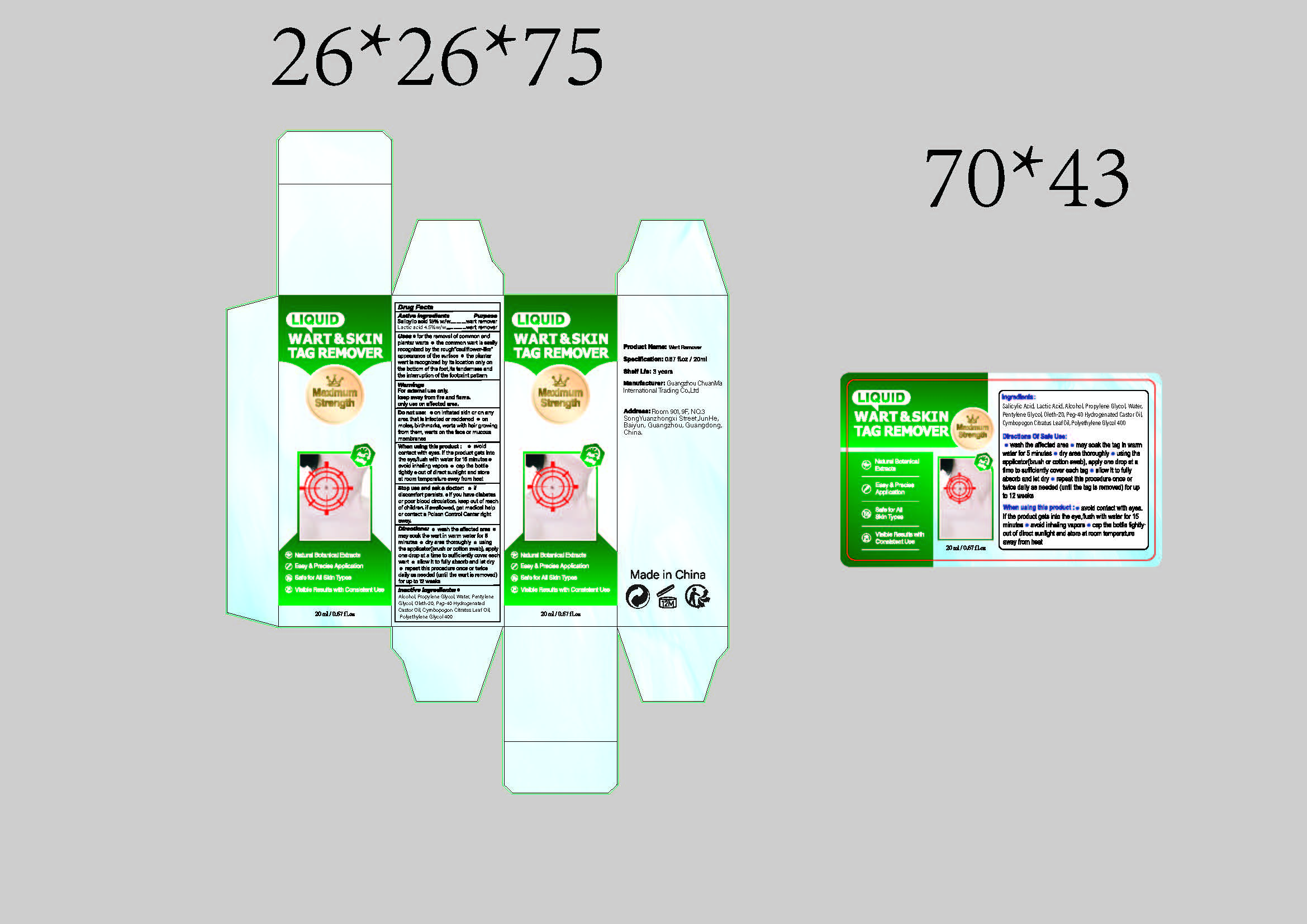
Drug Facts
Composition & Product
Identifiers & Packaging
Description
SALICYLIC ACID 15.00% LACTIC ACID 4.50%
Medication Information
Warnings and Precautions
WarningsFor external use only.keep away from fire and flame.only use on affected area.
Indications and Usage
for the removal of common andplantar warts o the common wart is easilyrecognized by the rough"cauliflower-like"appearance of the surface o the plantarwart is recognized by its location only onthe bottom of the foot,its tenderness andthe interruption of the footprint pattern
Dosage and Administration
wash the affected areamay soak the wart in warm water for 5minutes e dry area thoroughly e usingthe applicator(brush or cotton swab), applyone drop at a time to sufficiently cover eachwart e allow it to fully absorb and let dryrepeat this procedure once or twicedaily as needed (until the wart is removed)for up to 12 weeks
Description
SALICYLIC ACID 15.00% LACTIC ACID 4.50%
Section 50565-1
keep out of reachof children, if swallowed, get medical helpor contact a Poison Control Center rightaway.
Section 50567-7
When using this product:e avoidcontact with eyes. lf the product gets intothe eye,flush with water for 15 minutes avoid inhaling vapors o cap the bottletightly e out of direct sunlight and storeat room temperature away from heat
Section 50570-1
Do not use: on irritated skin or on anyarea that is infected or reddened o onmoles, birthmarks, warts with hair growingfrom them, warts on the face or mucousmembranes
Section 51727-6
Inactive ingredients: ®Alcohol, Propylene Glycol, Water, PentyleneGlycol, Oleth-20, Peg-40 HydrogenatedCastor Oil, Cymbopogon Citratus Leaf Oil,Polyethylene Glycol 400
Section 51945-4
Section 55105-1
Wart remover
Section 55106-9
SALICYLIC ACID 15.00%
LACTIC ACID 4.50%
Structured Label Content
Indications and Usage (34067-9)
for the removal of common andplantar warts o the common wart is easilyrecognized by the rough"cauliflower-like"appearance of the surface o the plantarwart is recognized by its location only onthe bottom of the foot,its tenderness andthe interruption of the footprint pattern
Dosage and Administration (34068-7)
wash the affected areamay soak the wart in warm water for 5minutes e dry area thoroughly e usingthe applicator(brush or cotton swab), applyone drop at a time to sufficiently cover eachwart e allow it to fully absorb and let dryrepeat this procedure once or twicedaily as needed (until the wart is removed)for up to 12 weeks
Warnings and Precautions (34071-1)
WarningsFor external use only.keep away from fire and flame.only use on affected area.
Section 50565-1 (50565-1)
keep out of reachof children, if swallowed, get medical helpor contact a Poison Control Center rightaway.
Section 50567-7 (50567-7)
When using this product:e avoidcontact with eyes. lf the product gets intothe eye,flush with water for 15 minutes avoid inhaling vapors o cap the bottletightly e out of direct sunlight and storeat room temperature away from heat
Section 50570-1 (50570-1)
Do not use: on irritated skin or on anyarea that is infected or reddened o onmoles, birthmarks, warts with hair growingfrom them, warts on the face or mucousmembranes
Section 51727-6 (51727-6)
Inactive ingredients: ®Alcohol, Propylene Glycol, Water, PentyleneGlycol, Oleth-20, Peg-40 HydrogenatedCastor Oil, Cymbopogon Citratus Leaf Oil,Polyethylene Glycol 400
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Wart remover
Section 55106-9 (55106-9)
SALICYLIC ACID 15.00%
LACTIC ACID 4.50%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:34.876911 · Updated: 2026-03-14T23:09:26.831955