These Highlights Do Not Include All The Information Needed To Use Pemetrexed For Injection Safely And Effectively. See Full Prescribing Information For Pemetrexed For Injection.
39219e4b-80a6-4d4f-96a3-1202e542d050
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Pemetrexed for injection is a folate analog metabolic inhibitor indicated: in combination with pembrolizumab and platinum chemotherapy, for the initial treatment of patients with metastatic non-squamous non-small cell lung cancer (NSCLC), with no EGFR or ALK genomic tumor aberrations. ( 1.1 ) in combination with cisplatin for the initial treatment of patients with locally advanced or metastatic, non-squamous (NSCLC). ( 1.1 ) as a single agent for the maintenance treatment of patients with locally advanced or metastatic, non-squamous NSCLC whose disease has not progressed after four cycles of platinum-based first-line chemotherapy. ( 1.1 ) as a single agent for the treatment of patients with recurrent, metastatic non-squamous, NSCLC after prior chemotherapy. ( 1.1 ) Limitations of Use: Pemetrexed for injection is not indicated for the treatment of patients with squamous cell, non-small cell lung cancer. ( 1.1 ) initial treatment, in combination with cisplatin, of patients with malignant pleural mesothelioma whose disease is unresectable or who are otherwise not candidates for curative surgery. ( 1.2 )
Indications and Usage
Pemetrexed for injection is a folate analog metabolic inhibitor indicated: in combination with pembrolizumab and platinum chemotherapy, for the initial treatment of patients with metastatic non-squamous non-small cell lung cancer (NSCLC), with no EGFR or ALK genomic tumor aberrations. ( 1.1 ) in combination with cisplatin for the initial treatment of patients with locally advanced or metastatic, non-squamous (NSCLC). ( 1.1 ) as a single agent for the maintenance treatment of patients with locally advanced or metastatic, non-squamous NSCLC whose disease has not progressed after four cycles of platinum-based first-line chemotherapy. ( 1.1 ) as a single agent for the treatment of patients with recurrent, metastatic non-squamous, NSCLC after prior chemotherapy. ( 1.1 ) Limitations of Use: Pemetrexed for injection is not indicated for the treatment of patients with squamous cell, non-small cell lung cancer. ( 1.1 ) initial treatment, in combination with cisplatin, of patients with malignant pleural mesothelioma whose disease is unresectable or who are otherwise not candidates for curative surgery. ( 1.2 )
Dosage and Administration
The recommended dose of pemetrexed for injection administered with pembrolizumab and platinum chemotherapy in patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater is 500 mg/m 2 as an intravenous infusion over 10 minutes, administered after pembrolizumab and prior to platinum chemotherapy, on Day 1 of each 21-day cycle. ( 2.1 ) The recommended dose of pemetrexed for injection, administered as a single agent or with cisplatin, in patients with creatinine clearance of 45 mL/minute or greater is 500 mg/m 2 as an intravenous infusion over 10 minutes on Day 1 of each 21-day cycle. ( 2.1 , 2.2 ) Initiate folic acid 400 mcg to 1000 mcg orally, once daily, beginning 7 days prior to the first dose of pemetrexed for injection and continue until 21 days after the last dose of pemetrexed for injection. ( 2.4 ) Administer vitamin B 12 , 1 mg intramuscularly, 1 week prior to the first dose of pemetrexed for injection and every 3 cycles. ( 2.4 ) Administer dexamethasone 4 mg orally, twice daily the day before, the day of, and the day after pemetrexed for injection administration. ( 2.4 )
Warnings and Precautions
Myelosuppression: Can cause severe bone marrow suppression resulting in cytopenia and an increased risk of infection. Do not administer pemetrexed for injection when the absolute neutrophil count is less than 1500 cells/mm 3 and platelets are less than 100,000 cells/mm 3 . Initiate supplementation with oral folic acid and intramuscular vitamin B 12 to reduce the severity of hematologic and gastrointestinal toxicity of pemetrexed for injection. ( 2.4 , 5.1 ) Renal Failure: Can cause severe, and sometimes fatal, renal failure. Do not administer when creatinine clearance is less than 45 mL/min. ( 2.3 , 5.2 ) Bullous and Exfoliative Skin Toxicity: Permanently discontinue for severe and life-threatening bullous, blistering or exfoliating skin toxicity. ( 5.3 ) Interstitial Pneumonitis: Withhold for acute onset of new or progressive unexplained pulmonary symptoms. Permanently discontinue if pneumonitis is confirmed. ( 5.4 ) Radiation Recall: Can occur in patients who received radiation weeks to years previously; permanently discontinue for signs of radiation recall. ( 5.5 ) Embryo-Fetal Toxicity: Can cause fetal harm. Advise patients of the potential risk to a fetus and to use effective contraception. ( 5.7 , 8.1 , 8.3 )
Contraindications
Pemetrexed for injection is contraindicated in patients with a history of severe hypersensitivity reaction to pemetrexed [see Adverse Reactions ( 6.1 )].
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling: Myelosuppression [see Warnings and Precautions ( 5.1 )] Renal failure [see Warnings and Precautions ( 5.2 )] Bullous and exfoliative skin toxicity [see Warning and Precautions ( 5.3 )] Interstitial pneumonitis [see Warnings and Precautions ( 5.4 )] Radiation recall [see Warnings and Precautions ( 5.5 )]
Drug Interactions
Ibuprofen increased risk of pemetrexed for injection toxicity in patients with mild to moderate renal impairment. Modify the ibuprofen dosage as recommended for patients with a creatinine clearance between 45 mL/min and 79 mL/min. ( 2.5 , 5.6 , 7 )
Medication Information
Warnings and Precautions
Myelosuppression: Can cause severe bone marrow suppression resulting in cytopenia and an increased risk of infection. Do not administer pemetrexed for injection when the absolute neutrophil count is less than 1500 cells/mm 3 and platelets are less than 100,000 cells/mm 3 . Initiate supplementation with oral folic acid and intramuscular vitamin B 12 to reduce the severity of hematologic and gastrointestinal toxicity of pemetrexed for injection. ( 2.4 , 5.1 ) Renal Failure: Can cause severe, and sometimes fatal, renal failure. Do not administer when creatinine clearance is less than 45 mL/min. ( 2.3 , 5.2 ) Bullous and Exfoliative Skin Toxicity: Permanently discontinue for severe and life-threatening bullous, blistering or exfoliating skin toxicity. ( 5.3 ) Interstitial Pneumonitis: Withhold for acute onset of new or progressive unexplained pulmonary symptoms. Permanently discontinue if pneumonitis is confirmed. ( 5.4 ) Radiation Recall: Can occur in patients who received radiation weeks to years previously; permanently discontinue for signs of radiation recall. ( 5.5 ) Embryo-Fetal Toxicity: Can cause fetal harm. Advise patients of the potential risk to a fetus and to use effective contraception. ( 5.7 , 8.1 , 8.3 )
Indications and Usage
Pemetrexed for injection is a folate analog metabolic inhibitor indicated: in combination with pembrolizumab and platinum chemotherapy, for the initial treatment of patients with metastatic non-squamous non-small cell lung cancer (NSCLC), with no EGFR or ALK genomic tumor aberrations. ( 1.1 ) in combination with cisplatin for the initial treatment of patients with locally advanced or metastatic, non-squamous (NSCLC). ( 1.1 ) as a single agent for the maintenance treatment of patients with locally advanced or metastatic, non-squamous NSCLC whose disease has not progressed after four cycles of platinum-based first-line chemotherapy. ( 1.1 ) as a single agent for the treatment of patients with recurrent, metastatic non-squamous, NSCLC after prior chemotherapy. ( 1.1 ) Limitations of Use: Pemetrexed for injection is not indicated for the treatment of patients with squamous cell, non-small cell lung cancer. ( 1.1 ) initial treatment, in combination with cisplatin, of patients with malignant pleural mesothelioma whose disease is unresectable or who are otherwise not candidates for curative surgery. ( 1.2 )
Dosage and Administration
The recommended dose of pemetrexed for injection administered with pembrolizumab and platinum chemotherapy in patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater is 500 mg/m 2 as an intravenous infusion over 10 minutes, administered after pembrolizumab and prior to platinum chemotherapy, on Day 1 of each 21-day cycle. ( 2.1 ) The recommended dose of pemetrexed for injection, administered as a single agent or with cisplatin, in patients with creatinine clearance of 45 mL/minute or greater is 500 mg/m 2 as an intravenous infusion over 10 minutes on Day 1 of each 21-day cycle. ( 2.1 , 2.2 ) Initiate folic acid 400 mcg to 1000 mcg orally, once daily, beginning 7 days prior to the first dose of pemetrexed for injection and continue until 21 days after the last dose of pemetrexed for injection. ( 2.4 ) Administer vitamin B 12 , 1 mg intramuscularly, 1 week prior to the first dose of pemetrexed for injection and every 3 cycles. ( 2.4 ) Administer dexamethasone 4 mg orally, twice daily the day before, the day of, and the day after pemetrexed for injection administration. ( 2.4 )
Dosage Forms and Strengths
How Supplied
Pemetrexed for injection USP, is a white-to-light yellow or green-yellow lyophilized powder supplied in single-dose vials for reconstitution for intravenous infusion.
NDC 70069-834-01: Carton containing one (1) single-dose vial of 100 mg pemetrexed.
