39659808-ec63-3ccc-e063-6294a90ab611
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
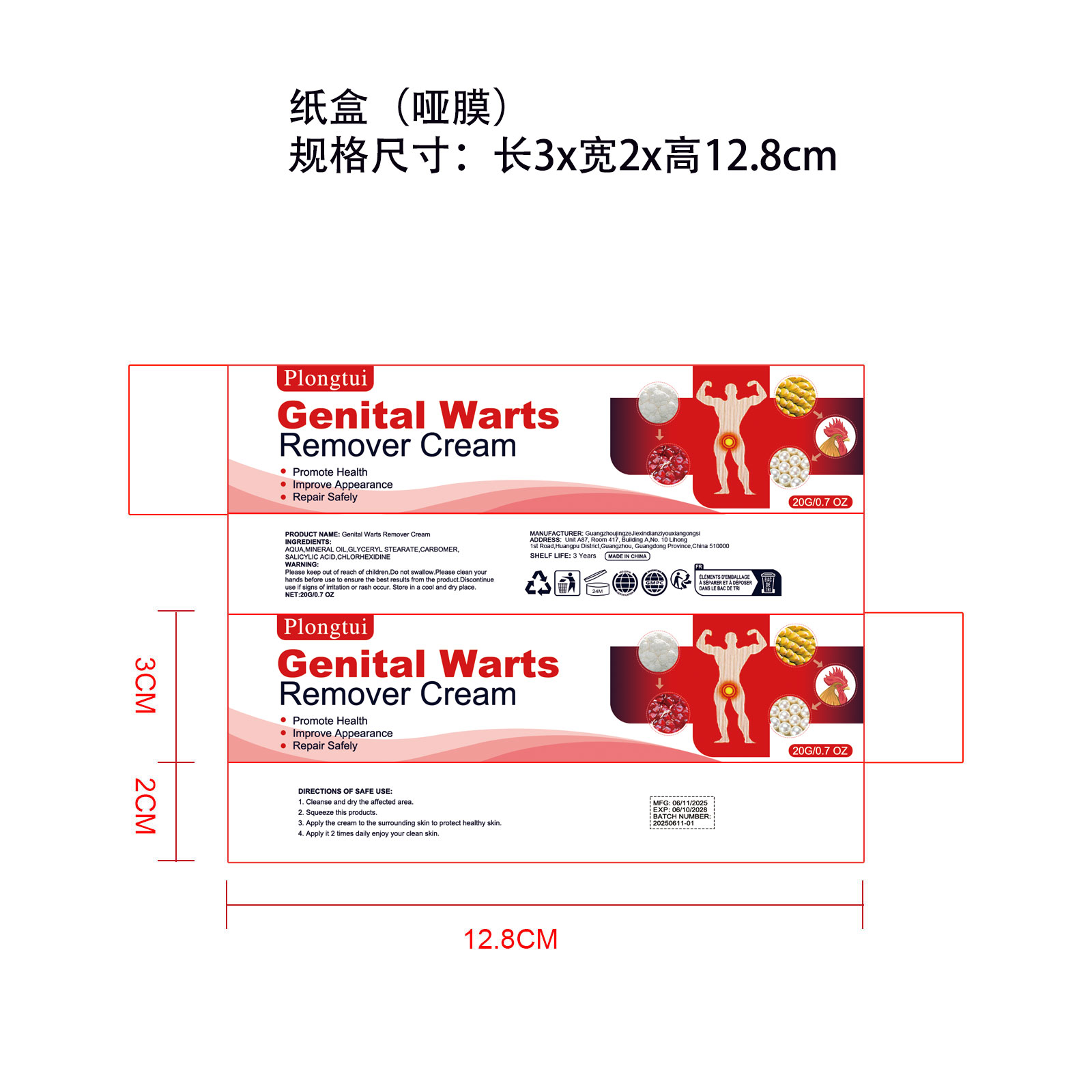
Composition & Product
Identifiers & Packaging
Description
Keep out of reach of children section
Medication Information
Warnings and Precautions
Keep out of reach of children
Indications and Usage
Genital Warts Remover
Dosage and Administration
20G/0.7 OZ
Description
Keep out of reach of children section
Section 50565-1
Keep out of reach of children section
Section 51727-6
AQUA,MINERAL OIL,GLYCERYL STEARATE,CARBOMER
Section 51945-4
Section 55105-1
Genital Warts Remover
Section 55106-9
SALICYLIC ACID,CHLORHEXIDINE
Structured Label Content
Indications and Usage (34067-9)
Genital Warts Remover
Dosage and Administration (34068-7)
20G/0.7 OZ
Warnings and Precautions (34071-1)
Keep out of reach of children
Section 50565-1 (50565-1)
Keep out of reach of children section
Section 51727-6 (51727-6)
AQUA,MINERAL OIL,GLYCERYL STEARATE,CARBOMER
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Genital Warts Remover
Section 55106-9 (55106-9)
SALICYLIC ACID,CHLORHEXIDINE
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:12:04.657456 · Updated: 2026-03-14T23:17:26.215261