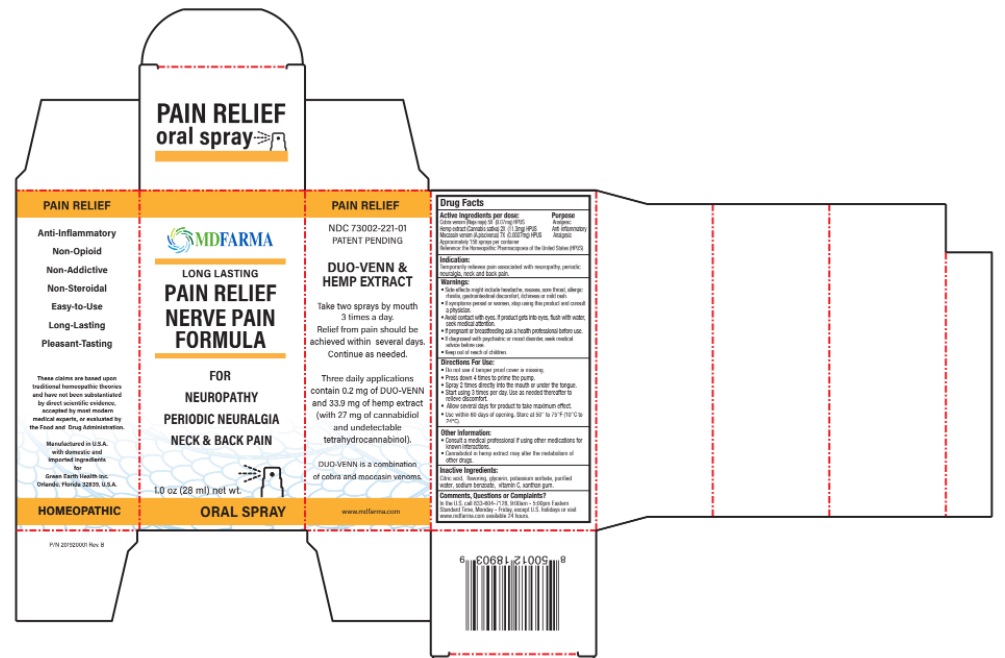
Drug Facts
399e4a4f-63a3-4c62-86a4-f37bf9700bcf
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Cobra venom (Naja naja) 5X (0.07mg) HPUS Analgesic Hemp extract (Cannabis sativa) 2X (11.3mg) HPUS Anti-inflammatory Moccasin venom (A.piscivorus) 7X (0.0007mg) HPUS Analgesic Approximately 158 sprays per container Reference: the Homeopathic Pharmacopoeia of the United States (HPUS)
Purpose
Cobra venom (Naja naja) 5X (0.07mg) HPUS Analgesic Hemp extract (Cannabis sativa) 2X (11.3mg) HPUS Anti-inflammatory Moccasin venom (A.piscivorus) 7X (0.0007mg) HPUS Analgesic Approximately 158 sprays per container Reference: the Homeopathic Pharmacopoeia of the United States (HPUS)
Medication Information
Purpose
Cobra venom (Naja naja) 5X (0.07mg) HPUS Analgesic Hemp extract (Cannabis sativa) 2X (11.3mg) HPUS Anti-inflammatory Moccasin venom (A.piscivorus) 7X (0.0007mg) HPUS Analgesic Approximately 158 sprays per container Reference: the Homeopathic Pharmacopoeia of the United States (HPUS)
Description
Cobra venom (Naja naja) 5X (0.07mg) HPUS Analgesic Hemp extract (Cannabis sativa) 2X (11.3mg) HPUS Anti-inflammatory Moccasin venom (A.piscivorus) 7X (0.0007mg) HPUS Analgesic Approximately 158 sprays per container Reference: the Homeopathic Pharmacopoeia of the United States (HPUS)
Warnings:
• Side effects might include headache, nausea, sore throat, allergic rhinitis, gastrointestinal discomfort, itchiness or mild rash.
• If symptoms persist or worsen, stop using this product and consult a physician.
• Avoid contact with eyes. If product gets into eyes, flush with water, seek medical attention.
• If pregnant or breastfeeding ask a health professional before use.
• If diagnosed with psychiatric or mood disorder, seek medical advice before use.
• Keep out of reach of children.
Indication:
Temporarily relieves pain associated with neuropathy, periodic neuralgia, neck & back pain
Product Label
Other Information:
- Consult medical professional if using other medications for known interactions.
- Cannabidiol in hemp extract may alter the metabolism of other drugs.
Directions for Use:
- Do not use if tamper proof cover is missing.
- Press down 4 times to prime the pump.
- Spray 2 times directly into the mouth or under the tongue.
- Start using 3 times per day. Use as needed thereafter to relieve discomfort.
- Allow several days for product to take maximum effect.
- Use within 60 days from opening. Store at 50° to 75°F (10°C to 24°C)
Inactive Ingredients:
Citric acid, flavoring, glycerin, potassium sorbate, purified water, sodium benzoate, vitamin C, xanthan gum.
Active Ingredients Purpose
Cobra venom (Naja naja) 5X (0.07mg) HPUS Analgesic
Hemp extract (Cannabis sativa) 2X (11.3mg) HPUS Anti-inflammatory
Moccasin venom (A.piscivorus) 7X (0.0007mg) HPUS Analgesic
Approximately 158 sprays per container
Reference: the Homeopathic Pharmacopoeia of the United States (HPUS)
Structured Label Content
Warnings:
• Side effects might include headache, nausea, sore throat, allergic rhinitis, gastrointestinal discomfort, itchiness or mild rash.
• If symptoms persist or worsen, stop using this product and consult a physician.
• Avoid contact with eyes. If product gets into eyes, flush with water, seek medical attention.
• If pregnant or breastfeeding ask a health professional before use.
• If diagnosed with psychiatric or mood disorder, seek medical advice before use.
• Keep out of reach of children.
Indication:
Temporarily relieves pain associated with neuropathy, periodic neuralgia, neck & back pain
Product Label (Product label)
Other Information:
- Consult medical professional if using other medications for known interactions.
- Cannabidiol in hemp extract may alter the metabolism of other drugs.
Directions for Use: (Directions For Use:)
- Do not use if tamper proof cover is missing.
- Press down 4 times to prime the pump.
- Spray 2 times directly into the mouth or under the tongue.
- Start using 3 times per day. Use as needed thereafter to relieve discomfort.
- Allow several days for product to take maximum effect.
- Use within 60 days from opening. Store at 50° to 75°F (10°C to 24°C)
Inactive Ingredients:
Citric acid, flavoring, glycerin, potassium sorbate, purified water, sodium benzoate, vitamin C, xanthan gum.
Active Ingredients Purpose
Cobra venom (Naja naja) 5X (0.07mg) HPUS Analgesic
Hemp extract (Cannabis sativa) 2X (11.3mg) HPUS Anti-inflammatory
Moccasin venom (A.piscivorus) 7X (0.0007mg) HPUS Analgesic
Approximately 158 sprays per container
Reference: the Homeopathic Pharmacopoeia of the United States (HPUS)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:12:05.425625 · Updated: 2026-03-14T23:17:27.453851