3a1cc0e1-fabf-52b9-e063-6394a90ae35e
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
menthol
Medication Information
Warnings and Precautions
If you are pregnant or breast-feeding, only use this medicine on the advice of your healthcare provider.Smoking can seriously harm your child.Try to stop smoking without using any nicotine replacement medicine.This medicine is believed to safer than smoking.However, the risks to your child from this medicine are not full known.
Indications and Usage
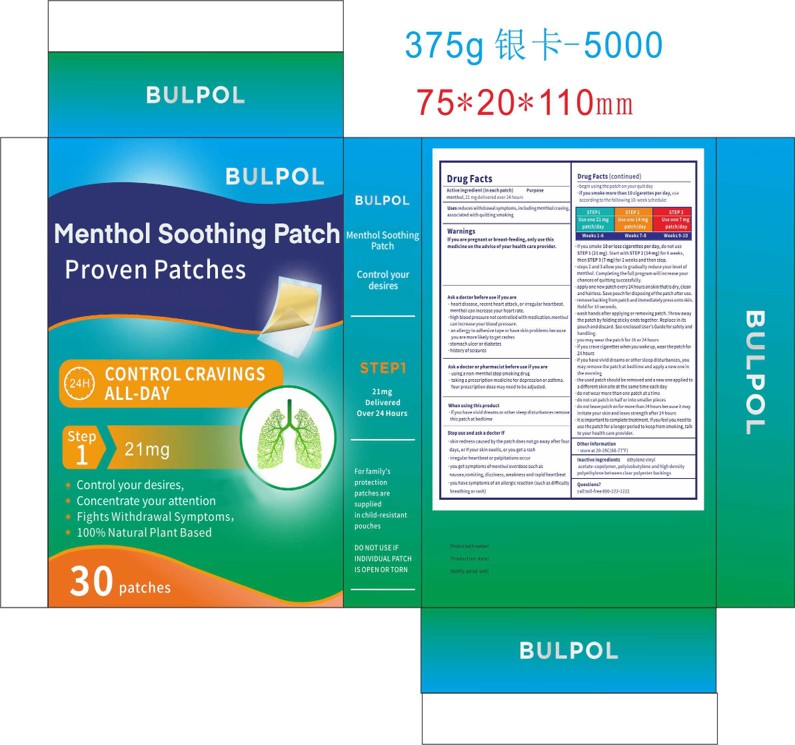
ifyou smoke 10 orless cigarettes per day, do not useSTEP 1 (21 mg). Start with STEP 2 (14 mg) for 6 weeks,then STEP 3 (7 mg) for 2 weeks and then stop.steps 2 and 3 allow you to gradually reduce yourlevel ofnicotine.Completingthe full program willincrease yourchances ofquittingsuccessfully.apply one new patch every 24 hours on skin that is dry, cleanand hairless. Save pouch for disposingofthe patch after use.remove backingfrom patch andimmediately press onto skin.Hold for 10 seconds.
Dosage and Administration
STEP1
Use one 21 mgpatch/day
Weeks 1-6
STEP 2
Use one 14 mg
patch/day
Weeks 7-8
STEP 3
Use one7mg
patch/day
Weeks9-10
Description
menthol
Section 50565-1
Keep out of reach of children and pets.
Used patches have enough nicotine to poison children and pets.If swallowed, get medical help or contact a Poison Control Center right away.Save pouch to use for patch di posal.Dispose of the used patches by folding sticky ends together and putting in pouch.
Section 50567-7
reduces withdrawal symptoms, including nicotine craving, associated with quitting smoking
Section 50569-3
Ask a doctor or pharmacist before use ifyou are"usinga non-nicotine stop smoking drugtaking a prescription medicine for depression or asthma.Your prescription dose may need to be adjusted.
Section 51727-6
nactive ingredientsethylene vinylacetate-copolymer, polyisobutylene and high densitypolyethylene between clearpolyester backings
Section 51945-4
Section 55105-1
Stop smoking aid
Section 55106-9
menthol
Structured Label Content
Indications and Usage (34067-9)
ifyou smoke 10 orless cigarettes per day, do not useSTEP 1 (21 mg). Start with STEP 2 (14 mg) for 6 weeks,then STEP 3 (7 mg) for 2 weeks and then stop.steps 2 and 3 allow you to gradually reduce yourlevel ofnicotine.Completingthe full program willincrease yourchances ofquittingsuccessfully.apply one new patch every 24 hours on skin that is dry, cleanand hairless. Save pouch for disposingofthe patch after use.remove backingfrom patch andimmediately press onto skin.Hold for 10 seconds.
Dosage and Administration (34068-7)
STEP1
Use one 21 mgpatch/day
Weeks 1-6
STEP 2
Use one 14 mg
patch/day
Weeks 7-8
STEP 3
Use one7mg
patch/day
Weeks9-10
Warnings and Precautions (34071-1)
If you are pregnant or breast-feeding, only use this medicine on the advice of your healthcare provider.Smoking can seriously harm your child.Try to stop smoking without using any nicotine replacement medicine.This medicine is believed to safer than smoking.However, the risks to your child from this medicine are not full known.
Section 50565-1 (50565-1)
Keep out of reach of children and pets.
Used patches have enough nicotine to poison children and pets.If swallowed, get medical help or contact a Poison Control Center right away.Save pouch to use for patch di posal.Dispose of the used patches by folding sticky ends together and putting in pouch.
Section 50567-7 (50567-7)
reduces withdrawal symptoms, including nicotine craving, associated with quitting smoking
Section 50569-3 (50569-3)
Ask a doctor or pharmacist before use ifyou are"usinga non-nicotine stop smoking drugtaking a prescription medicine for depression or asthma.Your prescription dose may need to be adjusted.
Section 51727-6 (51727-6)
nactive ingredientsethylene vinylacetate-copolymer, polyisobutylene and high densitypolyethylene between clearpolyester backings
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Stop smoking aid
Section 55106-9 (55106-9)
menthol
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:32.946400 · Updated: 2026-03-14T23:09:23.715399