Gavilyte™- G
3978822b-6473-4072-9f80-20ceb9bea355
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
GaviLyte-G is a combination of PEG 3350, an osmotic laxative, and electrolytes indicated for cleansing of the colon in preparation for colonoscopy and barium enema X-ray examination in adults. ( 1 )
Dosage and Administration
Preparation and Administration ( 2.1 ): Correct fluid and electrolyte abnormalities before treatment with Gavilyte-G. Reconstitute Gavilyte-G with water prior to ingestion. Do not take oral medications within 1 hour before the start or during administration of Gavilyte-G. ( 2.1 ) Do not take other laxatives while taking Gavilyte-G. Consume only clear liquids; avoid red and purple liquids. Consume water or other clear liquids up until 2 hours before the time of the colonoscopy. Do not consume solid food within 2 hours before starting Gavilyte-G. Adult Dosing Regimen ( 2.2 ): On day prior to colonoscopy, instruct patients to consume a light breakfast at least 2 hours before starting Gavilyte-G. Begin the recommended dosage regimen for Gavilyte-G early in the evening on the day before colonoscopy Drink reconstituted solution at a rate of 8 ounces every 10 minutes, until 4 liters are consumed, or rectal effluent is clear. For complete information on dosing, preparation and administration, see the full prescribing information. ( 2.1 , 2.2 )
Contraindications
Gastrointestinal (GI) obstruction ( 4 , 5.6 ) Bowel perforation ( 4 , 5.6 ) Toxic colitis or toxic megacolon ( 4 ) Gastric retention ( 4 ) Ileus (4) Hypersensitivity to components of Gavilyte-G ( 4 , 5.8 )
Warnings and Precautions
Risk of fluid and electrolyte abnormalities: Encourage adequate hydration, assess concurrent medications, and consider laboratory assessments prior to and after use. ( 5.1 , 5.2 , 7.1 ) Cardiac arrhythmias: Consider pre-dose and post-colonoscopy ECGs in patients at increased risk of serious cardiac arrhythmias. ( 5.2 ) Seizures: Use caution in patients with a history of seizures and patients at increased risk of seizure, including medications that lower the seizure threshold. ( 5.3 , 7.1 ) Patients with renal impairment or taking concomitant medications that affect renal function: Use caution, ensure adequate hydration and consider testing. ( 5.4 , 7.1 , 8.6) Mucosal ulcerations: Consider potential for mucosal ulcerations when interpreting colonoscopy findings in patients with known or suspected inflammatory bowel disease. ( 5.5 , 7.3 ) Patients at risk for aspiration: Observe during administration. ( 5.7 ) Hypersensitivity reactions including anaphylaxis: Inform patients to seek immediate medical care if symptoms occur. ( 5.8 )
Adverse Reactions
Most common adverse reactions are: nausea, abdominal fullness and bloating, abdominal cramps, vomiting and anal irritation ( 6 ) To report SUSPECTED ADVERSE REACTIONS, contact Lupin Pharmaceuticals, Inc. at 1-866-403-7592 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
Some drugs increase risks due to fluid and electrolyte changes ( 7.1 )
How Supplied
Gavilyte-G (polyethylene glycol 3350 and electrolytes for oral solution) is supplied in a 4-liter disposable jug containing 236 g polyethylene glycol 3350, 22.74 g sodium sulfate (anhydrous), 6.74 g sodium bicarbonate, 5.86 g sodium chloride and 2.97 g potassium chloride as a white to off white powder. When reconstituted with water to a volume of 4 liters, the solution contains 59 g/L PEG3350, 5.69 g/L sodium sulfate, 1.69 g/L sodium bicarbonate, 1.47 g/L sodium chloride and 0.743 g/L potassium chloride. Gavilyte-G 4 Liter Disposable Jug NDC 43386-090-19 Storage Store in sealed container at 15° to 30°C (59° to 86°F). Store reconstituted solution of Gavilyte-G at 2° to 8°C (36° to 46°F). Do not freeze [see Dosage and Administration ( 2.1 )].
Storage and Handling
Gavilyte-G (polyethylene glycol 3350 and electrolytes for oral solution) is supplied in a 4-liter disposable jug containing 236 g polyethylene glycol 3350, 22.74 g sodium sulfate (anhydrous), 6.74 g sodium bicarbonate, 5.86 g sodium chloride and 2.97 g potassium chloride as a white to off white powder. When reconstituted with water to a volume of 4 liters, the solution contains 59 g/L PEG3350, 5.69 g/L sodium sulfate, 1.69 g/L sodium bicarbonate, 1.47 g/L sodium chloride and 0.743 g/L potassium chloride. Gavilyte-G 4 Liter Disposable Jug NDC 43386-090-19 Storage Store in sealed container at 15° to 30°C (59° to 86°F). Store reconstituted solution of Gavilyte-G at 2° to 8°C (36° to 46°F). Do not freeze [see Dosage and Administration ( 2.1 )].
