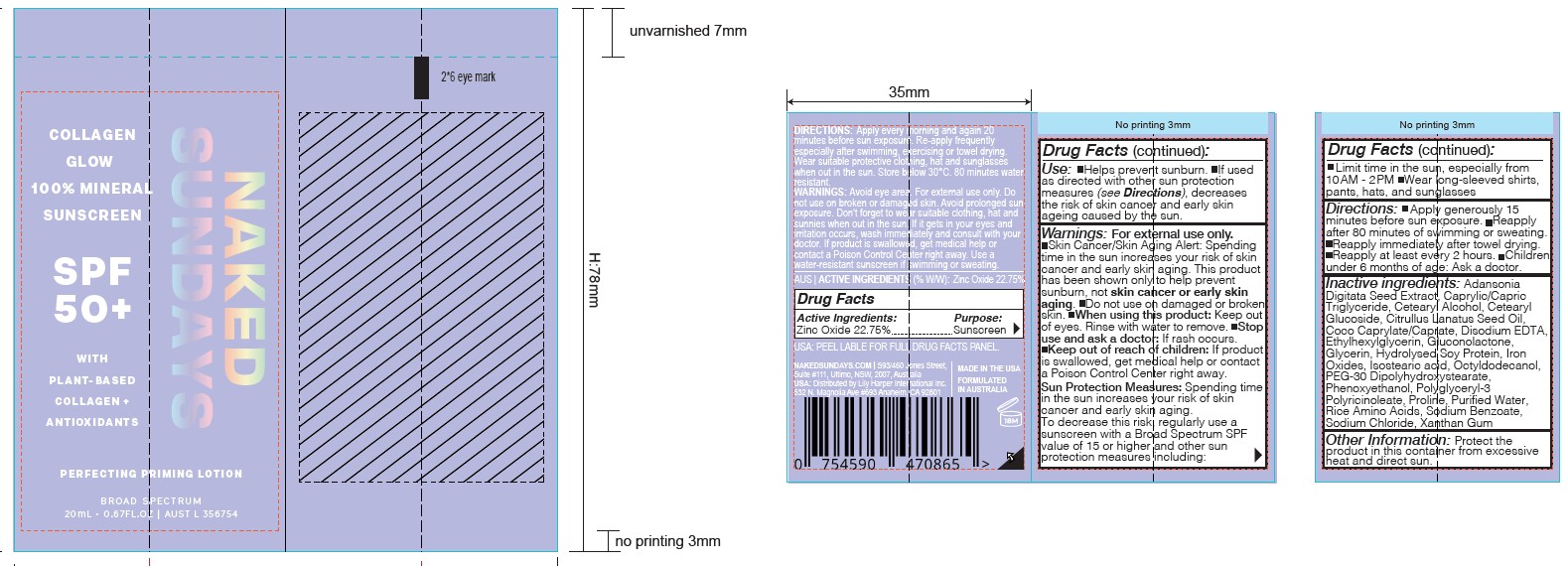
Naked Sundays Collagen Glow Mineral Sunscreen
388fb3db-29d2-bb29-e063-6394a90a4efb
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
ZINC OXIDE 22.75%
Purpose
Sunscreen
Medication Information
Purpose
Sunscreen
Description
ZINC OXIDE 22.75%
Uses
- Helps prevent sunburn.
- If used as directed with other sun protection measures (see directions), decreases the risk of skin cancer and early skin ageing caused by the sun.
Section 50565-1
Keep out of reach of children. If product is swallowed, get medical help or contact a Poison Control Center right away.
Section 51945-4
Warnings
For external use only.
- Do not use on damaged or broken skin.
- When using this product: keep out of eyes. Rinse with water to remove.
- Stop use and ask a doctor if: rash occurs.
- CONTAINS SODIUM BENZOATE.
Directions
- APPLY GENEROUSLY 15 MINUTES BEFORE SUN EXPOSURE.
- Reapply after 80 minutes of swimming or sweating.
- Reapply immediately after towel drying.
- Reapply at least every 2 hours.
- Children under 3 years of age: Ask a doctor.
SUN PROTECTION MEASURES:
Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- Limit time in the sun, especially from 10.00AM - 2.00PM
- Wear long-sleeved shirts, pants, hats, and sunglasses
Active Ingredient
ZINC OXIDE 22.75%
Other Information
Protect the product in this container from excessive heat and direct sun.