NDC 70069-835-01: Carton containing one (1) single-dose vial of 500 mg pemetrexed.
Contraindications
Pemetrexed for injection is contraindicated in patients with a history of severe hypersensitivity reaction to pemetrexed [see Adverse Reactions ( 6.1 )].
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling: Myelosuppression [see Warnings and Precautions ( 5.1 )] Renal failure [see Warnings and Precautions ( 5.2 )] Bullous and exfoliative skin toxicity [see Warning and Precautions ( 5.3 )] Interstitial pneumonitis [see Warnings and Precautions ( 5.4 )] Radiation recall [see Warnings and Precautions ( 5.5 )]
Drug Interactions
Ibuprofen increased risk of pemetrexed for injection toxicity in patients with mild to moderate renal impairment. Modify the ibuprofen dosage as recommended for patients with a creatinine clearance between 45 mL/min and 79 mL/min. ( 2.5 , 5.6 , 7 )
Description
Pemetrexed for injection is a folate analog metabolic inhibitor indicated: in combination with pembrolizumab and platinum chemotherapy, for the initial treatment of patients with metastatic non-squamous non-small cell lung cancer (NSCLC), with no EGFR or ALK genomic tumor aberrations. ( 1.1 ) in combination with cisplatin for the initial treatment of patients with locally advanced or metastatic, non-squamous (NSCLC). ( 1.1 ) as a single agent for the maintenance treatment of patients with locally advanced or metastatic, non-squamous NSCLC whose disease has not progressed after four cycles of platinum-based first-line chemotherapy. ( 1.1 ) as a single agent for the treatment of patients with recurrent, metastatic non-squamous, NSCLC after prior chemotherapy. ( 1.1 ) Limitations of Use: Pemetrexed for injection is not indicated for the treatment of patients with squamous cell, non-small cell lung cancer. ( 1.1 ) initial treatment, in combination with cisplatin, of patients with malignant pleural mesothelioma whose disease is unresectable or who are otherwise not candidates for curative surgery. ( 1.2 )
Section 42229-5
Vitamin Supplementation
- Initiate folic acid 400 mcg to 1000 mcg orally once daily, beginning 7 days before the first dose of pemetrexed for injection and continuing until 21 days after the last dose of pemetrexed for injection [see Warnings and Precautions ( 5.1)] .
- Administer vitamin B 12,1 mg intramuscularly, 1 week prior to the first dose of pemetrexed for injection and every 3 cycles thereafter. Subsequent vitamin B 12injections may be given the same day as treatment with pemetrexed for injection [see Warnings and Precautions ( 5.1)] . Do not substitute oral vitamin B 12 for intramuscular vitamin B 12 .
Section 42230-3
|
PATIENT INFORMATION
Pemetrexed for injection (pem-e-TREX-ed for in-jec-tion) for Intravenous Use |
|
What is Pemetrexed for injection?
Pemetrexed for injection is a prescription medicine used to treat:
Pemetrexed for injection is not for use for the treatment of people with squamous cell non-small cell lung cancer.
Pemetrexed for injection has not been shown to be safe and effective in children. |
|
Do not take pemetrexed for injection i
f you have had a severe allergic reaction to any medicine that contains pemetrexed.
|
Before taking pemetrexed for injection, tell your healthcare provider about all of your medical conditions, including if you:
Females who are able to become pregnant: Your healthcare provider will check to see if you are pregnant before you start treatment with pemetrexed for injection. You should use effective birth control (contraception) during treatment with pemetrexed for injection and for 6 months after the last dose. Tell your healthcare provider right away if you become pregnant or think you are pregnant during treatment with pemetrexed for injection. Maleswith female partners who are able to become pregnant should use effective birth control (contraception) during treatment with pemetrexed for injection and for 3 months after the last dose.
|
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Tell your healthcare provider if you have kidney problems and take a medicine that contains ibuprofen. You should avoid taking ibuprofen for 2 days before, the day of, and 2 days after receiving treatment with pemetrexed for injection. |
How is pemetrexed for injection given?
-
It is very important to take folic acid and vitamin B
12 during your treatment with pemetrexed for injection to lower your risk of harmful side effects.
- Take folic acid exactly as prescribed by your healthcare provider 1 time a day, beginning 7 days (1 week) before your first dose of pemetrexed for injection and continue taking folic acid until 21 days (3 weeks) after your last dose of pemetrexed for injection.
- Your healthcare provider will give you vitamin B 12 injections during treatment with pemetrexed for injection. You will get your first vitamin B 12 injection 7 days (1 week) before your first dose of pemetrexed for injection, and then every 3 cycles.
- Your healthcare provider will prescribe a medicine called corticosteroid for you to take 2 times a day for 3 days, beginning the day before each treatment with pemetrexed for injection.
- Pemetrexed for injection is given to you by intravenous (IV) infusion into your vein. The infusion is given over 10 minutes.
- Pemetrexed for injection is usually given 1 time every 21 days (3 weeks).
|
What are the possible side effects of pemetrexed for injection?
Pemetrexed for injection can cause serious side effects, including:
|
||
|
The most common side effects of pemetrexed for injection when given alone are:
|
||
|
|
|
|
The most common side effects of pemetrexed for injection when given with cisplatin are:
|
||
|
|
|
|
The most common side effects of pemetrexed for injection when given with pembrolizumab and platinum chemotherapy are:
|
||
|
|
|
| Pemetrexed for injection may cause fertility problems in males. This may affect your ability to father a child. It is not known if these effects are reversible. Talk to your healthcare provider if this is a concern for you.
Your healthcare provider will do blood test to check for side effects during treatment with pemetrexed for injection. Your healthcare provider may change your dose of pemetrexed for injection, delay treatment, or stop treatment if you have certain side effects. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the side effects of pemetrexed for injection. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088 General information about the safe and effective use of pemetrexed for injection. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your pharmacist or healthcare provider for information about pemetrexed for injection that is written for health professionals. What are the ingredients in pemetrexed for injection? Active ingredient: pemetrexed Inactive ingredients: mannitol, hydrochloric acid and/or sodium hydroxide may have been added to adjust pH. Manufactured for: Somerset Therapeutics, LLC. Somerset, NJ 08873 Made in India This Patient Information has been approved by the U.S. Food and Drug Administration. Revised: March 2025 |
Section 44425-7
Storage and Handling
Store at 25°C (77°F); excursions permitted to 15°C-30°C (59°F-86°F) [see USP Controlled Room Temperature].
Pemetrexed for injection USP is a hazardous drug. Follow applicable special handling and disposal procedures. 1
Section 51945-4
Label – Pemetrexed for Injection 100 mg single-dose vial
NDC 70069-834-01
Single-Dose Vial
Pemetrexed for Injection, USP
100 mg/vial
For intravenous use only.
Rx only
Manufactured for:
Somerset Therapeutics LLC
10 Overdosage
No drugs are approved for the treatment of pemetrexed for injection overdose. Based on animal studies, administration of leucovorin may mitigate the toxicities of pemetrexed for injection overdosage. It is not known whether pemetrexed is dialyzable.
15 References
- “OSHA Hazardous Drugs.” OSHA. [https://www.osha.gov/hazardous-drugs]
11 Description
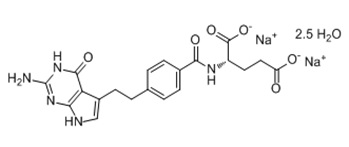
Pemetrexed for injection USP is a folate analog metabolic inhibitor. The drug substance, pemetrexed disodium hemi- pentahydrate, has the chemical name L-glutamic acid, N-[4-[2-(2-amino-4,7-dihydro-4- oxo-1H-pyrrolo[2,3-d] pyrimidin-5yl) ethyl] benzoyl]-, Disodium, hemi-pentahydrate with a molecular formula of C
20H
19N
5Na
2O
62.5 H
2O and a molecular weight of 516.37. The structural formula is as follows:
Pemetrexed for injection USP is a sterile white-to-light yellow or green-yellow lyophilized powder in single-dose vials to be reconstituted for intravenous infusion. Each 100-mg vial of pemetrexed for injection USP contains 100 mg pemetrexed (equivalent to 110.3 mg pemetrexed disodium hemi- pentahydrate) and 106 mg mannitol. Each 500 mg vial of pemetrexed for injection USP contains 500 mg pemetrexed (equivalent to 551.4 mg pemetrexed disodium hemi- pentahydrate) and 500 mg mannitol. Hydrochloric acid and/or sodium hydroxide may have been added to adjust pH.
1.2 Mesothelioma
Pemetrexed for injection is indicated, in combination with cisplatin, for the initial treatment of patients with malignant pleural mesothelioma whose disease is unresectable or who are otherwise not candidates for curative surgery.
