3866e2c1-051b-0b01-e063-6394a90af5cf
3866e2c1-051b-0b01-e063-6394a90af5cf
34390-5
HUMAN OTC DRUG LABEL
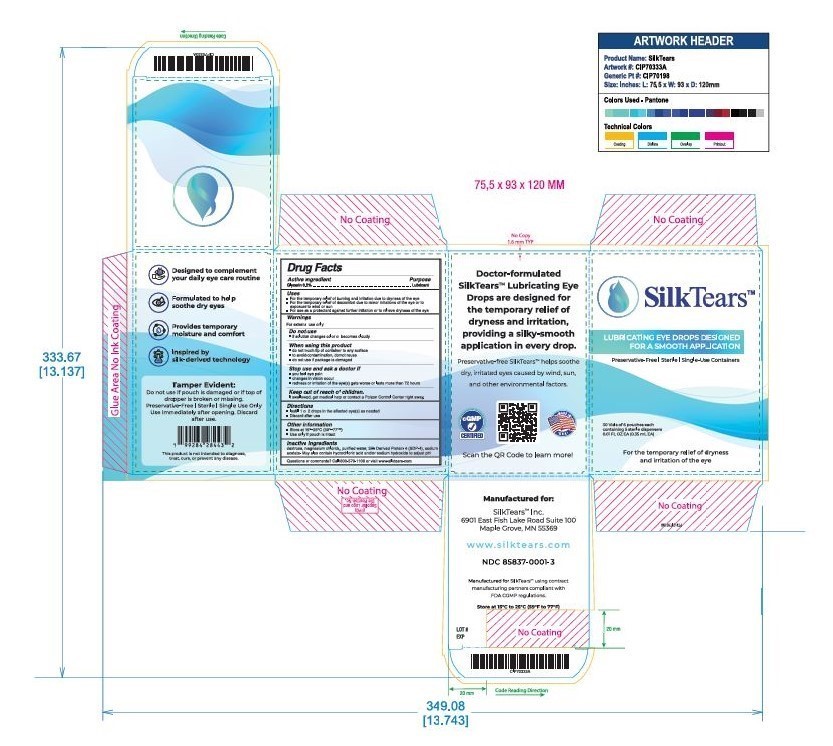
Drug Facts
Composition & Product
Identifiers & Packaging
Dosage and Administration
Instill 1 to 2 drops in the affected eye(s) as needed.
Purpose
Lubricant
Description
Lubricant
Medication Information
Warnings and Precautions
For external use only.
Do not use if solution changes color or becomes cloudy.
When using this product:
- do not touch tip of container to any surface to avoid contamination
- do not reuse
- once opened, discard
Stop use and ask a doctor if you experience:
- eye pain
- changes in vision
- continued redness or irritation
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Active Ingredient
Glycerin 0.2% – Lubricant
Indications and Usage
For the temporary relief of burning and irritation due to dryness of the eye.
Purpose
Lubricant
Dosage and Administration
Instill 1 to 2 drops in the affected eye(s) as needed.
Description
Lubricant
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6
Inactive ingredients: dextrose, magnesium chloride, purified water, Silk-Derived Protein-4 (SDP-4), sodium acetate. May also contain hydrochloric acid and/or sodium hydroxide to adjust PH
Section 51945-4
30 vials of 6 pouches each containing 5 sterile dispensers 0.01 FL OZ (0.35 mL EA)
Structured Label Content
Warnings and Precautions (34071-1)
For external use only.
Do not use if solution changes color or becomes cloudy.
When using this product:
- do not touch tip of container to any surface to avoid contamination
- do not reuse
- once opened, discard
Stop use and ask a doctor if you experience:
- eye pain
- changes in vision
- continued redness or irritation
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Active Ingredient (55106-9)
Glycerin 0.2% – Lubricant
Indications and Usage (34067-9)
For the temporary relief of burning and irritation due to dryness of the eye.
Purpose
Lubricant
Dosage and Administration
Instill 1 to 2 drops in the affected eye(s) as needed.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6 (51727-6)
Inactive ingredients: dextrose, magnesium chloride, purified water, Silk-Derived Protein-4 (SDP-4), sodium acetate. May also contain hydrochloric acid and/or sodium hydroxide to adjust PH
Section 51945-4 (51945-4)
30 vials of 6 pouches each containing 5 sterile dispensers 0.01 FL OZ (0.35 mL EA)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:32.500143 · Updated: 2026-03-14T23:09:21.323328