Description
Warnings and Precautions, Aspiration: ( 5.7 ) 05/2021
Medication Information
Recent Major Changes
Warnings and Precautions, Aspiration: (5.7) 05/2021
Warnings and Precautions
Risk of fluid and electrolyte abnormalities: Encourage adequate hydration, assess concurrent medications, and consider laboratory assessments prior to and after use. ( 5.1 , 5.2 , 7.1 ) Cardiac arrhythmias: Consider pre-dose and post-colonoscopy ECGs in patients at increased risk of serious cardiac arrhythmias. ( 5.2 ) Seizures: Use caution in patients with a history of seizures and patients at increased risk of seizure, including medications that lower the seizure threshold. ( 5.3 , 7.1 ) Patients with renal impairment or taking concomitant medications that affect renal function: Use caution, ensure adequate hydration and consider testing. ( 5.4 , 7.1 , 8.6) Mucosal ulcerations: Consider potential for mucosal ulcerations when interpreting colonoscopy findings in patients with known or suspected inflammatory bowel disease. ( 5.5 , 7.3 ) Patients at risk for aspiration: Observe during administration. ( 5.7 ) Hypersensitivity reactions including anaphylaxis: Inform patients to seek immediate medical care if symptoms occur. ( 5.8 )
Indications and Usage
GaviLyte-G is a combination of PEG 3350, an osmotic laxative, and electrolytes indicated for cleansing of the colon in preparation for colonoscopy and barium enema X-ray examination in adults. ( 1 )
Dosage and Administration
Preparation and Administration ( 2.1 ): Correct fluid and electrolyte abnormalities before treatment with Gavilyte-G. Reconstitute Gavilyte-G with water prior to ingestion. Do not take oral medications within 1 hour before the start or during administration of Gavilyte-G. ( 2.1 ) Do not take other laxatives while taking Gavilyte-G. Consume only clear liquids; avoid red and purple liquids. Consume water or other clear liquids up until 2 hours before the time of the colonoscopy. Do not consume solid food within 2 hours before starting Gavilyte-G. Adult Dosing Regimen ( 2.2 ): On day prior to colonoscopy, instruct patients to consume a light breakfast at least 2 hours before starting Gavilyte-G. Begin the recommended dosage regimen for Gavilyte-G early in the evening on the day before colonoscopy Drink reconstituted solution at a rate of 8 ounces every 10 minutes, until 4 liters are consumed, or rectal effluent is clear. For complete information on dosing, preparation and administration, see the full prescribing information. ( 2.1 , 2.2 )
Contraindications
Gastrointestinal (GI) obstruction ( 4 , 5.6 ) Bowel perforation ( 4 , 5.6 ) Toxic colitis or toxic megacolon ( 4 ) Gastric retention ( 4 ) Ileus (4) Hypersensitivity to components of Gavilyte-G ( 4 , 5.8 )
Adverse Reactions
Most common adverse reactions are: nausea, abdominal fullness and bloating, abdominal cramps, vomiting and anal irritation ( 6 ) To report SUSPECTED ADVERSE REACTIONS, contact Lupin Pharmaceuticals, Inc. at 1-866-403-7592 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
Some drugs increase risks due to fluid and electrolyte changes ( 7.1 )
Storage and Handling
Gavilyte-G (polyethylene glycol 3350 and electrolytes for oral solution) is supplied in a 4-liter disposable jug containing 236 g polyethylene glycol 3350, 22.74 g sodium sulfate (anhydrous), 6.74 g sodium bicarbonate, 5.86 g sodium chloride and 2.97 g potassium chloride as a white to off white powder. When reconstituted with water to a volume of 4 liters, the solution contains 59 g/L PEG3350, 5.69 g/L sodium sulfate, 1.69 g/L sodium bicarbonate, 1.47 g/L sodium chloride and 0.743 g/L potassium chloride. Gavilyte-G 4 Liter Disposable Jug NDC 43386-090-19 Storage Store in sealed container at 15° to 30°C (59° to 86°F). Store reconstituted solution of Gavilyte-G at 2° to 8°C (36° to 46°F). Do not freeze [see Dosage and Administration ( 2.1 )].
How Supplied
Gavilyte-G (polyethylene glycol 3350 and electrolytes for oral solution) is supplied in a 4-liter disposable jug containing 236 g polyethylene glycol 3350, 22.74 g sodium sulfate (anhydrous), 6.74 g sodium bicarbonate, 5.86 g sodium chloride and 2.97 g potassium chloride as a white to off white powder. When reconstituted with water to a volume of 4 liters, the solution contains 59 g/L PEG3350, 5.69 g/L sodium sulfate, 1.69 g/L sodium bicarbonate, 1.47 g/L sodium chloride and 0.743 g/L potassium chloride. Gavilyte-G 4 Liter Disposable Jug NDC 43386-090-19 Storage Store in sealed container at 15° to 30°C (59° to 86°F). Store reconstituted solution of Gavilyte-G at 2° to 8°C (36° to 46°F). Do not freeze [see Dosage and Administration ( 2.1 )].
Description
Warnings and Precautions, Aspiration: ( 5.7 ) 05/2021
Section 42229-5
GaviLyte-G is indicated for bowel cleansing prior to colonoscopy and barium enema X-ray examination in adults.
5.3 Seizures
There have been reports of generalized tonic-clonic seizures and/or loss of consciousness associated with use of bowel preparation products in patients with no prior history of seizures. The seizure cases were associated with electrolyte abnormalities (e.g., hyponatremia, hypokalemia, hypocalcemia, and hypomagnesemia) and low serum osmolality. The neurologic abnormalities resolved with correction of fluid and electrolyte abnormalities.