14.2 Mesothelioma
The efficacy of pemetrexed for injection was evaluated in Study JMCH (NCT00005636), a multicenter, randomized (1:1), single-blind study conducted in patients with MPM who had received no prior chemotherapy. Patients were randomized (n=456) to receive pemetrexed 500 mg/m
2 intravenously over 10 minutes followed 30 minutes later by cisplatin 75 mg/m
2 intravenously over two hours on Day 1 of each 21-day cycle or to receive cisplatin 75 mg/m
2 intravenously over 2 hours on Day 1 of each 21-day cycle; treatment continued until disease progression or intolerable toxicity. The study was modified after randomization and treatment of 117 patients to require that all patients receive folic acid 350 mcg to 1000 mcg daily beginning 1 to 3 weeks prior to the first dose of pemetrexed for injection and continuing until 1 to 3 weeks after the last dose, vitamin B
12 1000 mcg intramuscularly 1 to 3 weeks prior to first dose of pemetrexed for injection and every 9 weeks thereafter, and dexamethasone 4 mg orally, twice daily, for 3 days starting the day prior to each pemetrexed dose. Randomization was stratified by multiple baseline variables including KPS, histologic subtype (epithelial, mixed, sarcomatoid, other), and gender. The major efficacy outcome measure was overall survival and additional efficacy outcome measures were time to disease progression, overall response rate, and response duration.
A total of 448 patients received at least one dose of protocol-specified therapy; 226 patients were randomized to and received at least one dose of pemetrexed plus cisplatin, and 222 patients were randomized to and received cisplatin. Among the 226 patients who received cisplatin with pemetrexed for injection, 74% received full supplementation with folic acid and vitamin B
12 during study therapy, 14% were never supplemented, and 12% were partially supplemented. Across the study population, the median age was 61 years (range: 20 to 86 years); 81% were male; 92% were White, 5% were Hispanic or Latino, 3.1% were Asian, and <1% were other ethnicities; and 54% had a baseline KPS score of 90-100% and 46% had a KPS score of 70-80%. With regard to tumor characteristics, 46% had Stage IV disease, 31% Stage III, 15% Stage II, and 7% Stage I disease at baseline; the histologic subtype of mesothelioma was epithelial in 68% of patients, mixed in 16%, sarcomatoid in 10% and other histologic subtypes in 6%. The baseline demographics and tumor characteristics of the subgroup of fully supplemented patients was similar to the overall study population.
The efficacy results from Study JMCH are summarized in
Table 18and
Figure 9.
|
Efficacy Parameter
|
All Randomized and Treated Patients
(N=448) |
Fully Supplemented
Patients (N=331) |
||
|
Pemetrexed for injection /Cisplatin
(N=226) |
Cisplatin
(N=222) |
Pemetrexed for injection /Cisplatin
(N=168) |
Cisplatin
(N=163) |
|
| Median overall survival (months)
|
12.1
|
9.3
|
13.3
|
10.0
|
| (95% CI)
|
(10.0-14.4)
|
(7.8-10.7)
|
(11.4-14.9)
|
(8.4-11.9)
|
| Hazard ratio
a
|
0.77
|
0.75
|
||
| Log rank p-value
|
0.020
|
NA
b
|
a Hazard ratios are not adjusted for stratification variables.
bNot a pre-specified analysis.
Figure 9: Kaplan-Meier Curves for Overall Survival in Study JMCH
Based upon prospectively defined criteria (modified Southwest Oncology Group methodology) the objective tumor response rate for pemetrexed for injection plus cisplatin was greater than the objective tumor response rate for cisplatin alone. There was also improvement in lung function (forced vital capacity) in the pemetrexed for injection plus cisplatin arm compared to the control arm.
5.2 Renal Failure
Pemetrexed for injection can cause severe, and sometimes fatal, renal toxicity. The incidences of renal failure in clinical studies in which patients received pemetrexed with cisplatin were: 2.1% in Study JMDB and 2.2% in Study JMCH. The incidence of renal failure in clinical studies in which patients received pemetrexed as a single agent ranged from 0.4% to 0.6% (Studies JMEN, PARAMOUNT, and JMEI [
see Adverse Reactions (6.1)].
Determine creatinine clearance before each dose and periodically monitor renal function during treatment with pemetrexed for injection.
Withhold pemetrexed for injection in patients with a creatinine clearance of less than 45 mL/minute [
see Dosage and Administration (
2.3
)].
8.4 Pediatric Use
The safety and effectiveness of pemetrexed for injection in pediatric patients have not been established.
The safety and pharmacokinetics of pemetrexed for injection were evaluated in two clinical studies conducted in pediatric patients with recurrent solid tumors (NCT00070473 N=32 and NCT00520936 N=72). Patients in both studies received concomitant vitamin B12 and folic acid supplementation and dexamethasone.
No tumor responses were observed. Adverse reactions observed in pediatric patients were similar to those observed in adults.
Single-dose pharmacokinetics of pemetrexed were evaluated in 22 patients age 4 to 18 years enrolled in NCT00070473 were within range of values in adults.
8.5 Geriatric Use
Of the 3,946 patients enrolled in clinical studies of pemetrexed for injection, 34% were 65 and over and 4% were 75 and over. No overall differences in effectiveness were observed between these patients and younger patients. The incidences of Grade 3-4 anemia, fatigue, thrombocytopenia, hypertension, and neutropenia were higher in patients 65 years of age and older as compared to younger patients: in at least one of five randomized clinical trials. [
see Adverse Reactions (
6.1
) and Clinical Studies (
14.1
,
14.2
)].
4 Contraindications
Pemetrexed for injection is contraindicated in patients with a history of severe hypersensitivity reaction to pemetrexed [see Adverse Reactions ( 6.1)].
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling:
7 Drug Interactions
2.3 Renal Impairment
- Pemetrexed for injection dosing recommendations are provided for patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater [ see Dosage and Administration ( 2.1 , 2.2)]. There is no recommended dose for patients whose creatinine clearance is less than 45 mL/min [ see Use in Specific Populations ( 8.6 )].
5.5 Radiation Recall
Radiation recall can occur with pemetrexed for injection in patients who have received radiation weeks to years previously. Monitor patients for inflammation or blistering in areas of previous radiation treatment. Permanently discontinue pemetrexed for injection for signs of radiation recall.
12.2 Pharmacodynamics
Pemetrexed inhibited the in vitro growth of mesothelioma cell lines (MSTO-211H, NCI-H2052) and showed synergistic effects when combined with cisplatin.
Based on population pharmacodynamic analyses, the depth of the absolute neutrophil counts (ANC) nadir correlates with the systemic exposure to pemetrexed and supplementation with folic acid and vitamin B 12. There is no cumulative effect of pemetrexed exposure on ANC nadir over multiple treatment cycles.
1 Indications and Usage
Pemetrexed for injection is a folate analog metabolic inhibitor indicated:
- in combination with pembrolizumab and platinum chemotherapy, for the initial treatment of patients with metastatic non-squamous non-small cell lung cancer (NSCLC), with no EGFR or ALK genomic tumor aberrations. ( 1.1)
- in combination with cisplatin for the initial treatment of patients with locally advanced or metastatic, non-squamous (NSCLC). ( 1.1)
- as a single agent for the maintenance treatment of patients with locally advanced or metastatic, non-squamous NSCLC whose disease has not progressed after four cycles of platinum-based first-line chemotherapy. ( 1.1)
- as a single agent for the treatment of patients with recurrent, metastatic non-squamous, NSCLC after prior chemotherapy. ( 1.1)
Limitations of Use:Pemetrexed for injection is not indicated for the treatment of patients with squamous cell, non-small cell lung cancer. (
1.1)
- initial treatment, in combination with cisplatin, of patients with malignant pleural mesothelioma whose disease is unresectable or who are otherwise not candidates for curative surgery. ( 1.2)
12.1 Mechanism of Action
Pemetrexed for injection is a folate analog metabolic inhibitor that disrupts folate-dependent metabolic processes essential for cell replication. In vitro studies show that pemetrexed inhibits thymidylate synthase (TS), dihydrofolate reductase, and glycinamide ribonucleotide formyltransferase (GARFT), which are folate- dependent enzymes involved in the de novo biosynthesis of thymidine and purine nucleotides. Pemetrexed is taken into cells by membrane carriers such as the reduced folate carrier and membrane folate binding protein transport systems. Once in the cell, pemetrexed is converted to polyglutamate forms by the enzyme folylpolyglutamate synthetase. The polyglutamate forms are retained in cells and are inhibitors of TS and GARFT.
5.7 Embryo Fetal Toxicity
Based on findings from animal studies and its mechanism of action, pemetrexed can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, intravenous administration of pemetrexed to pregnant mice during the period of organogenesis was teratogenic, resulting in developmental delays and increased malformations at doses lower than the recommended human dose of 500 mg/m
2. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with pemetrexed and for 6 months after the last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with pemetrexed and for 3 months after the last dose
[see Use in Specific Populations (
8.1, 8.3
) and Clinical Pharmacology (
12.1
)].