Inactive Ingredients
CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) ,COCO-CAPRYLATE (UNII: 4828G836N6) ,EDETATE DISODIUM (UNII: 7FLD91C86K),GLUCONOLACTONE (UNII: WQ29KQ9POT),ADANSONIA DIGITATA SEED (UNII: 2936P60TPX), ORYZA SATIVA WHOLE (UNII: 84IVV0906Z),HYDROLYZED SOY PROTEIN (ENZYMATIC; 2000 MW) (UNII: 1394NXB9L6),ISOSTEARIC ACID (UNII: X33R8U0062),TRICAPRIN (UNII: O1PB8EU98M),OCTYLDODECANOL (UNII: 461N1O614Y),PEG-30 DIPOLYHYDROXYSTEARATE (UNII: 9713Q0S7FO),POLYGLYCERYL-3 RICINOLEATE (UNII: MZQ63P0N0W)
PROLINE (UNII: 9DLQ4CIU6V),WATER (UNII: 059QF0KO0R),SODIUM BENZOATE (UNII: OJ245FE5EU),SODIUM CHLORIDE (UNII: 451W47IQ8X),ETHYLHEXYLGLYCERIN (UNII: 147D247K3P),PHENOXYETHANOL (UNII: HIE492ZZ3T)
CETEARYL GLUCOSIDE (UNII: 09FUA47KNA)
Structured Label Content
Uses (USES)
- Helps prevent sunburn.
- If used as directed with other sun protection measures (see directions), decreases the risk of skin cancer and early skin ageing caused by the sun.
Section 50565-1 (50565-1)
Keep out of reach of children. If product is swallowed, get medical help or contact a Poison Control Center right away.
Section 51945-4 (51945-4)
Purpose
Sunscreen
Warnings (WARNINGS)
For external use only.
- Do not use on damaged or broken skin.
- When using this product: keep out of eyes. Rinse with water to remove.
- Stop use and ask a doctor if: rash occurs.
- CONTAINS SODIUM BENZOATE.
Directions (DIRECTIONS)
- APPLY GENEROUSLY 15 MINUTES BEFORE SUN EXPOSURE.
- Reapply after 80 minutes of swimming or sweating.
- Reapply immediately after towel drying.
- Reapply at least every 2 hours.
- Children under 3 years of age: Ask a doctor.
SUN PROTECTION MEASURES:
Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- Limit time in the sun, especially from 10.00AM - 2.00PM
- Wear long-sleeved shirts, pants, hats, and sunglasses
Active Ingredient (ACTIVE INGREDIENT)
ZINC OXIDE 22.75%
Other Information (OTHER INFORMATION)
Protect the product in this container from excessive heat and direct sun.
Inactive Ingredients (INACTIVE INGREDIENTS)
CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) ,COCO-CAPRYLATE (UNII: 4828G836N6) ,EDETATE DISODIUM (UNII: 7FLD91C86K),GLUCONOLACTONE (UNII: WQ29KQ9POT),ADANSONIA DIGITATA SEED (UNII: 2936P60TPX), ORYZA SATIVA WHOLE (UNII: 84IVV0906Z),HYDROLYZED SOY PROTEIN (ENZYMATIC; 2000 MW) (UNII: 1394NXB9L6),ISOSTEARIC ACID (UNII: X33R8U0062),TRICAPRIN (UNII: O1PB8EU98M),OCTYLDODECANOL (UNII: 461N1O614Y),PEG-30 DIPOLYHYDROXYSTEARATE (UNII: 9713Q0S7FO),POLYGLYCERYL-3 RICINOLEATE (UNII: MZQ63P0N0W)
PROLINE (UNII: 9DLQ4CIU6V),WATER (UNII: 059QF0KO0R),SODIUM BENZOATE (UNII: OJ245FE5EU),SODIUM CHLORIDE (UNII: 451W47IQ8X),ETHYLHEXYLGLYCERIN (UNII: 147D247K3P),PHENOXYETHANOL (UNII: HIE492ZZ3T)
CETEARYL GLUCOSIDE (UNII: 09FUA47KNA)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:56.564500 · Updated: 2026-03-14T23:10:01.237919