Use caution when prescribing GaviLyte-G for patients with a history of seizures and in patients at increased risk of seizure, such as patients taking medications that lower the seizure threshold (e.g., tricyclic antidepressants), patients withdrawing from alcohol or benzodiazepines, or patients with known or suspected hyponatremia. [see Drug Interactions (7.1)]
8.1 Pregnancy
Animal reproduction studies have not been conducted with GaviLyte-G. It is also not known whether GaviLyte-G can cause fetal harm when administered to a pregnant woman or can affect reproductive capacity. GaviLyte-G should be given to a pregnant woman only if clearly needed.
11 Description
Gavilyte-G is a combination of polyethylene glycol 3350, an osmotic laxative, and electrolytes (sodium sulfate, sodium chloride, sodium bicarbonate and potassium chloride) for oral solution supplied in a 4 liter disposable jug containing 236 g polyethylene glycol 3350, 22.74 g sodium sulfate (anhydrous), 6.74 g sodium bicarbonate, 5.86 g sodium chloride, and 2.97 g potassium chloride as a white to off-white powder.
Polyethylene Glycol 3350, NF
Sodium Sulfate, USP
The chemical name is Na2SO4. The average Molecular Weight is 142.04. The structural formula is:
Sodium Bicarbonate, USP
The chemical name is NaHCO3. The average Molecular Weight is 84.01. The structural formula is:
Sodium Chloride, USP
The chemical name is NaCl. The average Molecular Weight: 58.44. The structural formula is:
Na+ Cl-
Potassium Chloride, USP
The chemical name is KCl. The average Molecular Weight: 74.55. The structural formula is:
K-Cl
5.7 Aspiration
Use with caution in patients with impaired gag reflex, unconscious, or semiconscious patients, and patients prone to regurgitation or aspiration. Observe these patients during administration of GaviLyte-G, especially if it is administered via nasogastric tube.
Do not combine GaviLyte-G with starch-based thickeners [see Dosage and Administration (2.1)]. Polyethylene glycol (PEG), a component of GaviLyte-G, when mixed with starch-thickened liquids reduces the viscosity of the starch-thickened liquid. When a PEG-based product used for another indication was mixed in starch-based pre-thickened liquids used in patients with dysphagia, thinning of the liquid occurred and cases of chocking and potential aspiration were reported.
8.4 Pediatric Use
Safety and effectiveness of GaviLyte-G in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of GaviLyte-G did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
2.2 Dosage Regimen
Instruct adult patients that on the day before the colonoscopy procedure, they may consume a light breakfast at least 2 hours before starting Gavilyte-G. Begin the recommended dosage regiment for Gavilyte-G early in the evening on the day before colonoscopy.
Instruct patients to take Gavilyte-G in conjunction with clear liquids as follows:
This preparation can be used with or without the lemon flavor pack.
The pharmacist will add the lemon flavor pack prior to dispensing
(see packet instructions)
4 Liter Jug
- Fill the supplied container containing the Gavilyte-G powder with lukewarm drinking water to the 4-liter fill line
- Do not add any other ingredients, flavors, etc.
- After capping the container, shake vigorously several times to ensure that the ingredients are dissolved.
- Drink at a rate of 8 ounces every 10 minutes until the entire contents are consumed or the rectal effluent is clear. A loose watery bowel movement should result in approximately one hour.
- After reconstitution, keep solution refrigerated 2° to 8°C (36° to 46°F). Do not freeze. Use within 48 hours, discard unused portion.
Administration via a Nasogastric Tube For patients with a nasogastric tube, administer the reconstituted Gavilyte-G solution at a rate of 20 to 30 mL per minute (1.2 to 1.8 liters per hour).
4 Contraindications
6 Adverse Reactions
Most common adverse reactions are: nausea, abdominal fullness and bloating, abdominal cramps, vomiting and anal irritation (6)
To report SUSPECTED ADVERSE REACTIONS, contact Lupin Pharmaceuticals, Inc. at 1-866-403-7592 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
7 Drug Interactions
Some drugs increase risks due to fluid and electrolyte changes (7.1)
8.3 Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when GaviLyte-G is administered to a nursing woman.
5.4 Renal Impairment
Use caution when prescribing GaviLyte-G for patients with impaired renal function or patients taking concomitant medications that may affect renal function (such as diuretics, angiotensin converting enzyme inhibitors, angiotensin receptor blockers, or non-steroidal anti-inflammatory drugs) [see Drug Intersections (7.1)]. Advise these patients of the importance of adequate hydration, and consider performing baseline and post-colonoscopy laboratory tests (electrolytes, creatinine, and BUN) in these patients. [see Use is Specific Populations (8.6)].
8.6 Renal Impairment
Use Gavilyte-G with caution in patients with renal impairment or patients taking concomitant medications that may affect renal function [see Drug Interactions (7.1)]. These patients may be at risk for renal injury. Advise these patients of the importance of adequate hydration before, during and after use of Gavilyte-G and consider performing baseline and post-colonoscopy laboratory tests (electrolytes, creatinine, and BUN) in these patients [see Warnings and Precautions (5.4)].
12.2 Pharmacodynamics
GaviLyte-G induces as diarrhea which rapidly cleanses the bowel, usually within four hours.