5 Warnings and Precautions
- Myelosuppression: Can cause severe bone marrow suppression resulting in cytopenia and an increased risk of infection. Do not administer pemetrexed for injection when the absolute neutrophil count is less than 1500 cells/mm 3and platelets are less than 100,000 cells/mm 3. Initiate supplementation with oral folic acid and intramuscular vitamin B 12to reduce the severity of hematologic and gastrointestinal toxicity of pemetrexed for injection. ( 2.4, 5.1)
- Renal Failure: Can cause severe, and sometimes fatal, renal failure. Do not administer when creatinine clearance is less than 45 mL/min. ( 2.3, 5.2)
- Bullous and Exfoliative Skin Toxicity: Permanently discontinue for severe and life-threatening bullous, blistering or exfoliating skin toxicity. ( 5.3)
- Interstitial Pneumonitis: Withhold for acute onset of new or progressive unexplained pulmonary symptoms. Permanently discontinue if pneumonitis is confirmed. ( 5.4)
- Radiation Recall: Can occur in patients who received radiation weeks to years previously; permanently discontinue for signs of radiation recall. ( 5.5)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise patients of the potential risk to a fetus and to use effective contraception. ( 5.7, 8.1, 8.3)
2 Dosage and Administration
- The recommended dose of pemetrexed for injection administered with pembrolizumab and platinum chemotherapy in patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater is 500 mg/m 2as an intravenous infusion over 10 minutes, administered after pembrolizumab and prior to platinum chemotherapy, on Day 1 of each 21-day cycle. ( 2.1)
- The recommended dose of pemetrexed for injection, administered as a single agent or with cisplatin, in patients with creatinine clearance of 45 mL/minute or greater is 500 mg/m 2as an intravenous infusion over 10 minutes on Day 1 of each 21-day cycle. ( 2.1, 2.2)
- Initiate folic acid 400 mcg to 1000 mcg orally, once daily, beginning 7 days prior to the first dose of pemetrexed for injection and continue until 21 days after the last dose of pemetrexed for injection. ( 2.4)
- Administer vitamin B 12, 1 mg intramuscularly, 1 week prior to the first dose of pemetrexed for injection and every 3 cycles. ( 2.4)
- Administer dexamethasone 4 mg orally, twice daily the day before, the day of, and the day after pemetrexed for injection administration. ( 2.4)
3 Dosage Forms and Strengths
For injection: 100 mg or 500 mg pemetrexed for injection as a white to light-yellow or green-yellow lyophilized powder in single-dose vials for reconstitution.
5.4 Interstitial Pneumonitis
Serious interstitial pneumonitis, including fatal cases, can occur with pemetrexed for injection treatment. Withhold pemetrexed for injection for acute onset of new or progressive unexplained pulmonary symptoms such as dyspnea, cough, or fever pending diagnostic evaluation. If pneumonitis is confirmed, permanently discontinue pemetrexed.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of pemetrexed for injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System— immune-mediated hemolytic anemia
Gastrointestinal— colitis, pancreatitis
General Disorders and Administration Site Conditions— edema
Injury, poisoning, and procedural complications— radiation recall
Respiratory— interstitial pneumonitis
Skin— Serious and fatal bullous skin conditions, Stevens-Johnson syndrome, and toxic epidermal necrolysis
8 Use in Specific Populations
Lactation: Advise not to breastfeed. ( 8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reactions rates cannot be directly compared to rates in other clinical trials and may not reflect the rates observed in clinical practice.
In clinical trials, the most common adverse reactions (incidence ≥20%) of pemetrexed for injection, when administered as a single agent, are fatigue, nausea, and anorexia. The most common adverse reactions (incidence ≥20%) of pemetrexed for injection, when administered in combination with cisplatin are vomiting, neutropenia, anemia, stomatitis/pharyngitis, thrombocytopenia, and constipation. The most common adverse reactions (incidence ≥20%) of pemetrexed for injection, when administered in combination with pembrolizumab and platinum chemotherapy, are fatigue/asthenia, nausea, constipation, diarrhea, decreased appetite, rash, vomiting, cough, dyspnea, and pyrexia.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling ( Patient Information).
2.7 Preparation for Administration
- Pemetrexed for injection is a hazardous drug. Follow applicable special handling and disposal procedures. 1
- Calculate the dose of pemetrexed for injection and determine the number of vials needed.
- Reconstitute pemetrexed for injection to achieve a concentration of 25 mg/mL as follows:
- Reconstitute each 100-mg vial with 4.2 mL of 0.9% Sodium Chloride Injection, USP (preservative-free)
- Reconstitute each 500-mg vial with 20 mL of 0.9% Sodium Chloride Injection, USP (preservative-free)
- Do not use calcium-containing solutions for reconstitution.
- Gently swirl each vial until the powder is completely dissolved. The resulting solution is clear and ranges in color from colorless to yellow or green-yellow. FURTHER DILUTION IS REQUIRED prior to administration.
- Store reconstituted, preservative-free product under refrigerated conditions [2-8°C (36-46°F)] for no longer than 24 hours from the time of reconstitution. Discard vial after 24 hours.
- Inspect reconstituted product visually for particulate matter and discoloration prior to further dilution. If particulate matter is observed, discard vial.
- Withdraw the calculated dose of pemetrexed for injection from the vial(s) and discard vial with any unused portion.
- Further dilute pemetrexed for injection with 0.9% Sodium Chloride Injection (preservative-free) to achieve a total volume of 100 mL for intravenous infusion.
- Store diluted, reconstituted product under refrigerated conditions [2-8°C (36-46°F)] for no more than 24 hours from the time of reconstitution. Discard after 24 hours.
8.6 Patients With Renal Impairment
Pemetrexed for injection is primarily excreted by the kidneys. Decreased renal function results in reduced clearance and greater exposure (AUC) to pemetrexed for injection compared with patients with normal renal function [
Warnings and Precautions (
5.2
,
5.6
) and Clinical Pharmacology (
12.3
)]. No dose is recommended for patients with creatinine clearance less than 45 mL/min [
see Dosage and Administration (
2.3
)].
2.2 Recommended Dosage for Mesothelioma
- The recommended dose of pemetrexed for injection when administered with cisplatin in patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater is 500 mg/m 2as an intravenous infusion over 10 minutes on Day 1 of each 21-day cycle until disease progression or unacceptable toxicity.
5.3 Bullous and Exfoliative Skin Toxicity
Serious and sometimes fatal, bullous, blistering and exfoliative skin toxicity, including cases suggestive of Stevens-Johnson Syndrome/Toxic epidermal necrolysis can occur with pemetrexed for injection. Permanently discontinue pemetrexed for injection for severe and life-threatening bullous, blistering or exfoliating skin toxicity.
2.1 Recommended Dosage for Non Squamous Nsclc
- The recommended dose of pemetrexed for injection when administered with pembrolizumab and platinum chemotherapy for the initial treatment of metastatic non-squamous NSCLC in patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater is 500 mg/m 2as an intravenous infusion over 10 minutes administered after pembrolizumab and prior to carboplatin or cisplatin on Day 1 of each 21-day cycle for 4 cycles. Following completion of platinum-based therapy, treatment with pemetrexed for injection with or without pembrolizumab is administered until disease progression or unacceptable toxicity. Please refer to the full prescribing information for pembrolizumab and for carboplatin or cisplatin.
- The recommended dose of pemetrexed for injection when administered with cisplatin for initial treatment of locally advanced or metastatic non-squamous NSCLC in patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater is 500 mg/m 2as an intravenous infusion over 10 minutes administered prior to cisplatin on Day 1 of each 21-day cycle for up to six cycles in the absence of disease progression or unacceptable toxicity.
- The recommended dose of pemetrexed for injection for maintenance treatment of non-squamous NSCLC in patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater is 500 mg/m 2as an intravenous infusion over 10 minutes on Day 1 of each 21-day cycle until disease progression or unacceptable toxicity after four cycles of platinum-based first-line chemotherapy.
- The recommended dose of pemetrexed for injection for treatment of recurrent non-squamous NSCLC in patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater is 500 mg/m 2as an intravenous infusion over 10 minutes on Day 1 of each 21-day cycle until disease progression or unacceptable toxicity.
2.6 Dosage Modifications for Adverse Reactions
Obtain complete blood count on Days 1, 8, and 15 of each cycle. Assess creatinine clearance prior to each cycle. Do not administer pemetrexed for injection if the creatinine clearance is less than 45 mL/min.
Delay initiation of the next cycle of pemetrexed for injection until:
- recovery of non-hematologic toxicity to Grade 0-2,
- absolute neutrophil count (ANC) is 1500 cells/mm 3or higher, and
- platelet count is 100,000 cells/mm 3or higher.
Upon recovery, modify the dosage of pemetrexed for injection in the next cycle as specified in Table 1.
For dosing modifications for cisplatin, carboplatin, or pembrolizumab, refer to their prescribing information .
|
Toxicity in Most Recent Treatment Cycle
|
Pemetrexed for injection Dose Modification for Next Cycle
|
|
Myelosuppressive toxicity
[see Warnings and Precautions (
5.1)]
|
|
| ANC less than 500/mm
3
and
platelets greater than or equal to 50,000/mm
3
OR Platelet count less than 50,000/mm 3without bleeding. |
75% of previous dose
|
| Platelet count less than 50,000/mm
3with bleeding
|
50% of previous dose
|
| Recurrent Grade 3 or 4 myelosuppression after 2 dose reductions
|
Discontinue
|
|
Non-hematologic toxicity
|
|
| Any Grade 3 or 4 toxicities EXCEPT mucositis or neurologic toxicity
OR Diarrhea requiring hospitalization |
75% of previous dose
|
| Grade 3 or 4 mucositis
|
50% of previous dose
|
| Renal toxicity
[see Warnings and Precautions (
5.2)]
|
Withhold until creatinine clearance is 45 mL/min or greater
|
| Grade 3 or 4 neurologic toxicity
|
Permanently discontinue
|
| Recurrent Grade 3 or 4 non-hematologic toxicity after 2 dose reductions
|
Permanently discontinue
|
| Severe and life-threatening Skin Toxicity
[see Warnings and Precautions (
5.3)]
|
Permanently discontinue
|
| Interstitial Pneumonitis
[see Warnings and Precautions (
5.4)]
|
Permanently discontinue
|
8.3 Females and Males of Reproductive Potential
Based on animal data pemetrexed for injection can cause malformations and developmental delays when administered to a pregnant woman [see Use in Specific Populations (8.1)].
