Hemorrodil Unguento Plus
38443830-b7d8-4028-8878-8a0fde5e888a
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Anti-Itch
Medication Information
Purpose
Anti-Itch
Description
Drug Facts
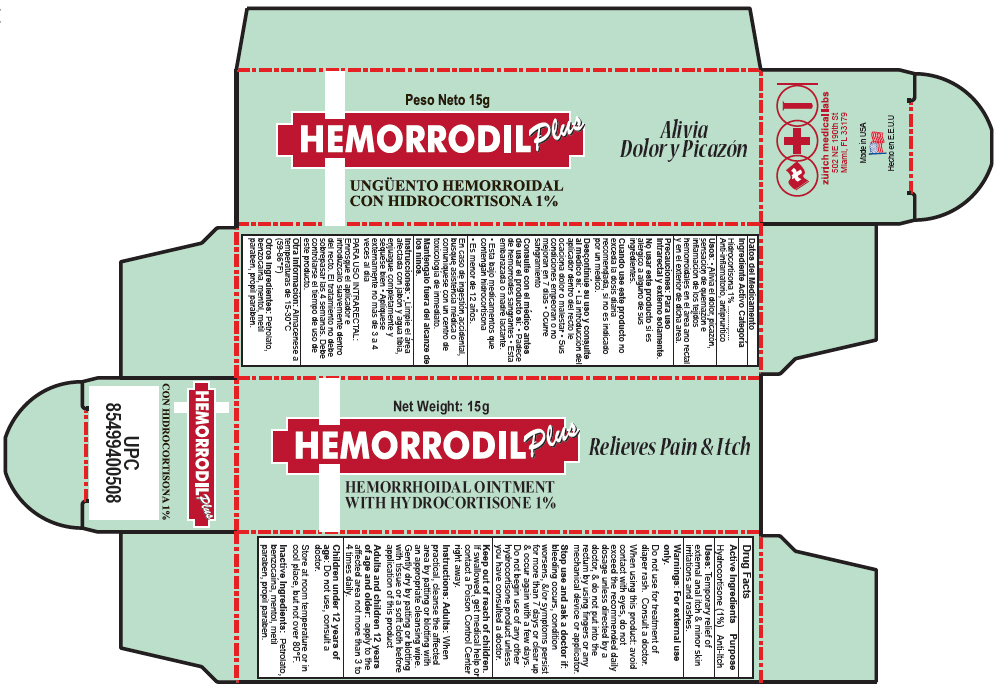
Uses
Temporary relief of external anal itch & minor skin irritations and rashes.
Adults
When practical, cleanse the affected area by patting or blotting with an appropriate cleansing wipe. Gently dry by patting or blotting with tissue or a soft cloth before application of this product
Section 42229-5
Drug Facts
Section 44425-7
Store at room temperature or in cool place, but not over 80°F.
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if: bleeding occurs, condition worsens, &/or symptoms persist for more than 7 days or clear up & occur again with a few days. Do not begin use of any other hydrocortisone product unless you have consulted a doctor.
Section 50567-7
When using this product: avoid contact with eyes, do not exceed the recommended daily dosage unless directed by a doctor, & do not put into the rectum by using fingers or any mechanical device or applicator.
Section 50570-1
Do not use for treatment of diaper rash. Consult a doctor.
Warnings
For external use only.
Active Ingredients
Hydrocortisone (1%)
Inactive Ingredients
Petrolato, benzocaína, mentol, metil paraben, propil paraben.
Children Under 12 Years of Age
Do not use, consult a doctor.
Principal Display Panel 15 G Tube Carton
Net Weight: 15g
HEMORRODIL Plus
Relieves Pain & Itch
HEMORRHOIDAL OINTMENT
WITH HYDROCORTISONE 1%
Structured Label Content
Uses
Temporary relief of external anal itch & minor skin irritations and rashes.
Adults
When practical, cleanse the affected area by patting or blotting with an appropriate cleansing wipe. Gently dry by patting or blotting with tissue or a soft cloth before application of this product
Section 42229-5 (42229-5)
Drug Facts
Section 44425-7 (44425-7)
Store at room temperature or in cool place, but not over 80°F.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if: bleeding occurs, condition worsens, &/or symptoms persist for more than 7 days or clear up & occur again with a few days. Do not begin use of any other hydrocortisone product unless you have consulted a doctor.
Section 50567-7 (50567-7)
When using this product: avoid contact with eyes, do not exceed the recommended daily dosage unless directed by a doctor, & do not put into the rectum by using fingers or any mechanical device or applicator.
Section 50570-1 (50570-1)
Do not use for treatment of diaper rash. Consult a doctor.
Purpose
Anti-Itch
Warnings
For external use only.
Active Ingredients
Hydrocortisone (1%)
Inactive Ingredients
Petrolato, benzocaína, mentol, metil paraben, propil paraben.
Children Under 12 Years of Age (Children under 12 years of age)
Do not use, consult a doctor.
Principal Display Panel 15 G Tube Carton (PRINCIPAL DISPLAY PANEL - 15 g Tube Carton)
Net Weight: 15g
HEMORRODIL Plus
Relieves Pain & Itch
HEMORRHOIDAL OINTMENT
WITH HYDROCORTISONE 1%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:45.815570 · Updated: 2026-03-14T22:53:42.713163