12.3 Pharmacokinetics
The pharmacokinetics of PEG 3350 following administration of GaviLyte-G were not assessed. Available pharmacokinetic information for oral PEG 3350 suggests that it is poorly absorbed.
1 Indications and Usage
GaviLyte-G is a combination of PEG 3350, an osmotic laxative, and electrolytes indicated for cleansing of the colon in preparation for colonoscopy and barium enema X-ray examination in adults. (1)
5.2 Cardiac Arrhythmias
There have been rare reports of serious arrhythmias associated with the use of ionic osmotic laxative products for bowel preparation. Use caution when prescribing GaviLyte-G for patients at increased risk of arrhythmias (e.g., patients with a history of prolonged QT, uncontrolled arrhythmias, recent myocardial infarction, unstable angina, congestive heart failure, or cardiomyopathy). Consider pre-dose and post-colonoscopy ECGs in patients at increased risk of serious cardiac arrhythmias.
7.3 Stimulant Laxatives
Concurrent use of stimulant laxatives and GaviLyte-G may increase the risk of mucosal ulceration or ischemic colitis. Avoid use of stimulant laxatives (e.g., bisacodyl, sodium picosulfate) while taking Gavilyte-G [see Warnings and Precautions (5.5)].
12.1 Mechanism of Action
The primary mode of action is thought to be through the osmotic effect of polyethylene glycol 3350 which causes water to be retained in the colon and produces a watery stool.
5 Warnings and Precautions
- Risk of fluid and electrolyte abnormalities: Encourage adequate hydration, assess concurrent medications, and consider laboratory assessments prior to and after use. (5.1, 5.2, 7.1)
- Cardiac arrhythmias: Consider pre-dose and post-colonoscopy ECGs in patients at increased risk of serious cardiac arrhythmias. (5.2)
- Seizures: Use caution in patients with a history of seizures and patients at increased risk of seizure, including medications that lower the seizure threshold. (5.3, 7.1)
- Patients with renal impairment or taking concomitant medications that affect renal function: Use caution, ensure adequate hydration and consider testing. (5.4, 7.1, 8.6)
- Mucosal ulcerations: Consider potential for mucosal ulcerations when interpreting colonoscopy findings in patients with known or suspected inflammatory bowel disease. (5.5, 7.3)
- Patients at risk for aspiration: Observe during administration. (5.7)
- Hypersensitivity reactions including anaphylaxis: Inform patients to seek immediate medical care if symptoms occur. (5.8)
2 Dosage and Administration
Preparation and Administration (2.1):
- Correct fluid and electrolyte abnormalities before treatment with Gavilyte-G.
- Reconstitute Gavilyte-G with water prior to ingestion.
- Do not take oral medications within 1 hour before the start or during administration of Gavilyte-G. (2.1)
- Do not take other laxatives while taking Gavilyte-G.
- Consume only clear liquids; avoid red and purple liquids.
- Consume water or other clear liquids up until 2 hours before the time of the colonoscopy.
- Do not consume solid food within 2 hours before starting Gavilyte-G.
Adult Dosing Regimen (2.2):
- On day prior to colonoscopy, instruct patients to consume a light breakfast at least 2 hours before starting Gavilyte-G.
- Begin the recommended dosage regimen for Gavilyte-G early in the evening on the day before colonoscopy
- Drink reconstituted solution at a rate of 8 ounces every 10 minutes, until 4 liters are consumed, or rectal effluent is clear.
- For complete information on dosing, preparation and administration, see the full prescribing information. (2.1, 2.2)
3 Dosage Forms and Strengths
For Oral Solution: 236 g polyethylene glycol 3350, 22.74 g sodium sulfate (anhydrous), 6.74 g sodium bicarbonate, 5.86 g sodium chloride, 2.97 g potassium chloride and 2 g favoring agent supplied in one 4 liter disposable jug. (3)
5.8 Hypersensitivity Reactions
Gavilyte-G contains PEG and may cause serious hypersensitivity reactions including anaphylaxis, angioedema, rash, urticaria, and pruritus [see Adverse Reactions (6 )]. Inform patients of the signs and symptoms of anaphylaxis and instruct them to seek immediate medical care should signs and symptoms occur.
17 Patient Counseling Information
Advise the patient to read the FDA-Approved Patient Labeling (Medication Guide and Instructions for Use).
Instruct patients:
- To reconstitute Gavilyte-G with water prior to ingestion.
- Not to take other laxatives while they are taking Gavilyte-G.
- Not to take oral medications within 1 hour before the start or during the administration of Gavilyte-G.
- To take only clear liquids but avoid red and purple liquids.
- To consume water or other clear liquids during the bowel preparation and after completion of the bowel preparation up until 2 hours before the time of the colonoscopy.
- To follow the directions in the Instructions for Use on how to prepare and administer the product.
- If they experience severe bloating, distention or abdominal pain, to slow or temporarily discontinue drinking the solution and to contact their healthcare provider.
- To contact their healthcare provider if they develop signs and symptoms of dehydration or if they experience altered consciousness or seizures. [see Warnings and Precautions (5.1, 5.2, 5.3, 5.4)].
- To discontinue administration of the solution and contact their healthcare provider if they develop symptoms of a hypersensitivity reaction [see Warnings and Precautions (5.8)].