Pregnancy Testing
Verify pregnancy status of females of reproductive potential prior to initiating pemetrexed for injection [see Use in Specific Populations (8.1)].
1.1 Non Squamous Non Small Cell Lung Cancer (nsclc)
Pemetrexed for injection is indicated:
- in combination with pembrolizumab and platinum chemotherapy, for the initial treatment of patients with metastatic non-squamous non-small cell lung cancer (NSCLC), with no EGFR or ALK genomic tumor aberrations.
- in combination with cisplatin for the initial treatment of patients with locally advanced or metastatic, non-squamous, NSCLC.
- as a single agent for the maintenance treatment of patients with locally advanced or metastatic, non-squamous NSCLC whose disease has not progressed after four cycles of platinum-based first-line chemotherapy.
- as a single agent for the treatment of patients with recurrent, metastatic non-squamous, NSCLC after prior chemotherapy.
Limitations of Use:Pemetrexed for injection is not indicated for the treatment of patients with squamous cell, non-small cell lung cancer [see Clinical Studies 14.1 ].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity studies have been conducted with pemetrexed. Pemetrexed was clastogenic in an in vivo micronucleus assay in mouse bone marrow but was not mutagenic in multiple in vitro tests (Ames assay, Chinese Hamster Ovary cell assay).
Pemetrexed administered intraperitoneally at doses of ≥0.1 mg/kg/day to male mice (approximately 0.0006 times the recommended human dose based on BSA) resulted in reduced fertility, hypospermia, and testicular atrophy.
5.6 Increased Risk of Toxicity With Ibuprofen in Patients With Renal Impairment
Exposure to pemetrexed is increased in patients with mild to moderate renal impairment who take concomitant ibuprofen, increasing the risks of adverse reactions of pemetrexed. In patients with creatinine clearances between 45 mL/min and 79 mL/min, avoid administration of ibuprofen for 2 days before, the day of, and 2 days following administration of pemetrexed. If concomitant ibuprofen use cannot be avoided, monitor patients more frequently for pemetrexed adverse reactions, including myelosuppression, renal, and gastrointestinal toxicity
[see Dosage and Administration (
2.5
),
Drug
Interactions (
7
), and Clinical Pharmacology (
12.3
)].
5.1 Myelosuppression and Increased Risk of Myelosuppression Without Vitamin Supplementation
Pemetrexed for injection can cause severe myelosuppression resulting in a requirement for transfusions and which may lead to neutropenic infection. The risk of myelosuppression is increased in patients who do not receive vitamin supplementation. In Study JMCH, incidences of Grade 3-4 neutropenia (38% versus 23%), thrombocytopenia (9% versus 5%), febrile neutropenia (9% versus 0.6%), and neutropenic infection (6% versus 0) were higher in patients who received pemetrexed plus cisplatin without vitamin supplementation as compared to patients who were fully supplemented with folic acid and vitamin B
12 prior to and throughout pemetrexed plus cisplatin treatment.
Initiate supplementation with oral folic acid and intramuscular vitamin B
12 prior to the first dose of pemetrexed; continue vitamin supplementation during treatment and for 21 days after the last dose of pemetrexed to reduce the severity of hematologic and gastrointestinal toxicity of pemetrexed for injection [s
ee Dosage and Administration (
2.4
)]. Obtain a complete blood count at the beginning of each cycle. Do not administer pemetrexed for injection until the ANC is at least 1500 cells/mm
3 and platelet count is at least 100,000 cells/mm
3. Permanently reduce pemetrexed for injection in patients with an ANC of less than 500 cells/mm
3 or platelet count of less than 50,000 cells/mm
3 in previous cycles [
see Dosage and Administration (
2.6
)].
In Studies JMDB and JMCH, among patients who received vitamin supplementation, incidence of Grade 3-4 neutropenia was 15% and 23%, the incidence of Grade 3-4 anemia was 6% and 4%, and incidence of Grade 3-4 thrombocytopenia was 4% and 5%, respectively. In Study JMCH, 18% of patients in the pemetrexed for injection arm required red blood cell transfusions compared to 7% of patients in the cisplatin arm [
see Adverse Reactions (
6.1
)]. In Studies JMEN, PARAMOUNT, and JMEI, where all patients received vitamin supplementation, incidence of Grade 3-4 neutropenia ranged from 3% to 5%, and incidence of Grade 3-4 anemia ranged from 3% to 5%.
2.5 Dosage Modification of Ibuprofen in Patients With Mild to Moderate Renal Impairment Receiving Pemetrexed for Injection
In patients with creatinine clearances between 45 mL/min and 79 mL/min, modify administration of ibuprofen as follows [see Warnings and Precautions ( 5.6), Drug Interactions ( 7) and Clinical Pharmacology ( 12.3)] :
- Avoid administration of ibuprofen for 2 days before, the day of, and 2 days following administration of pemetrexed for injection.
- Monitor patients more frequently for myelosuppression, renal, and gastrointestinal toxicity, if concomitant administration of ibuprofen cannot be avoided.
Structured Label Content
Dosage Forms and Strengths (34069-5)
How Supplied
Pemetrexed for injection USP, is a white-to-light yellow or green-yellow lyophilized powder supplied in single-dose vials for reconstitution for intravenous infusion.
NDC 70069-834-01: Carton containing one (1) single-dose vial of 100 mg pemetrexed.
NDC 70069-835-01: Carton containing one (1) single-dose vial of 500 mg pemetrexed.
Section 42229-5 (42229-5)
Vitamin Supplementation
- Initiate folic acid 400 mcg to 1000 mcg orally once daily, beginning 7 days before the first dose of pemetrexed for injection and continuing until 21 days after the last dose of pemetrexed for injection [see Warnings and Precautions ( 5.1)] .
- Administer vitamin B 12,1 mg intramuscularly, 1 week prior to the first dose of pemetrexed for injection and every 3 cycles thereafter. Subsequent vitamin B 12injections may be given the same day as treatment with pemetrexed for injection [see Warnings and Precautions ( 5.1)] . Do not substitute oral vitamin B 12 for intramuscular vitamin B 12 .
Section 42230-3 (42230-3)
|
PATIENT INFORMATION
Pemetrexed for injection (pem-e-TREX-ed for in-jec-tion) for Intravenous Use |
|
What is Pemetrexed for injection?
Pemetrexed for injection is a prescription medicine used to treat:
Pemetrexed for injection is not for use for the treatment of people with squamous cell non-small cell lung cancer.
Pemetrexed for injection has not been shown to be safe and effective in children. |
|
Do not take pemetrexed for injection i
f you have had a severe allergic reaction to any medicine that contains pemetrexed.
|
Before taking pemetrexed for injection, tell your healthcare provider about all of your medical conditions, including if you:
Females who are able to become pregnant: Your healthcare provider will check to see if you are pregnant before you start treatment with pemetrexed for injection. You should use effective birth control (contraception) during treatment with pemetrexed for injection and for 6 months after the last dose. Tell your healthcare provider right away if you become pregnant or think you are pregnant during treatment with pemetrexed for injection. Maleswith female partners who are able to become pregnant should use effective birth control (contraception) during treatment with pemetrexed for injection and for 3 months after the last dose.
|
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Tell your healthcare provider if you have kidney problems and take a medicine that contains ibuprofen. You should avoid taking ibuprofen for 2 days before, the day of, and 2 days after receiving treatment with pemetrexed for injection. |
How is pemetrexed for injection given?
-
It is very important to take folic acid and vitamin B
12 during your treatment with pemetrexed for injection to lower your risk of harmful side effects.
- Take folic acid exactly as prescribed by your healthcare provider 1 time a day, beginning 7 days (1 week) before your first dose of pemetrexed for injection and continue taking folic acid until 21 days (3 weeks) after your last dose of pemetrexed for injection.
- Your healthcare provider will give you vitamin B 12 injections during treatment with pemetrexed for injection. You will get your first vitamin B 12 injection 7 days (1 week) before your first dose of pemetrexed for injection, and then every 3 cycles.
- Your healthcare provider will prescribe a medicine called corticosteroid for you to take 2 times a day for 3 days, beginning the day before each treatment with pemetrexed for injection.
- Pemetrexed for injection is given to you by intravenous (IV) infusion into your vein. The infusion is given over 10 minutes.
- Pemetrexed for injection is usually given 1 time every 21 days (3 weeks).
|
What are the possible side effects of pemetrexed for injection?
Pemetrexed for injection can cause serious side effects, including:
|
||
|
The most common side effects of pemetrexed for injection when given alone are:
|
||
|
|
|
|
The most common side effects of pemetrexed for injection when given with cisplatin are:
|
||
|
|
|
|
The most common side effects of pemetrexed for injection when given with pembrolizumab and platinum chemotherapy are:
|
||
|
|
|
| Pemetrexed for injection may cause fertility problems in males. This may affect your ability to father a child. It is not known if these effects are reversible. Talk to your healthcare provider if this is a concern for you.
