Lidocaine 5_percent Cream
3953ccf4-f772-4f9c-ae7e-f373b6fe16dd
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
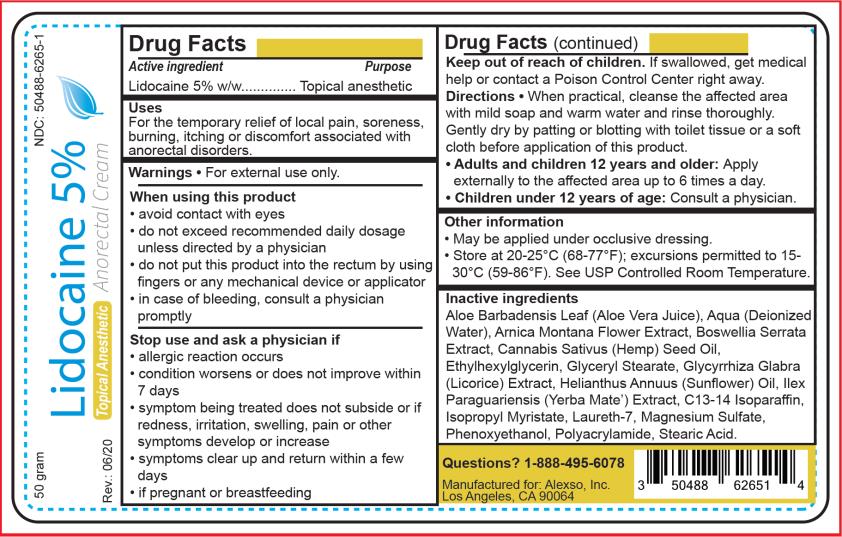
LIDOCAINE - L idocaine 5 % anorectal c ream Alexso, Inc. Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies. ---------- Lidocaine 5 % Anorectal Cream Drug Facts
Purpose
Topical anesthetic
Medication Information
Purpose
Topical anesthetic
Description
LIDOCAINE - L idocaine 5 % anorectal c ream Alexso, Inc. Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies. ---------- Lidocaine 5 % Anorectal Cream Drug Facts
Uses
For the temporary relief of local pain, soreness, burning, itching or discomfort associated with anorectal disorders.
Section 42229-5
LIDOCAINE
- L
idocaine 5
%
anorectal c
ream
Alexso, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Lidocaine 5 % Anorectal Cream
Drug Facts
Warnings
For external use only.
Directions
When practical, cleanse the affected area with mild soap and warm water and rinse thoroughly. Gently dry by patting or blotting with toilet tissue or a soft cloth before application of this product.
Adults and children 12 years and older: Apply externally to the affected area up to 6 times a day.
Children under 12 years of age: Consult a physician.
Active Ingredient
Lidocaine 5% w/w
Other Information
- May be applied under occlusive dressing.
- Store at 20-25°C (68-77°F); excursions permitted to 15-30°C (59-86°F). See USP Controlled Room Temperature.
Stop Use and Ask A
- allergic reaction occurs
- condition worsens or does not improve within 7 days
- symptom being treated does not subside or if redness, irritation, swelling, pain or other symptoms develop or increase
- symptoms clear up and return within a few days
- if pregnant or breastfeeding
Inactive Ingredients
Aloe Barbadensis Leaf (Aloe Vera Juice), Aqua (Deionized Water), Arnica Montana Flower Extract, Boswellia Serrata Extract, Cannabis Sativus (Hemp) Seed Oil, Ethylhexylglycerin, Glyceryl Stearate, Glycyrrhiza Glabra (Licorice) Extract, Helianthus Annuus (Sunflower) Oil, Ilex Paraguariensis (Yerba Mate) Extract, C13-14 Isoparaffin, Isopropyl Myristate, Laureth-7, Magnesium Sulfate, Phenoxyethanol, Polyacrylamide, Stearic Acid.
Principal Display Panel
Lidocaine 5 % C ream
NDC 50488-6265-1
Lidocaine 5 %
Anorectal Cream
Topical Anesthetic
50 grams
Manufactured for:
Alexso, Inc.
Los Angeles, CA 90064
When Using This Product
- avoid contact with eyes
- do not exceed recommended daily dosage unless directed by a physician
- do not put this product into the rectum by using fingers or any mechanical device or applicator
- in case of bleeding, consult a physician promptly
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
For the temporary relief of local pain, soreness, burning, itching or discomfort associated with anorectal disorders.
Section 42229-5 (42229-5)
LIDOCAINE
- L
idocaine 5
%
anorectal c
ream
Alexso, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Lidocaine 5 % Anorectal Cream
Drug Facts
Purpose
Topical anesthetic
Warnings
For external use only.
Directions
When practical, cleanse the affected area with mild soap and warm water and rinse thoroughly. Gently dry by patting or blotting with toilet tissue or a soft cloth before application of this product.
Adults and children 12 years and older: Apply externally to the affected area up to 6 times a day.
Children under 12 years of age: Consult a physician.
Active Ingredient (Active ingredient)
Lidocaine 5% w/w
Other Information (Other information)
- May be applied under occlusive dressing.
- Store at 20-25°C (68-77°F); excursions permitted to 15-30°C (59-86°F). See USP Controlled Room Temperature.
Stop Use and Ask A (Stop use and ask a)
- allergic reaction occurs
- condition worsens or does not improve within 7 days
- symptom being treated does not subside or if redness, irritation, swelling, pain or other symptoms develop or increase
- symptoms clear up and return within a few days
- if pregnant or breastfeeding
Inactive Ingredients (Inactive ingredients)
Aloe Barbadensis Leaf (Aloe Vera Juice), Aqua (Deionized Water), Arnica Montana Flower Extract, Boswellia Serrata Extract, Cannabis Sativus (Hemp) Seed Oil, Ethylhexylglycerin, Glyceryl Stearate, Glycyrrhiza Glabra (Licorice) Extract, Helianthus Annuus (Sunflower) Oil, Ilex Paraguariensis (Yerba Mate) Extract, C13-14 Isoparaffin, Isopropyl Myristate, Laureth-7, Magnesium Sulfate, Phenoxyethanol, Polyacrylamide, Stearic Acid.
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
Lidocaine 5 % C ream
NDC 50488-6265-1
Lidocaine 5 %
Anorectal Cream
Topical Anesthetic
50 grams
Manufactured for:
Alexso, Inc.
Los Angeles, CA 90064
When Using This Product (When using this product)
- avoid contact with eyes
- do not exceed recommended daily dosage unless directed by a physician
- do not put this product into the rectum by using fingers or any mechanical device or applicator
- in case of bleeding, consult a physician promptly
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:58:05.542524 · Updated: 2026-03-14T22:57:28.915858