LUPIN and the
are registered trademarks of Lupin Pharmaceuticals, Inc.Manufactured by:
Novel Laboratories, Inc.
Somerset, NJ 08873
Manufactured for:
Lupin Pharmaceuticals, Inc.
Naples FL, 34108
SAP code: 278923
Rev: 11/2024
16 How Supplied/storage and Handling
Gavilyte-G (polyethylene glycol 3350 and electrolytes for oral solution) is supplied in a 4-liter disposable jug containing 236 g polyethylene glycol 3350, 22.74 g sodium sulfate (anhydrous), 6.74 g sodium bicarbonate, 5.86 g sodium chloride and 2.97 g potassium chloride as a white to off white powder.
- When reconstituted with water to a volume of 4 liters, the solution contains 59 g/L PEG3350, 5.69 g/L sodium sulfate, 1.69 g/L sodium bicarbonate, 1.47 g/L sodium chloride and 0.743 g/L potassium chloride.
Gavilyte-G 4 Liter Disposable Jug NDC 43386-090-19
Storage
Store in sealed container at 15° to 30°C (59° to 86°F). Store reconstituted solution of Gavilyte-G at 2° to 8°C (36° to 46°F). Do not freeze [see Dosage and Administration (2.1)].
Package Label.principal Display Panel
GaviLyte-G
Container Label
Lemon Flavor Pack
7.2 Potential for Reduced Drug Absorption
Gavilyte-G can reduce the absorption of other administered drugs. Administer oral medications within one hour before the start of administration of Gavilyte-G [see Dosage and Administration (2.1)].
5.1 Serious Fluid and Serum Chemistry Abnormalities
Advise patients to hydrate adequately before, during, and after the use of GaviLyte-G. Use caution in patients with congestive heart failure when replacing fluids. If a patient develops significant vomiting or signs of dehydration including signs of orthostatic hypotension after taking GaviLyte-G, consider performing post-colonoscopy lab tests (electrolytes, creatinine, and BUN) and treat accordingly. Fluid and electrolyte disturbances can lead to serious adverse events including cardiac arrhythmias, seizures and renal impairment.Correct Fluid and electrolyte abnormalities before treatment with GaviLyte-G.
In addition, use caution when prescribing GaviLyte-G for patients who have conditions, or who are using medications, that increase the risk for fluid and electrolyte disturbances or may increase the risk of adverse events of seizure, arrhythmias, and renal impairment [see Drug Interactions (7.1) ]
5.5 Colonic Mucosal Ulcerations and Ischemic Colitis
Administration of osmotic laxative products may produce colonic mucosal aphthous ulcerations, and there have been reports of more serious cases of ischemic colitis requiring hospitalization. Concurrent use of stimulant laxatives and GaviLyte-G may increase this risk[see Drug Interactions (7.3)]. Consider the potential for mucosal ulcerations resulting from the bowel preparation when interpreting colonoscopy findings in patients with known or suspect inflammatory bowel disease (IBD).
2.1 Important Preparation and Administration Instructions
- Correct fluid and electrolyte abnormalities before treatment with Gavilyte-G [see Warnings and Precautions (5.1)].
- Reconstitute Gavilyte-G with water prior to ingestion, do not take undissolved Gavilyte-G [see Dosage and Administration (2.2)]. Do not reconstitute with other liquids and/or add starch-based thickeners to the mixing container [see Warnings and Precautions (5.7)].
- Do not take oral medications within 1 hour before the start of or during administration of Gavilyte-G [see Drug Interactions (7.2)].
- Do not take other laxatives while taking Gavilyte-G [see Drug Interactions (7.3)]. Consume only clear liquids, avoid red and purple liquids.
- Patients may consume water or other clear liquids during the bowel preparation and after completion of the bowel preparation up until 2 hours before the time of the colonoscopy.
- The solution is more palatable if chilled prior to administration.
- Do not consume solid food within 2 hours before starting Gavilyte-G. For the best results, do not consume solid food for 3 to 4 hours before drinking the solution.
- If severe bloating, distention or abdominal pain occurs, slow or temporarily discontinue Gavilyte-G until the symptoms abate.
5.6 Use in Patients With Significant Gastrointestinal Disease
If gastrointestinal obstruction or perforation is suspected, perform appropriate diagnostic studies to rule out these conditions before administering GaviLyte-G. [see Contraindications (4)].
Use with caution in patients with severe active ulcerative colitis.
7.1 Drugs That May Increase Risks Due to Fluid and Electrolyte Abnormalities
Use caution when prescribing GaviLyte-G for patients with conditions and/or who are using medications that increase the risk for fluid and electrolyte disturbances or may increase the risk of renal impairment, seizure, arrhythmias, and prolonged QT in the setting of fluid and electrolyte abnormalities. [see Warnings and Precautions (5.1, 5.2, 5.3, 5.4)]. Consider additional patient evaluations as appropriate.
Structured Label Content
Recent Major Changes (RECENT MAJOR CHANGES)
Warnings and Precautions, Aspiration: (5.7) 05/2021
Section 42229-5 (42229-5)
GaviLyte-G is indicated for bowel cleansing prior to colonoscopy and barium enema X-ray examination in adults.