Your healthcare provider will do blood test to check for side effects during treatment with pemetrexed for injection. Your healthcare provider may change your dose of pemetrexed for injection, delay treatment, or stop treatment if you have certain side effects. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the side effects of pemetrexed for injection. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088 General information about the safe and effective use of pemetrexed for injection. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your pharmacist or healthcare provider for information about pemetrexed for injection that is written for health professionals. What are the ingredients in pemetrexed for injection? Active ingredient: pemetrexed Inactive ingredients: mannitol, hydrochloric acid and/or sodium hydroxide may have been added to adjust pH. Manufactured for: Somerset Therapeutics, LLC. Somerset, NJ 08873 Made in India This Patient Information has been approved by the U.S. Food and Drug Administration. Revised: March 2025 |
Section 44425-7 (44425-7)
Storage and Handling
Store at 25°C (77°F); excursions permitted to 15°C-30°C (59°F-86°F) [see USP Controlled Room Temperature].
Pemetrexed for injection USP is a hazardous drug. Follow applicable special handling and disposal procedures. 1
Section 51945-4 (51945-4)
Label – Pemetrexed for Injection 100 mg single-dose vial
NDC 70069-834-01
Single-Dose Vial
Pemetrexed for Injection, USP
100 mg/vial
For intravenous use only.
Rx only
Manufactured for:
Somerset Therapeutics LLC
10 Overdosage (10 OVERDOSAGE)
No drugs are approved for the treatment of pemetrexed for injection overdose. Based on animal studies, administration of leucovorin may mitigate the toxicities of pemetrexed for injection overdosage. It is not known whether pemetrexed is dialyzable.
15 References (15 REFERENCES)
- “OSHA Hazardous Drugs.” OSHA. [https://www.osha.gov/hazardous-drugs]
11 Description (11 DESCRIPTION)
Pemetrexed for injection USP is a folate analog metabolic inhibitor. The drug substance, pemetrexed disodium hemi- pentahydrate, has the chemical name L-glutamic acid, N-[4-[2-(2-amino-4,7-dihydro-4- oxo-1H-pyrrolo[2,3-d] pyrimidin-5yl) ethyl] benzoyl]-, Disodium, hemi-pentahydrate with a molecular formula of C
20H
19N
5Na
2O
62.5 H
2O and a molecular weight of 516.37. The structural formula is as follows:
Pemetrexed for injection USP is a sterile white-to-light yellow or green-yellow lyophilized powder in single-dose vials to be reconstituted for intravenous infusion. Each 100-mg vial of pemetrexed for injection USP contains 100 mg pemetrexed (equivalent to 110.3 mg pemetrexed disodium hemi- pentahydrate) and 106 mg mannitol. Each 500 mg vial of pemetrexed for injection USP contains 500 mg pemetrexed (equivalent to 551.4 mg pemetrexed disodium hemi- pentahydrate) and 500 mg mannitol. Hydrochloric acid and/or sodium hydroxide may have been added to adjust pH.
1.2 Mesothelioma
Pemetrexed for injection is indicated, in combination with cisplatin, for the initial treatment of patients with malignant pleural mesothelioma whose disease is unresectable or who are otherwise not candidates for curative surgery.
14.2 Mesothelioma
The efficacy of pemetrexed for injection was evaluated in Study JMCH (NCT00005636), a multicenter, randomized (1:1), single-blind study conducted in patients with MPM who had received no prior chemotherapy. Patients were randomized (n=456) to receive pemetrexed 500 mg/m
2 intravenously over 10 minutes followed 30 minutes later by cisplatin 75 mg/m
2 intravenously over two hours on Day 1 of each 21-day cycle or to receive cisplatin 75 mg/m
2 intravenously over 2 hours on Day 1 of each 21-day cycle; treatment continued until disease progression or intolerable toxicity. The study was modified after randomization and treatment of 117 patients to require that all patients receive folic acid 350 mcg to 1000 mcg daily beginning 1 to 3 weeks prior to the first dose of pemetrexed for injection and continuing until 1 to 3 weeks after the last dose, vitamin B
12 1000 mcg intramuscularly 1 to 3 weeks prior to first dose of pemetrexed for injection and every 9 weeks thereafter, and dexamethasone 4 mg orally, twice daily, for 3 days starting the day prior to each pemetrexed dose. Randomization was stratified by multiple baseline variables including KPS, histologic subtype (epithelial, mixed, sarcomatoid, other), and gender. The major efficacy outcome measure was overall survival and additional efficacy outcome measures were time to disease progression, overall response rate, and response duration.
A total of 448 patients received at least one dose of protocol-specified therapy; 226 patients were randomized to and received at least one dose of pemetrexed plus cisplatin, and 222 patients were randomized to and received cisplatin. Among the 226 patients who received cisplatin with pemetrexed for injection, 74% received full supplementation with folic acid and vitamin B
12 during study therapy, 14% were never supplemented, and 12% were partially supplemented. Across the study population, the median age was 61 years (range: 20 to 86 years); 81% were male; 92% were White, 5% were Hispanic or Latino, 3.1% were Asian, and <1% were other ethnicities; and 54% had a baseline KPS score of 90-100% and 46% had a KPS score of 70-80%. With regard to tumor characteristics, 46% had Stage IV disease, 31% Stage III, 15% Stage II, and 7% Stage I disease at baseline; the histologic subtype of mesothelioma was epithelial in 68% of patients, mixed in 16%, sarcomatoid in 10% and other histologic subtypes in 6%. The baseline demographics and tumor characteristics of the subgroup of fully supplemented patients was similar to the overall study population.
The efficacy results from Study JMCH are summarized in
Table 18and
Figure 9.
|
Efficacy Parameter
|
All Randomized and Treated Patients
(N=448) |
Fully Supplemented
Patients (N=331) |
||
|
Pemetrexed for injection /Cisplatin
(N=226) |
Cisplatin
(N=222) |
Pemetrexed for injection /Cisplatin
(N=168) |
Cisplatin
(N=163) |
|
| Median overall survival (months)
|
12.1
|
9.3
|
13.3
|
10.0
|
| (95% CI)
|
(10.0-14.4)
|
(7.8-10.7)
|
(11.4-14.9)
|
(8.4-11.9)
|
| Hazard ratio
a
|
0.77
|
0.75
|
||
| Log rank p-value
|
0.020
|
NA
b
|
a Hazard ratios are not adjusted for stratification variables.
bNot a pre-specified analysis.
Figure 9: Kaplan-Meier Curves for Overall Survival in Study JMCH
Based upon prospectively defined criteria (modified Southwest Oncology Group methodology) the objective tumor response rate for pemetrexed for injection plus cisplatin was greater than the objective tumor response rate for cisplatin alone. There was also improvement in lung function (forced vital capacity) in the pemetrexed for injection plus cisplatin arm compared to the control arm.
5.2 Renal Failure
Pemetrexed for injection can cause severe, and sometimes fatal, renal toxicity. The incidences of renal failure in clinical studies in which patients received pemetrexed with cisplatin were: 2.1% in Study JMDB and 2.2% in Study JMCH. The incidence of renal failure in clinical studies in which patients received pemetrexed as a single agent ranged from 0.4% to 0.6% (Studies JMEN, PARAMOUNT, and JMEI [
see Adverse Reactions (6.1)].
Determine creatinine clearance before each dose and periodically monitor renal function during treatment with pemetrexed for injection.
Withhold pemetrexed for injection in patients with a creatinine clearance of less than 45 mL/minute [
see Dosage and Administration (
2.3
)].
8.4 Pediatric Use
The safety and effectiveness of pemetrexed for injection in pediatric patients have not been established.
The safety and pharmacokinetics of pemetrexed for injection were evaluated in two clinical studies conducted in pediatric patients with recurrent solid tumors (NCT00070473 N=32 and NCT00520936 N=72). Patients in both studies received concomitant vitamin B12 and folic acid supplementation and dexamethasone.
No tumor responses were observed. Adverse reactions observed in pediatric patients were similar to those observed in adults.
Single-dose pharmacokinetics of pemetrexed were evaluated in 22 patients age 4 to 18 years enrolled in NCT00070473 were within range of values in adults.
8.5 Geriatric Use
Of the 3,946 patients enrolled in clinical studies of pemetrexed for injection, 34% were 65 and over and 4% were 75 and over. No overall differences in effectiveness were observed between these patients and younger patients. The incidences of Grade 3-4 anemia, fatigue, thrombocytopenia, hypertension, and neutropenia were higher in patients 65 years of age and older as compared to younger patients: in at least one of five randomized clinical trials. [
see Adverse Reactions (
6.1
) and Clinical Studies (
14.1
,
14.2
)].
4 Contraindications (4 CONTRAINDICATIONS)
Pemetrexed for injection is contraindicated in patients with a history of severe hypersensitivity reaction to pemetrexed [see Adverse Reactions ( 6.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the labeling:
7 Drug Interactions (7 DRUG INTERACTIONS)
2.3 Renal Impairment
- Pemetrexed for injection dosing recommendations are provided for patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater [ see Dosage and Administration ( 2.1 , 2.2)]. There is no recommended dose for patients whose creatinine clearance is less than 45 mL/min [ see Use in Specific Populations ( 8.6 )].
5.5 Radiation Recall
Radiation recall can occur with pemetrexed for injection in patients who have received radiation weeks to years previously. Monitor patients for inflammation or blistering in areas of previous radiation treatment. Permanently discontinue pemetrexed for injection for signs of radiation recall.
12.2 Pharmacodynamics
Pemetrexed inhibited the in vitro growth of mesothelioma cell lines (MSTO-211H, NCI-H2052) and showed synergistic effects when combined with cisplatin.