5.3 Seizures
There have been reports of generalized tonic-clonic seizures and/or loss of consciousness associated with use of bowel preparation products in patients with no prior history of seizures. The seizure cases were associated with electrolyte abnormalities (e.g., hyponatremia, hypokalemia, hypocalcemia, and hypomagnesemia) and low serum osmolality. The neurologic abnormalities resolved with correction of fluid and electrolyte abnormalities.
Use caution when prescribing GaviLyte-G for patients with a history of seizures and in patients at increased risk of seizure, such as patients taking medications that lower the seizure threshold (e.g., tricyclic antidepressants), patients withdrawing from alcohol or benzodiazepines, or patients with known or suspected hyponatremia. [see Drug Interactions (7.1)]
8.1 Pregnancy
Animal reproduction studies have not been conducted with GaviLyte-G. It is also not known whether GaviLyte-G can cause fetal harm when administered to a pregnant woman or can affect reproductive capacity. GaviLyte-G should be given to a pregnant woman only if clearly needed.
11 Description (11 DESCRIPTION)
Gavilyte-G is a combination of polyethylene glycol 3350, an osmotic laxative, and electrolytes (sodium sulfate, sodium chloride, sodium bicarbonate and potassium chloride) for oral solution supplied in a 4 liter disposable jug containing 236 g polyethylene glycol 3350, 22.74 g sodium sulfate (anhydrous), 6.74 g sodium bicarbonate, 5.86 g sodium chloride, and 2.97 g potassium chloride as a white to off-white powder.
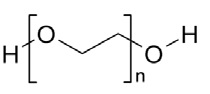
Polyethylene Glycol 3350, NF
Sodium Sulfate, USP
The chemical name is Na2SO4. The average Molecular Weight is 142.04. The structural formula is:
Sodium Bicarbonate, USP
The chemical name is NaHCO3. The average Molecular Weight is 84.01. The structural formula is:
Sodium Chloride, USP
The chemical name is NaCl. The average Molecular Weight: 58.44. The structural formula is:
Na+ Cl-
Potassium Chloride, USP
The chemical name is KCl. The average Molecular Weight: 74.55. The structural formula is:
K-Cl
5.7 Aspiration
Use with caution in patients with impaired gag reflex, unconscious, or semiconscious patients, and patients prone to regurgitation or aspiration. Observe these patients during administration of GaviLyte-G, especially if it is administered via nasogastric tube.
Do not combine GaviLyte-G with starch-based thickeners [see Dosage and Administration (2.1)]. Polyethylene glycol (PEG), a component of GaviLyte-G, when mixed with starch-thickened liquids reduces the viscosity of the starch-thickened liquid. When a PEG-based product used for another indication was mixed in starch-based pre-thickened liquids used in patients with dysphagia, thinning of the liquid occurred and cases of chocking and potential aspiration were reported.
8.4 Pediatric Use
Safety and effectiveness of GaviLyte-G in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of GaviLyte-G did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
2.2 Dosage Regimen
Instruct adult patients that on the day before the colonoscopy procedure, they may consume a light breakfast at least 2 hours before starting Gavilyte-G. Begin the recommended dosage regiment for Gavilyte-G early in the evening on the day before colonoscopy.
Instruct patients to take Gavilyte-G in conjunction with clear liquids as follows:
This preparation can be used with or without the lemon flavor pack.
The pharmacist will add the lemon flavor pack prior to dispensing
(see packet instructions)
4 Liter Jug
- Fill the supplied container containing the Gavilyte-G powder with lukewarm drinking water to the 4-liter fill line
- Do not add any other ingredients, flavors, etc.
- After capping the container, shake vigorously several times to ensure that the ingredients are dissolved.
- Drink at a rate of 8 ounces every 10 minutes until the entire contents are consumed or the rectal effluent is clear. A loose watery bowel movement should result in approximately one hour.
- After reconstitution, keep solution refrigerated 2° to 8°C (36° to 46°F). Do not freeze. Use within 48 hours, discard unused portion.
Administration via a Nasogastric Tube For patients with a nasogastric tube, administer the reconstituted Gavilyte-G solution at a rate of 20 to 30 mL per minute (1.2 to 1.8 liters per hour).
4 Contraindications (4 CONTRAINDICATIONS)
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions are: nausea, abdominal fullness and bloating, abdominal cramps, vomiting and anal irritation (6)
To report SUSPECTED ADVERSE REACTIONS, contact Lupin Pharmaceuticals, Inc. at 1-866-403-7592 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
7 Drug Interactions (7 DRUG INTERACTIONS)
Some drugs increase risks due to fluid and electrolyte changes (7.1)
8.3 Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when GaviLyte-G is administered to a nursing woman.
5.4 Renal Impairment
Use caution when prescribing GaviLyte-G for patients with impaired renal function or patients taking concomitant medications that may affect renal function (such as diuretics, angiotensin converting enzyme inhibitors, angiotensin receptor blockers, or non-steroidal anti-inflammatory drugs) [see Drug Intersections (7.1)]. Advise these patients of the importance of adequate hydration, and consider performing baseline and post-colonoscopy laboratory tests (electrolytes, creatinine, and BUN) in these patients. [see Use is Specific Populations (8.6)].