Based on population pharmacodynamic analyses, the depth of the absolute neutrophil counts (ANC) nadir correlates with the systemic exposure to pemetrexed and supplementation with folic acid and vitamin B 12. There is no cumulative effect of pemetrexed exposure on ANC nadir over multiple treatment cycles.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Pemetrexed for injection is a folate analog metabolic inhibitor indicated:
- in combination with pembrolizumab and platinum chemotherapy, for the initial treatment of patients with metastatic non-squamous non-small cell lung cancer (NSCLC), with no EGFR or ALK genomic tumor aberrations. ( 1.1)
- in combination with cisplatin for the initial treatment of patients with locally advanced or metastatic, non-squamous (NSCLC). ( 1.1)
- as a single agent for the maintenance treatment of patients with locally advanced or metastatic, non-squamous NSCLC whose disease has not progressed after four cycles of platinum-based first-line chemotherapy. ( 1.1)
- as a single agent for the treatment of patients with recurrent, metastatic non-squamous, NSCLC after prior chemotherapy. ( 1.1)
Limitations of Use:Pemetrexed for injection is not indicated for the treatment of patients with squamous cell, non-small cell lung cancer. (
1.1)
- initial treatment, in combination with cisplatin, of patients with malignant pleural mesothelioma whose disease is unresectable or who are otherwise not candidates for curative surgery. ( 1.2)
12.1 Mechanism of Action
Pemetrexed for injection is a folate analog metabolic inhibitor that disrupts folate-dependent metabolic processes essential for cell replication. In vitro studies show that pemetrexed inhibits thymidylate synthase (TS), dihydrofolate reductase, and glycinamide ribonucleotide formyltransferase (GARFT), which are folate- dependent enzymes involved in the de novo biosynthesis of thymidine and purine nucleotides. Pemetrexed is taken into cells by membrane carriers such as the reduced folate carrier and membrane folate binding protein transport systems. Once in the cell, pemetrexed is converted to polyglutamate forms by the enzyme folylpolyglutamate synthetase. The polyglutamate forms are retained in cells and are inhibitors of TS and GARFT.
5.7 Embryo Fetal Toxicity (5.7 Embryo-Fetal Toxicity)
Based on findings from animal studies and its mechanism of action, pemetrexed can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, intravenous administration of pemetrexed to pregnant mice during the period of organogenesis was teratogenic, resulting in developmental delays and increased malformations at doses lower than the recommended human dose of 500 mg/m
2. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with pemetrexed and for 6 months after the last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with pemetrexed and for 3 months after the last dose
[see Use in Specific Populations (
8.1, 8.3
) and Clinical Pharmacology (
12.1
)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Myelosuppression: Can cause severe bone marrow suppression resulting in cytopenia and an increased risk of infection. Do not administer pemetrexed for injection when the absolute neutrophil count is less than 1500 cells/mm 3and platelets are less than 100,000 cells/mm 3. Initiate supplementation with oral folic acid and intramuscular vitamin B 12to reduce the severity of hematologic and gastrointestinal toxicity of pemetrexed for injection. ( 2.4, 5.1)
- Renal Failure: Can cause severe, and sometimes fatal, renal failure. Do not administer when creatinine clearance is less than 45 mL/min. ( 2.3, 5.2)
- Bullous and Exfoliative Skin Toxicity: Permanently discontinue for severe and life-threatening bullous, blistering or exfoliating skin toxicity. ( 5.3)
- Interstitial Pneumonitis: Withhold for acute onset of new or progressive unexplained pulmonary symptoms. Permanently discontinue if pneumonitis is confirmed. ( 5.4)
- Radiation Recall: Can occur in patients who received radiation weeks to years previously; permanently discontinue for signs of radiation recall. ( 5.5)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise patients of the potential risk to a fetus and to use effective contraception. ( 5.7, 8.1, 8.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- The recommended dose of pemetrexed for injection administered with pembrolizumab and platinum chemotherapy in patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater is 500 mg/m 2as an intravenous infusion over 10 minutes, administered after pembrolizumab and prior to platinum chemotherapy, on Day 1 of each 21-day cycle. ( 2.1)
- The recommended dose of pemetrexed for injection, administered as a single agent or with cisplatin, in patients with creatinine clearance of 45 mL/minute or greater is 500 mg/m 2as an intravenous infusion over 10 minutes on Day 1 of each 21-day cycle. ( 2.1, 2.2)
- Initiate folic acid 400 mcg to 1000 mcg orally, once daily, beginning 7 days prior to the first dose of pemetrexed for injection and continue until 21 days after the last dose of pemetrexed for injection. ( 2.4)
- Administer vitamin B 12, 1 mg intramuscularly, 1 week prior to the first dose of pemetrexed for injection and every 3 cycles. ( 2.4)
- Administer dexamethasone 4 mg orally, twice daily the day before, the day of, and the day after pemetrexed for injection administration. ( 2.4)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For injection: 100 mg or 500 mg pemetrexed for injection as a white to light-yellow or green-yellow lyophilized powder in single-dose vials for reconstitution.
5.4 Interstitial Pneumonitis
Serious interstitial pneumonitis, including fatal cases, can occur with pemetrexed for injection treatment. Withhold pemetrexed for injection for acute onset of new or progressive unexplained pulmonary symptoms such as dyspnea, cough, or fever pending diagnostic evaluation. If pneumonitis is confirmed, permanently discontinue pemetrexed.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of pemetrexed for injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System— immune-mediated hemolytic anemia
Gastrointestinal— colitis, pancreatitis
General Disorders and Administration Site Conditions— edema
Injury, poisoning, and procedural complications— radiation recall
Respiratory— interstitial pneumonitis
Skin— Serious and fatal bullous skin conditions, Stevens-Johnson syndrome, and toxic epidermal necrolysis
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Advise not to breastfeed. ( 8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reactions rates cannot be directly compared to rates in other clinical trials and may not reflect the rates observed in clinical practice.
In clinical trials, the most common adverse reactions (incidence ≥20%) of pemetrexed for injection, when administered as a single agent, are fatigue, nausea, and anorexia. The most common adverse reactions (incidence ≥20%) of pemetrexed for injection, when administered in combination with cisplatin are vomiting, neutropenia, anemia, stomatitis/pharyngitis, thrombocytopenia, and constipation. The most common adverse reactions (incidence ≥20%) of pemetrexed for injection, when administered in combination with pembrolizumab and platinum chemotherapy, are fatigue/asthenia, nausea, constipation, diarrhea, decreased appetite, rash, vomiting, cough, dyspnea, and pyrexia.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling ( Patient Information).
2.7 Preparation for Administration
- Pemetrexed for injection is a hazardous drug. Follow applicable special handling and disposal procedures. 1
- Calculate the dose of pemetrexed for injection and determine the number of vials needed.
- Reconstitute pemetrexed for injection to achieve a concentration of 25 mg/mL as follows:
- Reconstitute each 100-mg vial with 4.2 mL of 0.9% Sodium Chloride Injection, USP (preservative-free)
- Reconstitute each 500-mg vial with 20 mL of 0.9% Sodium Chloride Injection, USP (preservative-free)
- Do not use calcium-containing solutions for reconstitution.
- Gently swirl each vial until the powder is completely dissolved. The resulting solution is clear and ranges in color from colorless to yellow or green-yellow. FURTHER DILUTION IS REQUIRED prior to administration.
- Store reconstituted, preservative-free product under refrigerated conditions [2-8°C (36-46°F)] for no longer than 24 hours from the time of reconstitution. Discard vial after 24 hours.
- Inspect reconstituted product visually for particulate matter and discoloration prior to further dilution. If particulate matter is observed, discard vial.
- Withdraw the calculated dose of pemetrexed for injection from the vial(s) and discard vial with any unused portion.
- Further dilute pemetrexed for injection with 0.9% Sodium Chloride Injection (preservative-free) to achieve a total volume of 100 mL for intravenous infusion.
- Store diluted, reconstituted product under refrigerated conditions [2-8°C (36-46°F)] for no more than 24 hours from the time of reconstitution. Discard after 24 hours.
8.6 Patients With Renal Impairment (8.6 Patients with Renal Impairment)
Pemetrexed for injection is primarily excreted by the kidneys. Decreased renal function results in reduced clearance and greater exposure (AUC) to pemetrexed for injection compared with patients with normal renal function [
Warnings and Precautions (
5.2
,
5.6
) and Clinical Pharmacology (
12.3
)]. No dose is recommended for patients with creatinine clearance less than 45 mL/min [
see Dosage and Administration (
2.3
)].
2.2 Recommended Dosage for Mesothelioma
- The recommended dose of pemetrexed for injection when administered with cisplatin in patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater is 500 mg/m 2as an intravenous infusion over 10 minutes on Day 1 of each 21-day cycle until disease progression or unacceptable toxicity.
5.3 Bullous and Exfoliative Skin Toxicity
Serious and sometimes fatal, bullous, blistering and exfoliative skin toxicity, including cases suggestive of Stevens-Johnson Syndrome/Toxic epidermal necrolysis can occur with pemetrexed for injection. Permanently discontinue pemetrexed for injection for severe and life-threatening bullous, blistering or exfoliating skin toxicity.