8.6 Renal Impairment
Use Gavilyte-G with caution in patients with renal impairment or patients taking concomitant medications that may affect renal function [see Drug Interactions (7.1)]. These patients may be at risk for renal injury. Advise these patients of the importance of adequate hydration before, during and after use of Gavilyte-G and consider performing baseline and post-colonoscopy laboratory tests (electrolytes, creatinine, and BUN) in these patients [see Warnings and Precautions (5.4)].
12.2 Pharmacodynamics
GaviLyte-G induces as diarrhea which rapidly cleanses the bowel, usually within four hours.
12.3 Pharmacokinetics
The pharmacokinetics of PEG 3350 following administration of GaviLyte-G were not assessed. Available pharmacokinetic information for oral PEG 3350 suggests that it is poorly absorbed.
1 Indications and Usage (1 INDICATIONS AND USAGE)
GaviLyte-G is a combination of PEG 3350, an osmotic laxative, and electrolytes indicated for cleansing of the colon in preparation for colonoscopy and barium enema X-ray examination in adults. (1)
5.2 Cardiac Arrhythmias
There have been rare reports of serious arrhythmias associated with the use of ionic osmotic laxative products for bowel preparation. Use caution when prescribing GaviLyte-G for patients at increased risk of arrhythmias (e.g., patients with a history of prolonged QT, uncontrolled arrhythmias, recent myocardial infarction, unstable angina, congestive heart failure, or cardiomyopathy). Consider pre-dose and post-colonoscopy ECGs in patients at increased risk of serious cardiac arrhythmias.
7.3 Stimulant Laxatives
Concurrent use of stimulant laxatives and GaviLyte-G may increase the risk of mucosal ulceration or ischemic colitis. Avoid use of stimulant laxatives (e.g., bisacodyl, sodium picosulfate) while taking Gavilyte-G [see Warnings and Precautions (5.5)].
12.1 Mechanism of Action
The primary mode of action is thought to be through the osmotic effect of polyethylene glycol 3350 which causes water to be retained in the colon and produces a watery stool.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Risk of fluid and electrolyte abnormalities: Encourage adequate hydration, assess concurrent medications, and consider laboratory assessments prior to and after use. (5.1, 5.2, 7.1)
- Cardiac arrhythmias: Consider pre-dose and post-colonoscopy ECGs in patients at increased risk of serious cardiac arrhythmias. (5.2)
- Seizures: Use caution in patients with a history of seizures and patients at increased risk of seizure, including medications that lower the seizure threshold. (5.3, 7.1)
- Patients with renal impairment or taking concomitant medications that affect renal function: Use caution, ensure adequate hydration and consider testing. (5.4, 7.1, 8.6)
- Mucosal ulcerations: Consider potential for mucosal ulcerations when interpreting colonoscopy findings in patients with known or suspected inflammatory bowel disease. (5.5, 7.3)
- Patients at risk for aspiration: Observe during administration. (5.7)
- Hypersensitivity reactions including anaphylaxis: Inform patients to seek immediate medical care if symptoms occur. (5.8)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Preparation and Administration (2.1):
- Correct fluid and electrolyte abnormalities before treatment with Gavilyte-G.
- Reconstitute Gavilyte-G with water prior to ingestion.
- Do not take oral medications within 1 hour before the start or during administration of Gavilyte-G. (2.1)
- Do not take other laxatives while taking Gavilyte-G.
- Consume only clear liquids; avoid red and purple liquids.
- Consume water or other clear liquids up until 2 hours before the time of the colonoscopy.
- Do not consume solid food within 2 hours before starting Gavilyte-G.
Adult Dosing Regimen (2.2):
- On day prior to colonoscopy, instruct patients to consume a light breakfast at least 2 hours before starting Gavilyte-G.
- Begin the recommended dosage regimen for Gavilyte-G early in the evening on the day before colonoscopy
- Drink reconstituted solution at a rate of 8 ounces every 10 minutes, until 4 liters are consumed, or rectal effluent is clear.
- For complete information on dosing, preparation and administration, see the full prescribing information. (2.1, 2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For Oral Solution: 236 g polyethylene glycol 3350, 22.74 g sodium sulfate (anhydrous), 6.74 g sodium bicarbonate, 5.86 g sodium chloride, 2.97 g potassium chloride and 2 g favoring agent supplied in one 4 liter disposable jug. (3)
5.8 Hypersensitivity Reactions
Gavilyte-G contains PEG and may cause serious hypersensitivity reactions including anaphylaxis, angioedema, rash, urticaria, and pruritus [see Adverse Reactions (6 )]. Inform patients of the signs and symptoms of anaphylaxis and instruct them to seek immediate medical care should signs and symptoms occur.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-Approved Patient Labeling (Medication Guide and Instructions for Use).
Instruct patients:
- To reconstitute Gavilyte-G with water prior to ingestion.
- Not to take other laxatives while they are taking Gavilyte-G.
- Not to take oral medications within 1 hour before the start or during the administration of Gavilyte-G.
- To take only clear liquids but avoid red and purple liquids.
- To consume water or other clear liquids during the bowel preparation and after completion of the bowel preparation up until 2 hours before the time of the colonoscopy.
- To follow the directions in the Instructions for Use on how to prepare and administer the product.
- If they experience severe bloating, distention or abdominal pain, to slow or temporarily discontinue drinking the solution and to contact their healthcare provider.
- To contact their healthcare provider if they develop signs and symptoms of dehydration or if they experience altered consciousness or seizures. [see Warnings and Precautions (5.1, 5.2, 5.3, 5.4)].