2.1 Recommended Dosage for Non Squamous Nsclc (2.1 Recommended Dosage for Non-Squamous NSCLC)
- The recommended dose of pemetrexed for injection when administered with pembrolizumab and platinum chemotherapy for the initial treatment of metastatic non-squamous NSCLC in patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater is 500 mg/m 2as an intravenous infusion over 10 minutes administered after pembrolizumab and prior to carboplatin or cisplatin on Day 1 of each 21-day cycle for 4 cycles. Following completion of platinum-based therapy, treatment with pemetrexed for injection with or without pembrolizumab is administered until disease progression or unacceptable toxicity. Please refer to the full prescribing information for pembrolizumab and for carboplatin or cisplatin.
- The recommended dose of pemetrexed for injection when administered with cisplatin for initial treatment of locally advanced or metastatic non-squamous NSCLC in patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater is 500 mg/m 2as an intravenous infusion over 10 minutes administered prior to cisplatin on Day 1 of each 21-day cycle for up to six cycles in the absence of disease progression or unacceptable toxicity.
- The recommended dose of pemetrexed for injection for maintenance treatment of non-squamous NSCLC in patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater is 500 mg/m 2as an intravenous infusion over 10 minutes on Day 1 of each 21-day cycle until disease progression or unacceptable toxicity after four cycles of platinum-based first-line chemotherapy.
- The recommended dose of pemetrexed for injection for treatment of recurrent non-squamous NSCLC in patients with a creatinine clearance (calculated by Cockcroft-Gault equation) of 45 mL/min or greater is 500 mg/m 2as an intravenous infusion over 10 minutes on Day 1 of each 21-day cycle until disease progression or unacceptable toxicity.
2.6 Dosage Modifications for Adverse Reactions
Obtain complete blood count on Days 1, 8, and 15 of each cycle. Assess creatinine clearance prior to each cycle. Do not administer pemetrexed for injection if the creatinine clearance is less than 45 mL/min.
Delay initiation of the next cycle of pemetrexed for injection until:
- recovery of non-hematologic toxicity to Grade 0-2,
- absolute neutrophil count (ANC) is 1500 cells/mm 3or higher, and
- platelet count is 100,000 cells/mm 3or higher.
Upon recovery, modify the dosage of pemetrexed for injection in the next cycle as specified in Table 1.
For dosing modifications for cisplatin, carboplatin, or pembrolizumab, refer to their prescribing information .
|
Toxicity in Most Recent Treatment Cycle
|
Pemetrexed for injection Dose Modification for Next Cycle
|
|
Myelosuppressive toxicity
[see Warnings and Precautions (
5.1)]
|
|
| ANC less than 500/mm
3
and
platelets greater than or equal to 50,000/mm
3
OR Platelet count less than 50,000/mm 3without bleeding. |
75% of previous dose
|
| Platelet count less than 50,000/mm
3with bleeding
|
50% of previous dose
|
| Recurrent Grade 3 or 4 myelosuppression after 2 dose reductions
|
Discontinue
|
|
Non-hematologic toxicity
|
|
| Any Grade 3 or 4 toxicities EXCEPT mucositis or neurologic toxicity
OR Diarrhea requiring hospitalization |
75% of previous dose
|
| Grade 3 or 4 mucositis
|
50% of previous dose
|
| Renal toxicity
[see Warnings and Precautions (
5.2)]
|
Withhold until creatinine clearance is 45 mL/min or greater
|
| Grade 3 or 4 neurologic toxicity
|
Permanently discontinue
|
| Recurrent Grade 3 or 4 non-hematologic toxicity after 2 dose reductions
|
Permanently discontinue
|
| Severe and life-threatening Skin Toxicity
[see Warnings and Precautions (
5.3)]
|
Permanently discontinue
|
| Interstitial Pneumonitis
[see Warnings and Precautions (
5.4)]
|
Permanently discontinue
|
8.3 Females and Males of Reproductive Potential
Based on animal data pemetrexed for injection can cause malformations and developmental delays when administered to a pregnant woman [see Use in Specific Populations (8.1)].
Pregnancy Testing
Verify pregnancy status of females of reproductive potential prior to initiating pemetrexed for injection [see Use in Specific Populations (8.1)].
1.1 Non Squamous Non Small Cell Lung Cancer (nsclc) (1.1 Non-Squamous Non-Small Cell Lung Cancer (NSCLC))
Pemetrexed for injection is indicated:
- in combination with pembrolizumab and platinum chemotherapy, for the initial treatment of patients with metastatic non-squamous non-small cell lung cancer (NSCLC), with no EGFR or ALK genomic tumor aberrations.
- in combination with cisplatin for the initial treatment of patients with locally advanced or metastatic, non-squamous, NSCLC.
- as a single agent for the maintenance treatment of patients with locally advanced or metastatic, non-squamous NSCLC whose disease has not progressed after four cycles of platinum-based first-line chemotherapy.
- as a single agent for the treatment of patients with recurrent, metastatic non-squamous, NSCLC after prior chemotherapy.
Limitations of Use:Pemetrexed for injection is not indicated for the treatment of patients with squamous cell, non-small cell lung cancer [see Clinical Studies 14.1 ].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity studies have been conducted with pemetrexed. Pemetrexed was clastogenic in an in vivo micronucleus assay in mouse bone marrow but was not mutagenic in multiple in vitro tests (Ames assay, Chinese Hamster Ovary cell assay).
Pemetrexed administered intraperitoneally at doses of ≥0.1 mg/kg/day to male mice (approximately 0.0006 times the recommended human dose based on BSA) resulted in reduced fertility, hypospermia, and testicular atrophy.
5.6 Increased Risk of Toxicity With Ibuprofen in Patients With Renal Impairment (5.6 Increased Risk of Toxicity with Ibuprofen in Patients with Renal Impairment)
Exposure to pemetrexed is increased in patients with mild to moderate renal impairment who take concomitant ibuprofen, increasing the risks of adverse reactions of pemetrexed. In patients with creatinine clearances between 45 mL/min and 79 mL/min, avoid administration of ibuprofen for 2 days before, the day of, and 2 days following administration of pemetrexed. If concomitant ibuprofen use cannot be avoided, monitor patients more frequently for pemetrexed adverse reactions, including myelosuppression, renal, and gastrointestinal toxicity
[see Dosage and Administration (
2.5
),
Drug
Interactions (
7
), and Clinical Pharmacology (
12.3
)].
5.1 Myelosuppression and Increased Risk of Myelosuppression Without Vitamin Supplementation (5.1 Myelosuppression and Increased Risk of Myelosuppression without Vitamin Supplementation)
Pemetrexed for injection can cause severe myelosuppression resulting in a requirement for transfusions and which may lead to neutropenic infection. The risk of myelosuppression is increased in patients who do not receive vitamin supplementation. In Study JMCH, incidences of Grade 3-4 neutropenia (38% versus 23%), thrombocytopenia (9% versus 5%), febrile neutropenia (9% versus 0.6%), and neutropenic infection (6% versus 0) were higher in patients who received pemetrexed plus cisplatin without vitamin supplementation as compared to patients who were fully supplemented with folic acid and vitamin B
12 prior to and throughout pemetrexed plus cisplatin treatment.
Initiate supplementation with oral folic acid and intramuscular vitamin B
12 prior to the first dose of pemetrexed; continue vitamin supplementation during treatment and for 21 days after the last dose of pemetrexed to reduce the severity of hematologic and gastrointestinal toxicity of pemetrexed for injection [s
ee Dosage and Administration (
2.4
)]. Obtain a complete blood count at the beginning of each cycle. Do not administer pemetrexed for injection until the ANC is at least 1500 cells/mm
3 and platelet count is at least 100,000 cells/mm
3. Permanently reduce pemetrexed for injection in patients with an ANC of less than 500 cells/mm
3 or platelet count of less than 50,000 cells/mm
3 in previous cycles [
see Dosage and Administration (
2.6
)].
In Studies JMDB and JMCH, among patients who received vitamin supplementation, incidence of Grade 3-4 neutropenia was 15% and 23%, the incidence of Grade 3-4 anemia was 6% and 4%, and incidence of Grade 3-4 thrombocytopenia was 4% and 5%, respectively. In Study JMCH, 18% of patients in the pemetrexed for injection arm required red blood cell transfusions compared to 7% of patients in the cisplatin arm [
see Adverse Reactions (
6.1
)]. In Studies JMEN, PARAMOUNT, and JMEI, where all patients received vitamin supplementation, incidence of Grade 3-4 neutropenia ranged from 3% to 5%, and incidence of Grade 3-4 anemia ranged from 3% to 5%.
2.5 Dosage Modification of Ibuprofen in Patients With Mild to Moderate Renal Impairment Receiving Pemetrexed for Injection (2.5 Dosage Modification of Ibuprofen in Patients with Mild to Moderate Renal Impairment Receiving Pemetrexed for injection)
In patients with creatinine clearances between 45 mL/min and 79 mL/min, modify administration of ibuprofen as follows [see Warnings and Precautions ( 5.6), Drug Interactions ( 7) and Clinical Pharmacology ( 12.3)] :
- Avoid administration of ibuprofen for 2 days before, the day of, and 2 days following administration of pemetrexed for injection.
- Monitor patients more frequently for myelosuppression, renal, and gastrointestinal toxicity, if concomitant administration of ibuprofen cannot be avoided.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:05.149983 · Updated: 2026-03-14T22:26:18.292818