- To discontinue administration of the solution and contact their healthcare provider if they develop symptoms of a hypersensitivity reaction [see Warnings and Precautions (5.8)].
LUPIN and the
are registered trademarks of Lupin Pharmaceuticals, Inc.Manufactured by:
Novel Laboratories, Inc.
Somerset, NJ 08873
Manufactured for:
Lupin Pharmaceuticals, Inc.
Naples FL, 34108
SAP code: 278923
Rev: 11/2024
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Gavilyte-G (polyethylene glycol 3350 and electrolytes for oral solution) is supplied in a 4-liter disposable jug containing 236 g polyethylene glycol 3350, 22.74 g sodium sulfate (anhydrous), 6.74 g sodium bicarbonate, 5.86 g sodium chloride and 2.97 g potassium chloride as a white to off white powder.
- When reconstituted with water to a volume of 4 liters, the solution contains 59 g/L PEG3350, 5.69 g/L sodium sulfate, 1.69 g/L sodium bicarbonate, 1.47 g/L sodium chloride and 0.743 g/L potassium chloride.
Gavilyte-G 4 Liter Disposable Jug NDC 43386-090-19
Storage
Store in sealed container at 15° to 30°C (59° to 86°F). Store reconstituted solution of Gavilyte-G at 2° to 8°C (36° to 46°F). Do not freeze [see Dosage and Administration (2.1)].
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
GaviLyte-G
Container Label
Lemon Flavor Pack
7.2 Potential for Reduced Drug Absorption
Gavilyte-G can reduce the absorption of other administered drugs. Administer oral medications within one hour before the start of administration of Gavilyte-G [see Dosage and Administration (2.1)].
5.1 Serious Fluid and Serum Chemistry Abnormalities
Advise patients to hydrate adequately before, during, and after the use of GaviLyte-G. Use caution in patients with congestive heart failure when replacing fluids. If a patient develops significant vomiting or signs of dehydration including signs of orthostatic hypotension after taking GaviLyte-G, consider performing post-colonoscopy lab tests (electrolytes, creatinine, and BUN) and treat accordingly. Fluid and electrolyte disturbances can lead to serious adverse events including cardiac arrhythmias, seizures and renal impairment.Correct Fluid and electrolyte abnormalities before treatment with GaviLyte-G.
In addition, use caution when prescribing GaviLyte-G for patients who have conditions, or who are using medications, that increase the risk for fluid and electrolyte disturbances or may increase the risk of adverse events of seizure, arrhythmias, and renal impairment [see Drug Interactions (7.1) ]
5.5 Colonic Mucosal Ulcerations and Ischemic Colitis
Administration of osmotic laxative products may produce colonic mucosal aphthous ulcerations, and there have been reports of more serious cases of ischemic colitis requiring hospitalization. Concurrent use of stimulant laxatives and GaviLyte-G may increase this risk[see Drug Interactions (7.3)]. Consider the potential for mucosal ulcerations resulting from the bowel preparation when interpreting colonoscopy findings in patients with known or suspect inflammatory bowel disease (IBD).
2.1 Important Preparation and Administration Instructions
- Correct fluid and electrolyte abnormalities before treatment with Gavilyte-G [see Warnings and Precautions (5.1)].
- Reconstitute Gavilyte-G with water prior to ingestion, do not take undissolved Gavilyte-G [see Dosage and Administration (2.2)]. Do not reconstitute with other liquids and/or add starch-based thickeners to the mixing container [see Warnings and Precautions (5.7)].
- Do not take oral medications within 1 hour before the start of or during administration of Gavilyte-G [see Drug Interactions (7.2)].
- Do not take other laxatives while taking Gavilyte-G [see Drug Interactions (7.3)]. Consume only clear liquids, avoid red and purple liquids.
- Patients may consume water or other clear liquids during the bowel preparation and after completion of the bowel preparation up until 2 hours before the time of the colonoscopy.
- The solution is more palatable if chilled prior to administration.
- Do not consume solid food within 2 hours before starting Gavilyte-G. For the best results, do not consume solid food for 3 to 4 hours before drinking the solution.
- If severe bloating, distention or abdominal pain occurs, slow or temporarily discontinue Gavilyte-G until the symptoms abate.
5.6 Use in Patients With Significant Gastrointestinal Disease (5.6 Use in Patients with Significant Gastrointestinal Disease)
If gastrointestinal obstruction or perforation is suspected, perform appropriate diagnostic studies to rule out these conditions before administering GaviLyte-G. [see Contraindications (4)].
Use with caution in patients with severe active ulcerative colitis.
7.1 Drugs That May Increase Risks Due to Fluid and Electrolyte Abnormalities (7.1 Drugs that May Increase Risks Due to Fluid and Electrolyte Abnormalities)
Use caution when prescribing GaviLyte-G for patients with conditions and/or who are using medications that increase the risk for fluid and electrolyte disturbances or may increase the risk of renal impairment, seizure, arrhythmias, and prolonged QT in the setting of fluid and electrolyte abnormalities. [see Warnings and Precautions (5.1, 5.2, 5.3, 5.4)]. Consider additional patient evaluations as appropriate.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:29.030605 · Updated: 2026-03-14T22:33:18.370731